

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (12): 2427.doi: 10.7503/cjcu2015405
收稿日期:2015-05-20
出版日期:2015-12-10
发布日期:2015-11-19
作者简介:联系人简介: 叶志文, 男, 博士, 教授, 博士生导师, 主要从事药物中间体方面的研究. E-mail:基金资助:
AN Dong1, YE Zhiwen1,*( ), ZHOU Mi2
), ZHOU Mi2
Received:2015-05-20
Online:2015-12-10
Published:2015-11-19
Contact:
YE Zhiwen
E-mail:yezw@njust.edu.cn
Supported by:摘要:
以甲基-α-D-吡喃葡萄糖、 甘氨酸和缬氨酸为起始原料, 设计合成了多种新型结构的氨基酸甘油糖脂衍生物(27~34), 并对其结构进行了表征. 采用四甲基偶氮唑蓝比色法(MTT法)研究了该类化合物对人乳腺癌细胞(MCF-7)、 人肝癌细胞(HcpG-2)和人白血病细胞(K562)的体外抑制活性. 初步研究结果表明, 在测试浓度范围内, 化合物27(IC50=3.53, 4.71, 4.13 μmol/L), 28(IC50=4.35, 4.57, 5.79 μmol/L), 31(IC50=3.91, 3.73, 4.54 μmol/L)和32(IC50=5.17, 5.52, 5.93 μmol/L)对癌细胞具有较好的抑制作用, 且对正常细胞无毒性. 此类型糖脂可能成为很好的抗肿瘤药物原料化合物.
中图分类号:
TrendMD:
安东, 叶志文, 周密. 新型氨基酸甘油糖脂的合成及抗肿瘤活性. 高等学校化学学报, 2015, 36(12): 2427.
AN Dong, YE Zhiwen, ZHOU Mi. Synthesis and Anti-tumor Activity of Novel Amino Acid Glyceroglycolipid†. Chem. J. Chinese Universities, 2015, 36(12): 2427.
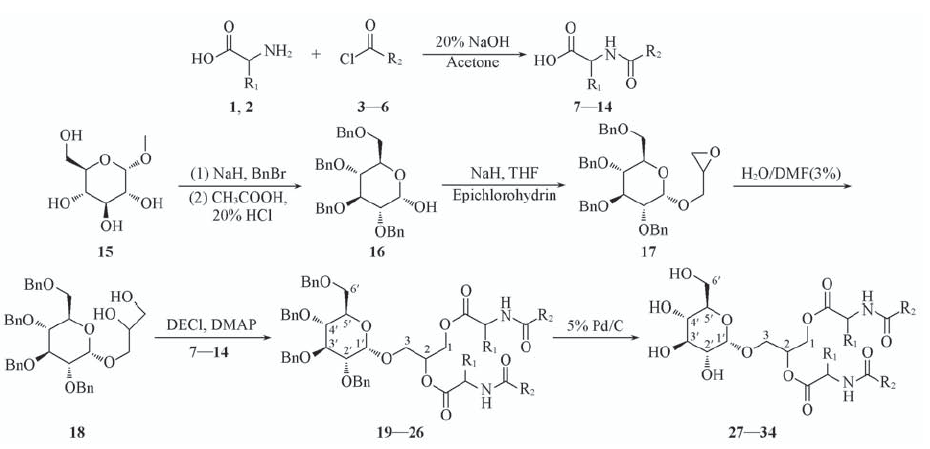
Scheme 1 Synthetic routes of title compounds 27—34 7, 19, 27: R1=H, R2=C9H19; 8, 20, 28: R1=H, R2=C11H23; 9, 21, 29: R1=H, R2=C13H27; 10, 22, 30: R1=H, R2=C15H31; 11, 23, 31: R1=CH(CH3)2, R2=C9H19; 12, 24, 32: R1=CH(CH3)2, R2=C11H23; 13, 25, 33: R1=CH(CH3)2, R2=C13H27; 14, 26, 34: R1=CH(CH3)2, R2=C15H31
| Compd. | m. p.(ref.)/℃ | Compd. | m. p.(ref.)/℃ |
|---|---|---|---|
| 7 | 112.3—113.7(112.3—113.8[ | 11 | 105.5—106.7(105.8—106.9[ |
| 8 | 118.3—119.1(117.5—119.3[ | 12 | 103.3—105.1(103.8—105.3[ |
| 9 | 120.2—121.7(119.1—120.6[ | 13 | 85.1—86.9(86.4—87.7[ |
| 10 | 121.3—122.6(120.4—121.7[ | 14 | 82.1—83.3(83.1—84.4[ |
Table 1 Melting points for compounds 7—14
| Compd. | m. p.(ref.)/℃ | Compd. | m. p.(ref.)/℃ |
|---|---|---|---|
| 7 | 112.3—113.7(112.3—113.8[ | 11 | 105.5—106.7(105.8—106.9[ |
| 8 | 118.3—119.1(117.5—119.3[ | 12 | 103.3—105.1(103.8—105.3[ |
| 9 | 120.2—121.7(119.1—120.6[ | 13 | 85.1—86.9(86.4—87.7[ |
| 10 | 121.3—122.6(120.4—121.7[ | 14 | 82.1—83.3(83.1—84.4[ |
| Compd. | Appearance | Yield(%) | m. p./℃ | [α |
|---|---|---|---|---|
| 19 | White solid | 81.2 | 93.5—95.3 | +4.2 |
| 20 | White solid | 82.3 | 71.5—73.1 | +3.9 |
| 21 | Pale oil | 73.9 | — | +4.4 |
| 22 | Pale oil | 52.6 | — | +5.1 |
| 23 | Yellow liquid | 77.3 | — | +7.3 |
| 24 | Pale liquid | 68.1 | — | +6.9 |
| 25 | Oily liquid | 61.3 | — | +8.1 |
