

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (10): 20220169.doi: 10.7503/cjcu20220169
唐元晖1,2, 李春玉1,2, 林亚凯2( ), 张春晖1, 刘泽1, 余立新2, 王海辉2, 王晓琳2
), 张春晖1, 刘泽1, 余立新2, 王海辉2, 王晓琳2
收稿日期:2022-03-20
出版日期:2022-10-10
发布日期:2022-04-26
通讯作者:
林亚凯
E-mail:yk_lin@tsinghua.edu.cn
基金资助:
TANG Yuanhui1,2, LI Chunyu1,2, LIN Yakai2( ), ZHANG Chunhui1, LIU Ze1, YU Lixin2, WANG Haihui2, WANG Xiaolin2
), ZHANG Chunhui1, LIU Ze1, YU Lixin2, WANG Haihui2, WANG Xiaolin2
Received:2022-03-20
Online:2022-10-10
Published:2022-04-26
Contact:
LIN Yakai
E-mail:yk_lin@tsinghua.edu.cn
Supported by:摘要:
通过分子动力学(MD)模拟映射方法构建了符合聚醚砜(PES)刚性结构的耗散粒子动力学(DPD)简谐力场, 并研究了PES链段刚性对PES/N-甲基-2-吡咯烷酮(NMP)/水体系非溶剂致相分离(NIPS)过程的影响. 结果表明, 由于非溶剂和溶剂在两相界面上发生的质量交换, 导致在相界面处PES链段发生堆积, 形成了薄而致密的聚合物表层, 在PES溶液内部, 由于非溶剂的侵入导致体系发生了旋节相分离, 从而在整体上得到了明显的非对称结构; 同时, PES链段刚性的提升能够明显加快体系的相分离速度, 导致相界面处的PES薄层形成得更加快速, 薄层更加致密、 孔径更小, 而对内部的疏松结构影响较小; 此外, 结合不同力场下聚合物浓度对相分离过程的影响可以发现, 不同PES浓度下, 链段刚性的提升对相分离过程的特征和演变趋势没有造成根本性的影响, 与经典的弹簧力场的模拟结果在整体趋势上有相似性. 研究结果表明, 简谐力场能提升PES链段的刚性, 从而能更真实地模拟实际体系的非溶剂相分离法成膜过程.
中图分类号:
TrendMD:
唐元晖, 李春玉, 林亚凯, 张春晖, 刘泽, 余立新, 王海辉, 王晓琳. 链段刚性对非溶剂致相分离成膜过程影响的耗散粒子动力学模拟. 高等学校化学学报, 2022, 43(10): 20220169.
TANG Yuanhui, LI Chunyu, LIN Yakai, ZHANG Chunhui, LIU Ze, YU Lixin, WANG Haihui, WANG Xiaolin. Dissipative Particle Dynamics Simulation of the Effect of Polymer Chain Rigidity on Membranes Formation by Nonsolvent Induced Phase Separation Process. Chem. J. Chinese Universities, 2022, 43(10): 20220169.
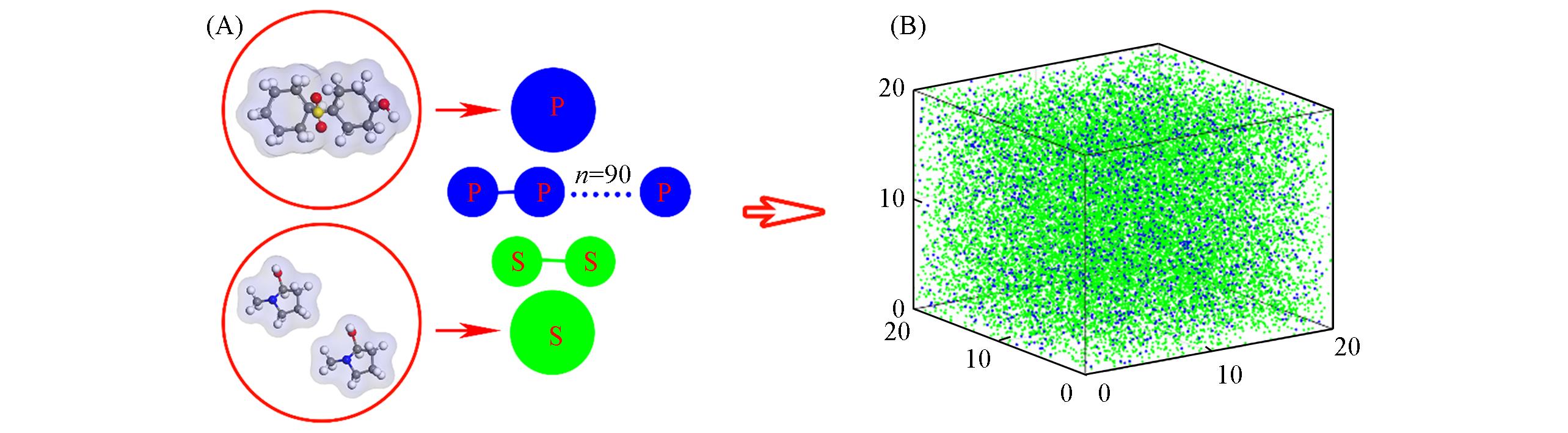
Fig.3 Schematics of the coarse?graining from the molecular level to the DPD particles(A) and a snapshot of the DPD equilibrium system of the PES/NMP solution(B)(A) P and S denote PES and NMP, respectively.
