

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (7): 1426.doi: 10.7503/cjcu20200090
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
收稿日期:2020-02-20
出版日期:2020-07-10
发布日期:2020-04-08
通讯作者:
朱守非
E-mail:sfzhu@nankai.edu.cn
基金资助:Received:2020-02-20
Online:2020-07-10
Published:2020-04-08
Contact:
Shoufei ZHU
E-mail:sfzhu@nankai.edu.cn
Supported by:摘要:
从不对称催化的角度总结了近年来碳硼成键反应的研究进展, 评述了这些反应各自的特点, 并对其未来研究进行了展望.
中图分类号:
TrendMD:
黄明耀, 朱守非. 催化不对称碳硼成键反应研究进展. 高等学校化学学报, 2020, 41(7): 1426.
HUANG Mingyao, ZHU Shoufei. Recent Advances of Catalytic Asymmetric C—B Bond Forming Reactions†. Chem. J. Chinese Universities, 2020, 41(7): 1426.
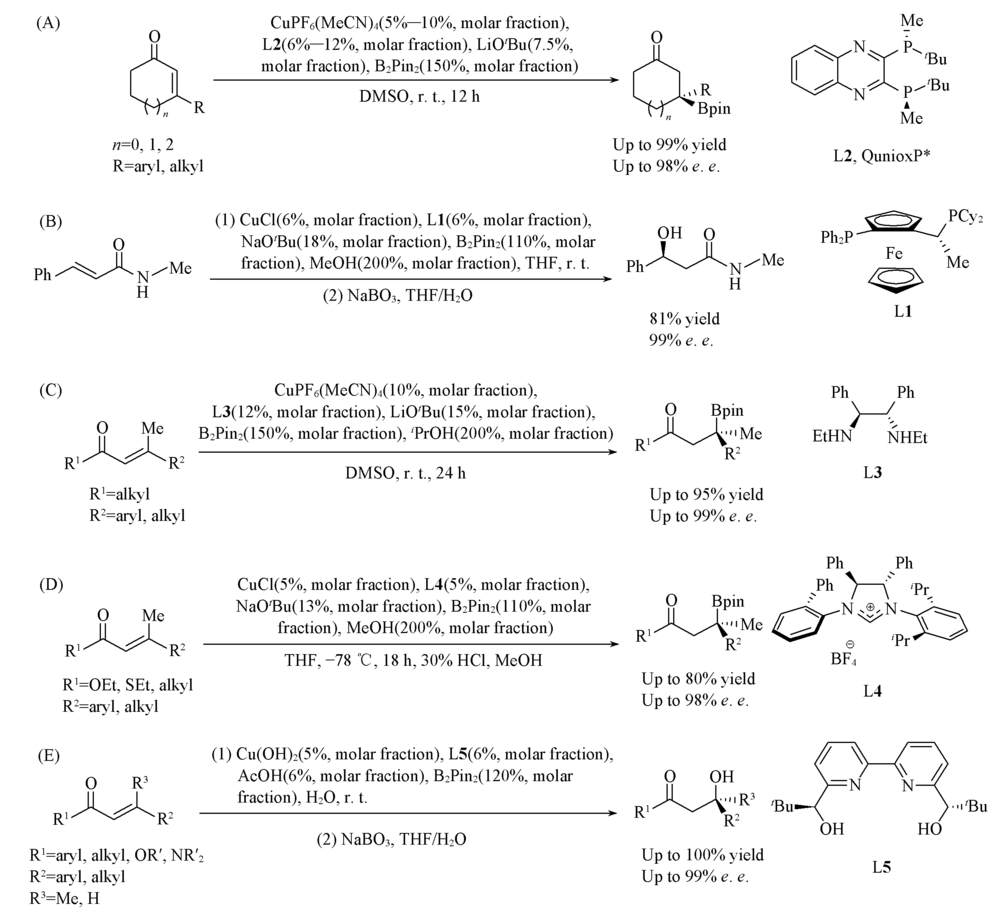
Scheme 4 Cu-catalyzed asymmetric protoboration of alkeneswith different ligands (A) Bisphosphorous ligand L2; (B) bisphosphorous ligand L1; (C) diamine ligands; (D) NHC ligands; (E) bipyridine ligands.