| 26 | Oily liquid | 47.7 | — | +6.3 |
| 27 | White solid | 92.4 | 107.5—109.1 | -33.7 |
| 28 | White solid | 93.1 | 82.7—83.5 | -33.9 |
| 29 | Yellow liquid | 83.1 | — | -52.7 |
| 30 | Yellow liquid | 71.9 | — | -34.3 |
| 31 | White solid | 82.9 | 63.2—64.7 | -43.3 |
| 32 | White liquid | 71.9 | — | -24.9 |
| 33 | White liquid | 74.3 | — | -32.7 |
| 34 | Yellow liquid | 51.7 | — | -24.3 |
Table 2 Appearance, yields, melting points and optical rotation of compounds 19—34
| Compd. | Appearance | Yield(%) | m. p./℃ | [α |
|---|---|---|---|---|
| 19 | White solid | 81.2 | 93.5—95.3 | +4.2 |
| 20 | White solid | 82.3 | 71.5—73.1 | +3.9 |
| 21 | Pale oil | 73.9 | — | +4.4 |
| 22 | Pale oil | 52.6 | — | +5.1 |
| 23 | Yellow liquid | 77.3 | — | +7.3 |
| 24 | Pale liquid | 68.1 | — | +6.9 |
| 25 | Oily liquid | 61.3 | — | +8.1 |
| 26 | Oily liquid | 47.7 | — | +6.3 |
| 27 | White solid | 92.4 | 107.5—109.1 | -33.7 |
| 28 | White solid | 93.1 | 82.7—83.5 | -33.9 |
| 29 | Yellow liquid | 83.1 | — | -52.7 |
| 30 | Yellow liquid | 71.9 | — | -34.3 |
| 31 | White solid | 82.9 | 63.2—64.7 | -43.3 |
| 32 | White liquid | 71.9 | — | -24.9 |
| 33 | White liquid | 74.3 | — | -32.7 |
| 34 | Yellow liquid | 51.7 | — | -24.3 |
| Compd. | ESI-MS, m/z, [M+Na]+ | Elemental analysis(%, calcd.) | ||
|---|---|---|---|---|
| C | H | N | ||
| 19 | 1059.73 | 71.02(70.63) | 7.88(8.16) | 2.79(2.70) |
| 20 | 1115.72 | 71.85(71.40) | 8.67(8.48) | 2.37(2.56) |
| 21 | 1171.79 | 72.47(72.09) | 8.93(8.77) | 2.29(2.44) |
| 22 | 1227.92 | 72.87(72.72) | 9.31(9.03) | 2.11(2.32) |
| 23 | 1143.76 | 71.93(71.75) | 8.31(8.63) | 2.37(2.50) |
| 24 | 1199.89 | 72.66(72.42) | 8.72(8.90) | 2.41(2.38) |
| 25 | 1255.61 | 73.25(73.02) | 9.02(9.15) | 2.47(2.27) |
| 26 | 1311.73 | 73.73(73.57) | 9.19(9.38) | 2.35(2.17) |
| 27 | 699.47 | 58.69(58.56) | 9.11(8.94) | 4.03(4.14) |
| 28 | 755.51 | 60.89(60.63) | 9.51(9.35) | 3.63(3.82) |
| 29 | 811.47 | 62.66(62.41) | 9.53(9.71) | 3.41(3.55) |
| 30 | 867.66 | 63.67(63.95) | 9.83(10.02) | 3.52(3.31) |
| 31 | 783.57 | 61.83(61.55) | 9.61(9.54) | 3.39(3.68) |
| 32 | 839.63 | 63.47(63.21) | 9.95(9.87) | 3.23(3.43) |
| 33 | 895.67 | 64.79(64.65) | 10.39(10.16) | 3.02(3.21) |
| 34 | 951.78 | 66.27(65.91) | 10.13(10.41) | 3.27(3.01) |
Table 3 ESI-MS and elemental analysis data of compounds 19—34
