

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (5): 1552.doi: 10.7503/cjcu20200750
收稿日期:2020-10-15
出版日期:2021-05-10
发布日期:2021-01-27
通讯作者:
孙永明
E-mail:yongmingsun@hust.edu.cn
基金资助:
MAO Eryang1, WANG Li2, SUN Yongming1( )
)
Received:2020-10-15
Online:2021-05-10
Published:2021-01-27
Contact:
SUN Yongming
E-mail:yongmingsun@hust.edu.cn
Supported by:摘要:
现有的以石墨为负极的锂离子电池能量密度逐渐接近其理论极限. 基于合金化反应机制的高容量含锂负极材料LixMy(M为能够和锂发生合金化反应的元素)是一类新兴的负极材料, 具有数倍于石墨的储锂比容量, 且可以为电池提供活性锂源. 这些特性使其能够与高容量无锂正极材料(如S, O2, FeF3和V2O5等)相匹配, 构建下一代高比能锂离子电池新体系. 本文综述了近年来高容量合金基含锂负极材料(如LixSi, LixSn, Li3P和LixAl基系列材料)的研究进展, 分析了所面临的挑战, 概述了材料的合成与电极的制备方法, 并介绍了它们在常规锂离子电池、 锂离子-硫电池及锂离子-空气电池等多个全电池体系中的应用实例, 提出并举证了其电化学性能优化与调控的策略, 最后展望了未来的研究方向.
中图分类号:
TrendMD:
毛尔洋, 王莉, 孙永明. 锂离子电池高容量合金基含锂负极材料的研究进展. 高等学校化学学报, 2021, 42(5): 1552.
MAO Eryang, WANG Li, SUN Yongming. Advances in Alloy-based High-capacity Li-containing Anodes for Lithium-ion Batteries. Chem. J. Chinese Universities, 2021, 42(5): 1552.
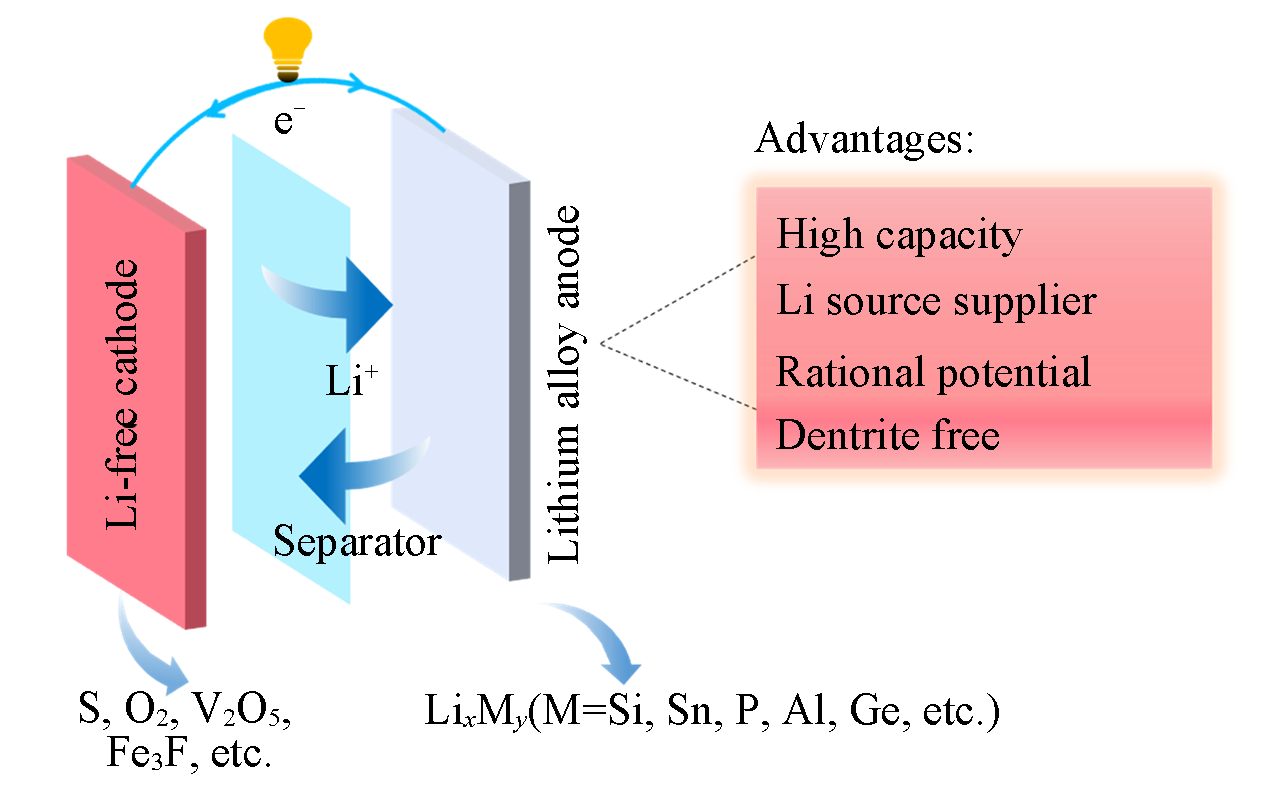
Fig.1 Novel battery mechanism coupling high?capacity Li?containing anode and Li?free cathode based on alloy?dealloy reaction, illustrating the advantages of utilizing high?capacity Li?containing anodes
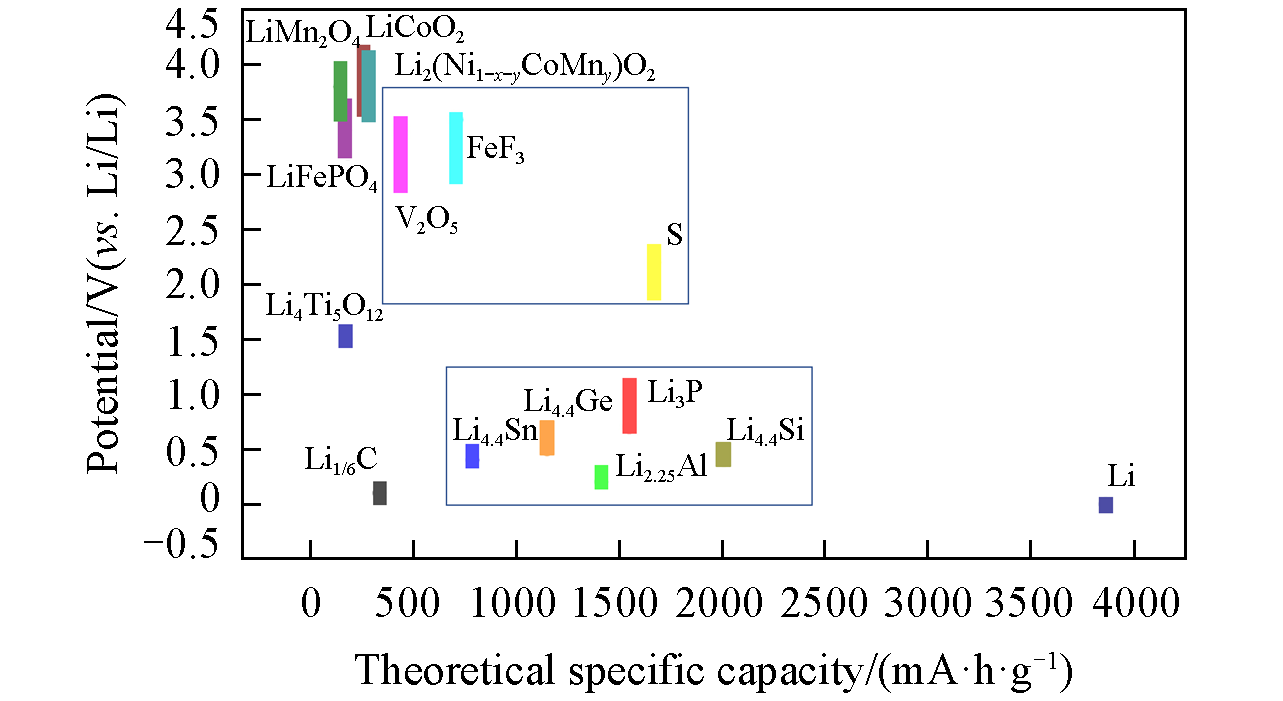
Fig.2 Theoretical specific capacity and potential(vs. Li/Li+) of various high?capa? city Li?containing anodes and Li?free cathodes, in contrast to the conventional intercalation?type electrodes
