

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (1): 268.doi: 10.7503/cjcu20200458
所属专题: 分子筛功能材料 2021年,42卷,第1期
收稿日期:2020-07-16
出版日期:2021-01-10
发布日期:2021-01-12
通讯作者:
李兰冬
E-mail:lild@nankai.edu.cn
基金资助:
LIU Shanshan1, CHAI Yuchao1, GUAN Naijia1,2, LI Landong1,2( )
)
Received:2020-07-16
Online:2021-01-10
Published:2021-01-12
Contact:
LI Landong
E-mail:lild@nankai.edu.cn
Supported by:摘要:
吸附分离技术与工艺在工业上具有重要意义. 常见的吸附剂包括沸石分子筛、 金属有机框架材料、 活性炭等材料. 分子筛具有比表面积大、 稳定性高、 生产成本低等优势, 可以满足吸附分离技术中高效、 节能和环保的需求, 是一种非常有应用前景的小分子混合物分离吸附剂. 本文综合评述了吸附分离领域中常用的吸附剂材料的特点和吸附分离机理与评价方法, 总结了分子筛在空气分离、 烃类分离、 二氧化碳吸附、 芳香硫化物脱除、 一氧化碳吸附、 氮氧化物吸附、 氢气储存吸附及氢同位素分离等领域的应用, 并对基于分子筛膜的小分子混合物分离现状进行了介绍. 此外, 本文还系统分析了分子筛对不同混合物的吸附分离性能与其拓扑结构、 骨架组成及改性方法之间的关系, 并对未来的研究前景进行了展望.
中图分类号:
TrendMD:
刘珊珊, 柴玉超, 关乃佳, 李兰冬. 分子筛材料在小分子吸附分离中的应用. 高等学校化学学报, 2021, 42(1): 268.
LIU Shanshan, CHAI Yuchao, GUAN Naijia, LI Landong. Small Molecule Adsorption and Separation on Zeolites. Chem. J. Chinese Universities, 2021, 42(1): 268.
| Zeolite | Adsorbate | Adsorption capacity/(mmol·g-1) | Temperature/K | Pressure/kPa | Reference |
|---|---|---|---|---|---|
| Na?A | O2 | 0.10* | 298 | 100 | [ |
| Na?X | N2 | 0.35 | 293 | 100 | [ |
| Na?X | Ar | 0.11 | 293 | 105 | [ |
| Na?X | CH4 | 0.58 | 293 | 99 | [ |
| Na?X | CO2 | 4.64 | 293 | 97 | [ |
| Na?X | CO | 0.93 | 293 | 100 | [ |
| Na?X | C2H6 | 2.70 | 298 | 100 | [ |
| Na?X | C2H4 | 3.32 | 298 | 100 | [ |
| Na?Y | C2H2 | 4.96 | 298 | 100 | [ |
| Na?Y | C2H4 | 3.84 | 298 | 100 | [ |
| ZSM?58 | C3H6 | 1.65 | 298 | 100 | [ |
| ZSM?58 | C3H8 | 0.18 | 298 | 100 | [ |
| Na?Y | NO | 0.04* | 323 | 50 | [ |
| Na?X | SO2 | 2.80 | 323 | 43 | [ |
| Na,Ca?A | N2O | 4.25* | 298 | 100 | [ |
| K?A | H2O | 13.45 | 293 | 2 | [ |
| Si?CHA | H2S | 2.9* | 298 | 100 | [ |
Table 1 Adsorption capacity of typical zeolites toward different gases
| Zeolite | Adsorbate | Adsorption capacity/(mmol·g-1) | Temperature/K | Pressure/kPa | Reference |
|---|---|---|---|---|---|
| Na?A | O2 | 0.10* | 298 | 100 | [ |
| Na?X | N2 | 0.35 | 293 | 100 | [ |
| Na?X | Ar | 0.11 | 293 | 105 | [ |
| Na?X | CH4 | 0.58 | 293 | 99 | [ |
| Na?X | CO2 | 4.64 | 293 | 97 | [ |
| Na?X | CO | 0.93 | 293 | 100 | [ |
| Na?X | C2H6 | 2.70 | 298 | 100 | [ |
| Na?X | C2H4 | 3.32 | 298 | 100 | [ |
| Na?Y | C2H2 | 4.96 | 298 | 100 | [ |
| Na?Y | C2H4 | 3.84 | 298 | 100 | [ |
| ZSM?58 | C3H6 | 1.65 | 298 | 100 | [ |
| ZSM?58 | C3H8 | 0.18 | 298 | 100 | [ |
| Na?Y | NO | 0.04* | 323 | 50 | [ |
| Na?X | SO2 | 2.80 | 323 | 43 | [ |
| Na,Ca?A | N2O | 4.25* | 298 | 100 | [ |
| K?A | H2O | 13.45 | 293 | 2 | [ |
| Si?CHA | H2S | 2.9* | 298 | 100 | [ |
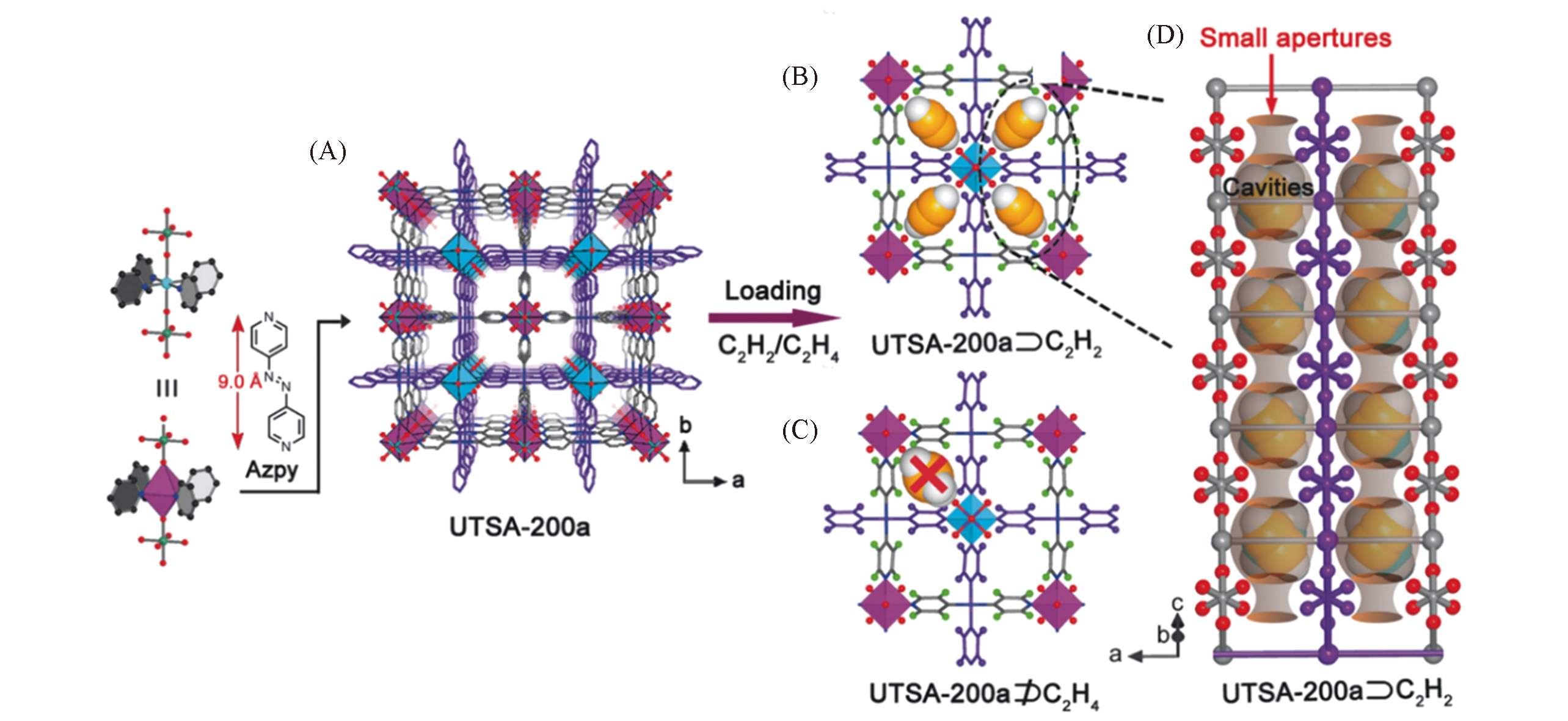
Fig.2 Structure description of UTSA?200a[44](A) Channel structure of UTSA-200a reveals a pores size of 0.34 nm;(B) DFT-D-calculated C2H2 adsorption models in UTSA-200a, revealing that this pore size enables the passage of C2H2 molecules; (C) simulated C2H4 adsorption in UTSA-200a indicating that the C2H4 molecules are too large to pass through the pores; (D) schematic illustration of ideal molecular sieves based on the structure of UTSA-200a?C2H2, in which larger cavities suitable for strongly binding C2H2 molecules are interconnected by narrow apertures that serve as sieves for C2H4 but not for C2H2. Color code: Cu(turquoise), Si(dark green), F(red), N(blue), C(gray), and H(green spheres).Copyright 2017, Wiley-VCH.