| Compd. | ESI-MS, m/z, [M+Na]+ | Elemental analysis(%, calcd.) | ||
|---|---|---|---|---|
| C | H | N | ||
| 19 | 1059.73 | 71.02(70.63) | 7.88(8.16) | 2.79(2.70) |
| 20 | 1115.72 | 71.85(71.40) | 8.67(8.48) | 2.37(2.56) |
| 21 | 1171.79 | 72.47(72.09) | 8.93(8.77) | 2.29(2.44) |
| 22 | 1227.92 | 72.87(72.72) | 9.31(9.03) | 2.11(2.32) |
| 23 | 1143.76 | 71.93(71.75) | 8.31(8.63) | 2.37(2.50) |
| 24 | 1199.89 | 72.66(72.42) | 8.72(8.90) | 2.41(2.38) |
| 25 | 1255.61 | 73.25(73.02) | 9.02(9.15) | 2.47(2.27) |
| 26 | 1311.73 | 73.73(73.57) | 9.19(9.38) | 2.35(2.17) |
| 27 | 699.47 | 58.69(58.56) | 9.11(8.94) | 4.03(4.14) |
| 28 | 755.51 | 60.89(60.63) | 9.51(9.35) | 3.63(3.82) |
| 29 | 811.47 | 62.66(62.41) | 9.53(9.71) | 3.41(3.55) |
| 30 | 867.66 | 63.67(63.95) | 9.83(10.02) | 3.52(3.31) |
| 31 | 783.57 | 61.83(61.55) | 9.61(9.54) | 3.39(3.68) |
| 32 | 839.63 | 63.47(63.21) | 9.95(9.87) | 3.23(3.43) |
| 33 | 895.67 | 64.79(64.65) | 10.39(10.16) | 3.02(3.21) |
| 34 | 951.78 | 66.27(65.91) | 10.13(10.41) | 3.27(3.01) |
| Compd. | 1H NMR(500 MHz, CDCl3), δ |
|---|---|
| 19 | 7.33—7.27(m, 18H, ArH), 7.15—7.14(m, 2H, ArH), 6.08—6.01(m, 2H, 2NH), 5.60—5.58(m, 1H, 1'-H), 4.93—4.91(m, 1H, 2-H), 4.86—4.84(m, 2H, 4'-PhCH2), 4.83—4.82(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.73—4.72(m, 1H, 3'b-PhCH), 4.58—4.56(m, 1H, 6'a-PhCH), 4.32—4.31(m, 1H, 6'b-PhCH), 4.31—4.27(m, 2H, 1-H), 4.27—4.26(m, 1H, 3a-H), 3.97—3.95(m, 1H, 6'a-CH), 3.88—3.87(m, 1H, 3b-H), 3.74—3.72(m, 1H, 5'-H), 3.64—3.63(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.58—3.56(m, 1H, 4'-H), 3.52—3.50(m, 2H, 6'-H), 2.23—2.16(m, 4H, 2CH2), 1.66—1.64(m, 2H, CH2), 1.63—1.61(m, 4H, 2CH2), 1.29—1.26[m, 24H, 2(CH2)6], 0.88—0.86(m, 6H, 2CH3) |
| 20 | 7.34—7.28(m, 18H, ArH), 7.16—7.14(m, 2H, ArH), 6.10—6.06(m, 2H, 2NH), 5.66—5.61(m, 1H, 1'-H), 4.95—4.92(m, 1H, 2-H), 4.88—4.86(m, 2H, 4'-PhCH2), 4.81—4.80(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.80—4.78(m, 1H, 3'b-PhCH), 4.63—4.61(m, 1H, 6'a-PhCH), 4.40—4.37(m, 1H, 6'b-PhCH), 4.34—4.30(m, 2H, 1-H), 4.29—4.26(m, 1H, 3a-H), 4.02—3.97(m, 1H, 6'a-PhCH), 3.95—3.93(m, 1H, 3b-H), 3.77—3.75(m, 1H, 5'-H), 3.67—3.65(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.59—3.57(m, 1H, 4'-H), 3.56—3.52(m, 2H, 6'-H), 2.27—2.20(m, 4H, 2CH2), 1.70—1.66(m, 2H, CH2), 1.65—1.63(m, 4H, 2CH2), 1.31—1.28[m, 32H, 2(CH2)8], 0.90—0.87(m, 6H, 2CH3) |