| Method | Bond length | Bond angle | ||
|---|---|---|---|---|
| Mean value/nm | Variance | Mean value/(°) | Variance | |
| MD | 0.945 | 1.35 | 116.28 | 53.59 |
| DPD | 0.939 | 3.43 | 116.69 | 59.23 |
Table 1 Mean and variance of bond lengths and angles in MD and DPD simulation
| Method | Bond length | Bond angle | ||
|---|---|---|---|---|
| Mean value/nm | Variance | Mean value/(°) | Variance | |
| MD | 0.945 | 1.35 | 116.28 | 53.59 |
| DPD | 0.939 | 3.43 | 116.69 | 59.23 |
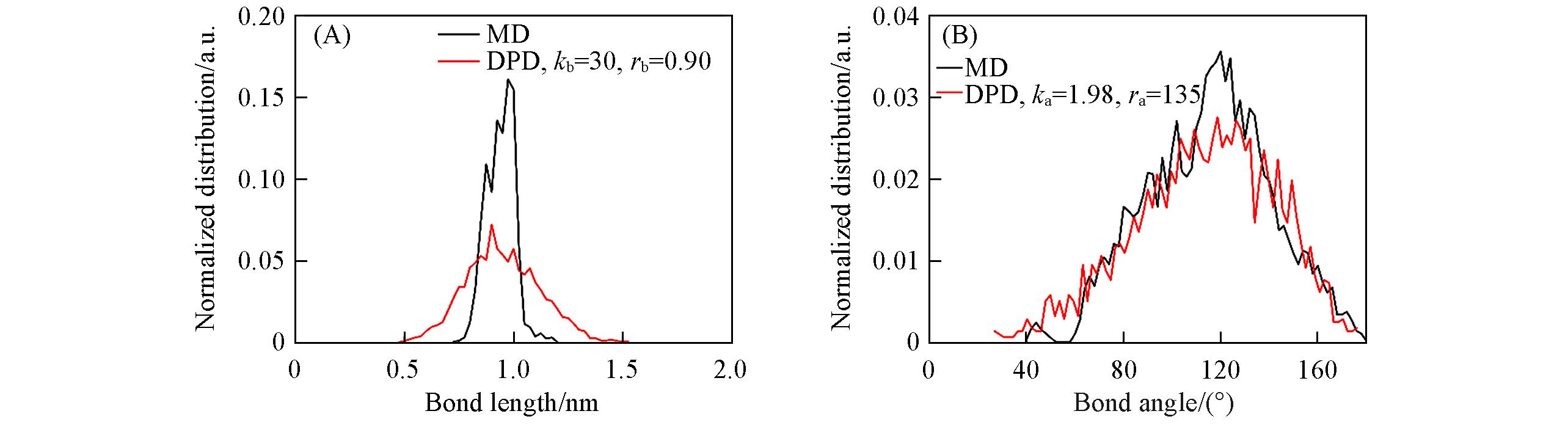
Fig.5 Distribution of bond length(A) and angle(B) between consecutive PES repeat unitsThe black lines are the MD simulation results and the red lines are the corresponding DPD simulation results.
| Species | Molecular volume/nm3 | Molar mass | Number/ particle |
|---|---|---|---|
| PES repeat unit(P) | 0.2 | 232.26 | 1 |
| NMP molecule(S) | 0.1 | 99.13 | 2 |
| Water molecule(C) | 0.02 | 18.02 | 10 |
Table 2 Molecular volume, molar mass, and the number of species per particle for PES, NMP and water
| Species | Molecular volume/nm3 | Molar mass | Number/ particle |
|---|---|---|---|
| PES repeat unit(P) | 0.2 | 232.26 | 1 |
| NMP molecule(S) | 0.1 | 99.13 | 2 |
| Water molecule(C) | 0.02 | 18.02 | 10 |
| Species | PES?H2O | PES?NMP | NMP?H2O |
|---|---|---|---|
| 2.18 | 0.16 | 0.27 | |
