

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (1): 21.doi: 10.7503/cjcu20200201
所属专题: 分子筛功能材料 2021年,42卷,第1期
收稿日期:2020-04-14
出版日期:2021-01-10
发布日期:2021-01-12
通讯作者:
肖丰收
E-mail:fsxiao@zju.edu.cn
基金资助:
WU Qinming, WANG Yeqing, MENG Xiangju, XIAO Fengshou( )
)
Received:2020-04-14
Online:2021-01-10
Published:2021-01-12
Contact:
XIAO Fengshou
E-mail:fsxiao@zju.edu.cn
Supported by:摘要:
目前针对沸石分子筛的晶化过程已经进行了大量研究, 但是近年来硅铝分子筛的合成显示其晶化过程超出了传统的沸石分子筛晶化理论, 特别表现在硅铝沸石分子筛的模板作用、 水的作用以及沸石晶体之间的转化等. 本文讨论了上述作用的本质, 并通过研究模板法作用提出了无有机模板法合成硅铝沸石的策略, 通过了解水的作用提出了无溶剂法合成沸石分子筛, 通过表征沸石之间的转化过程发现不仅低骨架密度可以向高骨架密度晶体转化, 而且高骨架密度也可以向低骨架密度晶体转化.
中图分类号:
TrendMD:
吴勤明, 王叶青, 孟祥举, 肖丰收. 硅铝沸石分子筛晶化过程再思考. 高等学校化学学报, 2021, 42(1): 21.
WU Qinming, WANG Yeqing, MENG Xiangju, XIAO Fengshou. Reconsideration of Crystallization Process for Aluminosilicate Zeolites. Chem. J. Chinese Universities, 2021, 42(1): 21.
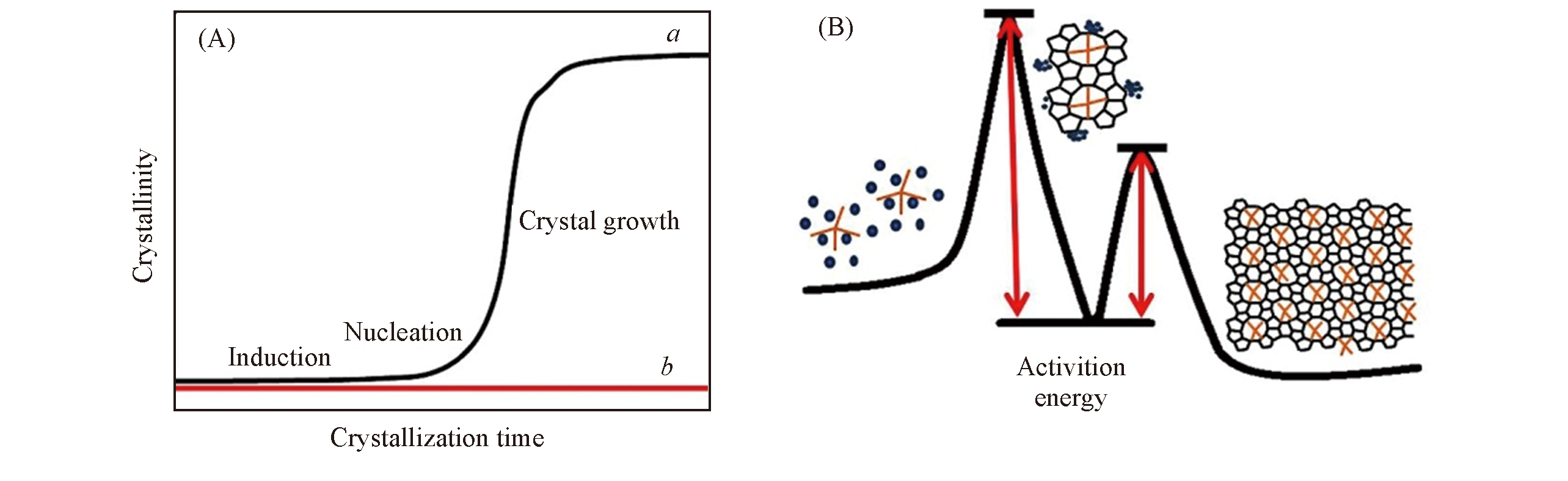
Fig.1 X-Ray crystallization curves of pure silica MFI zeolite synthesized with(a) and without(b) TPA+(A) and scheme for the crystallization of zeolites synthesized with organic templates(B)[22]Copyright 2017, Elsevier.