| 21 | 7.31—7.27(m, 18H, ArH), 7.14—7.12(m, 2H, ArH), 6.10—6.09(m, 2H, 2NH), 5.61—5.59(m, 1H, 1'-H), 4.93—4.91(m, 1H, 2-H), 4.86—4.84(m, 2H, 4'-PhCH2), 4.83—4.82(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.73—4.72(m, 1H, 3'b-PhCH), 4.58—4.56(m, 1H, 6'a-PhCH), 4.32—4.31(m, 1H, 6'b-PhCH), 4.31—4.27(m, 2H, 1-H), 4.27—4. 26(m, 1H, 3a-H), 3.97—3.95(m, 1H, 6'a-PhCH), 3.88—3.87(m, 1H, 3b-H), 3.74—3.72(m, 1H, 5'-H), 3.64—3.63(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.58—3.56(m, 1H, 4'-H), 3.52—3.50(m, 2H, 6'-H), 2.23—2.16(m, 4H, 2CH2), 1.66—1.64(m, 2H, CH2), 1.63—1.61(m, 4H, 2CH2), 1.29—1.26[m, 40H, 2(CH2)10], 0.88—0.86(m, 6H, 2CH3) |
| 22 | 7.32—7.28(m, 18H, ArH), 7.17—7.15(m, 2H, ArH), 6.08—6.01(m, 2H, 2NH), 5.60—5.58(m, 1H, 1'-H), 4.95—4.93(m, 1H, 2-H), 4.87—4.85(m, 2H, 4'-PhCH2), 4.84—4.82(m, 1H, 3'a-PhCH), 4.78—4.76(m, 2H, 2'-PhCH2), 4.75—4.73(m, 1H, 3'b-PhCH), 4.60—4.58(m, 1H, 6'a-PhCH), 4.35—4.32(m, 1H, 6'b-PhCH), 4.31—4.28(m, 2H, 1-H), 4.27—4.25(m, 1H, 3a-H), 3.98—3.96(m, 1H, 6'a-PhCH), 3.90—3.89(m, 1H, 3b-H), 3.77—3.74(m, 1H, 5'-H), 3.66—3.64(m, 1H, 3'-H), 3.63—3.61(m, 1H, 2'-H), 3.60—3.58(m, 1H, 4'-H), 3.57—3.54 |
| 22 | (m, 2H, 6'-H), 2.22—2.19(m, 4H, 2CH2), 1.68—1.65(m, 2H, CH2), 1.64—1.61(m, 4H, 2CH2), 1.30—1.27[m, 48H, 2(CH2)12], 0.89—0.85(m, 6H, 2CH3) |
| 23 | 7.35—7.26(m, 18H, ArH), 7.27—7.22(m, 2H, ArH), 6.08—6.00(m, 2H, 2NH), 5.61—5.60(m, 1H, 1'-H), 5.40—5.39(m, 1H, 2-H), 5.31—5.24(m, 2H, 4'-PhCH2), 4.83—4.82(m, 2H, 3'-PhCH), 4.81—4.80(m, 2H, 2'-PhCH2), 4.64—4.61(m, 2H, 6'-PhCH2), 4.60—4.59(m, 2H, 1-H), 4.56—4.55(m, 2H, 2COCHNH), 4.49—4.47(m, 1H, 3-H), 3.72—3.70(m, 1H, 5'-H), 3.56—3.54(m, 1H, 3'-H), 3.50—3.48(m, 1H, 2'-H), 3.47—3.45(m, 2H, 6'-H), 3.44—3.42(m, 1H, 4'-H), 2.31—2.27[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.67—1.65(m, 4H, 2CH2), 1.29—1.27[m, 24H, 2(CH2)6], 0.90—0.87(m, 18H, 6CH3) |
| 24 | 7.36—7.27(m, 18H, ArH), 7.25—7.23(m, 2H, ArH), 6.12—6.07(m, 2H, 2NH), 5.65—5.62(m, 1H, 1'-H), 5.44—5.41(m, 1H, 2-H), 5.35—5.27(m, 2H, 4'-PhCH2), 4.87—4.85(m, 2H, 3'-PhCH), 4.83—4.81(m, 2H, 2'-PhCH2), 4.65—4.62(m, 2H, 6'-PhCH2), 4.61—4.58(m, 2H, 1-H), 4.57—4.55(m, 2H, 2COCHNH), 4.51—4.49(m, 1H, 3-H), 3.75—3.73(m, 1H, 5'-H), 3.60—3.58(m, 1H, 3'-H), 3.53—3.50(m, 1H, 2'-H), 3.49—3.47(m, 2H, 6'-H), 3.45—3.43(m, 1H, 4'-H), 2.32—2.28[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.68—1.66(m, 4H, 2CH2), 1.30—1.28[m, 32H, 2(CH2)8], 0.93—0.88(m, 18H, 6CH3) |