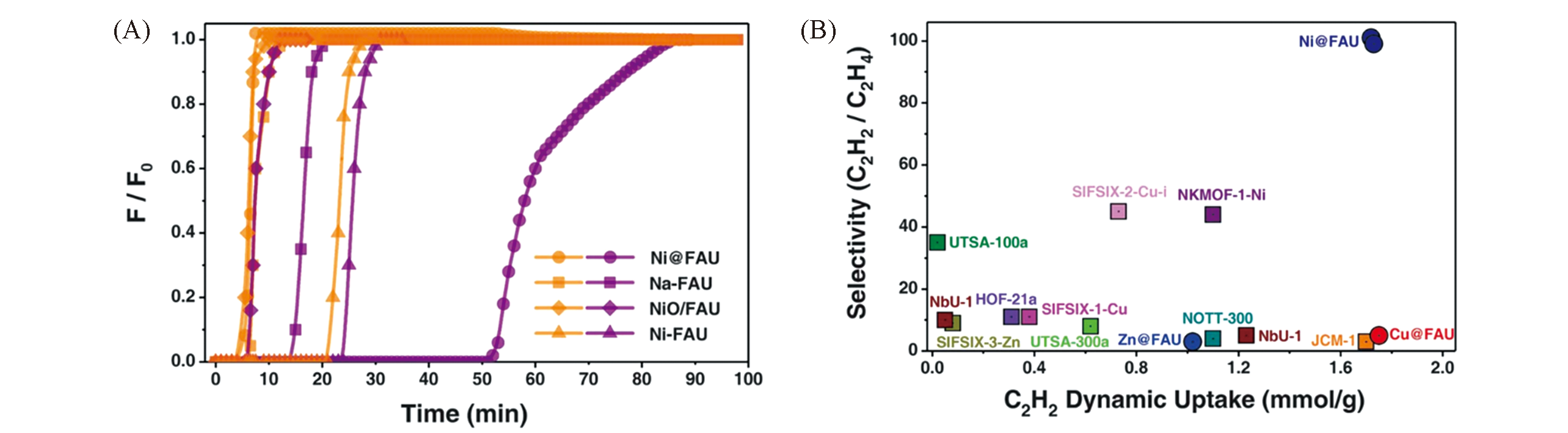
Fig.5 C2H2/C2H4 separation performance of Ni@FAU[10](A) Column breakthrough curves for a C2H2/C2H4(2%/2%) mixture using various zeolite samples at 298 K. C2H2 and C2H4 are shown in purple and orange, respectively.F, flow rate; F0, initial flow rate.(B) Plot of C2H2/C2H4 dynamic selectivity against C2H2 dynamic uptake under ambient conditions with state-of-theart sorbent materials.Copyright 2020, American Association for the Advancement of Science
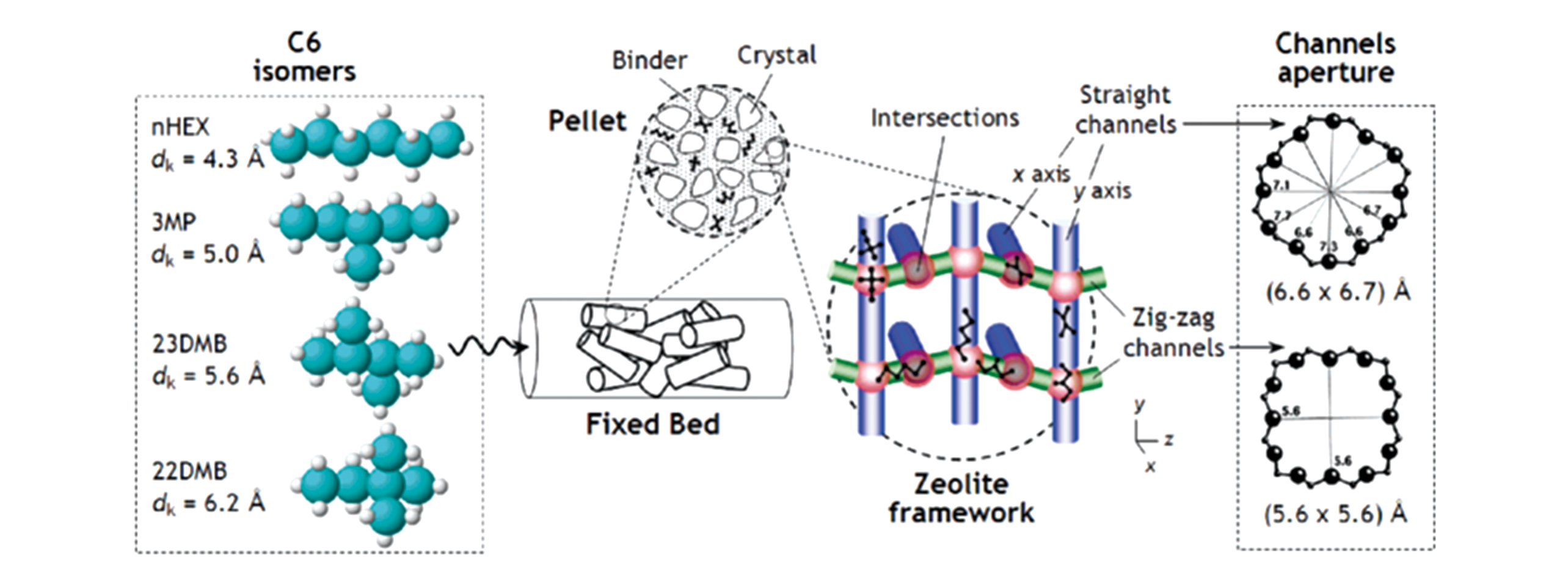
Fig.6 A 3D view of hexane isomers and the respective kinetic diameters[66] and a perspective view of the zeolite BEA framework with the respective 12?ring pore apertures[64]Copyright 2006, American Chemical Society.