Fig.2 TEM images of Beta-SDS samples crystallized for 1, 4, 8, and 18.5 h at 140 ℃ by addition of 10.3% Beta seeds(Si/Al molar ratio: 10.2) in the starting aluminosilicate gels[23]Copyright 2011, Royal Society of Chemistry.
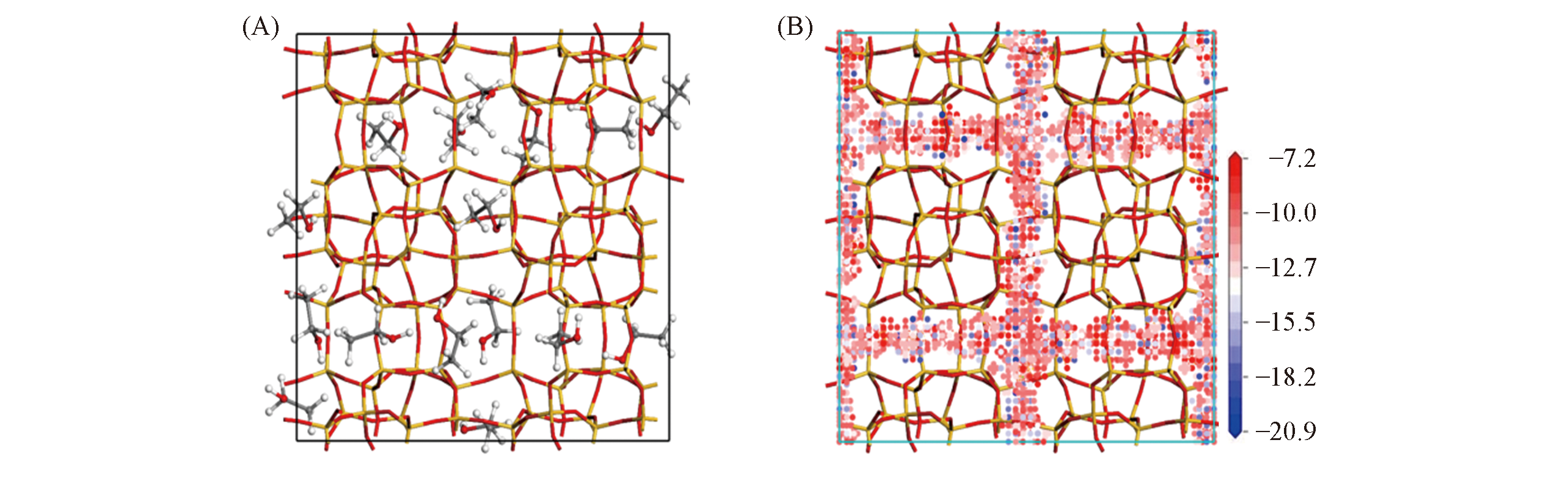
Fig.3 Position of ethanol adsorbing in the channel of S-1 zeolite(A) and isodensity surface colored by potential energy for ethanol in silicalite-1 zeolite(B)[35]Blue represents low potential energy/(kcal·mol-1). Copyright 2019, Wiley.