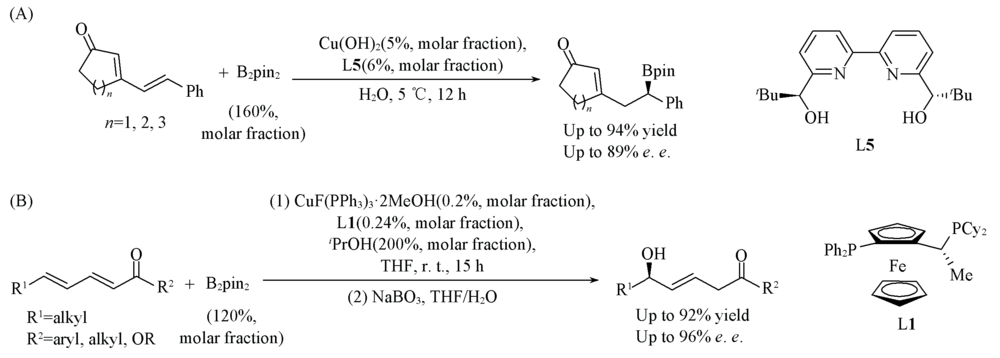
Scheme 5 Cu(Ⅱ)/L5-catalyzed asymmetric 1,6-protoboration of α,β,γ,δ-unsaturated compounds(A) and Cu(Ⅰ)/L1-catalyzed asymmetric 1,6-protoboration of α,β,γ,δ-unsaturated compounds(B)
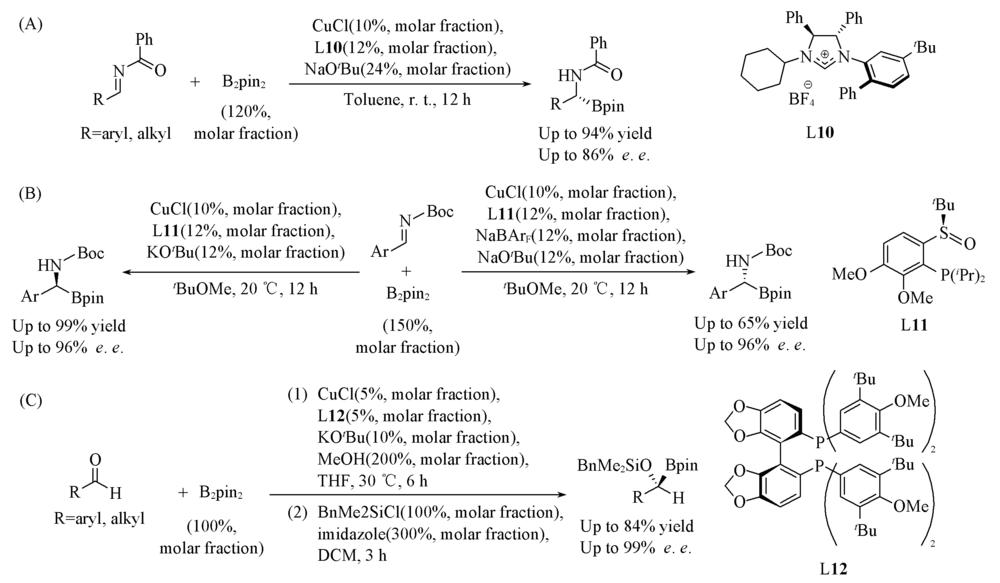
Scheme 9 Cu(Ⅰ)/L10-catalyzed asymmetric protoboration of imines(A), Cu(Ⅰ)/L11-catalyzed asymmetric protoboration of imines(B) and Cu/L12-catalyzed asymmetric protoboration of aldehydes(C)
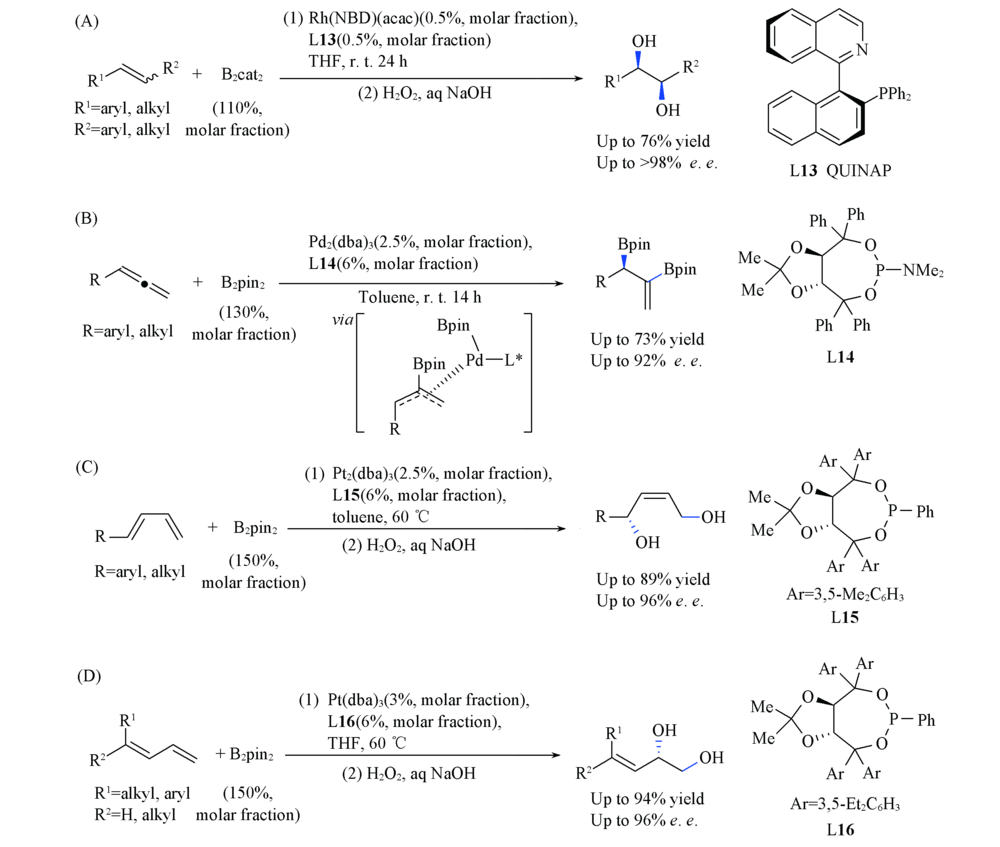
Scheme 12 Rh(Ⅰ)-catalyzed asymmetric diboration of 1,2-disubstituted alkenes(A), Pd(0)-catalyzed asymmetric diboration of allenes(B), Pt(0)-catalyzed 1,4-diboration of conjugated dienes(C) and Pt(0)-catalyzed 1,2-diboration of conjugated dienes(D)
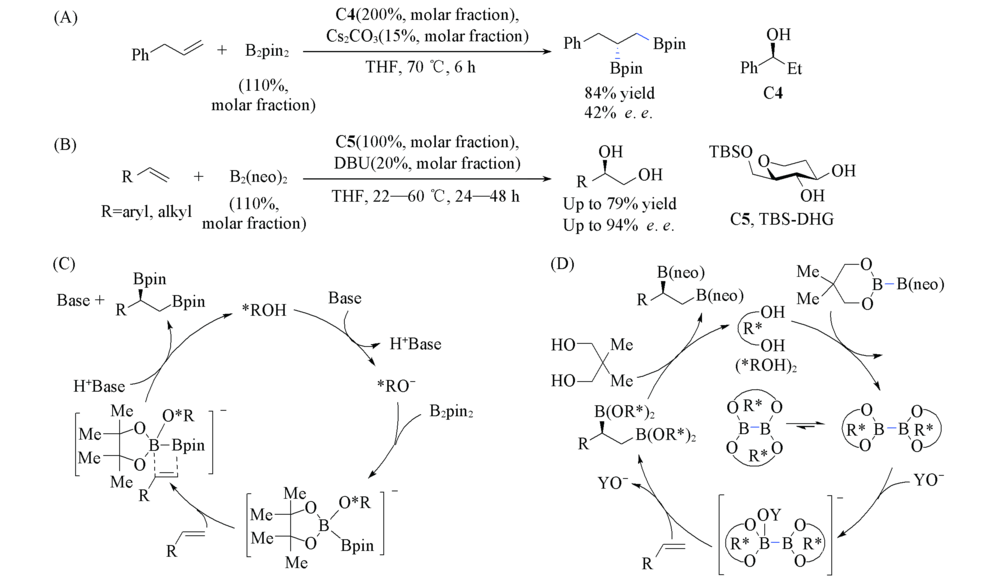
Scheme 14 Alcohol-catalyzed asymmetric diboration of alkenes (A) C4-catalyzed diboration; (B) C5-catalyzed diboration; (C) proposed mechanismof C4-catalyzed diboration; (D) proposed mechanismof C5-catalyzed diboration.