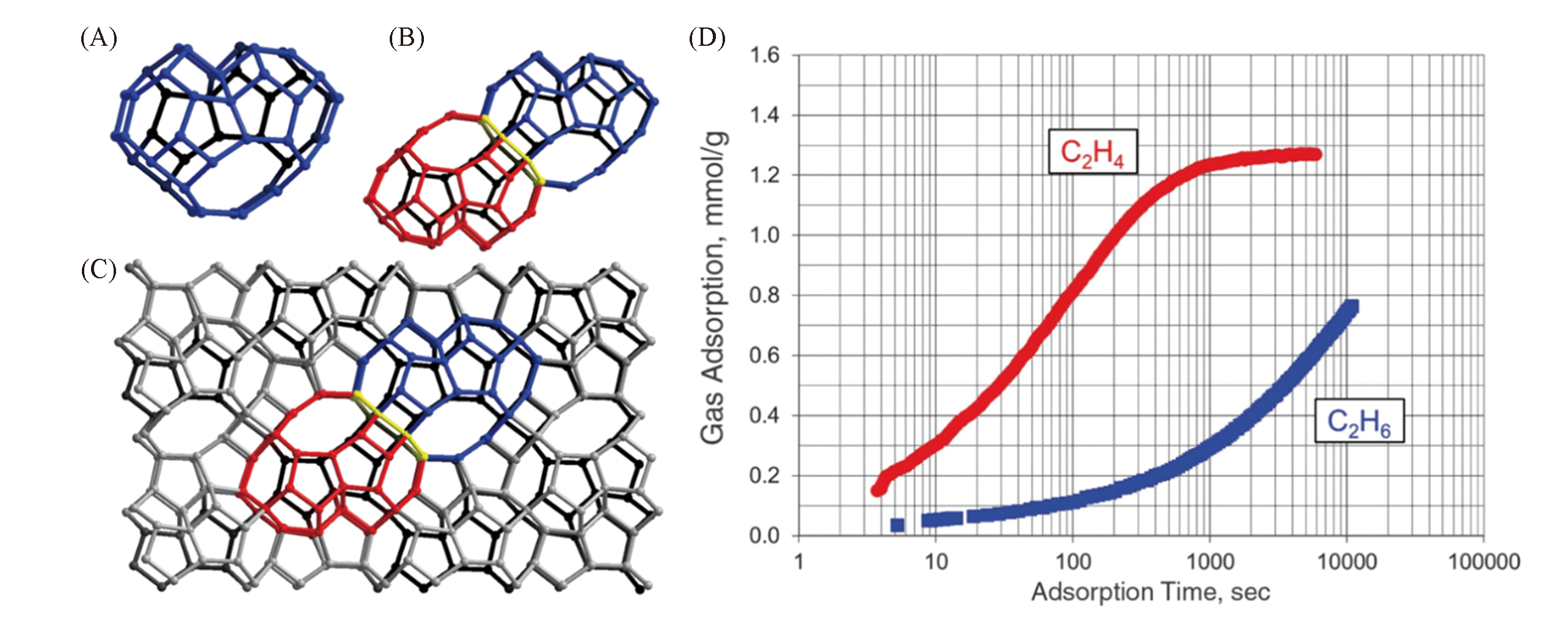
Fig.7 Description of the zeolitic structure of ITQ?55[72](A) A 48T heart-shaped cage;(B) dimeric 48T cages, the yellow ring indicates the interconnecting 8-ring;(C) projection along the b axis of the ITQ-55 structure(oxygen atoms are omitted for clarity; T atoms are in gray; two of the heart-shaped cavities are highlighted in blue and red, respectively); (D) adsorption of ethylene and ethane on small-crystal ITQ-55 at 0.45 bar and 0.6 bar, respectively.Copyright 2017, American Association for the Advancement of Science.
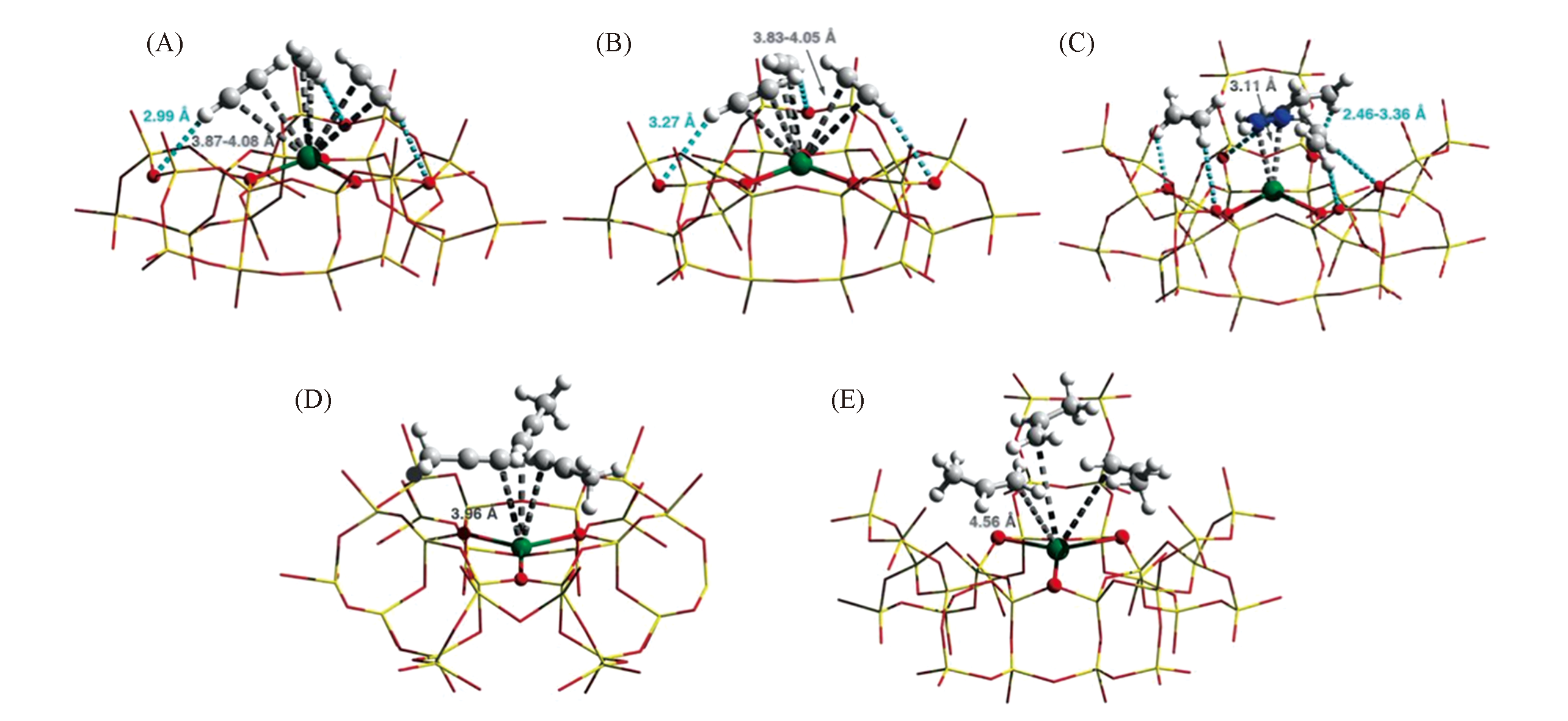
Fig.8 Views of crystal structures for the Ni@FAU zeolite as a function of gas loading[10]All structures were derived from Rietveld refinements of NPD data at 7 K[Si and Al: yellow; O: red; Ni: green; C: gray; D: white; C2D4(1) is highlighted in blue for clarity]. The host-guest interactions are highlighted by dashed lines, and the estimated standard deviation values for binding distances are typically within 0.02 to 0.08 ?. Views are of binding sites for adsorbed gas molecules in [Ni12Na20(Al44Si148O384)](C2D2)12(A), [Ni12Na20(Al44Si148O384)](C2D2)26(B), [Ni12Na20(Al44Si148O384)](C2D4)17(C), [Ni12Na20(Al44Si148O384)]·(C3D4)20(D), and [Ni12Na20(Al44Si148O384)](C3D6)26(E).Copyright 2020, American Association for the Advancement of Science.