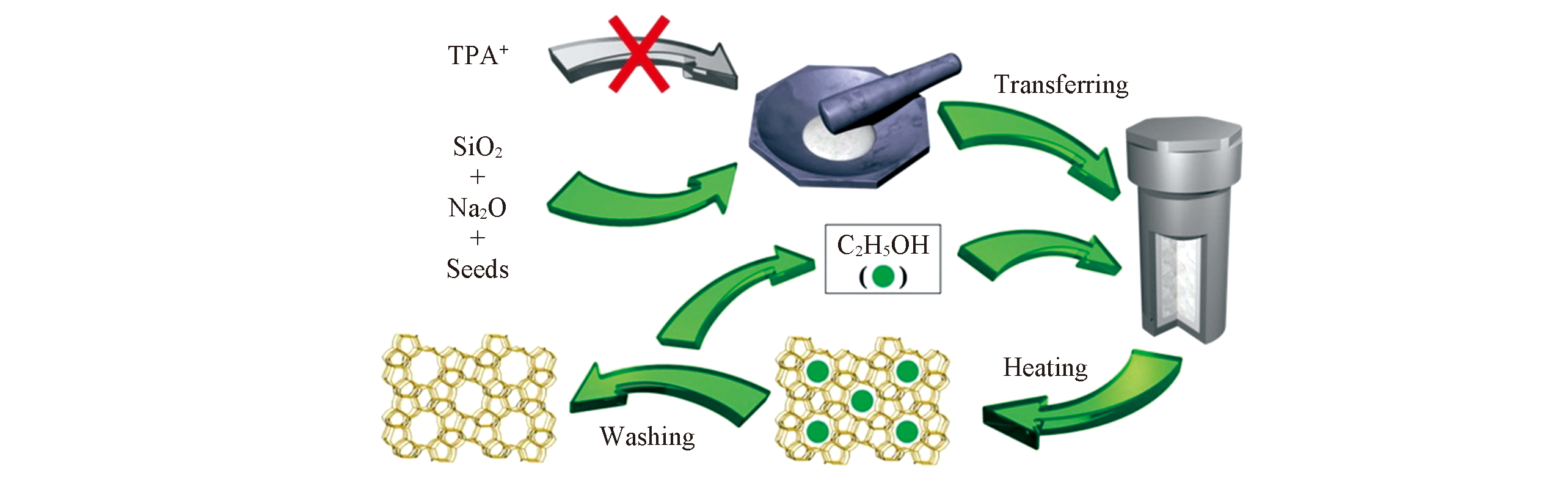
Fig.4 Representation of the synthesis of S-Si-ZSM-5 zeolite from a combination of zeolite seeding and alcohol filling in the absence of any organic templates[35]Copyright 2019, Wiley.
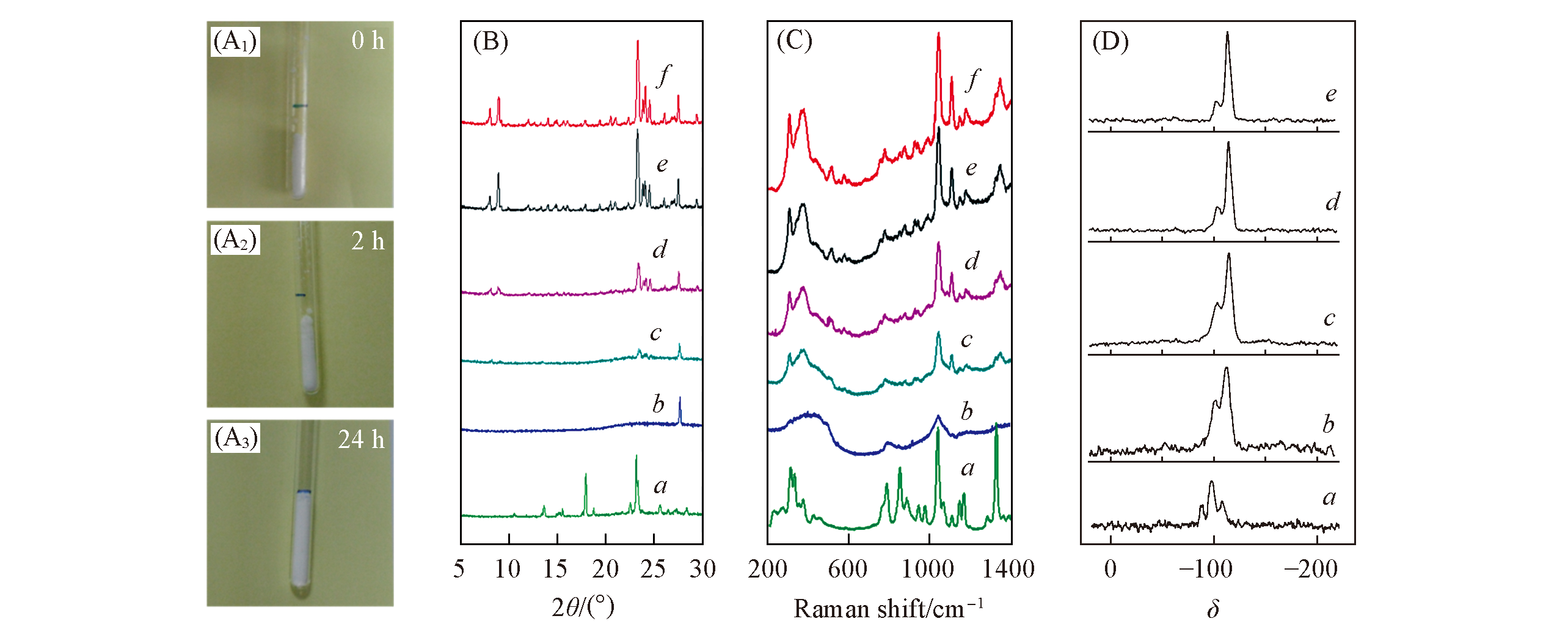
Fig.7 Investigation on the crystallization process in solvent-free synthesis of ZSM-5(A) Photographs of the samples crystallized at 0(A1), 2(A2) and 24 h(A3); (B) XRD patterns; (C) UV-Raman spectra; (D) 29Si NMR spectra of the samples crystallized at 0(a), 2(b), 10(c), 12(d), 18(e) and 24 h(f)[16].Copyright 2012, American Chemical Society.
