

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (10): 20220216.doi: 10.7503/cjcu20220216
田晓康1,2, 张青松1,2( ), 杨舒淋1,2, 白洁3, 陈冰洁1,2, 潘杰4, 陈莉1,2, 危岩5
), 杨舒淋1,2, 白洁3, 陈冰洁1,2, 潘杰4, 陈莉1,2, 危岩5
收稿日期:2022-04-06
出版日期:2022-10-10
发布日期:2022-06-11
通讯作者:
张青松
E-mail:zqs8011@163.com
基金资助:
TIAN Xiaokang1,2, ZHANG Qingsong1,2( ), YANG Shulin1,2, BAI Jie3, CHEN Bingjie1,2, PAN Jie4, CHEN Li1,2, WEI Yen5
), YANG Shulin1,2, BAI Jie3, CHEN Bingjie1,2, PAN Jie4, CHEN Li1,2, WEI Yen5
Received:2022-04-06
Online:2022-10-10
Published:2022-06-11
Contact:
ZHANG Qingsong
E-mail:zqs8011@163.com
Supported by:摘要:
微生物发酵作为一种新的制备多孔材料的方式, 将微生物发酵工程与发泡工程有机结合起来, 克服了传统制备方法需要特殊设备、 操作复杂、 后处理繁琐、 化学药品污染和成本昂贵等缺点, 受到了广泛关注.本文基于微生物发酵多孔材料的研究, 围绕多孔材料的定义和多孔水凝胶的分类及制备方式进行总结.针对微生物发酵诱导制备多孔材料的制备方法, 综合评述了该方法在染料吸附、 海水蒸发脱盐、 电磁屏蔽以及制备新型功能性生物材料等方面的应用.最后, 对微生物诱导制备多孔材料的未来发展进行了展望.
中图分类号:
TrendMD:
田晓康, 张青松, 杨舒淋, 白洁, 陈冰洁, 潘杰, 陈莉, 危岩. 微生物发酵诱导多孔材料: 制备方法和应用. 高等学校化学学报, 2022, 43(10): 20220216.
TIAN Xiaokang, ZHANG Qingsong, YANG Shulin, BAI Jie, CHEN Bingjie, PAN Jie, CHEN Li, WEI Yen. Porous Materials Inspired by Microbial Fermentation: Preparation Method and Application. Chem. J. Chinese Universities, 2022, 43(10): 20220216.
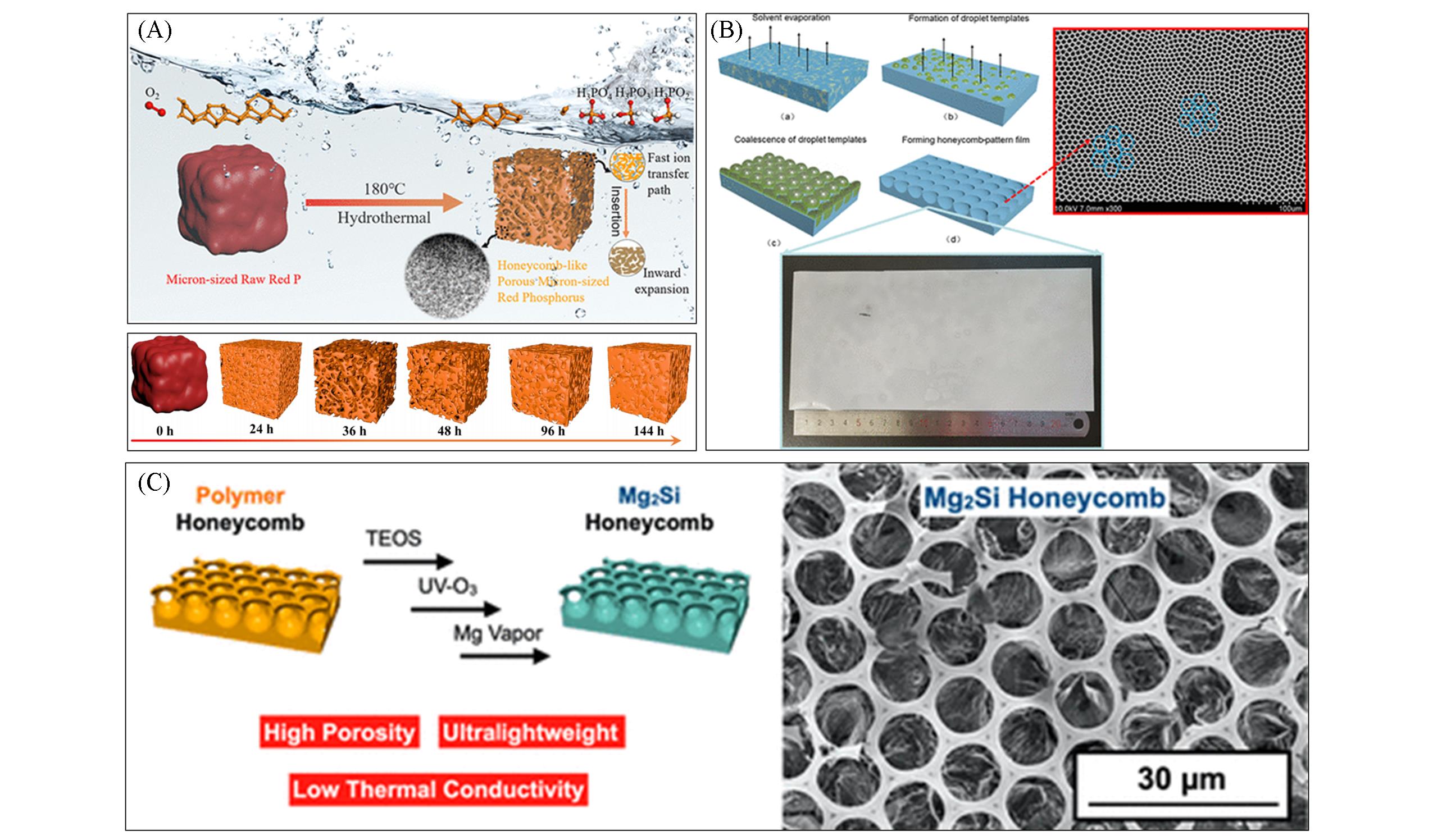
Fig.4 Honeycomb porous structures of different materials(A) A honeycomb porous red phosphorus(HPRP) was synthesized by hydrothermal method using the water suspension of micron-sized red phosphorus particles[11]; (B) polystyrene films with highly ordered honeycomb structures were prepared in normal indoor environment by solvent-induced phase separation[12]; (C) 1,2-polybutadiene honeycomb film was prepared by Mg steam annealing method to form Mg2Si honeycomb structure supported by amorphous carbon[13].(A) Copyright 2021, American Chemical Society; (B) Copyright 2021, American Chemical Society; (C) Copyright 2020, American Chemical Society.