| 32.62 | 25.56 | 25.94 |
Table 3 Flory-Huggins interaction parameter χij and the corresponding DPD repulsion parameter aij between different species
| Species | PES?H2O | PES?NMP | NMP?H2O |
|---|---|---|---|
| 2.18 | 0.16 | 0.27 | |
| 32.62 | 25.56 | 25.94 |
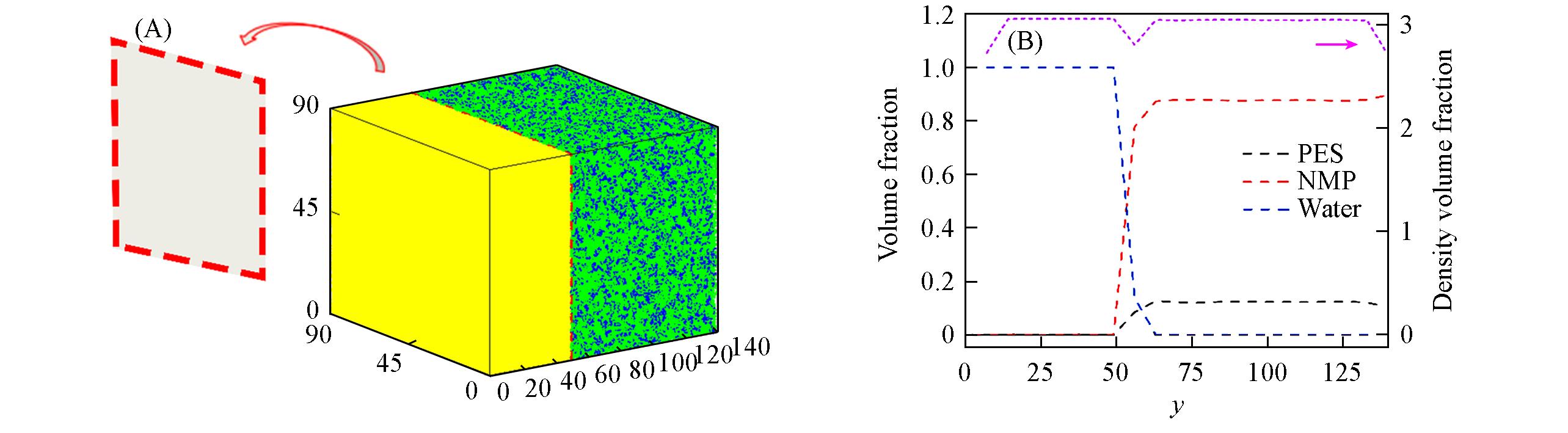
Fig.7 Schematics of the DPD simulation strategy for the membrane formation process via NIPS(A) Initial configuration of two separate systems: water(yellow) and PES(blue)/NMP(green) solution. The simulation of the NIPS process could be started when the artificial barrier(red/gray plane) between them was removed. (B) The initial profiles of volume fractions of the different species(black, red, blue) and total particle density(magenta) along the y axis with units of rc. The PES concentration in the solution is 12%(volume fraction).
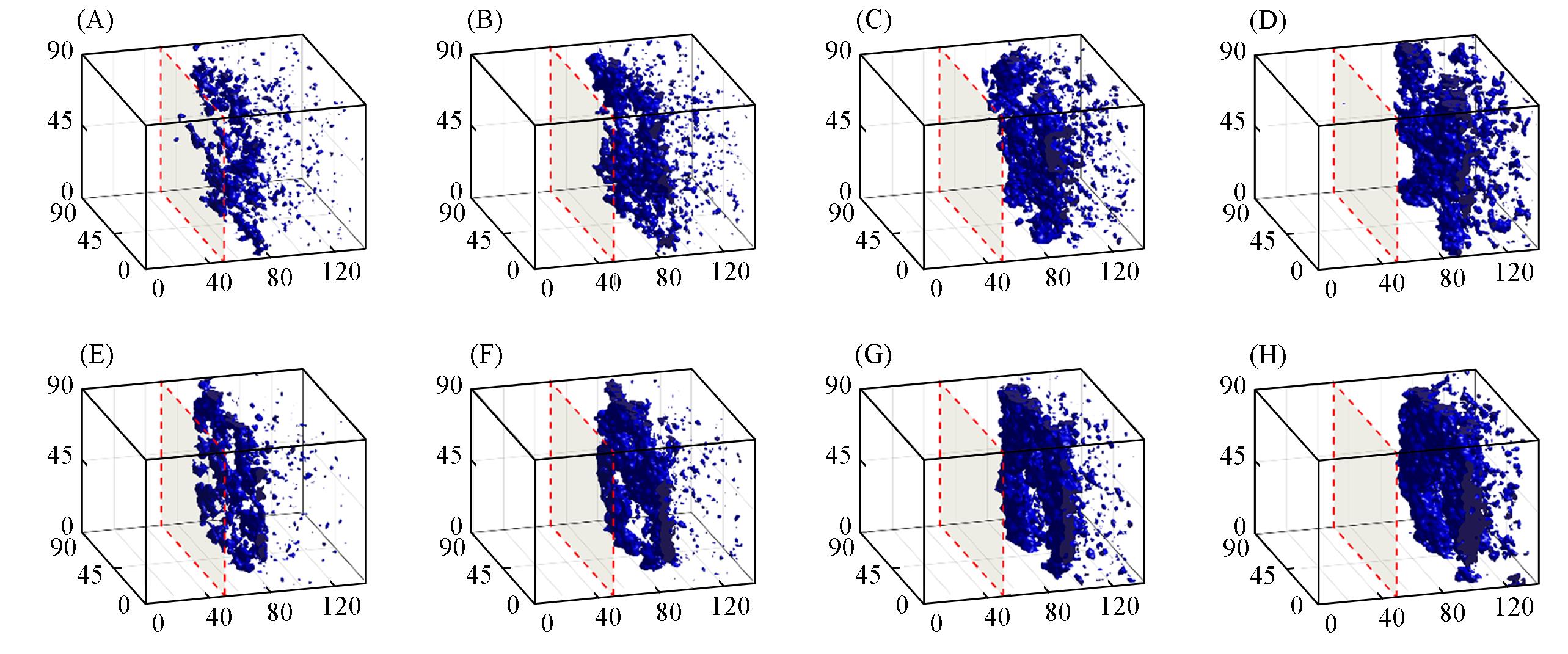
Fig.8 Morphology evolution of the systems with initial polymer volume fractions 12% in spring force field(A—D) and harmonic force field(E—H) at 2500τ(A, E), 5000τ(B, F), 7500τ(C, G) and 10000τ(D, H) during the DPD simulationsThe blue iso-surface represents a value of 45% local polymer volume fraction. Thus, the blue regions are considered as the polymer-rich phase and the white regions are regarded as the polymer-lean phase.