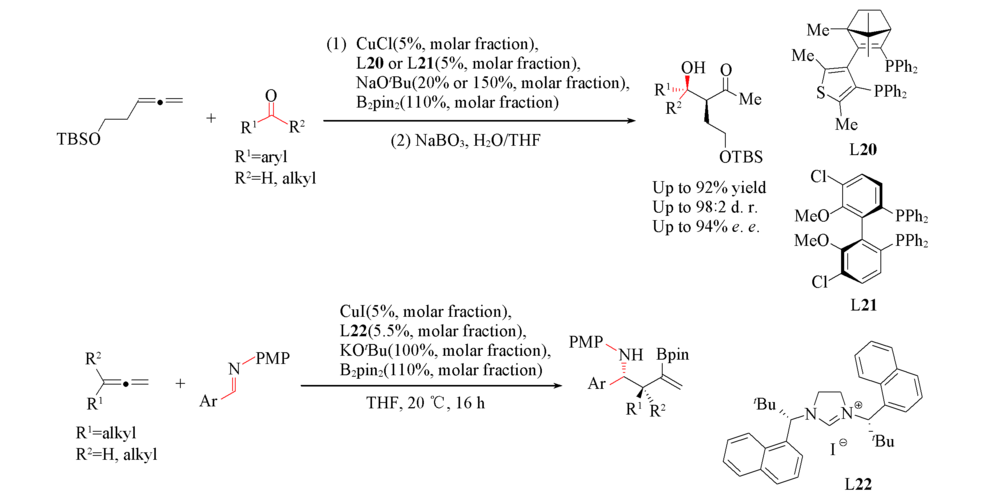
Scheme 18 Cu-catalyzed asymmetric carboration of allenes with aldehydes and ketones(A) and Cu-catalyzed asymmetric carboration of allenes with imines(B)
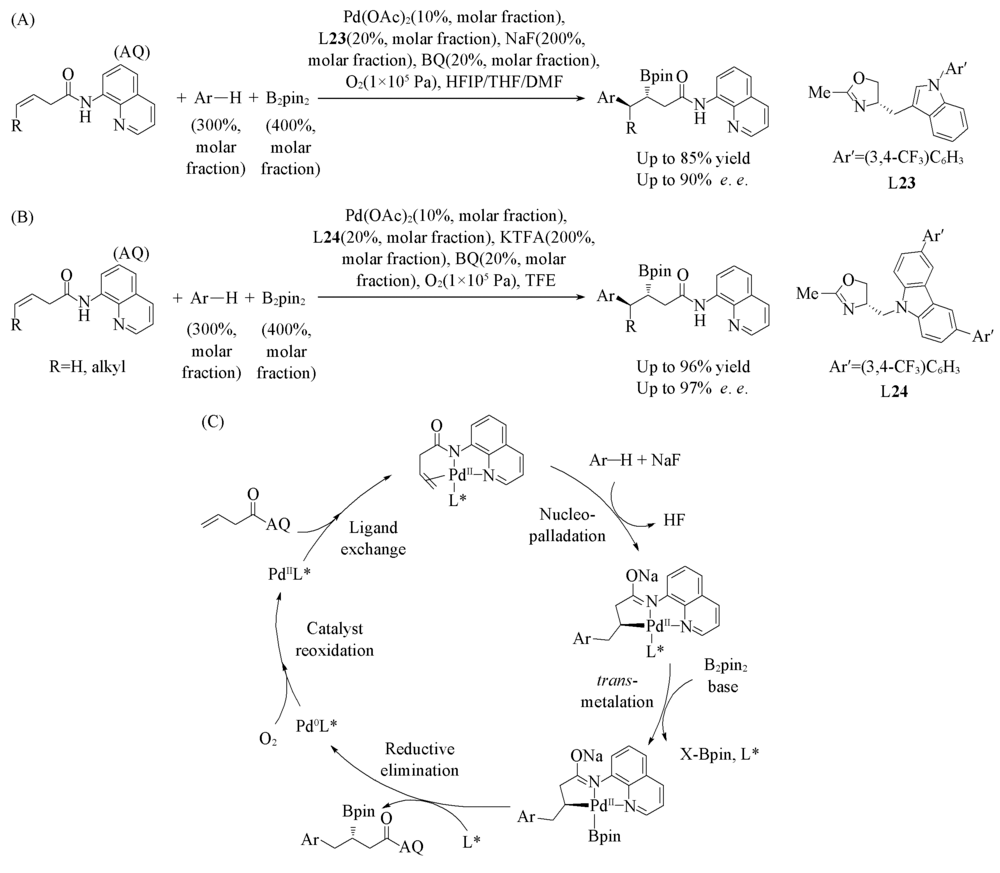
Scheme 19 Pd(Ⅱ)/L23-catalyzed asymmetric arylboration of alkenes(A), Pd(Ⅱ)/L24-catalyzed asymmetric arylboration of alkenes(B) and proposed mechanism(C)
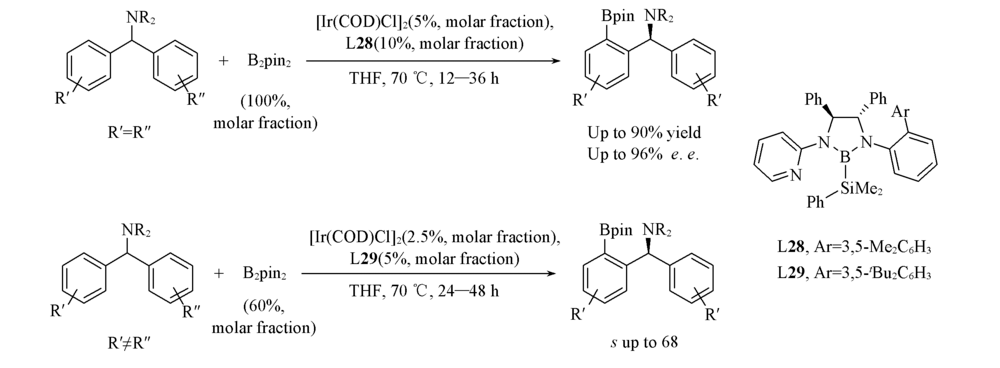
Scheme 23 Ir(Ⅲ)-catalyzed asymmetric C(sp2)—H borylation of diarylmethylamines(A) and Ir(Ⅲ)-catalyzed kinetic resolution through C(sp2)—H borylation of diarylmethylamines(B)
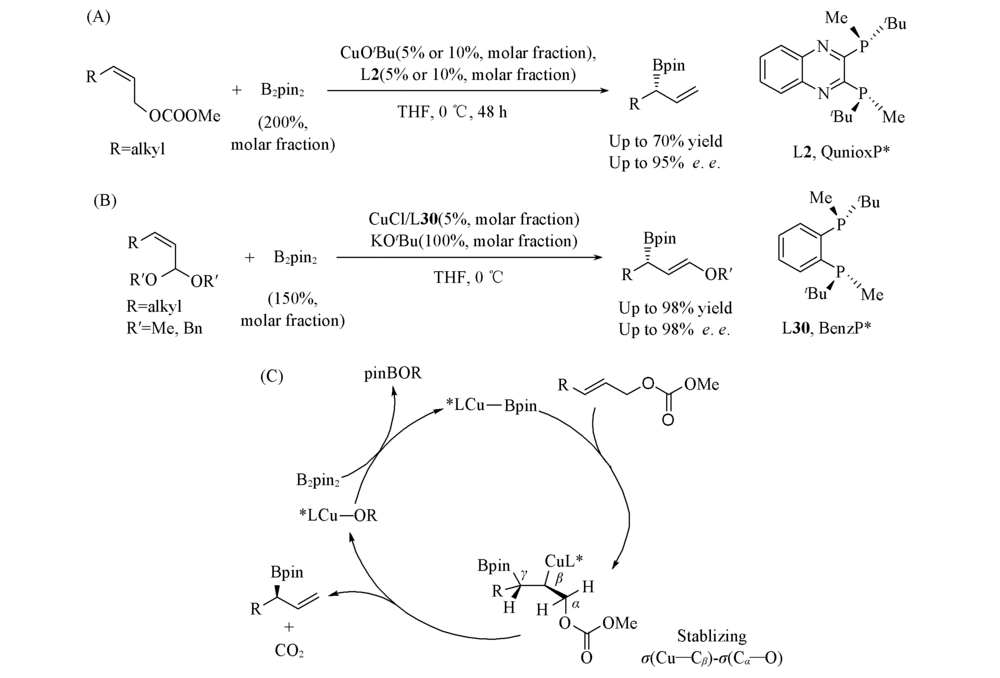
Scheme 24 Cu(Ⅰ)-catalyzed asymmetric borylation of allylic carbonates(A), Cu(Ⅰ)-catalyzed asymmetric borylation of (γ-alkoxyallyl)boronates(B) and proposed mechanism(C)
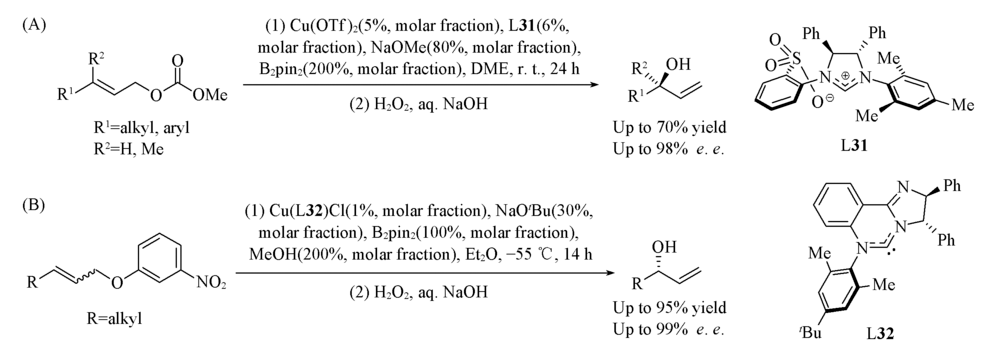
Scheme 25 Cu(Ⅰ)-catalyzed enantioselective borylation of allylic carbonates(A) and Cu(Ⅰ)-catalyzed asymmetric stereo-convergent borylation of allylic carbonates(B)
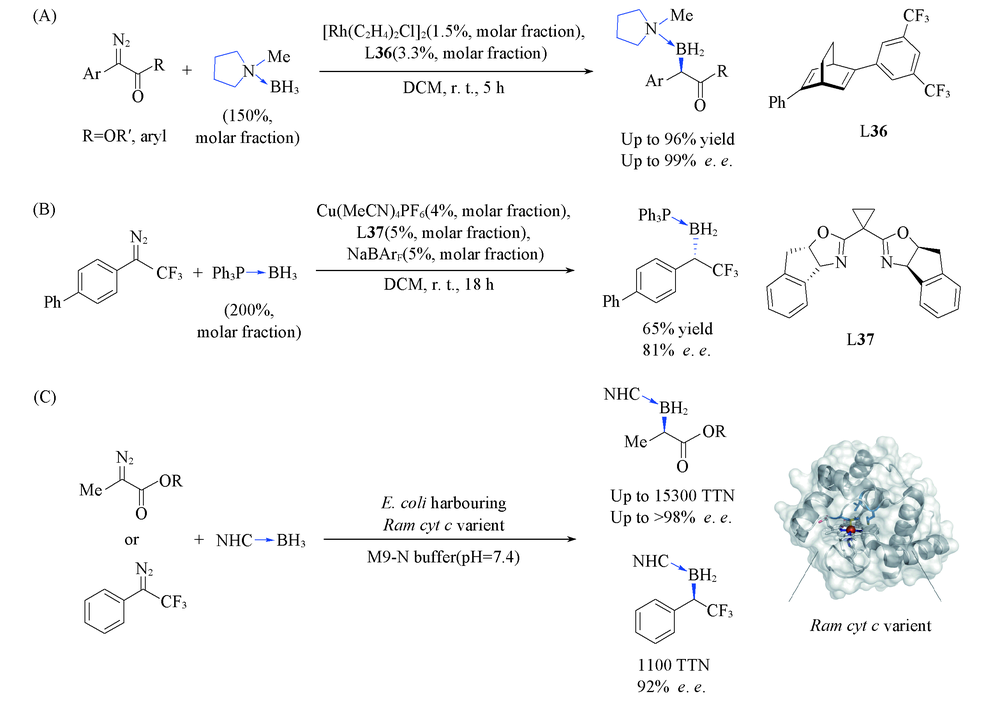
Scheme 30 Rh(Ⅲ)-catalyzed asymmetric B—H bond insertion reaction of diazoketones and diazoesters(A), Cu(Ⅰ)-catalyzed asymmetric B—H bond insertion reaction of α-trifluo methyl diazocompounds(B) and genetically programmed synthesis of chiral organoboranes(C)
| [1] | Miyaura N., Suzuki A., Chem. Rev., 1995, 95, 2457—2483 |
| [2] | Leonori D., Aggarwal V. K., Angew. Chem. Int. Ed., 2015, 54, 1082—1096 |
| [3] | Trippier P. C., MedChemComm, 2010, 1, 183—198 |
| [4] |
Valery M. D., Abed A. A. A. Q., Morris S., Chem. Rev., 2011, 111, 209—237
URL pmid: 21171664 |
| [5] |
Jäkle F., Chem. Rev., 2010, 110, 3985—4022
URL pmid: 20536123 |
| [6] |
William L. A. B., Brent S. S., Chem. Rev., 2016, 116, 1375—1397
URL pmid: 26367140 |
| [7] | Burgess K., Ohlmeyer M. J., J. Org. Chem., 1988, 53, 5178—5179 |
| [8] | Crudden C. M., Edwards D., Eur. J. Org. Chem., 2003, 2003, 4695—4712 |
| [9] | Collins B. S. L., Wilson C. M., Myers E. L., Aggarwal V. K., Angew. Chem. Int. Ed., 2017, 56, 11700—11733 |
| [10] | Chen J., Guo J., Lu Z., Chin. J. Chem., 2018, 36, 1075—1109 |
| [11] | Lee J. E., Yun J., Angew. Chem. Int. Ed., 2008, 47, 145—147 |