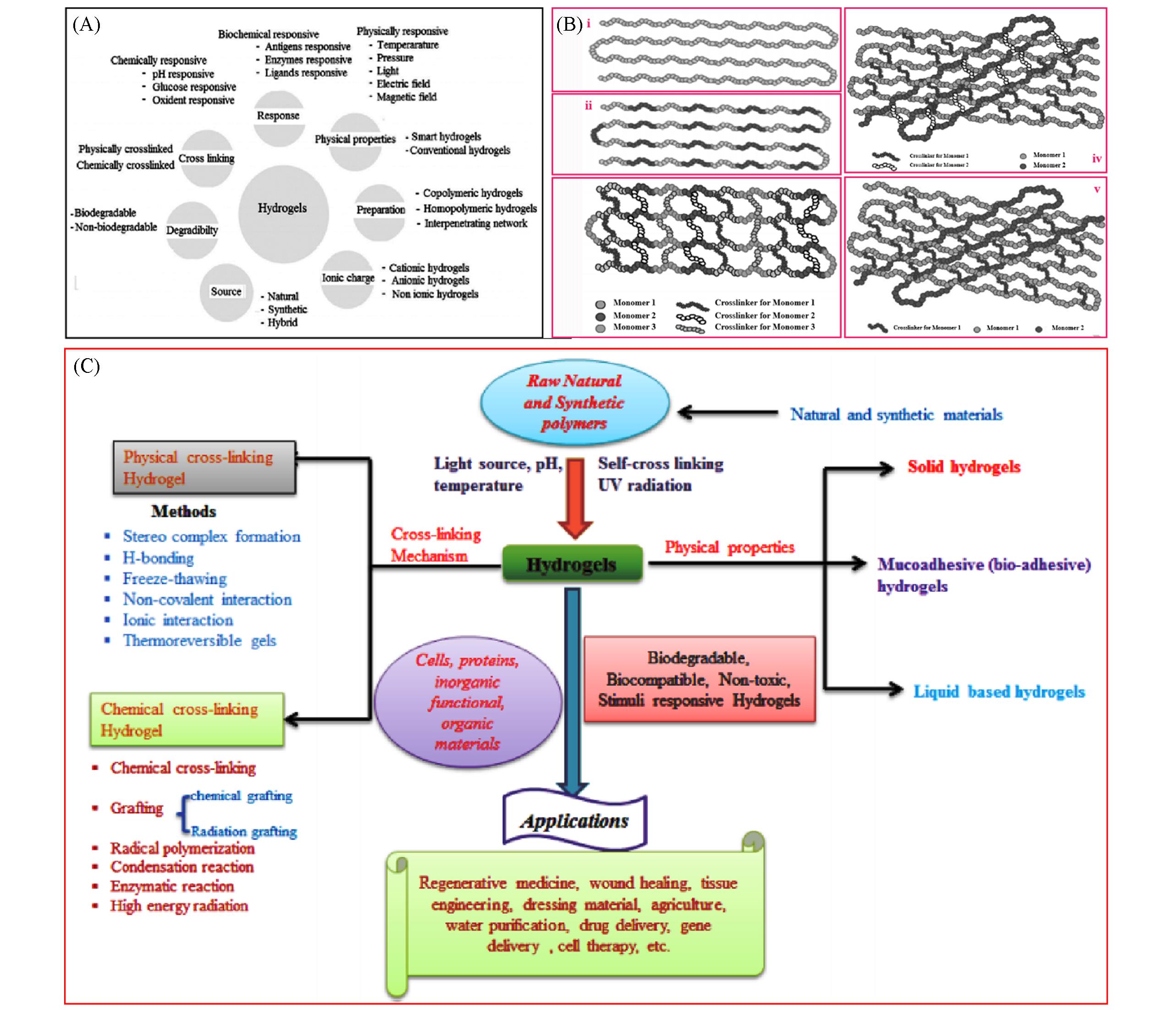
Fig.5 Classification of porous hydrogels in different ways(A) Classification of hydrogels based on the different properties[18]. (B) ⅰ. Homopolymer hydrogels; ⅱ. co-polymer hydrogels; ⅲ. multipolymer hydrogels; ⅳ. interpenetrating polymeric network(IPN) hydrogels; ⅴ. semi-Interpenetrating polymer network(semi-IPN) hydrogels[28]. (C) Hydrogel and their classification[29].(A) Copyright 2015, Elsevier; (B) Copyright 2015, Taylor & Francis; (C) Copyright 2020, Elsevier.
| Methods | Mechanism | Advantage | Disadvantage |
|---|---|---|---|
Freeze? drying[ | The well?free gel absorbs water after deep freezing, followed by vacuum drying to sublimate the solvent, thus leaving the hole. | Wide application, convenient operation, without any chemical solvent or heating, safety and green. | The size of holes depends on the growth form and growth rate of ice crystals at different temperatures, which is not easy to control. |
Phase separation[ | Different temperatures cause phase transformation, so that a polymer?rich phase and a polymer?poor phase are generated in the reaction solution, and phase separation occurs. The pores of the gel appear after the solvent is removed. | Compared with the traditional non?porous gel, the swelling rate is improved by 5 to 6 times, thereby providing advantages for the application of drug transmission, water absorption and sensor. | Poor universality; higher or lower heating rate will result in a closed pore structure, and it is difficult to control the pore size and porosity. |
| Supercritical fluids[ | Supercritical carbon dioxide(SC?CO2) was used to replace organic foaming agent, and foaming was carried out under a certain pressure and temperature. | The gas is non?toxic, stable, cheap and easy to obtain and many other advantages. | The unit is relatively complex and the melt temperature is relatively high (155—170 ℃). The cell size distribution is uniform only when the approp? riate melt temperature is reached. |
| Templating method[ | Regular template was added into the prepolymer solution for polymerization, and the pore size was determined by the template. | Porous gel with uniform distribution can be prepared. The pore size can be adjusted according to the content of surfactant in the emulsion. | When removing the template, a large number of organic solvents need to be used, which will pollute and cause solvent residue, so it is not widely used. |
| Pore?foaming agent[ | In the process of polymerization, the pore forming agent is added, and the gel is soaked in water or acid solution to dissolve the pore forming agent, leaving holes in its original position. | It can improve gel swelling and deswelling properties and improve product mechanical strength. | Processes such as dissolution, water washing, soaking, and removal of porogens are time?consuming and have problems with porogen residues. |
| Foaming[ | Adding gaseous substances to the polymer solution during the reaction, producing gases during gelation and leaving holes in the polymer after the reaction is completed. | The samples are diverse in size, low cost, pollution free and energy efficient. | Gelation was required to be synchronized with the addition of foaming agents to obtain well porous hydrogels, and the reaction conditions had an important influence on the gelation process. |
3D printing[ | Using CAD of 3D digital models, printing using adhesible materials, and many ways such as crosslinking by photocuring or regulation of environmental conditions, achieve rapid shaping of hydrogels. | Quick prototyping greatly reduces the difficulty of forming hydrogels with 3D complex structure. No special moulds and tools are required for the process. | High cost, low manufacturing efficiency, limited scale production, relatively poor accuracy, physical and chemical performance, limited use in actual projects. |