| 25 | 7.36—7.26(m, 18H, ArH), 7.28—7.24(m, 2H, ArH), 6.08—6.00(m, 2H, 2NH), 5.61—5.60(m, 1H, 1'-H), 5.40—5.39(m, 1H, 2-H), 5.31—5.24(m, 2H, 4'-PhCH2), 4.83—4.82(m, 2H, 3'-PhCH), 4.81—4.80(m, 2H, 2'-PhCH2), 4.64—4.61(m, 2H, 6'-PhCH2), 4.60—4.59(m, 2H, 1-H), 4.56—4.55(m, 2H, 2COCHNH), 4.49—4.47(m, 1H, 3-H), 3.72—3.70(m, 1H, 5'-H), 3.56—3.54(m, 1H, 3'-H), 3.50—3.48(m, 1H, 2'-H), 3.47—3.45(m, 2H, 6'-H), 3.53(m, 1H, 4'-H), 2.31—2.27[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.67—1.65(m, 4H, 2CH2), 1.29—1.27[m, 40H, 2(CH2)10], 0.90—0.87(m, 18H, 6CH3) |
| 26 | 7.37—7.28(m, 18H, ArH), 7.28—7.20(m, 2H, ArH), 6.09—6.02(m, 2H, 2NH), 5.63—5.61(m, 1H, 1'-H), 5.54—5.51(m, 1H, 2-H), 5.38—5.34(m, 2H, 4'-PhCH2), 4.89—4.85(m, 2H, 3'-PhCH), 4.83—4.80(m, 2H, 2'-PhCH2), 4.70—4.67(m, 2H, 6'-PhCH2), 4.65—4.62(m, 2H, 1-H), 4.58—4.56(m, 2H, 2COCHNH), 4.53—4.51(m, 1H, 3-H), 3.73—3.71(m, 1H, 5'-H), 3.60—3.56(m, 1H, 3'-H), 3.52—3.49(m, 1H, 2'-H), 3.48—3.46(m, 2H, 6'-H), 3.43—3.41(m, 1H, 4'-H), 2.32—2.28[m, 2H, 2CH(CH3)2], 2.27—2.23(m, 4H, 2NHCOCH2), 1.69—1.65(m, 4H, 2CH2), 1.31—1.28[m, 24H, 2(CH2)12], 0.92—0.88(m, 18H, 6CH3) |
Table 4 1H NMR data of compounds 19—26
| Compd. | 1H NMR(500 MHz, CDCl3), δ |
|---|---|
| 19 | 7.33—7.27(m, 18H, ArH), 7.15—7.14(m, 2H, ArH), 6.08—6.01(m, 2H, 2NH), 5.60—5.58(m, 1H, 1'-H), 4.93—4.91(m, 1H, 2-H), 4.86—4.84(m, 2H, 4'-PhCH2), 4.83—4.82(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.73—4.72(m, 1H, 3'b-PhCH), 4.58—4.56(m, 1H, 6'a-PhCH), 4.32—4.31(m, 1H, 6'b-PhCH), 4.31—4.27(m, 2H, 1-H), 4.27—4.26(m, 1H, 3a-H), 3.97—3.95(m, 1H, 6'a-CH), 3.88—3.87(m, 1H, 3b-H), 3.74—3.72(m, 1H, 5'-H), 3.64—3.63(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.58—3.56(m, 1H, 4'-H), 3.52—3.50(m, 2H, 6'-H), 2.23—2.16(m, 4H, 2CH2), 1.66—1.64(m, 2H, CH2), 1.63—1.61(m, 4H, 2CH2), 1.29—1.26[m, 24H, 2(CH2)6], 0.88—0.86(m, 6H, 2CH3) |
| 20 | 7.34—7.28(m, 18H, ArH), 7.16—7.14(m, 2H, ArH), 6.10—6.06(m, 2H, 2NH), 5.66—5.61(m, 1H, 1'-H), 4.95—4.92(m, 1H, 2-H), 4.88—4.86(m, 2H, 4'-PhCH2), 4.81—4.80(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.80—4.78(m, 1H, 3'b-PhCH), 4.63—4.61(m, 1H, 6'a-PhCH), 4.40—4.37(m, 1H, 6'b-PhCH), 4.34—4.30(m, 2H, 1-H), 4.29—4.26(m, 1H, 3a-H), 4.02—3.97(m, 1H, 6'a-PhCH), 3.95—3.93(m, 1H, 3b-H), 3.77—3.75(m, 1H, 5'-H), 3.67—3.65(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.59—3.57(m, 1H, 4'-H), 3.56—3.52(m, 2H, 6'-H), 2.27—2.20(m, 4H, 2CH2), 1.70—1.66(m, 2H, CH2), 1.65—1.63(m, 4H, 2CH2), 1.31—1.28[m, 32H, 2(CH2)8], 0.90—0.87(m, 6H, 2CH3) |