| [12] | Dang L., Lin Z., Marder T. B., Organometallics, 2008, 27, 4443—4454 |
| [13] |
Chen I. H., Yin L., Itano W., Kanai M., Shibasaki M., J. Am. Chem. Soc., 2009, 131, 11664—11665
URL pmid: 19653692 |
| [14] | Chea H., Sim H. S., Yun J., Adv. Synth. Catal., 2009, 351, 855—858 |
| [15] |
Calow A. D. J., Batsanov A. S., Pujol A., Solé C., Fernández E., Whiting A., Org. Lett., 2013, 15, 4810—4813
URL pmid: 23988035 |
| [16] | Kubota K., Hayama K., Iwamoto H., Ito H., Angew. Chem. Int. Ed., 2015, 54, 8809—8813 |
| [17] |
Wen L., Yue Z. T., Zhang H. Y., Chong Q. L., Meng F. K., Org. Lett., 2017, 19, 6610—6613
URL pmid: 29172547 |
| [18] |
Chen I. H., Kanai M., Shibasaki M., Org. Lett., 2010, 12, 4098—4101
URL pmid: 20722382 |
| [19] |
O’Brien J. M., Lee K. S., Hoveyda A. H., J. Am. Chem. Soc., 2010, 132, 10630—10633
URL pmid: 20681680 |
| [20] |
Jana A., Trzybiński D., Wozńiak K., Grela K., Chem. Eur. J., 2018, 24, 891—897
URL pmid: 29120504 |
| [21] | Kobayashi S., Xu P. Y., Endo T., Ueno M., Kitanosono K., Angew. Chem. Int. Ed., 2012, 51, 12763—12766 |
| [22] | Kitanosono K., Xu P. Y., Kobayashi S., Chem. Commun., 2013, 49, 8184—8186 |
| [23] |
Steven B. T., Joseph A. C., Webster L. S., Org. Lett., 2012, 14, 1918—1921
doi: 10.1021/ol300575d URL pmid: 22428578 |
| [24] | Luo Y. F., Roy I. D., Madec A. G. E., Lam H. W., Angew. Chem. Int. Ed. , 2014, 53, 4186—4190 |
| [25] | Ibrahem I., Breistein P., Córdova A., Angew. Chem. Int. Ed., 2011, 50, 12036—12046 |
| [26] |
Lee Y., Hoveyda A. H., J. Am. Chem. Soc., 2009, 131, 3160—3161
URL pmid: 19256564 |
| [27] |
Sasaki Y., Zhong C. M., Sawamura M., Ito H., J. Am. Chem. Soc., 2010, 132, 1226—1227
URL pmid: 20063883 |
| [28] |
Parra A., Amenos L., Guisan-Ceinos M., Lopez A., Ruano J. L. G., Tortosa M., J. Am. Chem. Soc., 2014, 136, 15833—15836
URL pmid: 25340304 |
| [29] | Guisan-Ceinos M., Parra A., Martin-Heras V., Tortosa M., Angew. Chem. Int. Ed., 2016, 55, 6969—6972 |
| [30] | Kubota K., Hayama K., Iwamoto H., Ito H., Angew. Chem. Int. Ed., 2015, 54, 8809—8813 |
| [31] |
Kubota K., Watanabe Y., Hayama K., Ito H., J. Am. Chem. Soc., 2016, 138, 4338—4341
URL pmid: 26967578 |