| Zeolite | MFI | *BEA | FAU | AEI |
|---|---|---|---|---|
| Framework density(T/1000 ?3) | 17.9 | 15.1 | 12.7 | 14.8 |
| Number of OSDA per unit cell | 4 | 4 | 4 | 4 |
| Stability energy OSDA?zeolite | 4.71 | -8.39 | -2.02 | -9.97 |
Table 1 Framework density of zeolites and the calculated energies (kJ/mol Si) between OSDA and zeolite framework[43]
| Zeolite | MFI | *BEA | FAU | AEI |
|---|---|---|---|---|
| Framework density(T/1000 ?3) | 17.9 | 15.1 | 12.7 | 14.8 |
| Number of OSDA per unit cell | 4 | 4 | 4 | 4 |
| Stability energy OSDA?zeolite | 4.71 | -8.39 | -2.02 | -9.97 |
| Zeolite | Channel size | Topological | SBET/(m2·g-1) | Vmicro/(cm3·g-1) |
|---|---|---|---|---|
| SSZ?39(AEI) | 0.38 nm×0.38 nm | 3-Dimensional | 587 | 0.26 |
Table 2 Framework properties of as-synthesized SSZ-39 zeolite[43]
| Zeolite | Channel size | Topological | SBET/(m2·g-1) | Vmicro/(cm3·g-1) |
|---|---|---|---|---|
| SSZ?39(AEI) | 0.38 nm×0.38 nm | 3-Dimensional | 587 | 0.26 |
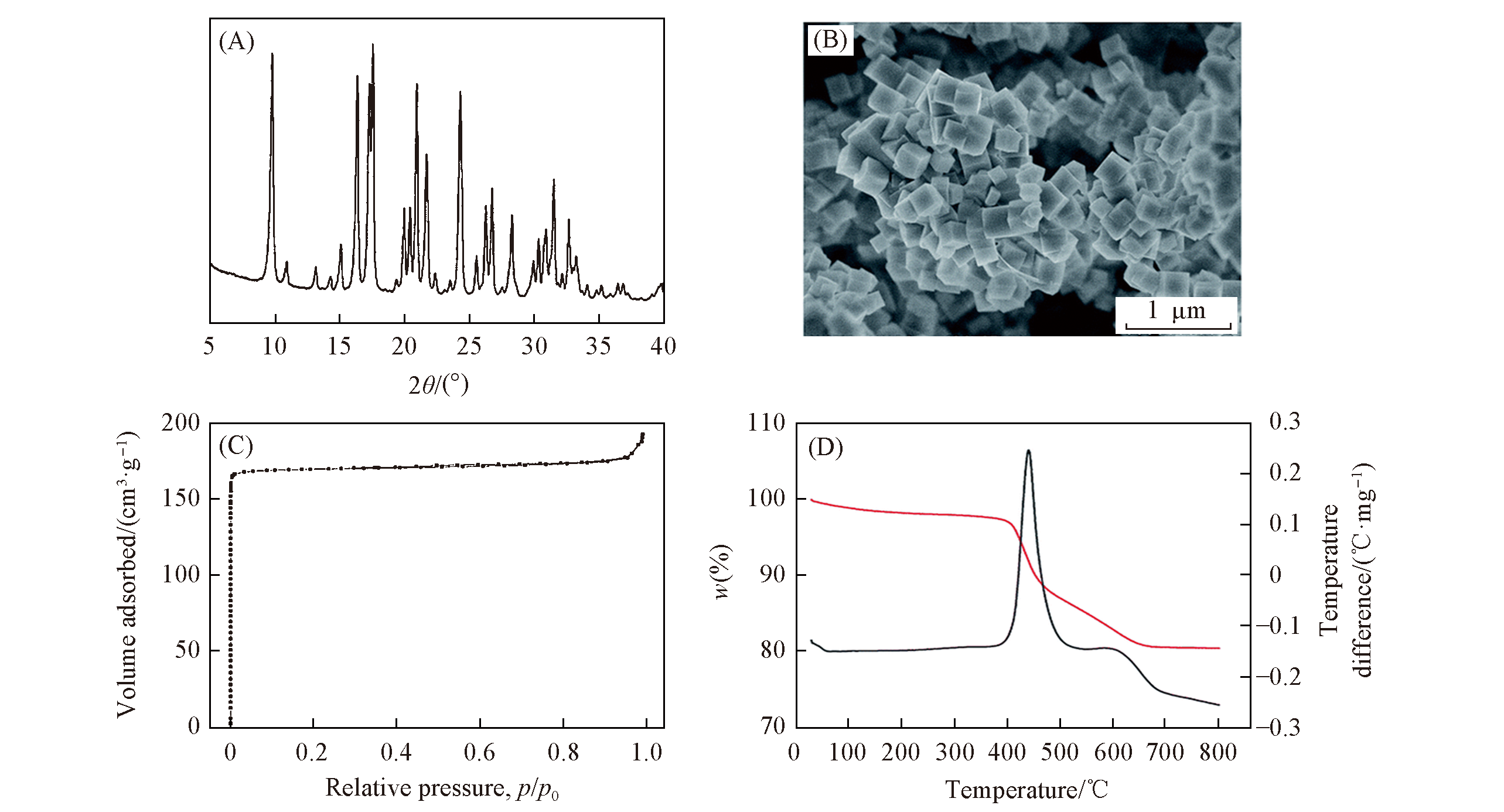
Fig.8 XRD pattern(A), SEM image(B), N2 sorption isotherm(C) and TG?DTA curves(D) of the Z?SSZ?39 zeolite sample from the transformation of ZSM?5 zeolite[43]Copyright 2019, Royal Society of Chemistry.