Method induced by microorganism | The MIH constructed by in?situ free radical polymerization and freeze?induced phase separation using CO2 generated from fermentation of microorganism and carbon source as foaming agent. | The microorganism has the advanta?ges of quick reaction, mild conditions, wide sources and low price. | The reproductive capacity of yeast is too strong for yeast to be completely remove from that porous hydrogel. |
Table 1 Comparison of preparation methods of Porous hydrogel
| Methods | Mechanism | Advantage | Disadvantage |
|---|---|---|---|
Freeze? drying[ | The well?free gel absorbs water after deep freezing, followed by vacuum drying to sublimate the solvent, thus leaving the hole. | Wide application, convenient operation, without any chemical solvent or heating, safety and green. | The size of holes depends on the growth form and growth rate of ice crystals at different temperatures, which is not easy to control. |
Phase separation[ | Different temperatures cause phase transformation, so that a polymer?rich phase and a polymer?poor phase are generated in the reaction solution, and phase separation occurs. The pores of the gel appear after the solvent is removed. | Compared with the traditional non?porous gel, the swelling rate is improved by 5 to 6 times, thereby providing advantages for the application of drug transmission, water absorption and sensor. | Poor universality; higher or lower heating rate will result in a closed pore structure, and it is difficult to control the pore size and porosity. |
| Supercritical fluids[ | Supercritical carbon dioxide(SC?CO2) was used to replace organic foaming agent, and foaming was carried out under a certain pressure and temperature. | The gas is non?toxic, stable, cheap and easy to obtain and many other advantages. | The unit is relatively complex and the melt temperature is relatively high (155—170 ℃). The cell size distribution is uniform only when the approp? riate melt temperature is reached. |
| Templating method[ | Regular template was added into the prepolymer solution for polymerization, and the pore size was determined by the template. | Porous gel with uniform distribution can be prepared. The pore size can be adjusted according to the content of surfactant in the emulsion. | When removing the template, a large number of organic solvents need to be used, which will pollute and cause solvent residue, so it is not widely used. |
| Pore?foaming agent[ | In the process of polymerization, the pore forming agent is added, and the gel is soaked in water or acid solution to dissolve the pore forming agent, leaving holes in its original position. | It can improve gel swelling and deswelling properties and improve product mechanical strength. | Processes such as dissolution, water washing, soaking, and removal of porogens are time?consuming and have problems with porogen residues. |
| Foaming[ | Adding gaseous substances to the polymer solution during the reaction, producing gases during gelation and leaving holes in the polymer after the reaction is completed. | The samples are diverse in size, low cost, pollution free and energy efficient. | Gelation was required to be synchronized with the addition of foaming agents to obtain well porous hydrogels, and the reaction conditions had an important influence on the gelation process. |
3D printing[ | Using CAD of 3D digital models, printing using adhesible materials, and many ways such as crosslinking by photocuring or regulation of environmental conditions, achieve rapid shaping of hydrogels. | Quick prototyping greatly reduces the difficulty of forming hydrogels with 3D complex structure. No special moulds and tools are required for the process. | High cost, low manufacturing efficiency, limited scale production, relatively poor accuracy, physical and chemical performance, limited use in actual projects. |