| [32] | Kubota K., Watanabe Y., Ito H., Adv. Synth. Catal., 2016, 358, 2379—2384 |
| [33] |
Zhang S. S., Zhao Y. S., Tian P., Lin G. Q., Synlett, 2013, 24, 437—442
doi: 10.1055/s-00000083 URL |
| [34] |
Wang D., Cao P., Wang B., Jia T., Lou Y. Z., Wang M., Liao J., Org. Lett., 2015, 17, 2420—2423
URL pmid: 25906191 |
| [35] |
Kubota K., Yamamoto E., Ito H., J. Am. Chem. Soc., 2015, 137, 420—424
URL pmid: 25494834 |
| [36] |
Wu H., Radomkit S O’Brien J. M., Hoveyda A. H., J. Am. Chem. Soc., 2012, 134, 8277—8285
URL pmid: 22559866 |
| [37] | Radomkit S., Hoveyda A. H., Angew. Chem. Int. Ed., 2014, 53, 3387—3397 |
| [38] |
Morgan J. B., Miller S. P., Morken J. P., J. Am. Chem. Soc., 2003, 125, 8702—8703
URL pmid: 12862446 |
| [39] | Trudeau S., Morgan J. B., Shrestha M., Morken J. P., J. Org. Chem. , 2005, 70, 9538—9544 |
| [40] |
Pelz N. F., Woodward A. R., Burks H. E., Sieber J. D., Morken J. P., J. Am. Chem. Soc., 2004, 126, 16328—16329
URL pmid: 15600327 |
| [41] |
Burks H. E., Liu S. B., Morken J. P., J. Am. Chem. Soc., 2007, 129, 8766—8773
doi: 10.1021/ja070572k URL |
| [42] |
Burks H. E., Kliman L. T., Morken J. P., J. Am. Chem. Soc., 2009, 131, 9134—9135
URL pmid: 19505078 |
| [43] | Kliman L. T., Mlynarski S. N., Ferris G. E., Morken J. P., Angew. Chem. Int. Ed., 2012, 51, 521—524 |
| [44] |
Kliman L. T., Mlynarski S. N., Morken J. P., J. Am. Chem. Soc., 2009, 131, 13210—13211
doi: 10.1021/ja9047762 URL |
| [45] |
Coombs J. R., Zhang L., Morken J. P., J. Am. Chem. Soc., 2014, 136, 16140—16143
doi: 10.1021/ja510081r URL |
| [46] |
Hong K., Morken J. P., J. Am. Chem. Soc., 2013, 135, 9252—9254
doi: 10.1021/ja402569j URL pmid: 23763463 |
| [47] |
Toribatake K., Nishiyama H., Angew. Chem. Int. Ed., 2013, 52, 11011—11015
doi: 10.1002/anie.201305181 URL |
| [48] |
Bonet A., Sole C., Gulyás H., Fernández E., Org. Biomol. Chem., 2012, 10, 6621—6623
URL pmid: 22811000 |
| [49] |
Fang L. C., Yan Y., Haeffner F., Morken J. P., J. Am. Chem. Soc., 2016, 138, 2508—2511
doi: 10.1021/jacs.5b13174 URL pmid: 26854364 |
| [50] | Ito H., Kosaka K., Nonoyama K., Sasaki Y., Sawamura M., Angew. Chem. Int. Ed. , 2008, 47, 7424—7427 |
| [51] |