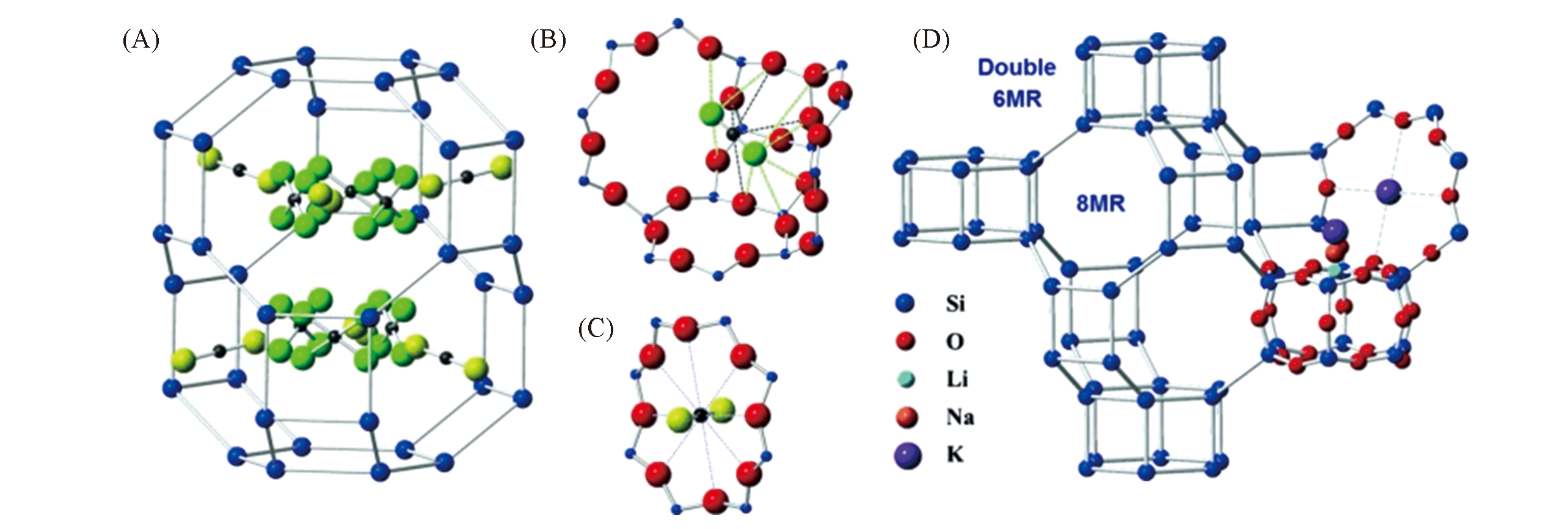
Fig.9 CO2 adsorption sites in pure silica chabazite and illustration of the chabazite structure and typical cation locations[92](A) CO2 adsorption sites in pure silica chabazite with framework oxygen atoms omitted for clarity(site A: 8MR, yellow oxygen atoms; site B: cage, green oxygen atoms); (B) detailed view of site B with short interaction distances to the red framework oxygens given by dashed lines; (C) detailed view of site A with CO2 in the center of the 8 MR; (D) illustration of the chabazite structure and typical cation locations(Li+: cyan, Na+: orange, and K+: purple) in the zeolite pores.Copyright 2014, Wiley-VCH.
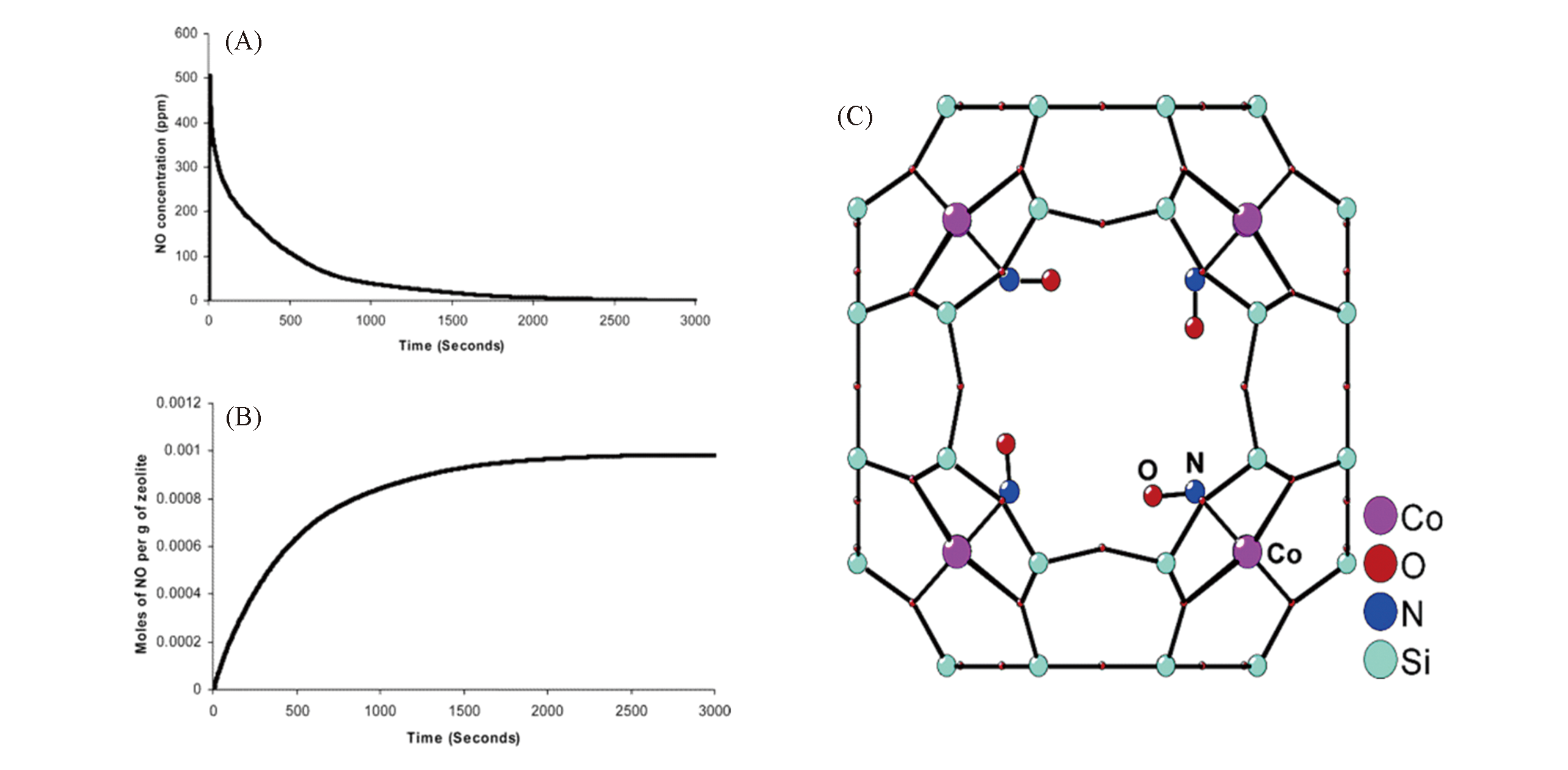
Fig.10 Two NO?release profiles for NO?loaded Co?exchanged zeolite?A(A, B) and structure of the cobalt?NO complex in zeolite?A as revealed by single?crystal X?ray diffraction(C)[116](A) NO concentration in the gas stream passing through the chemiluminescence detector versus time; (B) total amount of NO released by the material with time; (C) the cobalt is bound to three oxygen atoms of a six-ring unit in the zeolite framework and bonds to the nitrogen of a bent mononitrosyl ligand.Copyright 2006, American Chemical Society.