| 21 | 7.31—7.27(m, 18H, ArH), 7.14—7.12(m, 2H, ArH), 6.10—6.09(m, 2H, 2NH), 5.61—5.59(m, 1H, 1'-H), 4.93—4.91(m, 1H, 2-H), 4.86—4.84(m, 2H, 4'-PhCH2), 4.83—4.82(m, 1H, 3'a-PhCH), 4.77—4.75(m, 2H, 2'-PhCH2), 4.73—4.72(m, 1H, 3'b-PhCH), 4.58—4.56(m, 1H, 6'a-PhCH), 4.32—4.31(m, 1H, 6'b-PhCH), 4.31—4.27(m, 2H, 1-H), 4.27—4. 26(m, 1H, 3a-H), 3.97—3.95(m, 1H, 6'a-PhCH), 3.88—3.87(m, 1H, 3b-H), 3.74—3.72(m, 1H, 5'-H), 3.64—3.63(m, 1H, 3'-H), 3.63—3.59(m, 1H, 2'-H), 3.58—3.56(m, 1H, 4'-H), 3.52—3.50(m, 2H, 6'-H), 2.23—2.16(m, 4H, 2CH2), 1.66—1.64(m, 2H, CH2), 1.63—1.61(m, 4H, 2CH2), 1.29—1.26[m, 40H, 2(CH2)10], 0.88—0.86(m, 6H, 2CH3) |
| 22 | 7.32—7.28(m, 18H, ArH), 7.17—7.15(m, 2H, ArH), 6.08—6.01(m, 2H, 2NH), 5.60—5.58(m, 1H, 1'-H), 4.95—4.93(m, 1H, 2-H), 4.87—4.85(m, 2H, 4'-PhCH2), 4.84—4.82(m, 1H, 3'a-PhCH), 4.78—4.76(m, 2H, 2'-PhCH2), 4.75—4.73(m, 1H, 3'b-PhCH), 4.60—4.58(m, 1H, 6'a-PhCH), 4.35—4.32(m, 1H, 6'b-PhCH), 4.31—4.28(m, 2H, 1-H), 4.27—4.25(m, 1H, 3a-H), 3.98—3.96(m, 1H, 6'a-PhCH), 3.90—3.89(m, 1H, 3b-H), 3.77—3.74(m, 1H, 5'-H), 3.66—3.64(m, 1H, 3'-H), 3.63—3.61(m, 1H, 2'-H), 3.60—3.58(m, 1H, 4'-H), 3.57—3.54 |
| 22 | (m, 2H, 6'-H), 2.22—2.19(m, 4H, 2CH2), 1.68—1.65(m, 2H, CH2), 1.64—1.61(m, 4H, 2CH2), 1.30—1.27[m, 48H, 2(CH2)12], 0.89—0.85(m, 6H, 2CH3) |
| 23 | 7.35—7.26(m, 18H, ArH), 7.27—7.22(m, 2H, ArH), 6.08—6.00(m, 2H, 2NH), 5.61—5.60(m, 1H, 1'-H), 5.40—5.39(m, 1H, 2-H), 5.31—5.24(m, 2H, 4'-PhCH2), 4.83—4.82(m, 2H, 3'-PhCH), 4.81—4.80(m, 2H, 2'-PhCH2), 4.64—4.61(m, 2H, 6'-PhCH2), 4.60—4.59(m, 2H, 1-H), 4.56—4.55(m, 2H, 2COCHNH), 4.49—4.47(m, 1H, 3-H), 3.72—3.70(m, 1H, 5'-H), 3.56—3.54(m, 1H, 3'-H), 3.50—3.48(m, 1H, 2'-H), 3.47—3.45(m, 2H, 6'-H), 3.44—3.42(m, 1H, 4'-H), 2.31—2.27[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.67—1.65(m, 4H, 2CH2), 1.29—1.27[m, 24H, 2(CH2)6], 0.90—0.87(m, 18H, 6CH3) |