Method induced by microorganism | The MIH constructed by in?situ free radical polymerization and freeze?induced phase separation using CO2 generated from fermentation of microorganism and carbon source as foaming agent. | The microorganism has the advanta?ges of quick reaction, mild conditions, wide sources and low price. | The reproductive capacity of yeast is too strong for yeast to be completely remove from that porous hydrogel. |
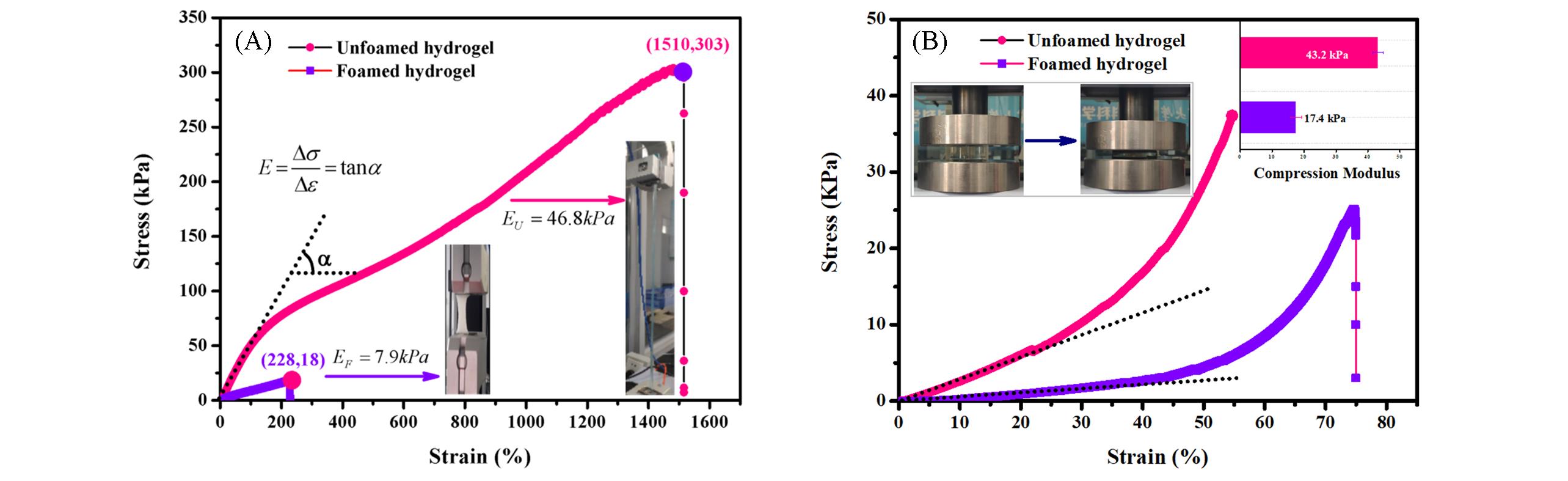
Fig.6 Mechanical strength of hydrogel before and after foaming[48](A) Stress-strain curves of tensile properties; (B) stress-strain curves of compression properties.
| Type | Pore structure parameter | Ref. | |||
|---|---|---|---|---|---|
| Porosity(%) | Density/(g?cm?3) | Specific surface area/(m2?g?1) | Pore size/μm | ||
| GO/CS/PVA composite sponge | 67.72—78.31 | 0.215—0.319 | — | — | [ |
| P(AAEA?co?AMPS) hydrogel | — | — | — | 30—120 | [ |
| Polyacrylamide(PAM) hydrogel | — | 0.33—0.76 | — | 1.04—2.48 | [ |
| Chitosan and alginate scaffolds | ~90 | — | — | 60—150 | [ |
| PEG?grafted SPH | — | — | — | 100—250 | [ |
| 3D Porous graphene oxide | — | 0.01—0.028 | ~206.8 | — | [ |
| Air?dryable graphene hydrogels | — | 0.0063—0.063 | — | 18—95 | [ |
| PVA/C porous hydrogels | ~54.1 | 0.3—0.85 | — | ~21.34 | [ |
| Chitosan?agarose cryogels | 80—85 | — | — | 85—100 | [ |
| PEG hydrogel scaffolds | 55—84 | — | — | 53—180 | [ |
| Supermacroporous cryogel | — | — | 84.9—194.9 | 10—200 | [ |
| Cryogels | — | — | ~192 | 10—100 | [ |
| Carbon aerogels | — | ~0.225 | 550—660 | 0.03—0.05 | [ |
| Hierarchical porous chitosan?carbon | 80—94 | — | 667—1743 | 0.00268—0.00358 | [ |
| Nanocomposite gels | — | — | ~17.7 | ~0.0033 | [ |
| Lightweight ceramic foam | 74.1—82.4 | 0.38—0.56 | — | 1500—5600 | [ |
Table 2 Pore structure parameters(porosity, density, specific surface area and pore size) of porous hydrogel
| Type | Pore structure parameter | Ref. | |||
|---|---|---|---|---|---|
| Porosity(%) | Density/(g?cm?3) | Specific surface area/(m2?g?1) | Pore size/μm | ||
| GO/CS/PVA composite sponge | 67.72—78.31 | 0.215—0.319 | — | — | [ |
| P(AAEA?co?AMPS) hydrogel | — | — | — | 30—120 | [ |
| Polyacrylamide(PAM) hydrogel | — | 0.33—0.76 | — | 1.04—2.48 | [ |
| Chitosan and alginate scaffolds | ~90 | — | — | 60—150 | [ |
| PEG?grafted SPH | — | — | — | 100—250 | [ |
| 3D Porous graphene oxide | — | 0.01—0.028 | ~206.8 | — | [ |
| Air?dryable graphene hydrogels | — | 0.0063—0.063 | — | 18—95 | [ |
| PVA/C porous hydrogels | ~54.1 | 0.3—0.85 | — | ~21.34 | [ |
| Chitosan?agarose cryogels | 80—85 | — | — | 85—100 | [ |
| PEG hydrogel scaffolds | 55—84 | — | — | 53—180 | [ |
| Supermacroporous cryogel | — | — | 84.9—194.9 | 10—200 | [ |
| Cryogels | — | — | ~192 | 10—100 | [ |
| Carbon aerogels | — | ~0.225 | 550—660 | 0.03—0.05 | [ |
| Hierarchical porous chitosan?carbon | 80—94 | — | 667—1743 | 0.00268—0.00358 | [ |
| Nanocomposite gels | — | — | ~17.7 | ~0.0033 | [ |
| Lightweight ceramic foam | 74.1—82.4 | 0.38—0.56 | — | 1500—5600 | [ |
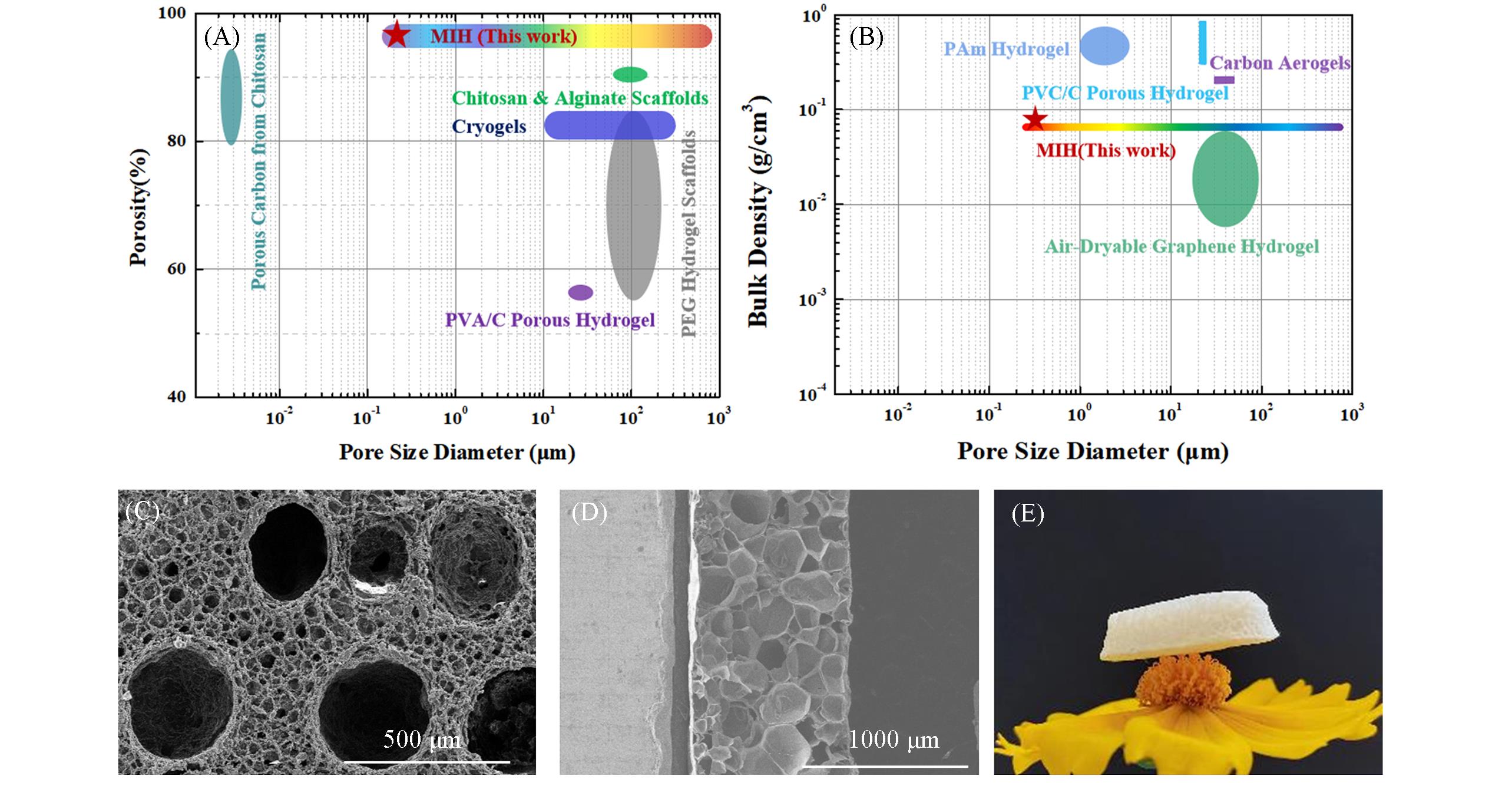
Fig.7 Comparison of pore structure parameters between porous gel materials inspired by Microbial Fermentation and other porous materials[48](A) Porosity vs. pore size; (B) bulk density vs. pore size; (C) scanning electron microscope surface morphology display of MIH; (D) scanning electron microscope section of MIH; (E) figure of low density MIH on stamens.
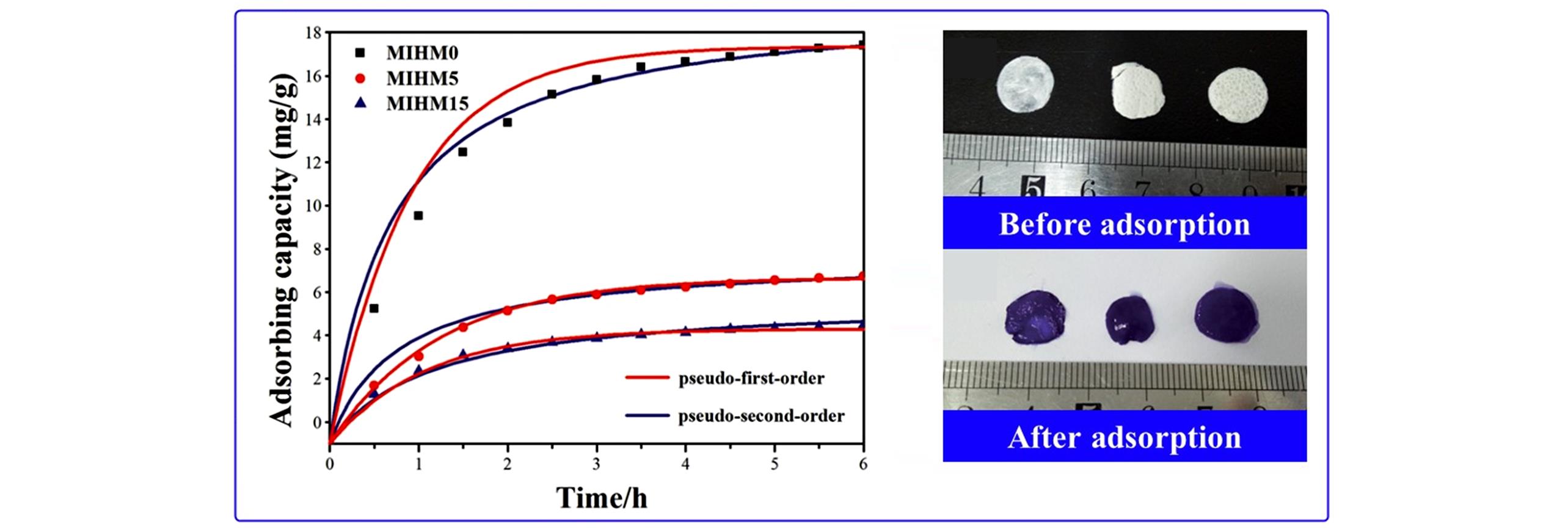
Fig.8 Effect of contact time on CV adsorption capacity(10 mg/L) of prepared MIHM hydrogel membranes[70]Copyright 2018, the Royal Society of Chemistry.