| 1 | Sholl D. S., Lively R. P., Nature, 2016, 532(435), 6—9 |
| 2 | Li J. R., Kuppler R. J., Zhou H. C., Chem. Soc. Rev., 2009, 38(5), 1477—1504 |
| 3 | Yang R. T., Adsorbents Fundamentals and Applications, John Wiley & Sons, Hoboken, 2004, 3—4 |
| 4 | Chen K. J., Scott H. S., Madden D. G., Pham T., Kumar A., Bajpai A., Lusi M., Forrest K. A., Space B., Perry J. J., Zaworotko M. J., Chem., 2016, 1(5), 753—765 |
| 5 | Cui X., Chen K., Xing H., Yang Q., Krishna R., Bao Z., Wu H., Zhou W., Dong X., Han Y., Li B., Ren Q., Zaworotko M. J., Chen B., Science, 2016, 353(6295), 141—144 |
| 6 | Sherry H. S., Science, 1966, 153(3735), 555—556 |
| 7 | Siriwardane R. V., Shen M. S., Fisher E. P., Energy & Fuels, 2003, 17(3), 571—576 |
| 8 | Park Y., Ju Y., Park D., Lee C. H., Chem. Eng. J., 2016, 292, 348—365 |
| 9 | Pu S., Wang J., Li L., Zhang Z., Bao Z., Yang Q., Yang Y., Xing H., Ren Q., Ind. Eng. Chem. Res., 2018, 57(5), 1645—1654 |
| 10 | Chai Y., Han X., Li W., Liu S., Yao S., Wang C., Shi W., da⁃Silva I., Manuel P., Cheng Y., Daemen L. D., Ramirez-Cuesta A. J., Tang C. C., Jiang L., Yang S., Guan N., Li L., Science, 2020, 368(6494), 1002—1006 |
| 11 | Selzer C., Werner A., Kaslel S., Ind. Eng. Chem. Res., 2018, 57, 6609—6617 |
| 12 | Yi H., Deng H., Tang X., Yu Q., Zhou X., Liu H., J. Hazard. Mater., 2012, 203–204, 111—117 |
| 13 | Wu T., Shen Y., Feng L., Tang Z., Zhang D., J. Chem. Eng. Data, 2019, 64(8), 3473—3482 |
| 14 | Kim K. M., Oh H. T., Lim S. J., Ho K., Park Y., Lee C. H., J. Chem. Eng. Data., 2016, 61(4), 1547—1554 |
| 15 | Maghsoudi H., Soltanieh M., Bozorgzadeh H., Mohamadalizadeh A., Adsorption, 2013, 19(5), 1045—1053 |
| 16 | Li H., Eddaoudi M., O’Keeffe M., Yaghi O. M., Nature, 1999, 402, 276—279 |
| 17 | Li Q., Zhang W., Miljanić O. Š., Sue C. H., Zhao Y. L., Liu L., Knobler C. B., Stoddart J. F., Yaghi O. M., Science, 2009, 325(5942), 855—859 |
| 18 | Chen Z., Li P., Anderson R., Wang X., Zhang X., Robison L., Redfern L. R., Moribe S., Islamoglu T., Gómez⁃Gualdrón D. A., Yildirim T., Stoddart J. F., Farha O. K., Science, 2020, 368(6488), 297—303 |
| 19 | Mafra L., Čendak T., Schneider S., Wiper P. V., Pires J., Gomes J. R. B., Pinto M. L., J. Am. Chem. Soc., 2017, 139(1), 389—408 |
| 20 | Espinal L., Wong⁃Ng W., Kaduk J. A., Allen A. J., Snyder C. R., Chiu C., Siderius D. W., Li L., Cockayne E., Espinal A. E., Suib S. L., J. Am. Chem. Soc., 2012, 134(18), 7944—7951 |
| 21 | Durá G., Budarin V. L., Castro⁃Osma J. A., Shuttleworth P. S., Quek S. C. Z., Clark J. H., North M., Angew. Chem. Int. Ed., 2016, 55(32), 9173—9177 |
| 22 | Richter H., Voss H., Kaltenborn N., Kämnitz S., Wollbrink A., Feldhoff A., Caro J., Roitsch, S., Voigt I., Angew. Chem. Int. Ed., 2017, 56(27), 7760—7763 |
| 23 | Koh D., McCool B. A., Deckman H. W., Lively R. P., Science, 2016, 353(6301), 804—807 |
| 24 | Bao Z., Xie D., Chang G., Wu H., Li L., Zhou W., Wang H., Zhang Z., Xing H., Yang Q., Zaworotko M. J., Ren Q., Chen B., J. Am. Chem. Soc., 2018, 140(13), 4596—4603 |
| 25 | Zhang X., Li L., Wang J. X., Wen H. M., Krishna R., Wu H., Zhou W., Chen Z. N., Li B., Qian G., Chen B., J. Am. Chem. Soc., 2020, 142(1), 633—640 |
| 26 | Lü J., Cao R., Angew. Chem. Int. Ed., 2016, 55(33), 9474—9480 |
| 27 | Furukawa H., Yaghi O. M., J. Am. Chem. Soc., 2009, 131(25), 8875—8883 |
| 28 | Zhao J., He G., Huang S., Villalobos L. F., Dakhchoune M., Bassas H., Agrawal K. V., Sci. Adv., 2019, 5(1), 1—10 |
| 29 | Qiu H., Xue M., Shen C., Zhang Z., Guo W., Adv. Mater., 2019, 31(42), 1—16 |
| 30 | Chowdhury S., Balasubramanian R., Sci. Rep., 2016, 6(21537), 1—10 |
| 31 | Liu M., Zhang L., Little M. A., Kapil V., Ceriotti M., Yang S., Ding L., Holden D. L., Balderas⁃Xicohténcatl R., He D., Clowes R., Chong S. Y., Schütz G., Chen L., Hirscher M., Cooper A. I., Science, 2019, 366(6465), 613—620 |
| 32 | Jin Y., Voss B. A., Noble R. D., Zhang W., Angew. Chem. Int. Ed., 2010, 49(36), 6348—6351 |
| 33 | Lee J. H., Lee H. J., Lim S. Y., Kim B. G., Choi J. W., J. Am. Chem. Soc., 2015, 137(22), 7210—7216 |
| 34 | Yoon J. W., Yoon T. U., Kim E. J., Kim A. R., Jung T. S., Han S. S., Bae Y. S., J. Hazard. Mater., 2018, 341, 321—327 |
| 35 | Cavalcanti L. P., Kalantzopoulos G. N., Eckert J., Knudsen K. D., Fossum J. O., Sci. Rep., 2018, 8(1), 1—6 |
| 36 | Herling M. M., Rieß M., Sato H., Li L., Martin T., Kalo H., Matsuda R., Kitagawa S., Breu J., Angew. Chem., 2018, 130(2), 573—577 |
| 37 | Rostami M., Mofarahi M., Karimzadeh R., Abedi D., J. Chem. Eng. Data, 2016, 61(7), 2638—2646 |
| 38 | Lin J. Y. S., Science, 2016, 353(6295), 8—10 |
| 39 | Kuznicki S. M., Bell V. A., Nair S., Hillhouse H. W., Jacubinas R. M., Braunbarth C. M., Toby B. H., Tsapatsis M., Nature, 2001, 412(6848), 720—724 |