| 1 | Davis M. E., Nature, 2002, 417, 813—821 |
| 2 | Corma A., Chem. Rev., 1999, 95, 599—614 |
| 3 | Li Y., Li L., Yu J. H., Chem, 2017, 3, 928—949 |
| 4 | Meng X. J., Xiao F. S., Chem. Rev., 2014, 114, 1521—1543 |
| 5 | Cundy C. S., Cox P. A., Chem. Rev.2003, 103, 663—701 |
| 6 | Breck D. W., Zeolite Molecular Sieves, Krieger Publishing Company, Malabar, 1984 |
| 7 | van Bekkum H., Flanigen E. M., Jacobs P. A., Jansen J. C., Introduction to Zeolite Science and Practice, Elsevier, Amsterdam, 2001 |
| 8 | Barrer R. M., Hydrothermal Chemistry of Zeolites, Academic Press, London, 1982 |
| 9 | Xu R. R., Pang W. Q., Yu J. H., Huo Q. S., Chen J. S., Chemistry of Zeolite and Related Porous Materials, Wiley, Singapore, 2007 |
| 10 | Xie B., Song J. W., Ren L. M., Ji Y. Y., Li J. X., Xiao F. S., Chem. Mater., 2008, 20, 4533—4535 |
| 11 | Ng E. P., Chateigner D., Bein T., Valtchev V., Mintova S., Science, 2012, 335, 70—73 |
| 12 | Itabashi K., Kaminura Y., Iyoki K., Shimojiama A, Okubo T., J. Am. Chem. Soc., 2012, 134, 11542—11549 |
| 13 | Yokoi T., Yoshioka M., Ima H., Tatsumi T., Angew. Chem. Int. Ed., 2009, 48, 9884—9887 |
| 14 | Awala H., Gilson J. P., Retoux R., Boullay P., Goupil J. M., Valtchev V., Mintova S., Nature Mater., 2015, 14, 447—451 |
| 15 | Feng G. D., Cheng P., Yan W. F., Boronat M., Li X., Su J. H., Wang J. Y., Li Y., Corma A., Xu R. R., Yu J. H., Science, 2016, 351, 1188—1191 |
| 16 | Ren L. M., Wu Q. M., Yang C. G., Zhu L. F., Li C. J., Zhang P. L., Zhang H. Y., Meng X. J., Xiao F. S., J. Am. Chem. Soc., 2012, 134, 15173—15176 |
| 17 | Wu Q. M., Wang X., Qi G. D., Guo Q., Pan S. X, Meng X. J., Xu J., Deng F., Fan F. T., Feng Z. C., Li C., Maurer S., Muller U., Xiao F. S., J. Am. Chem. Soc., 2014, 136, 4019—4025 |
| 18 | Wu Q. M., Liu X. L., Zhu L. F., Ding L. H., Gao P., Wang X., Pan S. X., Bian C. Q., Meng X. J., Xu J., Deng F., Maurer S., Muller U., Xiao F. S., J. Am. Chem. Soc., 2015, 137, 1052—1055 |
| 19 | Wu Q. M., Meng X. J., Gao X. H., Xiao F. S., Acc. Chem. Res., 2018, 51, 1396—1403 |
| 20 | Zhang B., Douthwaite M., Liu Q., Zhang C., Wu Q. F., Shi R. H., Wu P. X., Liu K., Wang Z. Q., Lin W. W., Cheng H. Y., Cheng H. Y., Da D., Zhao F. Y., Hutchings G. J., Green Chem., 2020, 22, 1630—1638 |
| 21 | Xu H., Chen W., Wu Q. M., Lei C., Zhang J., Han S. C., Zhang L., Zhu Q. Y., Meng X. J., Dai D., Maurer S., Parvulescu A. N., Muller U., Zhang W. P., Yokoi T., Bao X. H., Marler B., De Vos D. E., Kolb U., Zheng A. M., Xiao F. S., J. Mater. Chem. A, 2019, 7, 4420—4425 |