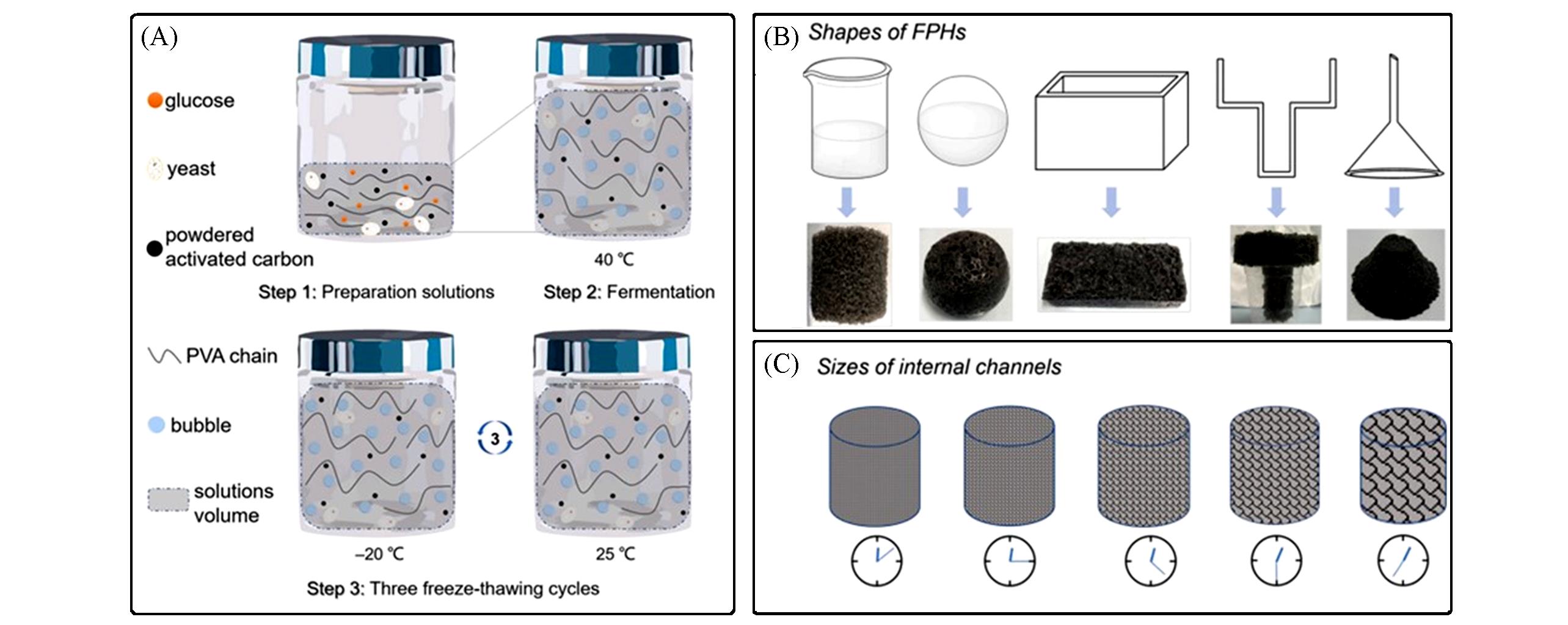
Fig.9 Schematic images of preparation process of FPHs(A), castability of FPHs using different shape of molds(B) and time?regulated internal?channel size of FPHs(C)[56]Copyright 2020, Elsevier.
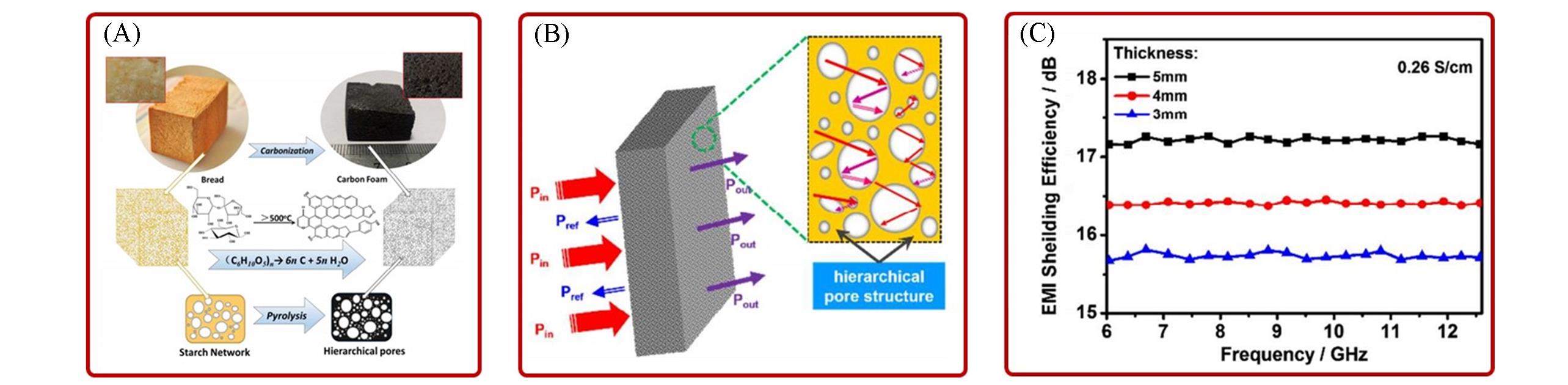
Fig.10 Raw diagram for the fabrication of carbon foam derived from bread(A), schematic representation of microwave transfer across the hierarchical pore structure of CF(B) and EMI shielding efficiency of carbon foam(carbonized at 1000 ℃)(C)[76]Copyright 2016, American Chemical Society.