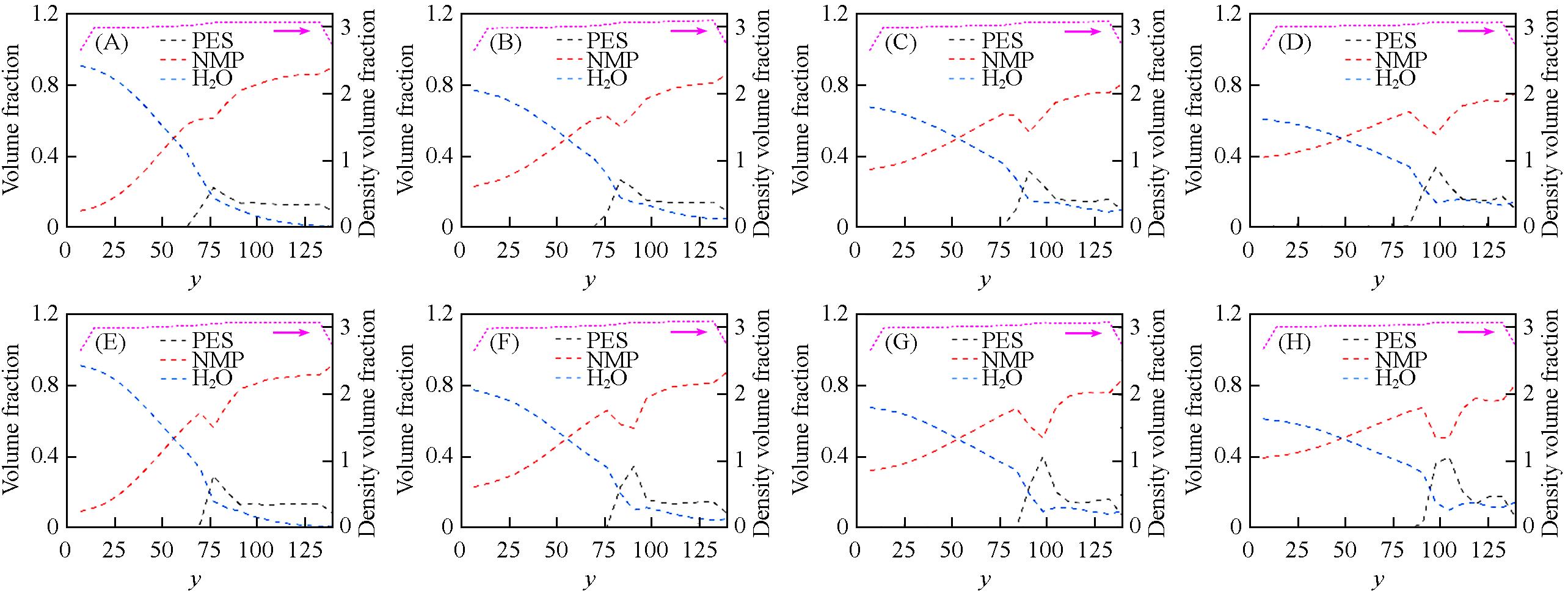
Fig.9 Profiles of volume fraction of PES(black), NMP(red) and H2O(blue) and total particle density(magenta) along the y axis in spring force field(A—D) and harmonic force field(E—H) at 2500τ(A, E), 5000τ(B, F), 7500τ(C, G) and 10000τ(D, H) during the DPD simulations
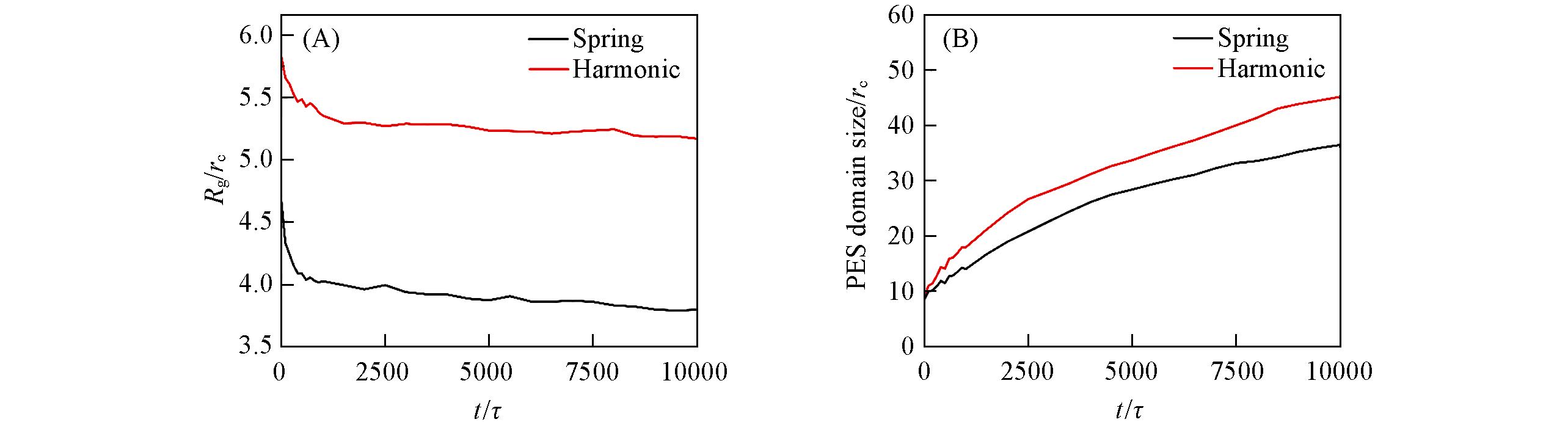
Fig.11 Evolution of radius of gyration Rg(A) and PES domain size(B) as functions of time for the systems in harmonic force field and spring force field
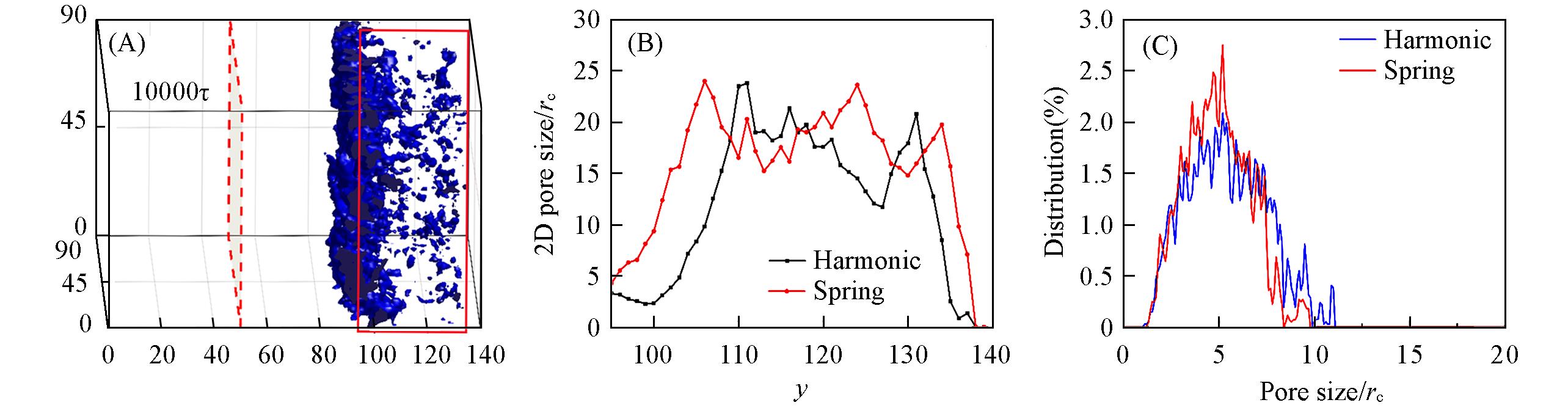