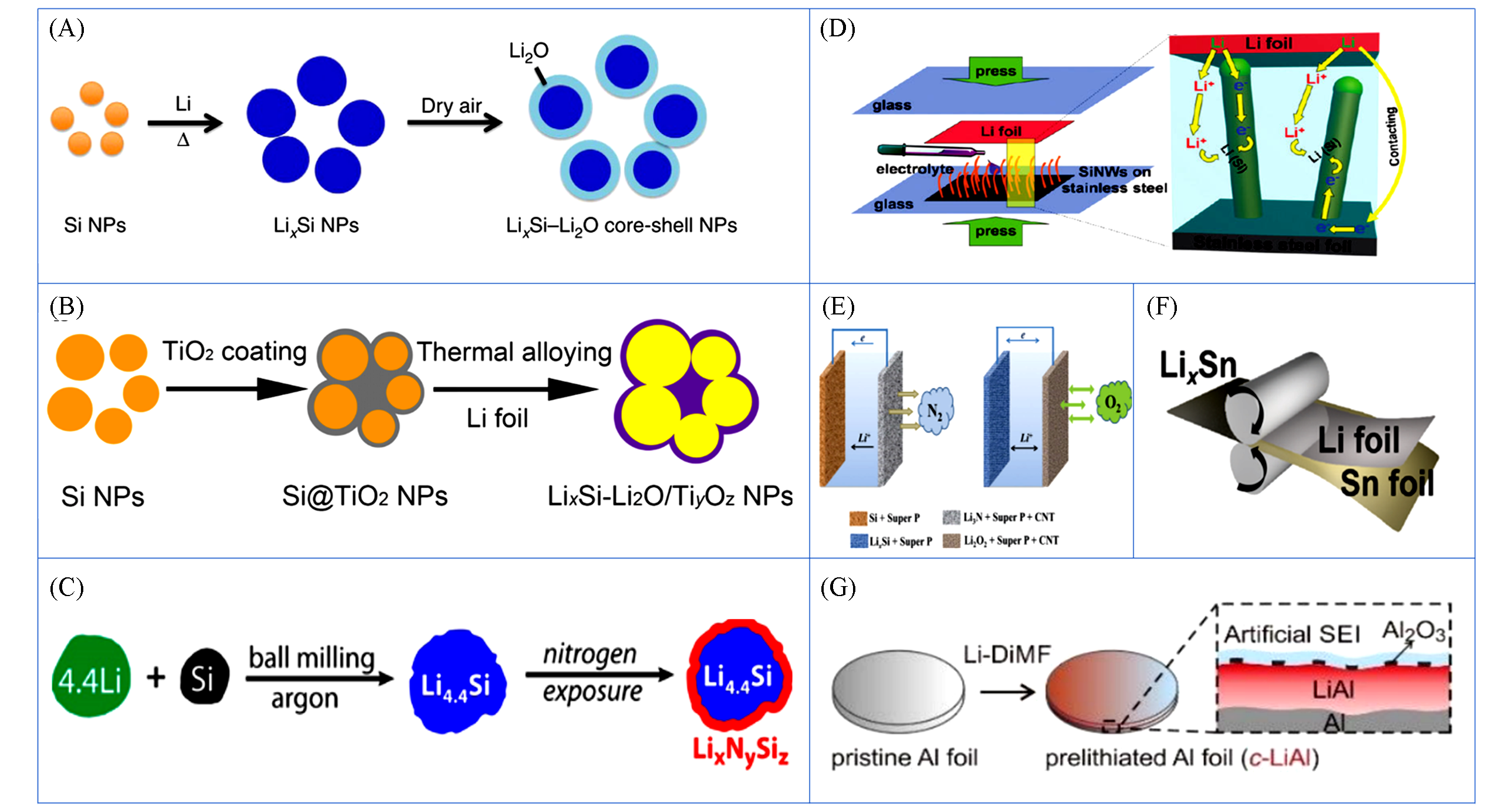
Fig.4 Various synthesis methods to prepare LixMy anodes materials(A) High-temperature melting-solidification method to prepare LixSi NPs using Si NPs and molten Li. A dense passivation layer is formed on the surface of LixSi NPs after exposure to trace amounts of oxygen, preventing the LixSi alloy from further oxidation in dry air and showing good air stability[65]. (B) A two-step approach of coating- lithiation strategy to prepare stable LixSi-Li2O/TiyOz core-shell NPs[70]. (C) High-energy ball milling fabrication for Li4.4Si@LixNySiz anode[67]. (D) Schematic illustration of electrochemical lithiation for Si nanowires(NWs) and the internal electron and Li+ pathways during the lithiation[61]. (E) Schematic illustration of a pre-loaded Li3N cathode as the Li source for in situ electrochemical Li-Si alloying and pared with O2 cathode[91]. (F) Roll-to-roll lithiation method for the preparation of LixSn, LixAl and LixSi/C anode via stacking and rolling[71]. (G) Chemical lithiation for the preparation of Li-Al anode using polishing Al foil in the Li-DiMF solution[93].(A) Copyright 2014, Springer Nature; (B) Copyright 2018, American Chemical Society; (C) Copyright 2017, American Chemical Society; (D) Copyright 2011, American Chemical Society; (E) Copyright 2018, Royal Society of Chemical; (F) Copyright 2019, Royal Society of Chemical; (G) Copyright 2020, Royal Society of Chemistry.