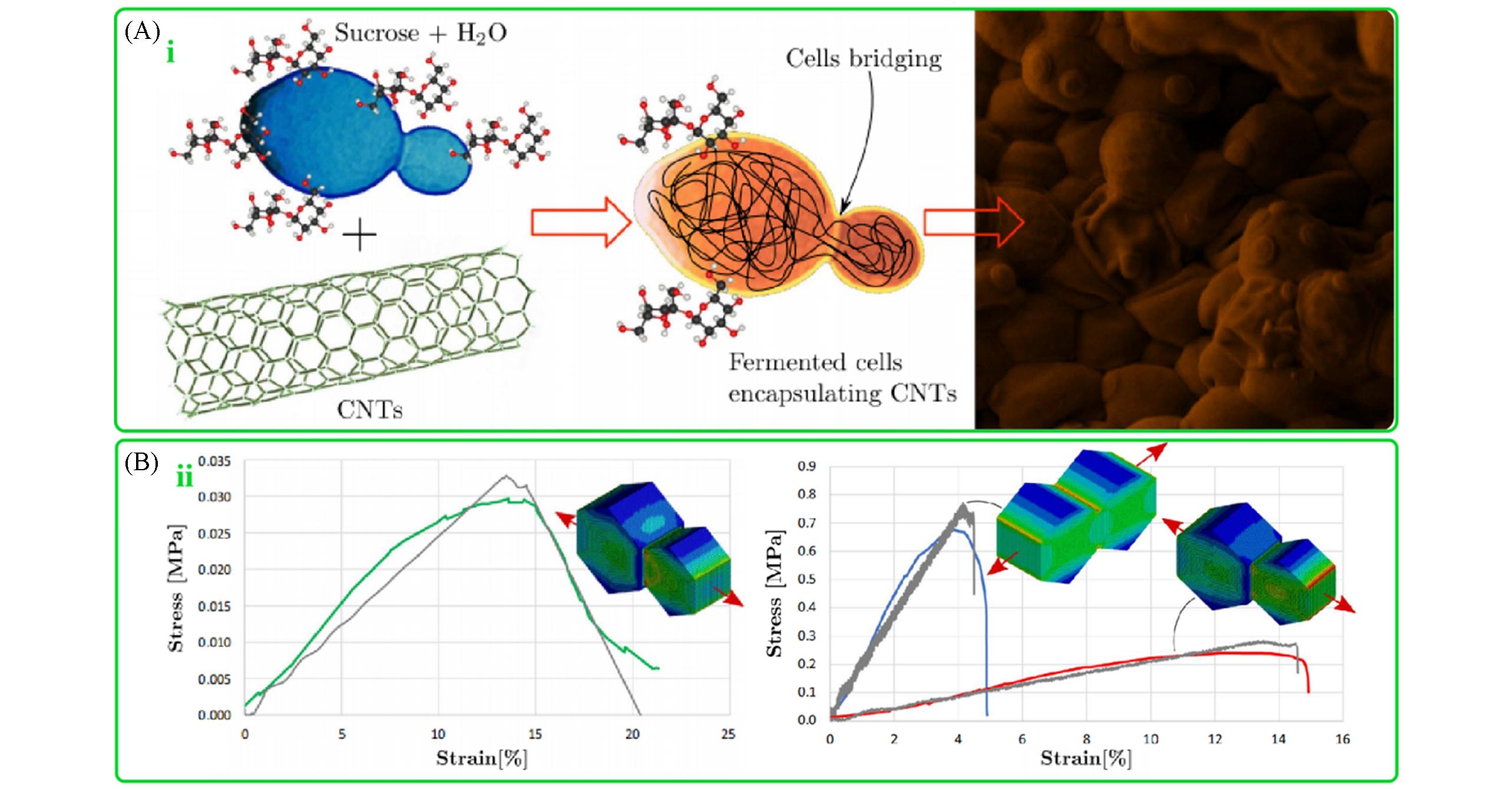
Fig.11 Schematic representation and appearance of the fermentation?mediated assembly of carbon nanotubes and yeast cells(A) and stress?strain curves obtained from tensile tests on fermented yeast sample(green curve) and yeast/CNT composites prepared before fermentation(blue curve) and after fermentation(red curve)(B)[77]Copyright 2016, Springer Nature.
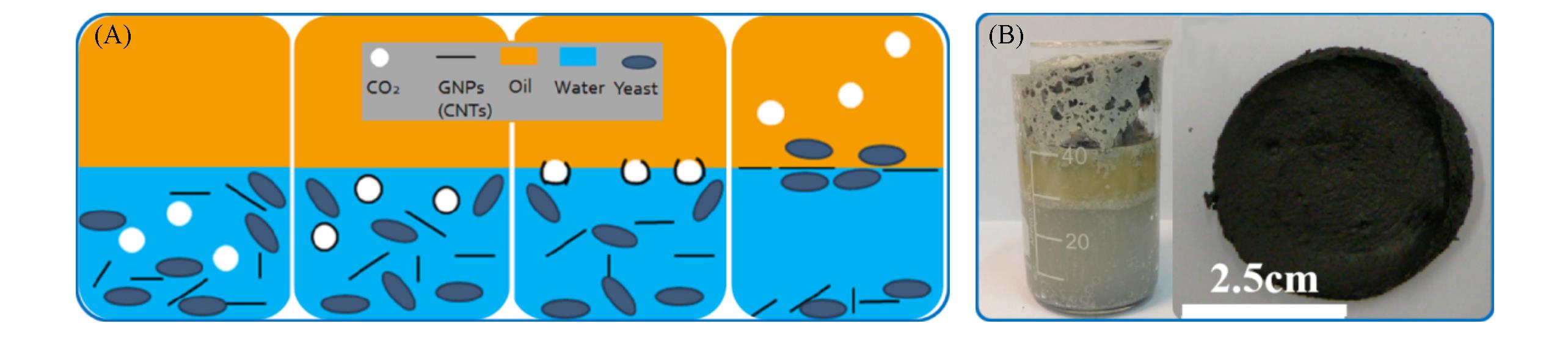
Fig.12 Schematic illustration of the assembly process of the GNPs(CNTs)(A) and optical microscope image of oil?water interface prepared with PEEK particles and GNPs(B)[78]Copyright 2017, American Chemical Society.
| 1 | Zou C. Y., Mod. Chem. Res., 2018, (3), 96—97 |
| 邹春悦. 当代化工研究, 2018, (3), 96—97 | |
| 2 | Tang H. P., Zhang Z. D., Rar. Met. Mater. Eng., 1997, (1), 1—6 |
| 汤慧平, 张正德. 稀有金属材料与工程, 1997, (1), 1—6 | |
| 3 | Davis M. E., Nature, 2002, 417, 813—821 |
| 4 | Ishizaki K., Komarneni S., Nanko M., Porous Mater., 1998, (4), 202—224 |
| 5 | Wei Y. N., Yang X. C., Sun H. K., Mater. Rev., 2006, 20(1), 10—13 |
| 韦亚南, 杨修春, 孙海阔. 材料导报, 2006, 20(1), 10—13 | |
| 6 | Gibson L. J., Ashby M. F., Cellular Solids: Structure and Properties, Cambridge University Press, London, 1997, 175—231 |
| 7 | Li J., Liu L. P., Chen H., Huang Z., Chinese J. Tissue Eng. Res., 2010, 14(9), 1521—1524 |
| 李捷, 刘玲萍, 陈宏, 黄震. 中国组织工程研究与临床康复, 2010, 14(9), 1521—1524 | |
| 8 | Liu G., Wang Y., Zhang Y., Xu X., Qi L., Wang H., Sci. Total. Environ., 2019, 652(20), 1366—1374 |
| 9 | Xue R. M., Donovan A., Zhang H. T., Ma Y. F., Adams C., Yang J., Hua B., Inniss E., Eichhoz T., Shi H. L., J. Environ. Sci., 2018, 64(2), 82—91 |
| 10 | Wan C. L., Ding S., Zhang C., Tan X. J., Zou W. G., Liu X., Yang X., Sep. Purif. Technol., 2017, 180, 1—12 |
| 11 | Zhu J. L., Liu Z. G., Wang W., Yue L. G., Li W. W., Zhang H. Y., Zhao L. G., Zheng H., Wang J. B., Li Y. Y., ACS Nano, 2021, 15(1), 1880—1892 |
| 12 | Cheng Q., Lin L. G., Deng X. S., Zheng T. T., Wang Q., Gao Y. X., Zhai X. F., Yang J., Ma W. S., Li X. Y., Zhang Y. Z., Ind. Eng. Chem. Res., 2021, 60(2), 898—907 |
| 13 | Yabu H., Matsuo Y., Yamada T., Maeda H., Matsui J., Chem. Mater., 2020, 32(23), 10176—10183 |
| 14 | Yuk H., Lu B. Y., Zhao X. H., Chem. Soc. Rev., 2019, 48(6), 1642—1667 |
| 15 | Zhang Y. S., Khademhosseini A., Science, 2017, 356(6337), 3627 |
| 16 | Caccavo D., Cascone S., Lamberti G., Barba A. A., Chem. Soc. Rev., 2018, 47(7), 2357—2373 |
| 17 | Thoniyot P., Tan M. J., Karim A. A., Young D. J., Loh X. J., Adv. Sci., 2015, 2(1/2), 1400010 |
| 18 | Ullah F., Othman M. B. H., Javed F., Ahmad Z., Akil H. M., Mater. Sci. Eng. C, 2015, 57, 414—433 |
| 19 | Patel A., Mequanint K., Hydrogel Biomaterials, InTech, London, 2007 |
| 20 | Laftah W. A., Hashim S., Ibrahim A. N., Polym.⁃Plast. Technol. Eng., 2011, 50(14), 1475—1486 |
| 21 | Misra G P., Gil E. S., Tao L. L., In the Biomedical Arena, Polymer Grafting and Crosslinking, Wiley, New York, 2009, 145—175 |
| 22 | Kopeek J., Yang J. Y., Acta Biomater., 2009, 5(3), 805—816 |
| 23 | Ahmed E. M., J. Adv. Res., 2015, 6(2), 105—121 |
| 24 | Zhao W., Xing J., Yang C., Liu Y. Y., Fu J., J. Chem. Technol. Biotechnol., 2013, 88(3), 327—339 |
| 25 | Iizawa T., Takea H., Maruta M., Ishido T., Sakohara S., J. Appl. Polym. Sci., 2010, 104(2), 842—850 |
| 26 | Yang L. B., Chu J. S., Fix J. A., Int. J. Pharm., 2002, 235(1/2), 1—15 |
| 27 | Zhai M. L., Li J., Yi M., Ha H. F., Radiat. Phys. Chem., 2000, 58(4), 397—400 |
| 28 | Singhal R., Gupta K., Polym.⁃Plast. Technol. Eng., 2015, 55(1), 55—70 |
| 29 | Sharma S., Tiwari S., Int. J. Biol. Macromol., 2020, 162, 737—747 |