Fig.12 Effects of harmonic force field and spring force field on the 2D and 3D pore size distributionsInitial polymer volume fraction was kept at 12%(volme fraction) in all cases. (A) Morphology of the systems at 10000τ, the red rectangle indicates the cutoff for pore size computation, pore sizes were computed only for this region; (B) average 2D(x?z plane) pore size; (C) 3D pore size distribution.
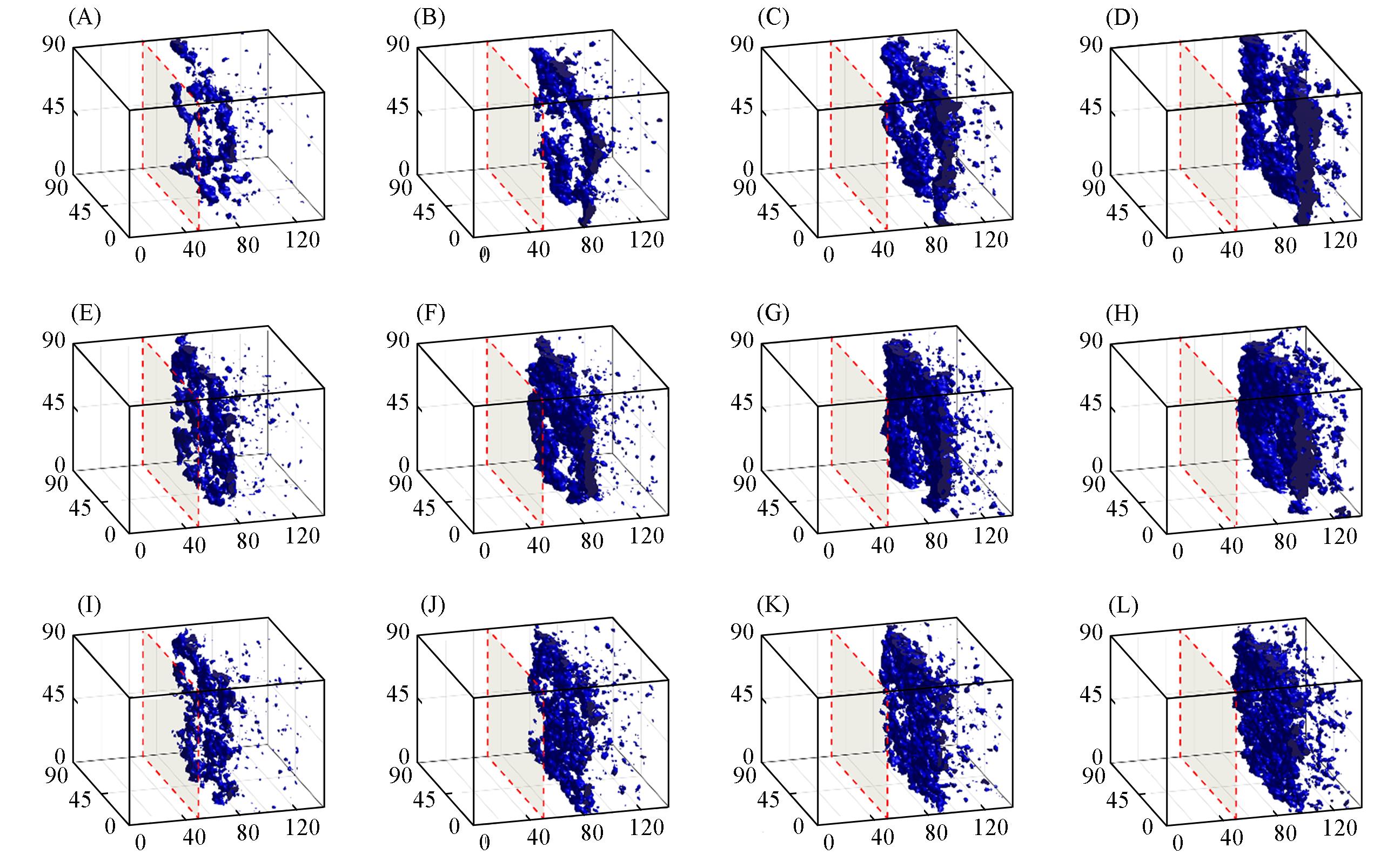
Fig.13 Morphology evolution of the systems with chain length n=90 and initial polymer volume