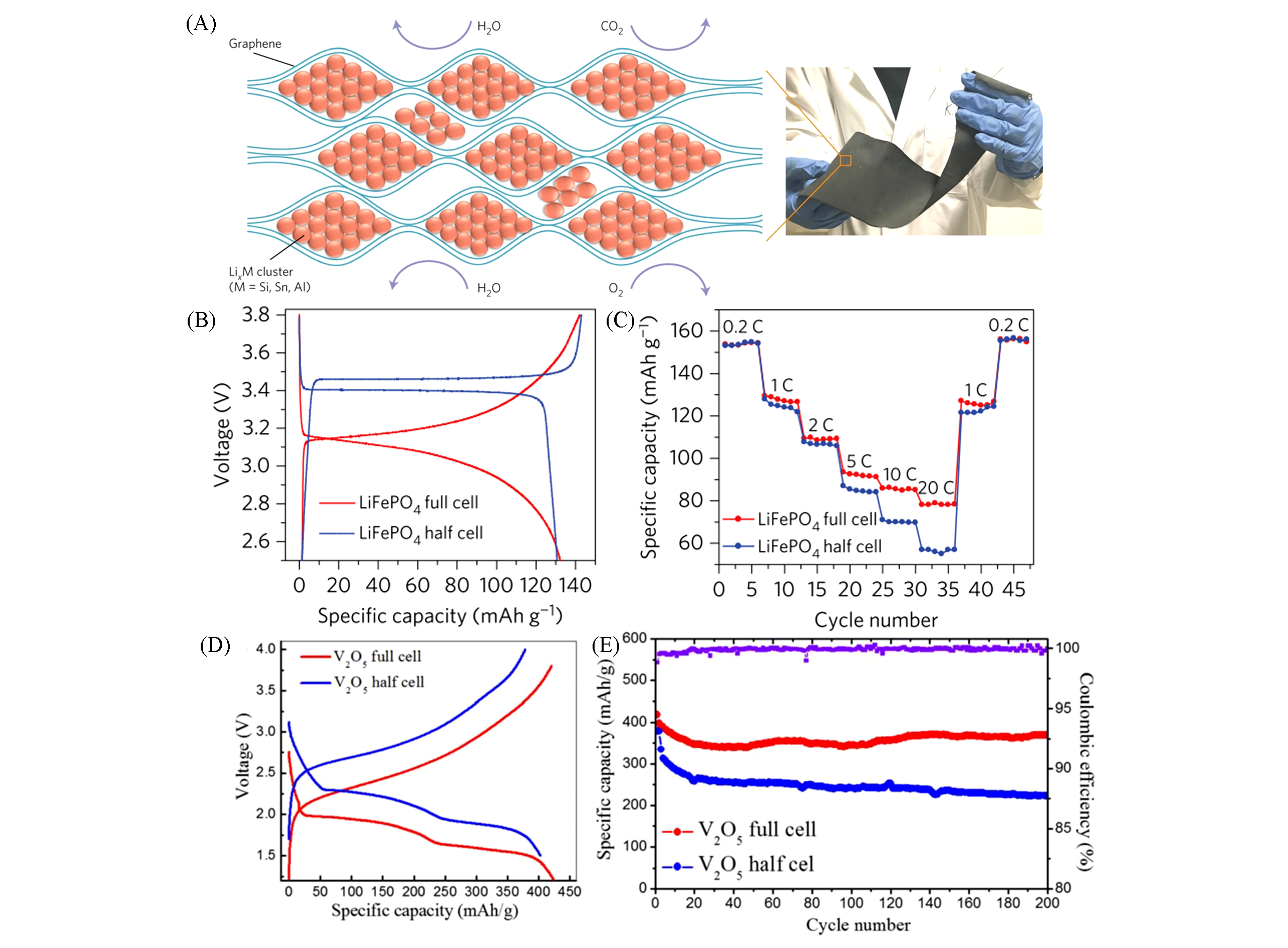
Fig.5 Air?stable and freestanding lithium alloy/graphene foil anodes[68](A) Unique foil structure comprising densely packed reactive LixM nanoparticles encapsulated by large graphene sheets; (B) voltage profiles; (C) rate capability of LixSi/graphene||LiFePO4 full cell and Li||LiFePO4(LiFePO4/Super P/PVDF mass ratio of 70∶20∶10, 1C=170 mA/g of LiFePO4); (D) first-cycle voltage profiles; (E) cycling performance of LixSi/graphene||V2O5 full cell(red) and Li||V2O5(V2O5 nanosheets/Super P/PVDF mass ratio of 70∶20∶10) half cell(blue) at the rate of 1C. The capacity and rate are both based on the mass of V2O5 nanosheets in the cathode. The mass ratio of anode to cathode is 1∶4. The purple curve is the Coulombic efficiency of the LixSi/graphene||V2O5 full cell.Copyright 2017, Springer Nature.