| 30 | Dong R. J., Pang Y., Su Y., Zhu X. Y., Biomater. Sci., 2015, 3(7), 937—954 |
| 31 | Xu X. F., Shen Y., Wang W., Sun C. M., Li C., Xiong Y. R., Tu J. S., Eur. J. Pharm. Biopharm., 2014, 88(3), 998—1004 |
| 32 | Loh X. J., Nguyen V. N., Kuo N., Li J., J. Mater. Chem., 2011, 21(7), 2246—2254 |
| 33 | Ho M. H., Kuo P. Y., Hsieh H. J., Hsien T. Y., Hou L. T., Lai J. Y., Wang D. M., Biomaterials, 2004, 25(1), 129—138 |
| 34 | Yuan J., Yi C. Q., Jiang H. Q., Liu F., Cheng G. J., ACS Appl. Polym. Mater, 2021, 3(2), 699—709 |
| 35 | Owusu⁃Nkwantabisah S., Gillmor J., ACS Appl. Mater. Interfaces, 2020, 12(40), 45657—45664 |
| 36 | Sahiner N., Prog. Polym. Sci., 2013, 38(9), 1329—1356 |
| 37 | Liu P. F., Peng Z., Zhang Q. S., React. Funct. Polym., 2017, 117, 43—51 |
| 38 | Guo J., Jin Y. C., Yang X. Q., Yu S. J., Yin S. W., Qi J. R., Food Hydrocolloids, 2013, 31(2), 220—260 |
| 39 | Leng Q., Lin J. M., Wu J. H., Xu K. Q., Huang M. L., Mater. Rev., 2011, 25(20), 28—31 |
| 冷晴, 林建明, 吴季怀, 许开卿, 黄妙良. 材料导报, 2011, 25(20), 28—31 | |
| 40 | Kabiri K., Omidian H., Zohuriaan⁃Mehr M. J., Polym. Int., 2003, 52(7), 1158—1164 |
| 41 | Qiao L. F., Li N., Luo L., He J. T., Lin Y., Li J. J., Yu L. M., Guo C., Murto P., Xu X. F., J. Mater. Chem. A, 2021, 9(15), 9692—9705 |
| 42 | Hinton T. J., Jallerat Q., Palchesko R. N., Park J. H., Grodzicki M. S., Shue H. J., Ramadan M. H., Sci. Adv., 2015, 1(9), e1500758 |
| 43 | Hong S., Sycks D., Cham H. F., Lin S., Lopez G. P., Guilak F., Leong K. W., Zhao X., Adv. Mater., 2015, 27(27), 4034—4034 |
| 44 | Zhu Z. J., Ng D. W. H., Park H. S., Mcalpine M. C., Nat. Rev. Mater., 2020, 6(1), 27—47 |
| 45 | Chen J., Park H., Park K., J. Biomed. Mater. Res., 2015, 44(1), 53—62 |
| 46 | Chen J., Park K., J. Control. Release, 2000, 65(1/2), 73—82 |
| 47 | Yang S. C., Park K. N., Rocca J. G., J. Bioact. Compat. Polym., 2004, 19(2), 81—100 |
| 48 | Tian X. K., Response Surface Optimization and Structural Evolution of Microorganism Inspired Hydrogel Induced by Stress, Tiangong University, Tianjin, 2021 |
| 田晓康. 微生物发酵水凝胶的响应面优化和力诱导下孔结构的演化, 天津: 天津工业大学, 2021 | |
| 49 | Xu X. Q., Tian M. W., Xi X. L., Zhu S. F., Qu L. J., China Dye. Finish., 2015, 41(14), 6—10 |
| 许晓琪, 田明伟, 袭祥龙, 朱士风, 曲丽君. 印染, 2015, 41(14), 6—10 | |
| 50 | Huang B. Y., Lin S. B., Zhang X., Li Y. L., Acta Polym. Sin., 2010, 10(5), 594—600 |
| 黄铂扬, 林松柏, 张弦, 李云龙. 高分子学报, 2010, 10(5), 594—600 | |
| 51 | Feng Q., Lu G. D., Liu Y. Q., Yan Q. Z., Ge C. C., Acta Polym. Sin., 2009, 1(7), 674—679 |
| 冯巧, 卢国冬, 刘玉琼, 燕青芝, 葛昌纯. 高分子学报, 2009, 1(7), 674—679 | |
| 52 | Ho M. H., Kuo P. Y., Hsieh H. J., Hsien T. Y., Hou L. T., Lai J. Y., Wang D. M., Biomaterials, 2004, 25(1), 129—138 |
| 53 | Huh K. M., Baek N., Park K., J. Bioact. Compat. Polym., 2005, 20(3), 231—243 |
| 54 | Yan H. Q., Huang D. G., Chen X. Q., Liu H. F., Feng Y. H., Zhao Z. D., Dai Z. H., Zhang X. Q., Lin Q., Polym. Bull., 2018, 75(3), 985—1000 |
| 55 | Yang H. S., Li Z. L., Sun G. Q., Jin X. T., Lu B., Zhang P. P., Lin T. Y., Qu L., Adv. Funct. Mater., 2019, 29(26), 1901917 |
| 56 | Liang X. C., Zhang X. J., Huang Q. C., Zhang H., Liu C. K., Liu Y. Z., Sol. Energy, 2020, 208, 778—786 |
| 57 | Bhat S., Tripathi A., Kumar A., J. R. Soc. Interface, 2011, 8(57), 540—554 |
| 58 | Lanasa S. M., Hoffecker I. T., Bryant S. J., J. Biomed. Mater. Res., Part B, 2011, 96B(2), 294—302 |
| 59 | Ingavle G. C., Baillie L. W. J., Zheng Y., Lis E. K., Savina I. N., Howell C. A., Mikhalosky S. V., Sandeman S. R., Biomaterials, 2015, 50(1), 140—153 |
| 60 | Hixon K. R., Lu T., Sell S. A., Acta Biomater., 2017, 62, 29—41 |