Zhong C., Kunii S., Kosaka Y., Sawamura M., Ito H., J. Am. Chem. Soc., 2010, 132, 11440—11442
doi: 10.1021/ja103783p URL pmid: 20684557 |
| [52] |
Meng F. K., McGrath K. P., Hoveyda A. H., Nature, 2014, 513, 367—374
URL pmid: 25230659 |
| [53] |
Jia T., Cao P., Wang B., Lou Y. Z., Yin X. M., Wang M., Liao J., J. Am. Chem. Soc., 2015, 137, 13760—13763
doi: 10.1021/jacs.5b09146 URL pmid: 26458555 |
| [54] |
Logan K. M., Brown M. K., Angew. Chem. Int. Ed., 2017, 56, 851—855
doi: 10.1002/anie.201609844 URL |
| [55] |
Chen B., Cao P., Liao Y., Wang M., Liao J., Org. Lett., 2018, 20, 1346—1349
URL pmid: 29461840 |
| [56] | Meng F. K., Jang H. J., Jung B., Hoveyda A. H., Angew. Chem. Int. Ed., 2013, 52, 5046—5051 |
| [57] | Yeung K., Ruscoe R. E., Rae J., Pulis A. P., Procter D. J., Angew. Chem. Int. Ed., 2016, 55, 11912—11916 |
| [58] | Jiang L. Y., Cao P., Wang M., Chen B., Wang B., Liao J., Angew. Chem. Int. Ed., 2016, 55, 13854—13858 |
| [59] | Li X. B., Meng F. K., Torker S., Shi Y., Hoveyda A. H., Angew. Chem. Int. Ed., 2016, 55, 9997—10002 |
| [60] |
Liu Z., Li X. H., Zeng T., Engle K. M., ACS Catal., 2019, 9, 3260—3265
URL pmid: 31799023 |
| [61] | Bai Z. B., Zheng S. J., Bai Z. Q., Song F. F., Wang H., Peng Q., Chen G., He G., ACS Catal., 2019, 9, 6502—6509 |
| [62] |
Matsuda N., Hirano K., Satoh T., Miura M., J. Am. Chem. Soc., 2013, 135, 4934—4937
doi: 10.1021/ja4007645 URL pmid: 23495912 |
| [63] | Sakae R., Hirano K., Satoh T., Miura M., Angew. Chem. Int. Ed.., 2015, 54, 613—617 |
| [64] |
Kato K., Hirano K., Miura M., Angew. Chem. Int. Ed.., 2016, 55, 14400—14404
doi: 10.1002/anie.v55.46 URL |
| [65] |
Mkhalid I. A. I., Barnard J. H., Marder T. B., Murphy J. M., Hartwig J. F., Chem. Rev., 2010, 110, 890—931
URL pmid: 20028025 |
| [66] |
He J., Shao Q., Wu Q. F., Yu J. Q., J. Am. Chem. Soc., 2017, 139, 3344—3347
doi: 10.1021/jacs.6b13389 URL pmid: 28209055 |
| [67] |
Shi Y. G., Gao Q., Xu S. M., J. Am. Chem. Soc., 2019, 141, 10599—10604
URL pmid: 31259545 |
| [68] | Su B., Zhou T. G., Xu P. L., Shi Z. J., Hartwig J. F., Angew. Chem. Int. Ed., 2017, 56, 7205—7208 |
| [69] |
Zou X. L., Zhao H. N., Li Y. W., Gao Q., Ke Z. F., Xu S. M., J. Am. Chem. Soc., 2019, 141, 5334—5342