| 40 | Yu L., Dong X., Gong Q., Acharya S. R., Lin Y., Wang H., Han Y., Thonhauser T., Li J., J. Am. Chem. Soc., 2020, 142, 6925—6929 |
| 41 | Chen K. J., Madden D. G., Pham T., Forrest K. A., Kumar A., Yang Q. Y., Xue W., Space B., Perry J. J., Zhang J. P., Chen X. M., Zaworotko M. J., Angew. Chem. Int. Ed., 2016, 55(35), 10268—10272 |
| 42 | Bao Z., Wang J., Zhang Z., Xing H., Yang Q., Yang Y., Wu H., Krishna R., Zhou W., Chen B., Ren Q., Angew. Chem. Int. Ed., 2018, 57(49), 16020—16025 |
| 43 | Aguado S., Bergeret G., Daniel C., Farrusseng D., J. Am. Chem. Soc., 2012, 134(36), 14635—14637 |
| 44 | Li B., Cui X., O’Nolan D., Wen H. M., Jiang M., Krishna R., Wu H., Lin R. B., Chen Y. S., Yuan D., Xing H., Zhou W., Ren Q., Qian G., Zaworotko M. J., Chen B., Adv. Mater., 2017, 29(47), 1—7 |
| 45 | Liao P. Q., Huang N. Y., Zhang W. X., Zhang J. P., Chen X. M., Science, 2017, 356(6343), 1193—1196 |
| 46 | Li K., Olson D. H., Seidel J., Emge T. J., Gong H., Zeng H., Li J., J. Am. Chem. Soc., 2009, 131(30), 10368—10369 |
| 47 | Li B., Zhang Y., Krishna R., Yao K., Han Y., Wu Z., Ma D., Shi Z., Pham T., Space B., Liu J., Thallapally P. K., Liu J., Chrzanowski M., Ma S., J. Am. Chem. Soc., 2014, 136(24), 8654—8660 |
| 48 | Cao D. W., Equilibrium Quantum Sieving Effect of Hydrogen Isotopes Separation by Microporous Metal Organic Framework Materials, China Academy of Engineering Physics, Mianyang, 2017(曹大伟. 微孔金属有机框架材料氢同位素热力学量子筛效应研究, 绵阳: 中国工程物理研究院, 2010) |
| 49 | Beenakker J. J. M., Borman V. D., Krylov S. Y., Chem. Phys. Lett., 1995, 232(4), 379—382 |
| 50 | Shang J., Li G., Singh R., Gu Q., Nairn K. M., Bastow T. J., Medhekar N., Doherty C. M., Hill A. J., Liu J. Z., Webley P. A., J. Am. Chem. Soc., 2012, 134(46), 19246—19253 |
| 51 | Baerdemaeker T. D., de Vos D., Nat. Chem., 2013, 5(2), 89—90 |
| 52 | Jiang J., Sandler S. I., Langmuir, 2006, 22(13), 5702—5707 |
| 53 | Myers A. L., Prausnitz J. M., AIChE J., 1965, 11(1), 121—127 |
| 54 | Walton K. S., Sholl D. S., AIChE J., 2015, 61(9), 2757—2762 |
| 55 | Bower J. K., Barpaga D., Prodinger S., Krishna R., Schaef H. T., McGrail B. P., Derewinski M. A., Motkuri R. K., ACS Appl. Mater. Interfaces, 2018, 10(17), 14287—14291 |
| 56 | Li L., Lin R. B., Krishna R., Li H., Xiang S., Wu H., Li J., Zhou W., Chen B., Science, 2018, 362(6413), 443—446 |
| 57 | Hassan M. M., Raghavan N. S., Ruthven D. M., Chem. Eng. Sci., 1987, 42(8), 2037—2043 |
| 58 | Shang Y., Wu J., Zhu J., Wang Y., Meng C., J. Alloys Compd., 2009, 478(1/2), 5—7 |
| 59 | Shang Y., Wu J., Zhu J., Wang Y., Liu R., Meng C., Mater. Res. Bull., 2010, 45(9), 1132—1134 |
| 60 | Baksh M. S. A., Kikkinides E. S., Yang R. T., Sep. Sci. Technol., 1992, 27(3), 277—294 |
| 61 | Yang R. T., Chen Y. D., Peck J. D., Chen N., Ind. Eng. Chem. Res., 1996, 35(9), 3093—3099 |
| 62 | Chao C. C., Millwood N. Y., Process for Separating Nitrogen from Mixtures Thereof with Less Polar Substances, 4859217, 1989⁃08⁃22 |
| 63 | Huddersman K., Klimczyk M., AIChE J., 1996, 42(2), 405—408 |
| 64 | Bárcia P. S., Silva J. A. C., Rodrigues A. E., Ind. Eng. Chem. Res., 2006, 45(12), 4316—4328 |
| 65 | Denayer J. F. M., Ocakoglu R. A., Arik I. C., Kirschhock C. E. A., Martens J. A., Baron G. V., Angew. Chem. Int. Ed., 2005, 44(3), 400—403 |
| 66 | Maloncy M. L., Gora L., Jansen J. C., Maschmeyer T., Ars Sep. Acta, 2003, 2, 18—28 |
| 67 | Bárcia P. S., Ferreira A., Gascon J., Aguado S., Silva J. A. C., Rodrigues A. E., Kapteijn F., Micropor.Mesopor. Mater., 2010, 128(1—3), 194—202 |
| 68 | Zhang H., Xiao Q., Guo X., Li N., Kumar P., Rangnekar N., Jeon M. Y,., Al⁃Thabaiti S., Narasimharao K., Basahel S. N., Topuz B., Onorato F. J., Macosko C. W., Mkhoyan K. A., Tsapatsis M., Angew. Chem. Int. Ed., 2016, 55(25), 7184—7187 |
| 69 | Funke H. H., Kovalchick M. G., Falconer J. L., Noble R. D., Ind. Eng. Chem. Res., 1996, 35(5), 1575—1582 |
| 70 | Mofarahi M., Salehi S. M., Adsorption, 2013, 19(1), 101—110 |
| 71 | Grande C. A., Rodrigues A. E., Ind. Eng. Chem. Res., 2005, 44(23), 8815—8829 |
| 72 | Bereciartua P. J., Cantín Á., Corma A., Jordá J. L., Palomino M., Rey F., Valencia S., Corcoran E. W., Kortunov P., Ravikovitch P. I., Burton A., Yoon C., Wang Y., Paur C., Guzman J., Bishop A. R., Casty G. L., Science, 2017, 358(6366), 1068—1071 |
| 73 | Padin J., Yang R. T., Munson C. L., Ind. Eng. Chem. Res.,1999, 38(10), 3614—3621 |
| 74 | Yang L., Cui X., Yang Q., Qian S., Wu H., Bao Z., Zhang Z., Ren Q., Zhou W., Chen B., Xing H., Adv. Mater., 2018, 30(10), 1—8 |
| 75 | Li L., Lin R. B., Krishna R., Wang X., Li B., Wu H., Li J., Zhou W., Chen B., J. Am. Chem. Soc., 2017, 139(23), 7733—7736 |