| 61 | Wu D. C., Fu R. W., Zhang S. T., Dresselhaus M. S., Dresselhaus G., Carbon, 2004, 42(10), 2033—2039 |
| 62 | Luo M., Wang X., Meng T. T., Yang P., Zhu Z. Q., Min H. H., Chen M. Z., Chen W. M., Zhou X. Y., Int. J. Biol. Macromol., 2020, 146, 453—461 |
| 63 | Kazutoshi H., Shoichi S., Satoshi T., Langmuir, 2018, 34(29), 8480—8488 |
| 64 | Chen J. H., Liu P. S., Sun J. X., Ceram. Int., 2020, 46(14), 22699—22708 |
| 65 | Zhang Q. S., Chen B. J., Tao L., Yan M. Y., Chen L., Wei Y., RSC Adv., 2014, 4(61), 32475—32481 |
| 66 | Chen B. J., Zhang S. H., Zhang Q. S., Mu Q. F., Deng L. L., Chen L., Wei Y., Tao L., Zhang X. Y., Wang K., RSC Adv., 2015, 5(112), 91937—91945 |
| 67 | Zhang M., Yang Y., Li C. T., Li B. J., Liu Z. Y., Eng. Plast. Appl., 2016, 44(11), 22—26 |
| 张敏, 杨鱼, 李成涛, 李八军, 刘智勇. 工程塑料应用, 2016, 44(11), 22—26 | |
| 68 | Shi X. N., Chen H., Zhang H. B., Dai H. X., Duan G. J., Acta Mater. Compo. Sin., 2018, 35(6), 1386—1394 |
| 施小宁, 陈晖, 张浩波, 戴红霞, 段国建. 复合材料学报, 2018, 35(6), 1386—1394 | |
| 69 | Nguyen P. Q., Courchesne N. M. D., Duraj⁃Thatte A., Praveschotinunt P., Joshi N. S., Adv. Mater., 2018, 30(19), 1870134 |
| 70 | Xing L. J., Li Z. G., Zhang Q. S., Zhang Y. X., Liu P. F., Zhang K. L., RSC Adv., 2018, 8(5), 2622—2631 |
| 71 | Wan Y., Zhang M., Weng Y. X., Li C. T., J. Chem. Ind. Eng., 2020, (4), 1828—1835 |
| 万豫, 张敏, 翁云宣, 李成涛. 化工学报, 2020, (4), 1828—1835 | |
| 72 | Shin Y. S., Liu J., Wang L. Q., Nie Z. M., Samuels W. D., Fryxell G. E., Exarhos G. J., Angew. Chem. Int. Ed., 2000, 39(15), 2702—2707 |
| 73 | Chen M. L., Zhu L., Dong Y. C., Li L. L., Liu J., ACS Sustainable Chem. Eng., 2016, 4(4), 2098—2106 |
| 74 | Luo W., Wei J., Deng Y. H., Li Y. H., Wang L. J., Zhao T., Jiang W., Mesoporous Mater., 2017, 32(1), 1—10 |
| 75 | Chen C. C., Chen H. R., Ye Z. Q., Shi J. L., Yu J. C., Acta Chim. Sin., 2012, 70(4), 423—428 |
| 76 | Yuan Y., Ding Y. J., Wang C. H., Xu F., Lin Z. S., Qin Y. Y., Li Y., Yang M. L., He X. D., Peng Q. Y., ACS Appl. Mater. Interfaces, 2016, 8(26), 16852—16861 |
| 77 | Valentini L., Bon S. B., Signetti S., Tripathi M., Iacob E., Pugno N. M., Sci. Rep., 2016, 6, 27031 |
| 78 | Valentini L., Bon S. B., Pugno N. M., ACS Appl. Mater. Interfaces, 2017, 27(24), 1606526 |
| [1] | 李祎頔, 田晓春, 李俊鹏, 陈立香, 赵峰. 半导体-微生物界面电子传递及其在环境领域的应用[J]. 高等学校化学学报, 2022, 43(6): 20220089. |
| [2] | 马鉴新, 刘晓东, 徐娜, 刘国成, 王秀丽. 一种具有发光传感、 安培传感和染料吸附性能的多功能Zn(II)配位聚合物[J]. 高等学校化学学报, 2022, 43(1): 20210585. |
| [3] | 高晓乐, 王家信, 李志芳, 李艳春, 杨冬花. 复合材料NiOx-ZSM-5的制备及微生物电解池催化析氢性能[J]. 高等学校化学学报, 2021, 42(9): 2886. |
| [4] | 余丽莎, 李丹, 肖丽萍, 范杰. 沸石与蛋白质的相互作用及生物医用功能[J]. 高等学校化学学报, 2021, 42(1): 311. |
| [5] | 华涛, 李胜男, 李凤祥, 王浩楠. 生物电化学系统降解多环芳烃萘及微生物群落研究[J]. 高等学校化学学报, 2019, 40(9): 1964. |
| [6] | 于承鑫, 刘洋洋, 张霞. Cu-BTC/PVDF杂化膜的原位掺杂制备及增强的染料吸附性能[J]. 高等学校化学学报, 2018, 39(7): 1384. |
| [7] | 代红艳, 杨慧敏, 刘宪, 简选, 郭敏敏, 曹乐乐, 梁镇海. MoS2/石墨烯复合阴极材料的制备及微生物电解池催化产氢性能[J]. 高等学校化学学报, 2018, 39(2): 351. |
| [8] | 韩瑞霞, 吕继涛, 张淑贞. 一种适用于复杂异相体系中羟基自由基定量检测的探针分子—香豆素[J]. 高等学校化学学报, 2018, 39(12): 2658. |
| [9] | 鲁冠秀, 刘庆文, 孔祥怡, 丛乐乐, 宋岩, 赵晴, 杨清彪, 李耀先. PVA/GA/β-CD纳米纤维的制备及染料吸附性能[J]. 高等学校化学学报, 2017, 38(8): 1465. |
| [10] | 马晓丹, 张志明, 于良民. 天然海水中聚吡咯膜的防微生物附着及防腐蚀性能[J]. 高等学校化学学报, 2016, 37(2): 373. |
| [11] | 付融冰, 杨兰琴, 冯雷雨, 郭伟. 氮掺杂石墨烯的一步法低温合成及用作微生物燃料电池阴极催化剂的产电特性[J]. 高等学校化学学报, 2014, 35(4): 825. |
| [12] | 翁强, 陈沛, 赵凤起, 高红旭, 陈新兵, 安忠维. 多级孔无铝Beta分子筛的合成与表征[J]. 高等学校化学学报, 2014, 35(2): 215. |
| [13] | 曹占平, 张景丽, 张宏伟. 电生物还原降解二氯酚的动力学特性及功能菌解析[J]. 高等学校化学学报, 2013, 34(4): 964. |
| [14] | 王井玉, 黄卫民, 王璇, 林海波, 陆海彦. 生物膜电极在以苯酚为燃料的微生物燃料电池中的应用[J]. 高等学校化学学报, 2013, 34(4): 975. |
| [15] | 陈炳鹏 王卓鹏 柳菁菁 王金成 于吉红. 分子筛在医学领域的应用及作用机制[J]. 高等学校化学学报, 2011, 32(3): 485. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||