| 24 | 7.36—7.27(m, 18H, ArH), 7.25—7.23(m, 2H, ArH), 6.12—6.07(m, 2H, 2NH), 5.65—5.62(m, 1H, 1'-H), 5.44—5.41(m, 1H, 2-H), 5.35—5.27(m, 2H, 4'-PhCH2), 4.87—4.85(m, 2H, 3'-PhCH), 4.83—4.81(m, 2H, 2'-PhCH2), 4.65—4.62(m, 2H, 6'-PhCH2), 4.61—4.58(m, 2H, 1-H), 4.57—4.55(m, 2H, 2COCHNH), 4.51—4.49(m, 1H, 3-H), 3.75—3.73(m, 1H, 5'-H), 3.60—3.58(m, 1H, 3'-H), 3.53—3.50(m, 1H, 2'-H), 3.49—3.47(m, 2H, 6'-H), 3.45—3.43(m, 1H, 4'-H), 2.32—2.28[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.68—1.66(m, 4H, 2CH2), 1.30—1.28[m, 32H, 2(CH2)8], 0.93—0.88(m, 18H, 6CH3) |
| 25 | 7.36—7.26(m, 18H, ArH), 7.28—7.24(m, 2H, ArH), 6.08—6.00(m, 2H, 2NH), 5.61—5.60(m, 1H, 1'-H), 5.40—5.39(m, 1H, 2-H), 5.31—5.24(m, 2H, 4'-PhCH2), 4.83—4.82(m, 2H, 3'-PhCH), 4.81—4.80(m, 2H, 2'-PhCH2), 4.64—4.61(m, 2H, 6'-PhCH2), 4.60—4.59(m, 2H, 1-H), 4.56—4.55(m, 2H, 2COCHNH), 4.49—4.47(m, 1H, 3-H), 3.72—3.70(m, 1H, 5'-H), 3.56—3.54(m, 1H, 3'-H), 3.50—3.48(m, 1H, 2'-H), 3.47—3.45(m, 2H, 6'-H), 3.53(m, 1H, 4'-H), 2.31—2.27[m, 2H, 2CH(CH3)2], 2.25—2.20(m, 4H, 2NHCOCH2), 1.67—1.65(m, 4H, 2CH2), 1.29—1.27[m, 40H, 2(CH2)10], 0.90—0.87(m, 18H, 6CH3) |
| 26 | 7.37—7.28(m, 18H, ArH), 7.28—7.20(m, 2H, ArH), 6.09—6.02(m, 2H, 2NH), 5.63—5.61(m, 1H, 1'-H), 5.54—5.51(m, 1H, 2-H), 5.38—5.34(m, 2H, 4'-PhCH2), 4.89—4.85(m, 2H, 3'-PhCH), 4.83—4.80(m, 2H, 2'-PhCH2), 4.70—4.67(m, 2H, 6'-PhCH2), 4.65—4.62(m, 2H, 1-H), 4.58—4.56(m, 2H, 2COCHNH), 4.53—4.51(m, 1H, 3-H), 3.73—3.71(m, 1H, 5'-H), 3.60—3.56(m, 1H, 3'-H), 3.52—3.49(m, 1H, 2'-H), 3.48—3.46(m, 2H, 6'-H), 3.43—3.41(m, 1H, 4'-H), 2.32—2.28[m, 2H, 2CH(CH3)2], 2.27—2.23(m, 4H, 2NHCOCH2), 1.69—1.65(m, 4H, 2CH2), 1.31—1.28[m, 24H, 2(CH2)12], 0.92—0.88(m, 18H, 6CH3) |
| Compd. | 1H NMR(500 MHz, DMSO), δ | 13C NMR(125 MHz, DMSO), δ |
|---|---|---|
| 27 | 8.21—8.23(m, 2H, 2NH), 5.12—5.05(m, 1H, 1'-H),5.04—4.88(m, 1H, 2-H), 4.88—4.78(m, 2H, 1-H), 4.69—4.62(m, 2H, 1COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.38—4.36(m, 1H, 2'-H), 4.25—4.06(m, 2H, 1COCH2NH), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.16—3.07(m, 2H, 6'-H), 2.11—2.09(m, 4H, 2NHC=OCH2), 1.48—1.46(m, 4H, CH2), 1.23—1.21(m, 24H, 12CH2), 0.85—0.82(m, 6H, 2CH3) | 173.22(NHC=O), 173.19(NHC=O),169.47(C=O), 169.45(C=O), 95.15(C1'), 79.26(C2'), 78.31(C2), 76.59(C3'), 72.83(C4'), 69.72(C5'), 67.74(C3), 64.95(C1), 60.80(C6'), 35.46, 31.67, 29.42, 29.33, 29.20, 29.08, 28.96, 25.56, 22.46, 14.30 |