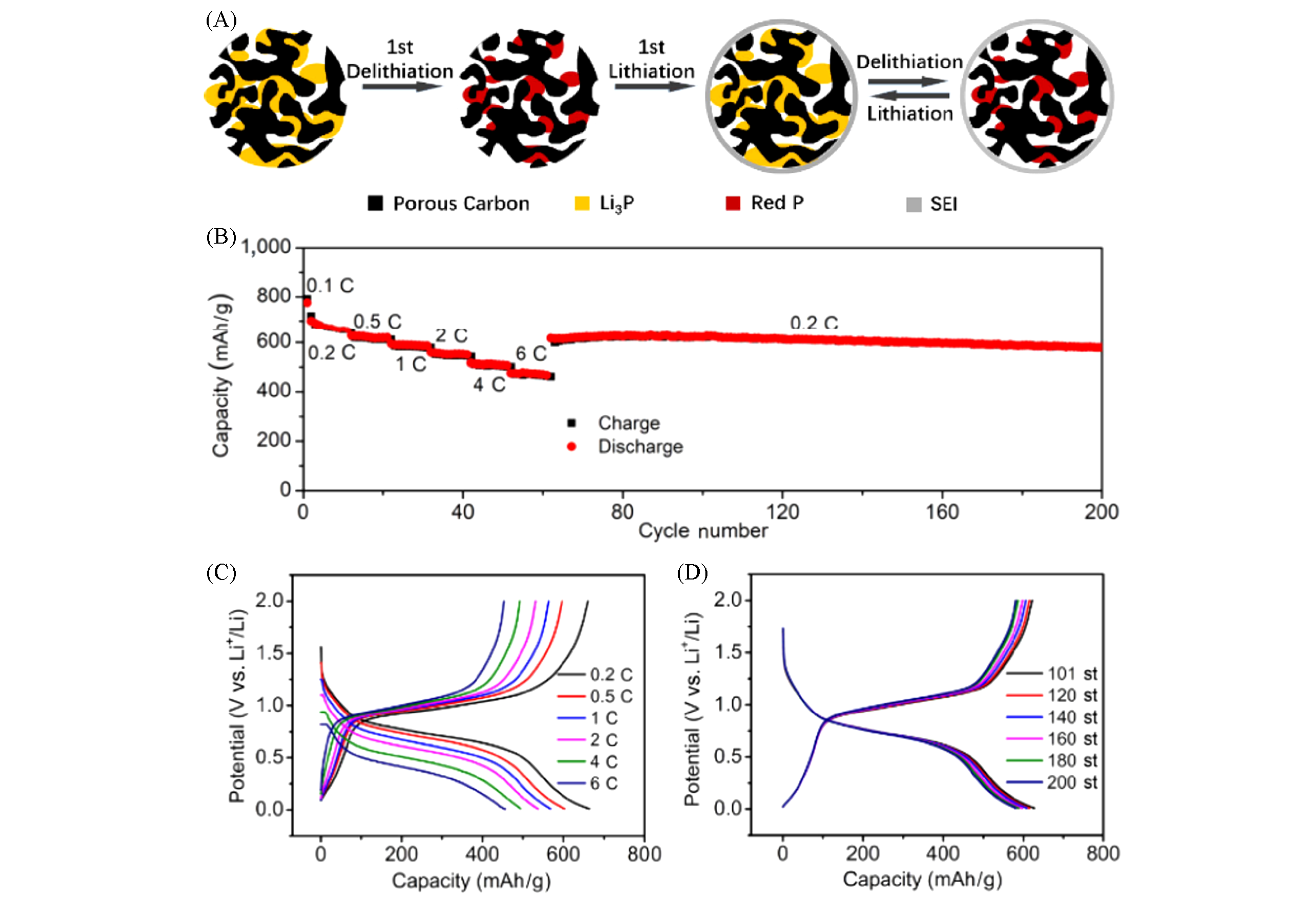
Fig.6 Li3P/C nanocomposite for high?capacity lithium?containing anode[72](A) Schematic illustrates that Li3P nanoclusters are embedded in the nanopores of the carbon particles. The interconnected carbon framework of the porous carbon works as the conductive skeleton for fast electron transport. The ultrafine particle size of Li3P enables fast electrochemical reactions. The volume change of Li3P/P active material is confined within the nanopores of the carbon particles and stable SEI layer is formed at the outer surface of the Li3P/C composite particle during cycling; (B) cycling of the Li3P/C electrode cycled at various lithiation current densities with a constant delithiation current density of 0.2C; (C), (D) the corresponding voltage-capacity plots. The galvanostatic charge/discharge measurement for Li3P/C||Li metal cells was carried out with the cut-off potential range of 0.01―2 V.Copyright 2020, Springer Nature.
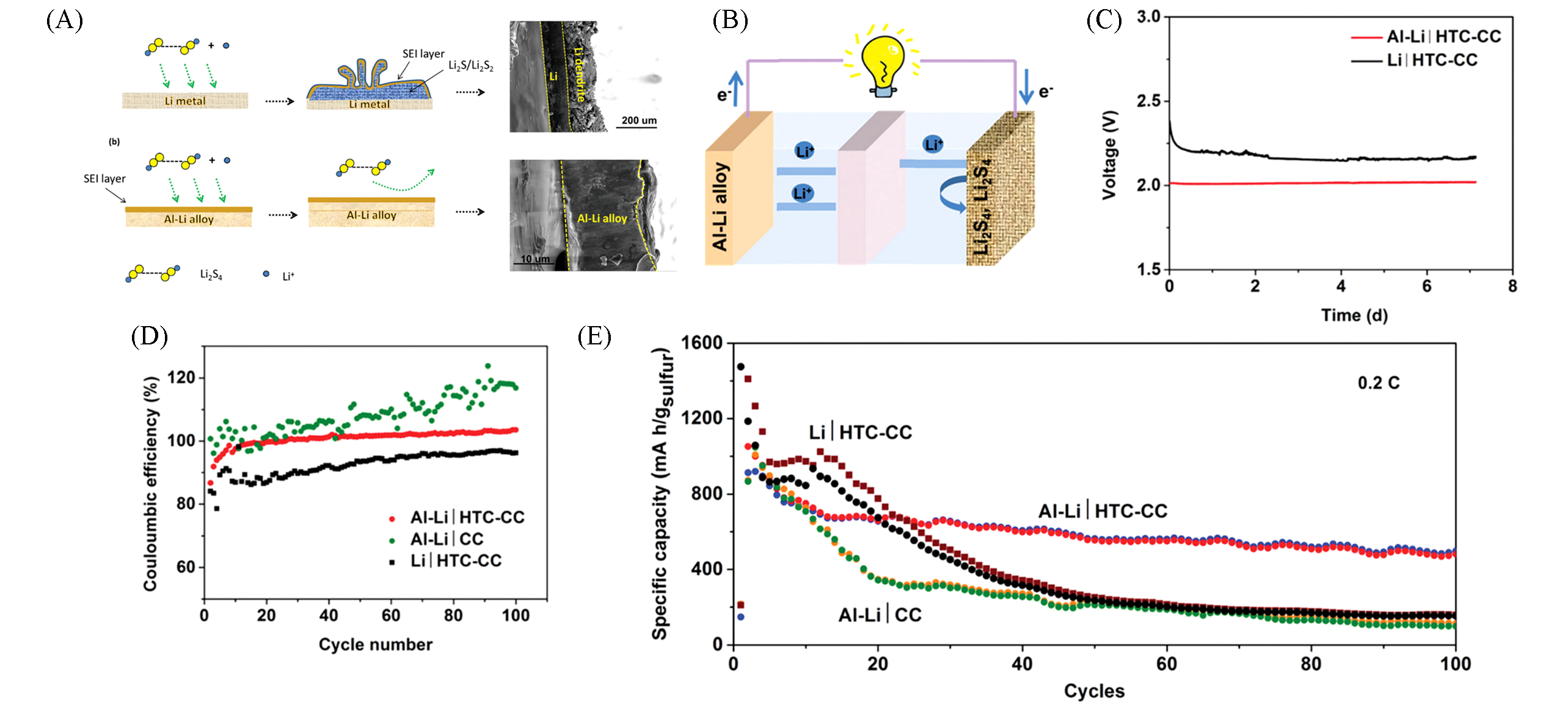
Fig.7 Li?Al alloy anodes for novel battery configuration[77](A) Working mechanism of Li metal and Li-Al alloy anodes. The interface stability of Al-Li alloy is superior to Li metal in polysulphide solution, which is attributed to the protection by the implantable SEI layer formed during the electrochemical alloying. The SEM image shows the dense cross-section of the immersed Al-Li alloy foil; (B) full cell Al-Li alloy||Li2S4-C(HTC-CC); (C) self-discharge test of the Al-Li||HTC-CC and Li||HTC-CC full cells; (D) coulombic efficiency; (E) capacities of the Al-Li||HTC-CC, Li||HTC-CC and Al-Li||CC full cells upon 100 cycles.Copyright 2018, Royal Society of Chemistry.