| 76 | Breck D. W., Eversole W. G., Milton R. M., Reed T. B., Thomas T. L., J. Am. Chem. Soc., 1956, 78(23), 5963—5972 |
| 77 | Zhu W., Kapteijn F., Moulijn J. A., Jansen J. C., Phys. Chem. Chem. Phys., 2000, 2(8), 1773—1779 |
| 78 | Tijsebaert B., Varszegi C., Gies H., Xiao F. S., Bao X., Tatsumi T., Müller U., De Vos D., Chem. Commun., 2008,2480—2482 |
| 79 | Takahashi A., Yang R. T., Munson C. L., Chinn D., Langmuir, 2001, 17(26), 8405—8413 |
| 80 | Chen S. J., Fu Y., Huang Y. X., Tao Z. C., Zhu M., Appl. Energy, 2016, 179, 329—337 |
| 81 | Mofarahi M., Gholipour F., Micropor. Mesopor. Mater., 2014, 200, 1—10 |
| 82 | Li Y., Yi H., Tang X., Li F., Yuan Q., Chem. Eng. J., 2013, 229, 50—56 |
| 83 | Pham T. D., Liu Q., Lobo R. F., Langmuir, 2013, 29(2), 832—839 |
| 84 | Arefi Pour A., Sharifnia S., Neishabori Salehi R., Ghodrati M., J. Nat. Gas Sci. Eng., 2015, 26, 1246—1253 |
| 85 | Bacsik Z., Cheung O., Vasiliev P., Hedin N., Appl. Energy, 2016, 162, 613—621 |
| 86 | Li G., Xiao P., Webley P., Zhang J., Singh R., Marshall M., Adsorption, 2008, 14(2/3), 415—422 |
| 87 | Montanari T., Finocchio E., Salvatore E., Garuti G., Giordano A., Pistarino C., Busca G., Energy, 2011, 36(1), 314—319 |
| 88 | Ke Q., Sun T., Wei X., Guo Y., Wang S., ChemSusChem, 2017, 10(21), 4207—4214 |
| 89 | Hudson M. R., Queen W. L., Mason J. A., Fickel D. W., Lobo R. F., Brown C. M., J. Am. Chem. Soc., 2012, 134(4), 1970—1973 |
| 90 | Sethia G., Somani R. S., Chand Bajaj H., RSC Adv., 2015, 5(17), 12773—12781 |
| 91 | Liu Q., Mace A., Bacsik Z., Sun J., Laaksonen A., Hedin N., Chem. Commun., 2010, 46(25), 4502—4504 |
| 92 | Pham T. D., Hudson M. R., Brown C. M., Lobo R. F., ChemSusChem, 2014, 7(11), 3031—3038 |
| 93 | Grajciar L., Čejka J., Zukal A., Otero Areán C., Turnes Palomino G., Nachtigall P., ChemSusChem, 2012, 5(10), 2011—2022 |
| 94 | Polisi M., Grand J., Arletti R., Barrier N., Komaty S., Zaarour M., Mintova S., Vezzalini G., J. Phys. Chem. C, 2019, 123(4), 2361—2369 |
| 95 | Stevens R. W., Siriwardane R. V., Logan J., Energy & Fuels, 2008, 22(5), 3070—3079 |
| 96 | Lozinska M. M., Mangano E., Mowat J. P. S., Shepherd A. M., Howe R. F., Thompson S. P., Parker J. E., Brandani S., Wright P. A., J. Am. Chem. Soc., 2012, 134(42), 17628—17642 |
| 97 | Georgieva V. M., Bruce E. L., Verbraeken M. C., Scott A. R., Casteel W. J., Brandani S., Wright P. A., J. Am. Chem. Soc., 2019, 141(32), 12744—12759 |
| 98 | Panda D., Kumar E. A., Singh S. K., Ind. Eng. Chem. Res., 2019, 58, 5301—5313 |
| 99 | Jadhav P. D., Chatti R. V., Biniwale R. B., Labhsetwar N. K., Devotta S., Rayalu S. S., Energy & Fuels, 2007, 21(6), 3555—3559 |
| 100 | Bezerra D. P., Silva F. W. M. D., Moura P. A. S. D., Sousa A. G. S., Vieira R. S., Rodriguez⁃Castellon E., Azevedo D. C. S., Appl. Surf. Sci., 2014, 314, 314—321 |
| 101 | Chatti R., Bansiwal A. K., Thote J. A., Kumar V., Jadhav P., Lokhande S. K., Biniwale R. B., Labhsetwar N. K., Rayalu S. S., Micropor. Mesopor. Mater., 2009, 121(1—3), 84—89 |
| 102 | Kim C., Cho H. S., Chang S., Cho S. J., Choi M., Energy Environ. Sci., 2016, 9(5), 1803—1811 |
| 103 | Huang Y. Y., J. Catal., 1973, 30(2), 187—194 |
| 104 | Xie Y., Zhang J., Qiu J., Tong X., Fu J., Yang G., Yan H., Tang Y., Adsorption, 1996, 3(1), 27—32 |
| 105 | Gao F., Wang Y., Wang S., Chem. Eng. J., 2016, 290, 418—427 |
| 106 | Yang R. T., Hernández⁃Maldonado A. J., Yang F. H., Science, 2003, 301(5629), 79—81 |
| 107 | Takahashi A., Yang F. H., Yang R. T. Ind. Eng. Chem. Res., 2002, 41, 2487—2496 |
| 108 | Hernández⁃Maldonado A. J., Yang R. T., J. Am. Chem. Soc., 2004, 126, 992—993 |
| 109 | Lee K. X., Valla J. A., Appl. Catal. B Environ., 2017, 201, 359—369 |
| 110 | Centi G., Generali P., Dall’olio L., Perathoner S., Rak Z., Ind. Eng. Chem. Res., 2000, 39(1), 131—137 |
| 111 | Rezaei F., Rownaghi A. A., Monjezi S., Lively R. P., Jones C. W., Energy & Fuels, 2015, 29(9), 5467—5486 |
| 112 | Deng H., Yi H., Tang X., Yu Q., Ning P., Yang L., Chem. Eng. J., 2012, 188, 77—85 |
| 113 | Ryou Y. S., Lee J., Cho S. J., Lee H., Kim C. H., Kim D. H., Appl. Catal. B Environ., 2017, 212, 140—149 |
| 114 | Lee J., Ryou Y. S., Cho S. J., Lee H., Kim C. H., Kim D. H., Appl. Catal. B Environ., 2018, 226, 71—82 |
| 115 | Ogura M., Itabashi K., Dedecek J., Onkawa T., Shimada Y., Kawakami K., Onodera K., Nakamura S., Okubo T., J. Catal., 2014, 315, 1—5 |
| 116 | Wheatley P. S., Butler A. R., Crane M. S., Fox S., Xiao B., Rossi A. G., Megson I. L., Morris R. E., J. Am. Chem. Soc., 2006, 128(2), 502—509 |
| 117 | Xing N., Wang X. P., Yu Q., Guo X. W., Chin. J. Catal.,2007, 28(3), 205—209(邢娜, 王新平, 于青, 郭新闻. 催化学报, 2007, 28(3), 205—209) |
| 118 | Despres J., Koebel M., Kröcher O., Elsener M., Wokaun A., Micropor. Mesopor. Mater., 2003, 58(2), 175—183 |