fractions of 8%(A—D), 12%(E—H), and 16%(I—L) at 2500τ(A, E, I), 5000τ(B, F, J), 7500τ(C, G, K) and 10000τ(D, H, L) during the DPD simulationsThe blue iso-surface represents a value of 45% local polymer volume fraction. Thus, the blue regions are considered as the polymer-rich phase and the white regions are considered as the polymer-lean phase.
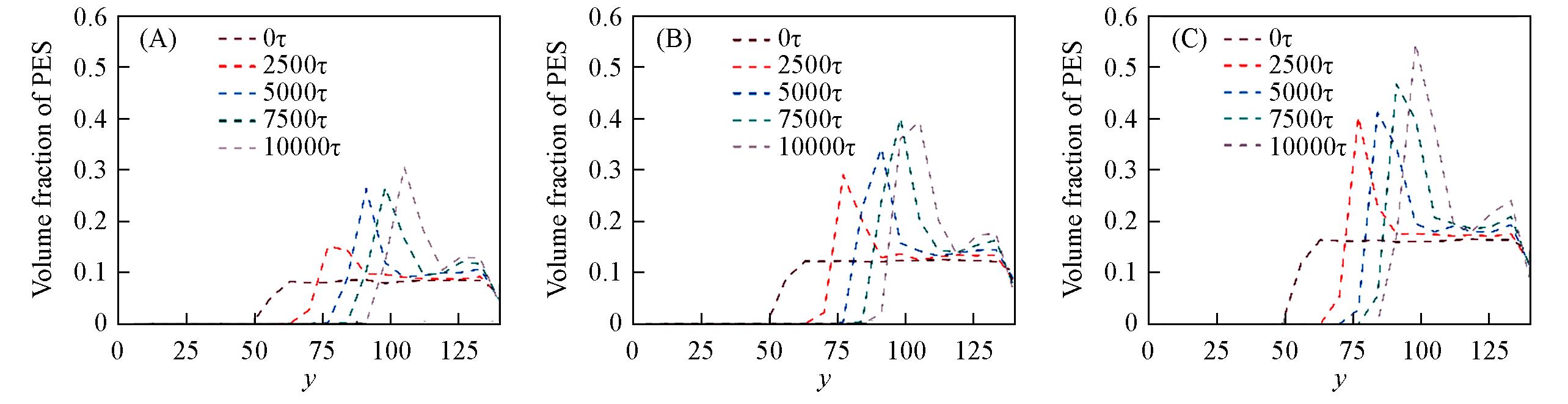
Fig.14 Profiles of volume fractions of PES along y axis at 0, 2500τ, 5000τ, 7500τ and 10000τInitial polymer volume fraction: (A) 8%; (B) 12%; (C) 16%.