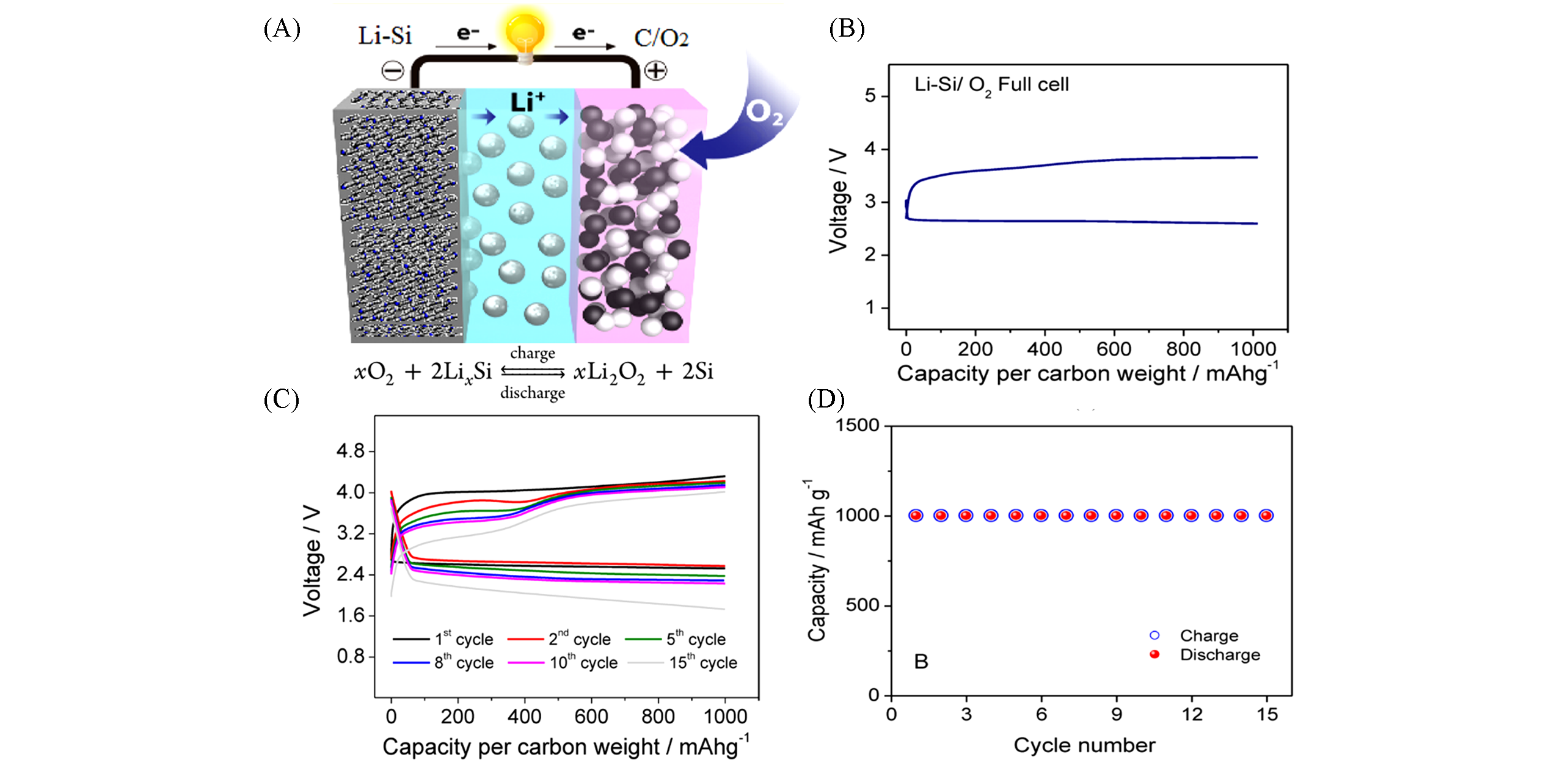
Fig.8 An initial prototype of lithium ion?air battery[63](A) Novel lithium ion-air battery consisting of a lithiated-silicon anode and a carbon-oxygen cathode; (B) voltage profiles for the first galvanostatic cycle(B) and the following cycles(C), and the corresponding capacity-cycle number plots for the lithiated-silicon/carbon-oxygen cell(D).Copyright 2012, American Chemical Society.