URL pmid: 30852888 |
| [70] |
Ito H., Ito S., Sasaki Y., Matsuura K., Sawamura M., J. Am. Chem. Soc., 2007, 129, 14856—14857
URL pmid: 17988133 |
| [71] |
Yamamoto E., Takenouchi Y., Ozaki T., Miya T., Ito H., J. Am. Chem. Soc., 2014, 136, 16515—16521
URL pmid: 25329863 |
| [72] |
Martinez A. G., Hoveyda A. H., J. Am. Chem. Soc., 2010, 132, 10634—10637
URL pmid: 20681681 |
| [73] |
Park J. K., Lackey H. H., Ondrusek B. A., McQuade D. T., J. Am. Chem. Soc., 2011, 133, 2410—2413
doi: 10.1021/ja1112518 URL pmid: 21291218 |
| [74] |
Ito H., Kunii S., Sawamura M ., Nat. Chem., 2010, 2, 972—976
URL pmid: 20966955 |
| [75] | Wang Z. B., Bachman S., Dudnik A. S., Fu G. C., Angew. Chem. Int. Ed., 2018, 57, 14529—14532 |
| [76] |
Cheng R. F., Li B. W., Wu J., Zhang J., Qiu Z. Z., Tang W. J., You S. L., Tang Y., Xie Z. W., J. Am. Chem. Soc., 2018, 140, 4508—4511
URL pmid: 29575894 |
| [77] |
Cheng Q. Q., Zhu S. F., Zhang Y. Z., Xie X. L., Zhou Q. L., J. Am. Chem. Soc., 2013, 135, 14094—14097
URL pmid: 24025045 |
| [78] |
Li X., Curran D. P., J. Am. Chem. Soc., 2013, 135, 12076—12081
URL pmid: 23865527 |
| [79] | Cheng Q. Q., Xu H., Zhu S. F., Zhou Q. L., Acta Chim. Sinica, 2015, 73, 326—329 |
| ( 程清卿, 许唤, 朱守非, 周其林. 化学学报, 2015, 73(4), 326—329) | |
| [80] |
Chen D., Zhang X., Qi W. Y., Xu B., Xu M. H., J. Am. Chem. Soc., 2015, 137, 5268—5271
doi: 10.1021/jacs.5b00892 URL pmid: 25726987 |
| [81] |
Hyde S., Veliks J., Liegault B., Grassi D., Taillefer M., Gouverneur V., Angew. Chem. Int. Ed., 2016, 55, 3785—3789
doi: 10.1002/anie.201511954 URL |
| [82] |
Kan S. B. J., Huang X. Y., Gumulyal Y., Chen K., Arnold F. A., Nature, 2017, 552, 132—136
doi: 10.1038/nature24996 URL pmid: 29186119 |
| [83] |
Pang Y., He Q., Li Z. Q., Yang J. M., Yu J. H., Zhu S. F., Zhou Q. L., J. Am. Chem. Soc., 2018, 140, 10663—10668
URL pmid: 30102528 |
| [84] |
Yang J. M., Li Z. Q., Li M. L., He Q., Zhu S. F., Zhou Q. L., J. Am. Chem. Soc., 2017, 139, 3784—3789
doi: 10.1021/jacs.6b13168 URL pmid: 28195708 |
| [85] | Yang J. M., Zhao Y. T., Li Z. Q., Gu X. S., Zhu S. F., Zhou Q. L., ACS Catal., 2018, 8, 7351—7355 |
| [86] |