| 1 | Nunes S. P., Culfaz⁃Emecen P. Z., Ramon G. Z., Visser T., Koops G. H., Jin W., Ulbricht M., J. Membr. Sci., 2020, 598, 117761 |
| 2 | Kausar A., Polym. Plast. Technol. Eng., 2017, 56(13), 1421—1437 |
| 3 | Wang D. M., Lai J. Y., Curr. Opin. Chem. Eng., 2013, 2(2), 229—237 |
| 4 | Tang Y. H., Lin Y. K., Ford D. M., Qian X., Cervellere M. R., Millett P. C., Wang X., J. Membr. Sci., 2021, 640, 119810 |
| 5 | Groot R. D., Warren P. B., J. Chem. Phys., 1997, 107(3), 4423 |
| 6 | Bai J. L., Liu D., Wang R., Chinese J. Polym. Sci., 2021, 39(9), 1217—1224 |
| 7 | Ruiz⁃Morales Y., Alvarez⁃Ramirez F., Energy Fuels, 2021, 35(11), 9294—9311 |
| 8 | Sofos F., Chatzoglou E., Liakopoulos A., Comput. Part. Mech., 2021, 1—15 |
| 9 | Wang X. L., Qian H. J., Chen L. J., Lu Z. Y., Li Z. S., J. Membr. Sci., 2008, 311(1/2), 251—258 |
| 10 | Lin H. H., Tang Y. H., Matsuyama H., Wang X. L., J. Membr. Sci., 2018, 548, 288—297 |
| 11 | Tang Y. H., Ledieu E., Cervellere M. R., Millett P. C., Ford D. M., Qian X., J. Membr. Sci., 2020, 599, 117826 |
| 12 | Wang Z. G., J. Chem. Phys., 2002, 117(1), 481—500 |
| 13 | van der Haven D. L. H., Kohler S., Schreiner E., In 't Veld P. J., J. Phys. Chem. B, 2021, 125(27), 7485—7498 |
| 14 | Wang J. M., Wolf R. M., Caldwell J. W., Kollman P. A., Case D. A., J. Comput. Chem., 2004, 25(9), 1157—1174 |
| 15 | Cornell W. D., Cieplak P., Bayly C. I., Gould I. R., Merz K. M., Ferguson D. M., Spellmeyer D. C., Fox T., Caldwell J. W., Kollman P. A., J. Am. Chem. Soc., 1995, 117(19), 5179—5197 |
| 16 | Vanommeslaeghe K., Hatcher E., Acharya C., Kundu S., Zhong S., Shim J., Darian E., Guvench O., Lopes P., Vorobyov I., Mackerell A. D. Jr., J. Comput. Chem., 2010, 31(4), 671—690 |
| 17 | Frisch M. J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A., Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas O., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision B.01, Gaussian Inc., Wallingford CT, 2010 |
| 18 | Case D. A., Betz R. M., Cerutti D. S., Cheatham T. E. Ⅲ, Darden T. A., Duke R. E., Giese T. J., Gohlke H., Goetz A. W., Homeyer N., Izadi S., Janowski P., Kaus J., Kovalenko A., Lee T. S., LeGrand S., Li P., Lin C., Luchko T., Luo R., Madej B., Mermelstein D., Merz K. M., Monard G., Nguyen H., Nguyen H. T., Omelyan I., Onufriev A., Roe D. R., Roitberg A., Sagui C., Simmerling C. L., Botello⁃Smith W. M., Swails J., Walker R. C., Wang J., Wolf R. M., Wu X., Xiao L., Kollman P. A., AMBER 2016, University of California, San Francisco, 2016 |
| 19 | Salomon‐Ferrer R., Götz A., Poole D., Le Grand S., Walker R., J. Chem. Theory. Comput., 2013, 9, 3878—3888 |
| 20 | Gotz A. W., Williamson M. J., Xu D., Poole D., Le Grand S., Walker R. C., J. Chem. Theory. Comput., 2012, 8(5), 1542—1555 |
| 21 | Le Grand S., Comput. Phys. Commun., 2013, 184, 374—380 |
| 22 | Wiebe H., Louwersheimer J., Weinberg N., Mol. Phys., 2015, 113(21), 3176—3181 |
| 23 | Luchko T., Gusarov S., Roe D. R., Simmerling C., Case D. A., Tuszynski J., Kovalenko A., J. Chem. Theory. Comput., 2010, 6(3), 607—624 |