| 1 | Tarascon J. M., Armand M., Nature,2001, 414(6861), 359—367 |
| 2 | Armand M., Tarascon J. M., Nature, 2008, 451(7179), 652—657 |
| 3 | Dunn B., Kamath H., Tarascon J. M., Science, 2011, 334(6058), 928—935 |
| 4 | Chu S., Cui Y., Liu N., Nat. Mater.,2017, 16(1), 16—22 |
| 5 | Whittingham M. S., Chem. Rev., 2004, 104(10), 4271—4301 |
| 6 | Goodenough J. B., Kim Y., Chem. Mater., 2010, 22(3), 587—603 |
| 7 | Goodenough J. B., Acc。 Chem. Res.,2013, 46(5), 1053—1061 |
| 8 | Thackeray M. M., Wolverton C., Isaacs E. D., Energy Environ. Sci.,2012, 5(7), 7854—7863 |
| 9 | Whittingham M. S., Chem. Rev., 2014, 114(23), 11414—11443 |
| 10 | Armstrong A. R., Bruce P. G., Nature,1996, 381(6582), 499—500 |
| 11 | Yabuuchi N., Ohzuku T., J. Power Sources,2003, 119, 171—174 |
| 12 | Okubo M., Hosono E., Kim J., Enomoto M., Kojima N., Kudo T., Zhou H., Honma I., J. Am. Chem. Soc.,2007, 129(23), 7444—7452 |
| 13 | Chan C. K., Peng H., Liu G., McIlwrath K., Zhang X. F., Huggins R. A., Cui Y., Nat. Nanotechnol., 2008, 3(1), 31—35 |
| 14 | Nitta N., Wu F., Lee J. T., Yushin G., Mater. Today,2015, 18(5), 252—264 |
| 15 | Mao E., Fu L., Liu W., Gao X., Liu J., Zhan R., J. Alloy. Compd.,2021, 850, 156815 |
| 16 | Kim H., Jeong G., Kim Y. U., Kim J. H., Park C. M., Sohn H. J., Chem. Soc. Rev., 2013, 42(23), 9011—9034 |
| 17 | Liu H., Cheng X. B., Huang J. Q., Kaskel S., Chou S., Park H. S., Zhang Q., ACS Mater. Lett., 2019, 1(2), 217—229 |
| 18 | Wan M., Kang S., Wang L., Lee H. W., Zheng G. W., Cui Y., Sun Y., Nat. Commun., 2020, 11(1), doi: 10.1038/s41467⁃020⁃14550⁃3 |
| 19 | Wu D. S., Zhou G., Mao E., Sun Y., Liu B., Wang L., Wang J., Shi F., Cui Y., Nano Res., 2020, 13(5), 1383—1388 |
| 20 | Ji X., Lee K. T., Nazar L. F., Nat. Mater., 2009, 8(6), 500—506 |
| 21 | Ji X., Nazar L. F., J. Mater. Chem., 2010, 20(44), 9821—9826 |
| 22 | Bruce P. G., Freunberger S. A., Hardwick L. J., Tarascon J. M., Nat. Mater., 2011, 11(1), 19—29 |
| 23 | Li Y., Wang W., Liu X., Mao E., Wang M., Li G., Fu L., Li Z., Eng A. Y. S., Seh Z. W., Sun Y., Energy Storage Mater., 2019, 23, 261—268 |
| 24 | Chan C. K., Peng H., Twesten R. D., Jarausch K., Zhang X. F., Cui Y., Nano Lett., 2007, 7(2), 490—495 |
| 25 | Chernova N. A., Roppolo M., Dillon A. C., Whittingham M. S., J. Mater. Chem.,2009, 19(17), 2526—2552 |
| 26 | Chen X., Zhu H., Chen Y. C., Shang Y., Cao A., Hu L., Rubloff G. W., ACS Nano, 2012, 6(9), 7948—7955 |
| 27 | Li H., Balaya P., Maier J., J. Electrochem. Soc., 2004, 151(11), A1878 |
| 28 | Poizot P., Laruelle S., Grugeon S., Dupont L., Tarascon J., Nature, 2000, 407(6803), 496—499 |
| 29 | Li T., Li L., Cao Y. L., Ai X. P., Yang H. X., J. Phy. Chem. C, 2010, 114(7), 3190—3195 |
| 30 | Wang F., Robert R., Chernova N. A., Pereira N., Omenya F., Badway F., Hua X., Ruotolo M., Zhang R., Wu L., Volkov V., Su D., Key B., Whittingham M. S., Grey C. P., Amatucci G. G., Zhu Y., Graetz J., J. Am. Chem. Soc., 2011, 133(46), 18828—18836 |
| 31 | Zheng G., Lee S. W., Liang Z., Lee H. W., Yan K., Yao H., Wang H., Li W., Chu S., Cui Y., Nat. Nanotechnol.,2014, 9(8), 618—623 |
| 32 | Sun Y., Liu N., Cui Y., Nat. Energy, 2016, 1(7), 1—12 |
| 33 | Cheng X. B., Zhang R., Zhao C. Z., Zhang Q., Chem. Rev., 2017, 117(15), 10403—10473 |
| 34 | Lin D., Liu Y., Cui Y., Nat. Nanotechnol.,2017, 12(3), 194—206 |
| 35 | Liu J., Bao Z., Cui Y., Dufek E. J., Goodenough J. B., Khalifah P., Li Q., Liaw B. Y., Liu P., Manthiram A., Nat. Energy, 2019, 4(3), 180—186 |
| 36 | Aurbach D., Zinigrad E., Cohen Y., Teller H., Solid State Ionics,2002, 148(3/4), 405—416 |
| 37 | Xu W., Wang J., Ding F., Chen X., Nasybulin E., Zhang Y., Zhang J. G., Energy Environ. Sci., 2014, 7(2), 513—537 |
| 38 | Albertus P., Babinec S., Litzelman S., Newman A., Nat. Energy,2018, 3(1), 16—21 |
| 39 | Dey A. N., J. Electrochem. Soc.,1971, 118(10), 1547—1549 |
| 40 | Wen C. J., Huggins R. A., J. Solid State Chem., 1981, 37(3), 271—278 |
| 41 | Boukamp B., Lesh G., Huggins R., J. Electrochem. Soc.,1981, 128(4), 725 |
| 42 | Wang J., King P., Huggins R., Solid State Ionics, 1986, 20(3), 185—189 |
| 43 | Wang J., Raistrick I., Huggins R. A., J. Electrochem. Soc.,1986, 133(3), 457 |
| 44 | Anani A., Crouch‐Baker S., Huggins R., J. Electrochem. Soc.,1987, 134(12), 3098 |
| 45 | Anani A., Crouch‐Baker S., Huggins R., J. Electrochem. Soc., 1988, 135(8), 2103 |
| 46 | Huggins R., Solid State Ionics, 1998, 113—115(1/2), 57—67 |
| 47 | Huggins R. A., J. Power Sources, 1999, 81, 13—19 |