| 22 | Wang Y. Q., Wu Q. M., Meng X. J., Xiao F. S., Engineering, 2017, 3, 567—574 |
| 23 | Xie B., Zhang H. Y., Yang C. G., Liu S., Ren L. M., Zhang L., Meng X. J., Yilmaz B., Muller U., Xiao F. S., Chem. Commun., 2011, 47, 3945—3947 |
| 24 | Zhang H. Y., Guo Q., Ren L., Yang C., Zhu L. F., Meng X. J., Li C. J., Xiao F. S., J. Mater. Chem., 2011, 21, 9494—9497 |
| 25 | Zhang H. Y., Yang C. G., Zhu L. F., Meng X. J., Yilmaz B., Müller U., Feyen M., Xiao F. S., Micropor. Mesopor. Mater., 2012, 155, 1—7 |
| 26 | Majano G., Delmotte L., Valtchev V., Mintova S., Chem. Mater., 2009, 21, 4184—4191 |
| 27 | Kamimura Y., Chaikittisilp W., Itabashi K., Shimojima A., Okubo T., Chem. Asian J., 2010, 5, 2182— 2191 |
| 28 | Iyoki K., Kamimura Y., Itabashi K., Shimojima A., Okubo T., Chem. Lett., 2010, 39, 730—731 |
| 29 | Kamimura Y., Itabashi K., Okubo T., Micropor. Mesopor. Mater., 2012, 147, 149—156 |
| 30 | Wu Q., Wang X., Meng X., Yang C., Liu Y., Jin Y., Yang Q., Xiao F. S., Micropor. Mesopor. Mater., 2014, 186, 106—112 |
| 31 | Yang C. G., Ren L. M., Zhang H. Y., Zhu L. F., Wang L., Meng X. J., Xiao F. S., J. Mater. Chem., 2012, 22, 12238—12245 |
| 32 | Zhang H. Y., Wang L., Zhang D. L., Meng X. J., Xiao F. S., Micropor. Mesopor. Mater., 2016, 233, 133—139 |
| 33 | Iyoki K., Takase M., Itabashi K., Muraoka K., Chaikittisilp W., Okubo T., Micropor. Mesopor. Mater., 2015, 215, 191—198 |
| 34 | Wu Q. M., Ma Y., Wang S., Meng X. J., Xiao F. S., Ind. Eng. Chem. Res., 2019, 58, 11653—11658 |
| 35 | Wu Q. M., Zhu L. F., Chu Y. Y., Liu X. L., Zhang C. S., Zhang J., Xu H., Xu J., Deng F., Feng Z. C., Meng X. J., Xiao F. S., Angew. Chem. Int. Ed., 2019, 58, 12138—12142 |
| 36 | Wu D., Xu X., Chen X. Q., Yu G., Zhang K., Qiu M. H., Xue W. J., Yang C. G., Liu Z. Y., Sun Y. H., ChemSusChem, 2019, 12, 3871—3877 |
| 37 | Soekiman C. N., Miyake K., Hirota Y., Uchida Y., Tanaka S., Miyamoto M., Nishiyama N., Micropor. Mesopor. Mater., 2019, 273, 273—275 |
| 38 | Zhang C. S., Wu Q. M., Lei C., Pan S. X., Bian C. Q., Wang L., Meng, X. J., Xiao F. S., Ind. Eng. Chem. Res., 2017, 56, 1450—1460 |
| 39 | Xiao Y. C., Sheng N., Chu Y. Y., Wang Y. Q., Wu Q. M., Liu X. L., Deng F., Meng X. J., Feng Z. C., Micropor. Mesopor. Mater., 2017, 237, 201—209 |
| 40 | Goel S., Zones S. I., Iglesia E., Chem. Mater., 2015, 27, 2056—2066 |