| 24 | Español P., Warren P., EPL, 1995, 30(4), 191—196 |
| 25 | Chen L. J., Qian H. J., Lu Z. Y., Li Z. S., Sun C. C., J. Phys. Chem. B, 2006, 110(47), 24093—24100 |
| 26 | Ortiz V., Nielsen S. O., Discher D. E., Klein M. L., Lipowsky R., Shillcock J., J. Phys. Chem. B, 2005, 109(37), 17708—17714 |
| 27 | Martinez L., Andrade R., Birgin E. G., Martinez J. M., J. Comput. Chem., 2009, 30(13), 2157—2164 |
| 28 | Zhu Y. L., Liu H., Li Z. W., Qian H. J., Milano G., Lu Z. Y., J. Comput. Chem., 2013, 34(25), 2197—2211 |
| 29 | Laradji M., Mouritsen O. G., Toxvaerd S., Zuckermann M. J., Phys. Rev. E, 1994, 50(2), 1243—1252 |
| 30 | Willems T. F., Rycroft C. H., Kazi M., Meza J. C., Haranczyk M., Micropor. Mesopor. Mat., 2012, 149(1), 134—141 |
| 31 | Xiao L. Y., Ji D. H., Phys. Bull., 2017, (9), 89—93 |
| 肖立勇, 纪登辉. 物理通报, 2017, (9), 89—93 | |
| 32 | Guillen G. R., Pan Y., Li M, Hoek E. M. V., Ind. Eng. Chem. Res., 2011, 50(7), 3798—3817 |
| 33 | Hwang J. R., Koo S. H., Kim J. H., Higuchi A., Tak T. M., J. Appl. Polym. Sci., 1996, 60(9), 1343—1348 |
| 34 | Mosqueda⁃Jimenez D. B., Narbaitz R. M., Matsuura T., Chowdhury G., Pleizier G., Santerre J. P., J. Membr. Sci., 2004, 231(1/2), 209—224 |
| 35 | Miyano T., Matsuura T., Sourirajan S., Chem. Eng. Commun., 2010, 95(1), 11—26 |
| 36 | King J. S., Boyer W., Wignall G. D., Ullman R., Macromol., 1985, 18(4), 709—718 |
| 37 | Kaznessis Y. N., Hill D. A., Maginn E. J., J. Chem. Phys., 1998, 109(12), 5078—5088 |
| 38 | Barzin J., Sadatnia B., Polym., 2007, 48(6), 1620—1631 |
| [1] | 吴同华, 岳喜贵, 梅笑寒, 梁留博, 彭鑫, 马友美, 张淑玲. 三明治结构多壁碳纳米管/聚醚醚酮电磁屏蔽复合材料的制备[J]. 高等学校化学学报, 2021, 42(8): 2627. |
| [2] | 朱雨昕, HARAGIRIMANA Alphonse, 陆瑶, BUREGEYA Ingabire Providence, 宁聪, 李娜, 胡朝霞, 陈守文. 填充型具有微孔结构的磺化聚芳醚砜/聚醚砜复合质子交换膜的制备及性能[J]. 高等学校化学学报, 2019, 40(5): 1051. |
| [3] | 郑秋光, 刘海亮, 肖长发. 热致相分离法偏二氯乙烯-氯乙烯共聚物多孔膜的制备及性能[J]. 高等学校化学学报, 2019, 40(4): 841. |
| [4] | 梁旭, 秦利娟, 张亚涛, 刘金盾. 二氧化硅接枝聚合磷酸胆碱/聚醚砜复合膜的制备及性能[J]. 高等学校化学学报, 2018, 39(3): 598. |
| [5] | 容婧婧, 马兰, 朱有亮, 黄以能, 孙昭艳. 表面图案诱导两嵌段共聚物形成穿孔层状结构的模拟[J]. 高等学校化学学报, 2018, 39(12): 2805. |
| [6] | 王莉, 王楚, 周健. 耗散粒子动力学模拟嵌段聚合物刷修饰纳米孔的pH响应门控[J]. 高等学校化学学报, 2018, 39(1): 85. |
| [7] | 宋庆松, 张敬男, 刘勇. 周期性变电场下熔体静电纺丝射流的介观模拟[J]. 高等学校化学学报, 2017, 38(6): 966. |
| [8] | 杨平辉, 朱嘉峰, 孙巍, 周婉蓉. 两亲性Janus粒子在蜂窝状多孔聚合物膜上的自组装性能[J]. 高等学校化学学报, 2017, 38(4): 678. |
| [9] | 高虹, 段月琴, 袁志好. 超亲水-水下超疏油PVDF-g-PAA多孔膜的制备及油水分离性能[J]. 高等学校化学学报, 2016, 37(6): 1208. |
| [10] | 徐凡华, 衡晓, 任建学, 周恒为. 配体链包覆纳米粒子的表面相分离及自组装的模拟研究[J]. 高等学校化学学报, 2016, 37(6): 1115. |
| [11] | 陶应勇, 张虚略, 胡朝霞, 张晶晶, 耿慧, 高颖, 袁祖凤, 毕慧平, 陈守文, 王连军. 侧链型含氟磺化聚醚砜/磺化聚酰亚胺共混质子交换膜的制备及性能[J]. 高等学校化学学报, 2016, 37(4): 793. |
| [12] | 刘林林, 杨忠志. 耗散粒子动力学与可极化ABEEM力场结合模拟嵌段共聚物自组装形态调控[J]. 高等学校化学学报, 2015, 36(11): 2179. |
| [13] | 余亮, 张亚涛, 刘金盾. 改性氧化石墨烯/聚醚砜杂化荷正电纳滤膜的制备及表征[J]. 高等学校化学学报, 2014, 35(5): 1100. |
| [14] | 刘晓晗, 郭洪霞. 长棒状T形三组分双亲分子相行为的耗散粒子动力学模拟[J]. 高等学校化学学报, 2014, 35(2): 440. |
| [15] | 谢宇, 吕中元, 孙昭艳, 安立佳. PEO-PPO-PEO水溶液体系胶束化及凝胶化行为的耗散粒子动力学模拟[J]. 高等学校化学学报, 2013, 34(6): 1454. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||