| 48 | Weydanz W., Wohlfahrt-Mehrens M., Huggins R. A., J. Power Sources,1999, 81, 237—242 |
| 49 | Yao N. P., Herédy L. A., Saunders R. C., J. Electrochem. Soc., 1971, 118(7), 1039 |
| 50 | Rao B., Francis R., Christopher H., J. Electrochem. Soc., 1977, 124(10), 1490 |
| 51 | Besenhard J. O., J. Electroanalytical Chem. Inter. Electrochem., 1978, 94(1), 77—81 |
| 52 | Garreau M., Thevenin J., Fekir M., J. Power Sources,1983, 9(3), 235—238 |
| 53 | Moshtev R., Zlatilova P., Puresheva B., Manev V., Kozawa A., J. Power Sources,1994, 51(3), 409—423 |
| 54 | Benedek R., Thackeray M. M., J. Power Sources,2002, 110(2), 406—411 |
| 55 | Thackeray M. M., Vaughey J. T., Fransson L. M. L., Jom, 2002, 54(3), 20—23 |
| 56 | Lai S. C., J. Electrochem. Soc., 2019, 123(8), 1196—1197 |
| 57 | Sharma R. A., Seefurth R. N., J. Electrochem. Soc.,1976, 123(12), 1763 |
| 58 | Seefurth R. N., Sharma R. A., J. Electrochem. Soc., 1977, 124(8), 1207 |
| 59 | Seefurth R. N., Sharma R. A., J. Electrochem. Soc., 1980, 127(5), 1101 |
| 60 | Hassoun J., Lee K. S., Sun Y. K., Scrosati B., J. Am. Chem. Soc.,2011, 133(9), 3139—3143 |
| 61 | Liu N., Hu L., McDowell M. T., Jackson A., Cui Y., ACS Nano, 2011, 5(8), 6487—6493 |
| 62 | Elazari R., Salitra G., Gershinsky G., Garsuch A., Panchenko A., Aurbach D., Electrochem. Commun., 2012, 14(1), 21—24 |
| 63 | Hassoun J., Jung H. G., Lee D. J., Park J. B., Amine K., Sun Y. K., Scrosati B., Nano Lett., 2012, 12(11), 5775—5779 |
| 64 | Ma R., Liu Y., He Y., Gao M., Pan H., J. Phys. Chem. Lett.,2012, 3(23), 3555—3558 |
| 65 | Zhao J., Lu Z., Liu N., Lee H.W., McDowell M. T., Cui Y., Nat. Commun., 2014, 5, 5088 |
| 66 | Wu S., Zhu K., Tang J., Liao K., Bai S., Yi J., Yamauchi Y., Ishida M., Zhou H., Energy Environ. Sci., 2016, 9(10), 3262—3271 |
| 67 | Li X., Kersey-Bronec F. E., Ke J., Cloud J. E., Wang Y., Ngo C., Pylypenko S., Yang Y., ACS Appl. Mater. Inter.,2017, 9(19), 16071—16080 |
| 68 | Zhao J., Zhou G., Yan K., Xie J., Li Y., Liao L., Jin Y., Liu K., Hsu P. C., Wang J., Cheng H. M., Cui Y., Nat. Nanotechnol.,2017, 12(10), 993—999 |
| 69 | Sun J., Lv R., Lv W., Yang Q. H., Amal R., Wang D. W., Energy Storage Mater.,2018, 15, 209—217 |
| 70 | Wang C., Han Y., Li S., Chen T., Yu J., Lu Z., ACS Appl. Mater. Inter.,2018, 10(15), 12750—12758 |
| 71 | Xu H., Li S., Zhang C., Chen X., Liu W., Zheng Y., Xie Y., Huang Y., Li J., Energy Environ. Sci., 2019, 12(10), 2991—3000 |
| 72 | Mao E., Wang W., Wan M., Wang L., He X., Sun Y., Nano Res., 2020, 13(4), 1122—1126 |
| 73 | Park C. M., Kim J. H., Kim H., Sohn H. J., Chem. Soc. Rev., 2010, 39(8), 3115—3141 |
| 74 | Obrovac M. N., Chevrier V. L., Chem. Rev., 2014, 114(23), 11444—11502 |
| 75 | Sun Y., Wang L., Li Y., Li Y., Lee H.R., Pei A., He X., Cui Y., Joule,2019, 3(4), 1080—1093 |
| 76 | Fan H., Chen B., Li S., Yu Y., Xu H., Jiang M., Huang Y., Li J., Nano Lett.,2020, 20(2), 896—904 |
| 77 | Sun J., Liang J., Liu J., Shi W., Sharma N., Lv W., Lv R., Yang Q. H., Amal R., Wang D. W., Energy Environ. Sci., 2018, 11(9), 2509—2520 |
| 78 | Cloud J. E., Wang Y., Yoder T. S., Taylor L. W., Yang Y., Angew. Chem. Int. Ed. Engl.,2014, 53(52), 14527—14532 |
| 79 | Luo F., Chu G., Huang J., Sun Y., Li H., Energy Storage Science and Technology, 2014, 3(2), 146—163(罗飞, 褚赓, 黄杰, 孙洋, 李泓. 储能科学与技术,2014, 3(2), 146—163) |
| 80 | Zhou J. H., Chu G., Lu H., Liu B. N., Luo F., Zheng J. Y., Chen S. M., Guo Y. G., Li H., Energy Storage Science and Technology, 2019, 8(1), 215—223(周军华, 褚赓, 陆浩, 刘柏男, 罗飞, 郑杰允, 陈仕谋, 郭玉国, 李泓. 储能科学与技术, 2019, 8(1), 215—223) |
| 81 | Zhao J., Lee H.W., Sun J., Yan K., Liu Y., Liu W., Lu Z., Lin D., Zhou G., Cui Y., Proc. Natl. Acad. Sci. USA,2016, 113(27), 7408—7413 |
| 82 | Zhao J., Liao L., Shi F., Lei T., Chen G., Pei A., Sun J., Yan K., Zhou G., Xie J., Liu C., Li Y., Liang Z., Bao Z., Cui Y., J. Am. Chem. Soc.,2017, 139(33), 11550—11558 |
| 83 | Zhao J., Sun J., Pei A., Zhou G., Yan K., Liu Y., Lin D., Cui Y., Energy Storage Mater., 2018, 10, 275—281 |
| 84 | Li S., Wang C., Yu J., Han Y., Lu Z., Energy Storage Mater., 2019, 20, 7—13 |
| 85 | Wang C., Yu J., Li S., Lu Z., Chem. Eng. J., 2019, 370, 1019—1026 |
| 86 | Tamori R., Machida N., Shigematsu T., J. Japan Soc. Powder and Powder Metallurgy,2001, 48(3), 267—273 |
| 87 | Cloud J. E., Wang Y., Li X., Yoder T. S., Yang Y., Yang Y., Inorg. Chem., 2014, 53(20), 11289—11297 |
| 88 | Hashimoto Y., Machida N., Shigematsu. T., Solid State Ionics, 2004, 175(1—4), 177—180 |