| 41 | Jon H., Ikawa N., Oumi Y., Sano T., Chem. Mater., 2008, 20, 4135—4131 |
| 42 | Li C., Moliner M., Corma A., Angew. Chem. Int. Ed., 2018, 57, 15330—15353 |
| 43 | Xu H., Chen W., Wu Q. M., Lei C., Zhang J., Han S. C., Zhang L., Zhu Q. Y., Meng X. J., Maurer S., Parvulescu A. N., Muller U., Zhang W. P., Yokoi T., Bao X., Marler B., De Vos D. E., Kolb U., Zheng A., Xiao F. S., J. Mater. Chem. A, 2019, 7, 4420—4425 |
| 44 | Zhang J., Chu Y. Y., Liu X. L., Xu H., Meng X. J., Feng Z. C., Xiao F. S., Chinese J. Catal., 2019, 40, 1854—1859 |
| 45 | Bian C. Q., Zhang C. S., Pan S. X., Chen F., Zhang W. P., Meng X. J., Maurer S., Dai D., Parvulescu A. N., Muller U., Xiao F. S., J. Mater. Chem. A, 2017, 5, 2613—2618 |
| 46 | Tian P., Wei Y. X., Ye M., Liu Z. M., ACS Catal., 2015, 5, 1922—1938 |
| 47 | Beale A. M., Gao F., Lezcano-Gonzalez I., Peden C. H. F., Szanyi J., Chem. Soc. Rev., 2015, 44, 7371—7405 |
| 48 | Lin Y. C., Chang F. T., J. Hazard. Mater., 2009, 164, 517—526 |
| [1] | 梁宇, 刘欢, 宫丽阁, 王春晓, 王春梅, 于凯, 周百斌. 联咪唑修饰{SiW12O40}杂化物的合成及超级电容性能[J]. 高等学校化学学报, 2022, 43(1): 20210556. |
| [2] | 张会双, 高延晓, 王秋娴, 李向南, 刘文凤, 杨书廷. CTAB辅助水热合成高镍三元材料LiNi0.6Co0.2Mn0.2O2及其高低温性能研究[J]. 高等学校化学学报, 2021, 42(3): 819. |
| [3] | 王冶, 张晓思, 孙丽婧, 李冰, 刘琳, 杨淼, 田鹏, 刘仲毅, 刘中民. 有机硅烷辅助合成特殊形貌SAPO分子筛[J]. 高等学校化学学报, 2021, 42(3): 683. |
| [4] | 王健羽, 张强, 闫文付, 于吉红. 羟基自由基在沸石分子筛合成中的作用[J]. 高等学校化学学报, 2021, 42(1): 11. |
| [5] | 刘亚冰,李明阳,田戈,阿拉腾沙嘎,裴桐鹤,聂婧思. 基于2-氨基吡啶的两个簇基超分子化合物的合成、 结构及催化性能[J]. 高等学校化学学报, 2020, 41(5): 995. |
| [6] | 卓孟宁,李飞,蒋浩,陈倩文,李鹏,王立章. SnO2/GDE阴极的制备及电催化还原CO2产甲酸性能[J]. 高等学校化学学报, 2020, 41(3): 530. |
| [7] | 董乐, 黄星亮, 任俊杰, 代小平, 刘宗俨, 田洪锋, 王志东, 吴晓彤. 硅溶胶粒度与分布对高硅铝比镁碱沸石合成的影响机理[J]. 高等学校化学学报, 2020, 41(11): 2449. |
| [8] | 周海, 陈豪, 郭娅, 康敏. 介孔Co3O4多面体的制备及电化学性能[J]. 高等学校化学学报, 2019, 40(7): 1374. |
| [9] | 高宁萧, 徐玉龙, 刘勇. 豆奶粉提取碳点及含碳点荧光纳米纤维的制备[J]. 高等学校化学学报, 2019, 40(3): 555. |
| [10] | 潘帅, 胡校兵, 宋润民, 解丽丽, 朱志刚, 郑嘹赢. 醋酸钾溶液体系中α-Fe2O3纳米微球的离子液体辅助合成及气敏性能研究[J]. 高等学校化学学报, 2018, 39(8): 1631. |
| [11] | 夏坤, 周丹, 杨赟, 杨水金, 夏清华. 高度均一的AlPO4-11微晶的高效合成及晶化过程研究[J]. 高等学校化学学报, 2018, 39(8): 1624. |
| [12] | 宋伟, 王力群, 曾双利, 王莉, 范勇, 徐家宁. 镉配位聚合物的原位水热合成、晶体结构及荧光性质[J]. 高等学校化学学报, 2018, 39(7): 1406. |
| [13] | 华承贺, 马红超, 董晓丽, 张秀芳. α-Bi2O3纳米管/氮掺杂碳量子点复合材料的合成及光催化性能[J]. 高等学校化学学报, 2018, 39(2): 200. |
| [14] | 冯慧杰, 郑文君. MoS2多级纳米结构的合成及锂离子电池性能[J]. 高等学校化学学报, 2017, 38(7): 1134. |
| [15] | 李兴盛, 赫文秀, 张永强, 于慧颖, 李子庆. β-Ni(OH)2纳米线的相转化法制备及电化学性能[J]. 高等学校化学学报, 2017, 38(2): 261. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||