Nelson H. M., Williams B. D., Miro M., Toste F. D., J. Am. Chem. Soc., 2015, 137, 3213—3216
doi: 10.1021/jacs.5b00344 URL pmid: 25723255 |
| [87] |
Huang Y., del Pozo J., Torker S., Hoveyda A. H., J. Am. Chem. Soc., 2018, 140, 2643—2655
doi: 10.1021/jacs.7b13296 URL pmid: 29417810 |
| [88] | Sang H. L., Yu S., Ge S., Org. Chem. Front., 2018, 5, 1284—1287 |
| [89] |
Gao D. W., Xiao Y. Y., Liu M. Y., Liu Z., Karunananda M. K., Chen J. S., Engle K. M., ACS Catal., 2018, 8, 3650—3654
doi: 10.1021/acscatal.8b00626 URL pmid: 30740261 |
| [90] | Buñuel N., Cárdenas D. J., Eur. J. Org. Chem., 2016, 2016, 5446—5464 |
| [91] |
Buñuel N., Cárdenas D. J., Chem. Eur. J., 2018, 24, 11239—11244
doi: 10.1002/chem.201800659 URL pmid: 29575256 |
| [1] | 穆宏文, 吴昊, 高莹莹, 金瑛, 王黎明. 有机催化吲哚碳环上的不对称Friedel⁃Crafts烷基化反应[J]. 高等学校化学学报, 2022, 43(2): 20210571. |
| [2] | 许聪, 胡文浩. 手性螺环配体及催化剂在不对称催化反应中的应用研究进展[J]. 高等学校化学学报, 2020, 41(10): 2153. |
| [3] | 郭庆君. 手性磷酰胺类配体不对称催化串联反应合成手性3-取代苯酞化合物[J]. 高等学校化学学报, 2019, 40(10): 2104. |
| [4] | 李霄, 高立国, 弓莹, 马亚军, 马向荣. 杂双金属复合物ZABDP催化芳香酮与芳香醛的直接不对称Aldol反应[J]. 高等学校化学学报, 2017, 38(5): 778. |
| [5] | 李鑫, 韩玉, 谈柏轩, 王斌, 程津培. 苯并呋喃-2,3-二酮和丙酮的不对称Aldol反应[J]. 高等学校化学学报, 2014, 35(9): 1908. |
| [6] | 刘健, 石鑫. 利用"Click"反应制备含有(R)-(+)-BINOL的有机聚合物及其钛配合物在不对称加成反应中的催化性能[J]. 高等学校化学学报, 2013, 34(5): 1052. |
| [7] | 张庆友, 张丹丹, 索净洁, 李静亚, 龙海林, 许禄. 不对称反应的对映体过量值预测[J]. 高等学校化学学报, 2012, 33(07): 1413. |
| [8] | 张东岩, 汪权, 陈福欣, 袁野, 王锐. C2轴对称樟脑磺酰胺基醇配体催化环己烯乙炔对酮的不对称加成反应[J]. 高等学校化学学报, 2008, 29(9): 1750. |
| [9] | 任奇志, 丁晓健, 原鲜霞, 王爱琴 . 单面桥联手性金属卟啉的合成、构象分析及不对称催化氧化性能[J]. 高等学校化学学报, 2007, 28(11): 2128. |
| [10] | 樊保敏,谢建华,周章涛,张齐,涂永强,周其林 . 手性螺环单磷配体在不对称氢甲酰化反应中的应用 |
| [11] | 范青华, 杨夕强, 刘国华, 陈晓闽, 陈新滋. 新型树状结构手性联二萘酚衍生物的合成及催化性能研究[J]. 高等学校化学学报, 2003, 24(2): 274. |
| [12] | 周懿波, 段学欣, 周其林. 新型手性氢化喹啉噁唑啉配体的合成[J]. 高等学校化学学报, 2001, 22(S1): 130. |
| [13] | 彭云贵, 冯小明, 王斌, 李智, 杨桂树, 蒋耀忠. 手性Salen-Cu(Ⅱ)和噁唑啉-Cu(Ⅱ)配合物催化2-苯基环己酮的不对称Baeyer-Villiger反应研究[J]. 高等学校化学学报, 2001, 22(2): 223. |
| [14] | 李枝蓬, 汤方毅, 伍新燕, 周其林, 陈新滋. 钯催化烯丙基取代反应中手性配体的“甲基效应”[J]. 高等学校化学学报, 2001, 22(12): 2032. |
| [15] | 吕守茂, 周海兵, 张骥, 谢如刚, 周忠远, 陈新滋, 杨登贵. N-方酰麻黄碱配体的合成及催化前手性芳酮的不对称还原反应[J]. 高等学校化学学报, 2001, 22(11): 1846. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