| 89 | Deng H., Qiu F., Li X., Qin H., Zhao S., He P., Zhou H., Electrochem. Commun., 2017, 78, 11—15 |
| 90 | Shen C., Ge M., Zhang A., Fang X., Liu Y., Rong J., Zhou C., Nano Energy, 2016, 19, 68—77 |
| 91 | Zhang T., Yang J., Zhu J., Zhou J., Xu Z., Wang J., Qiu F., He P., Chem. Commun.(Camb), 2018, 54(9), 1069—1072 |
| 92 | Xu H., Li S., Chen X., Zhang C., Liu W., Fan H., Yu Y., Huang Y., Li J., Adv. Energy Mater., 2019, 9(42), 1902150 |
| 93 | Huang Y., Liu C., Wei F., Wang G., Xiao L., Lu J., Zhuang L., J. Mater. Chem. A, 2020, 8(36), 18715—18720 |
| 94 | Elia G.A., Bresser D., Reiter J., Oberhumer P., Sun Y. K., Scrosati B., Passerini S., Hassoun J., ACS Appl. Mater. Inter., 2015, 7(40), 22638—22643 |
| 95 | Hirshberg D., Sharon D.,de La Llave E., Afri M., Frimer A. A., Kwak W. J., Sun Y. K., Aurbach D., ACS Appl. Mater. Inter., 2017, 9(5), 4352—4361 |
| 96 | Zhang T., Xu Z., Guo Y., Liang C., Wang J., Yang J., J. Alloy. Compd.,2019, 806, 335—342 |
| 97 | Fu L., Wan M., Zhang B., Yuan Y., Jin Y., Wang W., Wang X., Li Y., Wang L., Jiang J., Lu J., Sun Y., Adv. Mater., 2020, 32(29), 2000952 |
| 98 | Hassoun J., Kim J., Lee D. J., Jung H. G., Lee S. M., Sun Y. K., Scrosati B., J. Power Sources, 2012, 202, 308—313 |
| 99 | Hou C., Song X., Yang K., Wang H., Liu X., Nie Z., Mater. Today Energy, 2018, 7, 122—128 |
| 100 | Ren J. G., Wang K., He X. M., Jiang C. Y., Wan C. R., Pu W. H., Progress in Chemistry, 2005, (4), 597—603(任建国, 王科, 何向明, 姜长印, 万春荣, 蒲薇华. 化学进展, 2005, (4), 597—603) |
| 101 | Vaughey J. T., Kepler K. D., Benedek R., Thackeray M. M., Electrochem. Commun.,1999, 1(11), 517—521 |
| 102 | Zaghib K., Gauthier M., Armand M., J. Power Sources, 2003, 119—121, 76—83 |
| 103 | Iwamura S., Nishihara H., Ono Y., Morito H., Yamane H., Nara H., Osaka T., Kyotani T., Sci. Rep.,2015, 5(1), 8085 |
| 104 | Tang W. S., Chotard J. N., Janot R., J. Electrochem. Soc.,2013, 160(8), A1232—A1240 |
| 105 | Guo Z., Dong X., Wang Y., Xia Y., Chem. Commun.(Camb),2015, 51(4), 676—678 |
| 106 | Qin L., Zhai D., Lv W., Yang W., Huang J., Yao S., Cui J., Chong W. G., Huang J. Q., Kang F., Kim J. K., Yang Q. H., Nano Energy, 2017, 40, 258—263 |
| 107 | Zhang Y., Jiao Y., Lu L., Wang L., Chen T., Peng H., Angew. Chem. Int. Ed. Engl.,2017, 56(44), 13741—13746 |
| 108 | Yang J., Winter M., Besenhard J., Solid State Ionics,1996, 90(1—4), 281—287 |
| 109 | Chumak I., Hinterstein M., Ehrenberg H., Prog. Solid State Chem., 2014, 42(4), 149—156 |
| 110 | Jiang M., Yu Y., Fan H., Xu H., Zheng Y., Huang Y., Li S., Li J., ACS Appl. Mater. Inter., 2019, 11(17), 15656—15661 |
| 111 | Huang C. K., Surampudi S., Attia A. I., Halpert G., Anode for Rechargeable Ambient Temperature Lithium Cells, USA 5294503, 1994⁃03⁃15 |
| 112 | Zhao J., Lu Z., Wang H., Liu W., Lee H. W., Yan K., Zhuo D., Lin D., Liu N., Cui Y., J. Am. Chem. Soc., 2015, 137(26), 8372—8375 |
| 113 | Ikeda K., Terada S., Mandai T., Ueno K., Dokko K., Watanabe M., Electrochem.,2015, 83(10), 914—917 |
| 114 | Agostini M., Hassoun J., Sci. Rep.,2015, 5(1), 7591 |
| [1] | 贾洋刚, 邵霞, 程婕, 王朋朋, 冒爱琴. 赝电容控制型钙钛矿高熵氧化物La(Co0.2Cr0.2Fe0.2Mn0.2Ni0.2)O3负极材料的制备及储锂性能[J]. 高等学校化学学报, 2022, 43(8): 20220157. |
| [2] | 鲍俊全, 郑仕兵, 苑旭明, 史金强, 孙田将, 梁静. 有机盐PTO(KPD)2作为高性能锂离子电池正极材料的研究[J]. 高等学校化学学报, 2021, 42(9): 2911. |
| [3] | 李辉阳, 朱思颖, 李莎, 张桥保, 赵金保, 张力. 锂离子电池硅氧化物负极首次库伦效率的影响因素与提升策略[J]. 高等学校化学学报, 2021, 42(8): 2342. |
| [4] | 卓增庆, 潘锋. 基于软X射线光谱的锂电池材料的电子结构与演变的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2332. |
| [5] | 吴卓彦, 李至, 赵旭东, 王倩, 陈顺鹏, 常兴华, 刘志亮. 一步法高效制备纳米Si/C复合材料及其在高性能锂离子电池中的应用[J]. 高等学校化学学报, 2021, 42(8): 2500. |
| [6] | 易聪华, 苏华坚, 钱勇, 李琼, 杨东杰. 木质素纳米炭的制备及作为锂离子电池负极的性能研究[J]. 高等学校化学学报, 2021, 42(6): 1807. |
| [7] | 詹迎新, 石鹏, 张学强, 魏俊宇, 张乾魁, 黄佳琦. 锂金属负极亲锂骨架的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1569. |
| [8] | 石颖, 胡广剑, 吴敏杰, 李峰. 低温等离子体在锂离子电池材料中的应用[J]. 高等学校化学学报, 2021, 42(5): 1315. |
| [9] | 王弈艨, 刘凯, 王保国. 高镍三元正极材料的表面包覆策略[J]. 高等学校化学学报, 2021, 42(5): 1514. |
| [10] | 刘铁峰, 张奔, 盛欧微, 佴建威, 王垚, 刘育京, 陶新永. 硅负极黏结剂的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1446. |
| [11] | 王任衡, 肖哲, 李艳, 孙一翎, 范姝婷, 郑俊超, 钱正芳, 贺振江. 固相烧结法制备锂离子电池正极材料Li2FeP2O7及其电化学性能研究[J]. 高等学校化学学报, 2021, 42(4): 1299. |
| [12] | 孙全虎, 卢天天, 何建江, 黄长水. 含异原子石墨炔基电极材料的研究进展[J]. 高等学校化学学报, 2021, 42(2): 366. |
| [13] | 周战, 马录芳, 谭超良. 层状钒青铜纳米片的制备及其锂离子电池阳极材料性能[J]. 高等学校化学学报, 2021, 42(2): 662. |
| [14] | 高小雅, 左自成, 李玉良. 石墨炔电化学电池界面构筑[J]. 高等学校化学学报, 2021, 42(2): 321. |
| [15] | 韩慕瑶, 赵丽娜, 孙洁. 硅及硅基负极材料的研究进展[J]. 高等学校化学学报, 2021, 42(12): 3547. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||