| 119 | Chang X., Lu G., Guo Y., Wang Y., Guo Y., Micropor. Mesopor. Mater., 2013, 165, 113—120 |
| 120 | Zecchina A., Bordiga S., Vitillo J. G., Ricchiardi G., Lamberti C., Spoto G., Bjørgen M., Lillerud K. P., J. Am. Chem. Soc., 2005, 127(17), 6361—6366 |
| 121 | Langmi H. W., Walton A., Al⁃Mamouri M. M., Johnson S. R., Book D., Speight J. D., Edwards P. P., Gameson I., Anderson P. A., Harris I. R., J. Alloys Compd., 2003, 356/357, 710—715 |
| 122 | Ipek B., Pollock R. A., Brown C. M., Uner D., Lobo. R. F., J. Phys. Chem. C, 2018, 122, 540—548 |
| 123 | Niimura S., Fujimori T., Minami D., Hattori Y., Abrams L., Corbin D., Hata K., Kaneko K., J. Am. Chem. Soc., 2012, 134(45), 18483—18486 |
| 124 | Gao L. G., Zhang R. M., Xu X., Truhlar D. G., J. Am. Chem. Soc., 2019, 141(34), 13635—13642 |
| 125 | Zhao X., Wang Y., Li D. S., Bu X., Feng P., Adv. Mater., 2018, 30(37), 1—34 |
| 126 | Shah M. S., Tsapatsis M., Siepmann J. I., Angew. Chem. Int. Ed., 2016, 55(20), 5938—5942 |
| 127 | Sharma P., Song J. S., Han M. H., Cho C. H., Sci. Rep., 2016, 6(22734), 1—26 |
| 128 | Song Z., Huang Y., Xu W. L., Wang L., Bao Y., Li S., Yu M., Sci. Rep., 2015, 5(13981), 1—7 |
| 129 | Bai P., Jeon M. Y., Ren L., Knight C., Deem M. W., Tsapatsis M., Siepmann J. I., Nat. Commun., 2015, 6, 1—9 |
| 130 | Kim J., Lin L. C., Swisher J. A., Haranczyk M., Smit B., J. Am. Chem. Soc., 2012, 134(46), 18940—18943 |
| 131 | Wang L., Liu W., Zhang B., Mod. Chem. Ind., 2018, 38(12), 2—6(王连霞, 刘伟, 张宝泉. 现代化工, 2018, 38(12), 2—6) |
| 132 | Kita H., Horii K., Ohtoshi Y., Tanaka K., Okamoto K. I., J. Mater. Sci. Lett., 1995, 14(3), 206—208 |
| 133 | Caro J., Noack M., Micropor. Mesopor. Mater., 2008, 115(3), 215—233 |
| 134 | Caro J., Noack M., Kölsch P., Adsorption, 2005, 11(3/4), 215—227 |
| 135 | Xu C., Zhou C., Wang S., Huang A., Chinese Chem. Lett., 2019, 30(6), 1204—1206 |
| 136 | Cao Y., Wang M., Xu Z. L., Ma X. H., Xue S. M., ACS Appl. Mater. Interfaces, 2016, 8(38), 25386—25395 |
| 137 | Huang B., Liu Q., Caro J., Huang A., J. Memb. Sci., 2014, 455, 200—206 |
| 138 | Xu K., Jin H., Wang L., Liu Y., Zhou C., Caro J., Huang A., Sep. Sci. Technol.,2018, 53(11), 1741—1751 |
| 139 | Feng X., Zong Z., Elsaidi S. K., Jasinski J. B., Krishna R., Thallapally P. K., Carreon M. A., J. Am. Chem. Soc., 2016, 138(31), 9791—9794 |
| 140 | Zhou M., Korelskiy D., Ye P., Grahn M., Hedlund J., Angew. Chem. Int. Ed., 2014, 53(13), 3492—3495 |
| 141 | Xu X. C., Cheng M. J., Yang W. S., Lin L. W., Science in China(Series B), 1998, 28(3), 247—251(徐晓春, 程谟杰, 杨维慎, 林励吾. 中国科学(B辑), 1998, 28(3), 247—251) |
| 142 | Lai Z., Bonilla G., Diaz I., Nery J. G., Sujaoti K., Amat M. A., Kokkoli E., Terasaki O., Thompson R. W., Tsapatsis M., Vlachos D. G., Science, 2003, 300(5618), 456—460 |
| 143 | Agrawal K. V., Topuz B., Pham T. C. T., Nguyen T. H., Sauer N., Rangnekar N., Zhang H., Narasimharao K., Basahel S. N., Francis L. F., Macosko C. W., Al⁃Thabaiti S., Tsapatsis M., Yoon K. B., Adv. Mater., 2015, 27(21), 3243—3249 |
| 144 | Huang A., Liang F., Steinbach F., Gesing T. M., Caro J., J. Am. Chem. Soc., 2010, 132(7), 2140—2141 |
| [1] | 姜宏斌, 代文臣, 张娆, 徐晓晨, 陈捷, 杨光, 杨凤林. Co3O4/UiO-66@α-Al2O3陶瓷膜对VOCs废气的分离催化性能[J]. 高等学校化学学报, 2022, 43(6): 20220025. |
| [2] | 戴卫, 侯华, 王宝山. 七氟异丁腈负离子结构与反应活性的理论研究[J]. 高等学校化学学报, 2022, 43(6): 20220044. |
| [3] | 郝宏蕾, 孟繁雨, 李若钰, 李迎秋, 贾明君, 张文祥, 袁晓玲. 生物质基氮掺杂多孔炭材料的制备及对水中亚甲基蓝的吸附性能[J]. 高等学校化学学报, 2022, 43(6): 20220055. |
| [4] | 王红宁, 黄丽, 清江, 马腾洲, 蒋伟, 黄维秋, 陈若愚. 香蒲基生物炭的活化及对VOCs吸附的应用[J]. 高等学校化学学报, 2022, 43(4): 20210824. |
| [5] | 孟祥龙, 杨歌, 郭海玲, 刘晨光, 柴永明, 王纯正, 郭永梅. 纳米分子筛的合成及硫化氢吸附性能[J]. 高等学校化学学报, 2022, 43(3): 20210687. |
| [6] | 陈潇禄, 袁珍闫, 仲迎春, 任浩. 机械球磨制备三苯胺基PAF-106s及C2烃吸附性质[J]. 高等学校化学学报, 2022, 43(3): 20210771. |
| [7] | 靳科研, 白璞, 李小龙, 张佳楠, 闫文付. 新型Mg-Al吸附剂去除压水堆核电厂废水中高浓度硼[J]. 高等学校化学学报, 2022, 43(2): 20210516. |
| [8] | 王寿柏, 吴修明, 束辰, 钟敏, 黄卫, 颜德岳. 含叔丁基聚酰亚胺均质膜的气体分离性能[J]. 高等学校化学学报, 2022, 43(11): 20220357. |
| [9] | 谭乐见, 仲宣树, 王锦, 刘宗建, 张爱英, 叶霖, 冯增国. β-环糊精的低临界溶解温度现象及其在有序纳米孔道片晶制备中的应用[J]. 高等学校化学学报, 2022, 43(11): 20220405. |
| [10] | 郑美琪, 毛方琪, 孔祥贵, 段雪. 类水滑石材料在核废水处理领域的应用[J]. 高等学校化学学报, 2022, 43(10): 20220456. |
| [11] | 田晓康, 张青松, 杨舒淋, 白洁, 陈冰洁, 潘杰, 陈莉, 危岩. 微生物发酵诱导多孔材料: 制备方法和应用[J]. 高等学校化学学报, 2022, 43(10): 20220216. |
| [12] | 唐元晖, 李春玉, 林亚凯, 张春晖, 刘泽, 余立新, 王海辉, 王晓琳. 链段刚性对非溶剂致相分离成膜过程影响的耗散粒子动力学模拟[J]. 高等学校化学学报, 2022, 43(10): 20220169. |
| [13] | 张弛, 孙福兴, 朱广山. 双金属同构金属-有机框架材料CAU-21-Al/M的合成、 氮气吸附及复合膜性能[J]. 高等学校化学学报, 2022, 43(1): 20210578. |
| [14] | 马鉴新, 刘晓东, 徐娜, 刘国成, 王秀丽. 一种具有发光传感、 安培传感和染料吸附性能的多功能Zn(II)配位聚合物[J]. 高等学校化学学报, 2022, 43(1): 20210585. |
| [15] | 柳雪广, 杨晓珊, 马菁菁, 刘伟生. 铕基金属有机框架材料从混合染料中选择性分离亚甲基蓝[J]. 高等学校化学学报, 2022, 43(1): 20210715. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||