| 28 | 8.23—8.21(m, 2H, 2NH), 5.72—5.71(m, 1H, 2-H), 5.36—5.34(m, 1H, 1'-H), 5.27—5.26(m, 1H, 3'-OH), 5.11—5.10(m, 1H, 4'-OH), 5.02—5.01(m, 1H, 2'-OH), 4.63—4.60(m, 1H, 6'-OH), 4.39—4.37(m, 2H, 1-H), 4.25—4.21(m, 2H, 1COCH2NH), 4.20—4.18(m, 1H, 4'-H), 3.90—3.87(m, 2H, 1COCH2NH), 3.85—3.83(m, lH, 3-H), 3.81—3.73(m, 1H, 2'-H), 3.65—3.62(m, 1H, 5'-H), 3.47—3.43(m, 1H, 3'-H), 3.22—3.21(m, 2H, 6'-H), 2.24—2.13(m, 4H, 2NHC=OCH2), 1.67—1.59(m, 4H, NHC=OCH2CH2), 1.26—1.25(m, 32H, 16CH2), 0.89—0.86(m, 6H, 2CH3) | 173.53(NHC=O), 173.42(NHC=O),171.53(C=O), 171.52(C=O), 95.67(C1'), 78.64(C2'), 78.59(C2), 76.94(C3'), 73.30(C4'), 73.19(C5'), 70.16(C3), 61.25(C1), 56.92(C6'), 39.48, 35.83, 35.76, 32.08, 29.82, 29.73, 29.60, 29.48, 29.36, 29.33, 25.97, 22.87(CH2), 17.69, 14.68(CH3) |
| 29 | 8.25—8.24(m, 2H, 2NH), 5.12—5.05(m, 1H, 1'-H), 4.96—4.94(m, 1H, 2-H), 4.89—4.87(m, 2H, 1-H), 4.78—4.70(m, 2H, 1COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.44—4.39(m, 1H, 2'-H), 4.25—4.06(m, 1H, 1COCH2NH), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.15—3.05(m, 2H, 6'-H), 2.09—2.06(m, | 173.29(NHC=O), 173.24(NHC=O), 169.55(C=O), 169.50(C=O), 95.24(C1'),79.55(C2'), 78.41(C2), 76.70(C3'), 72.93(C4'), 70.25(C5'), 68.82(C3), 60.90(C1), 56.96(C6'), 35.55, 31.76, |
| 29 | 4H, 2NHC=OCH2), 1.46—1.45(m, 4H, CH2), 1.28—1.25(m, 40H, 20CH2), 0.84—0.82(m, 6H, 2CH3) | 29.52, 29.47, 29.41, 29.29, 29.16, 29.06, 25.65, 22.55, 14.41 |
| 30 | 8.21—8.23(m, 2H, 2NH), 5.11—5.03(m, 1H, 1'-H), 4.95—4.93(m, 1H, 2-H), 4.82—4.70(m, 2H, 1-H), 4.69—4.62(m, 4H, 2COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.38—4.36(m, 1H, 2'-H), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.16—3.07(m, 2H, 6'-H), 2.11—2.09(m, 4H, 2CH2), 1.48—1.46(m, 4H, CH2), 1.23—1.21(m, 48H, 24CH2), 0.85—0.82(m, 6H, 2CH3) | 173.29(NHC=O), 173.24(NHC=O), 169.54(C=O), 169.22(C=O), 95.24(C1'),80.01(C2'), 78.40(C2), 76.68(C3'), 72.92(C4'), 69.82(C5'), 68.57(C3), 60.89(C1), 56.77(C6'), 35.55, 31.76, 29.52, 29.47, 29.41, 29.29, 29.16, 29.06, 25.65, 22.55, 14.41 |
| 31 | 8.25—8.20(m, 2H, 2NH), 5.58—5.51(m, 1H, 1'-H), 5.34—5.30(m, 1H, 2-H), 5.25—5.22(m, 1H, 3'-OH), 5.21—5.12(m, 1H, 6'-OH), 5.03—5.02(m, 1H, 2'-OH), 4.68—4.66(m, 1H, 4'-OH), 4.58—4.41(m, 2H, 1-H), 3.91—4.06(m, 2H, 2COCHN), 3.65—3.63(m, 1H, 4'-H), 3.46—3.44(m, 1H, 2'-H), 3.19—3.17(m, 1H, 5'-H), 3.16—3.14(m, 1H, 3'-H), 3.12—3.09(m, 2H, 6'-H), 2.23—2.12(m, 6H, 2CH, 2CH2), 1.60—1.61(m, 4H, CH), 1.28—1.21(m, 24H, 12CH2), 0.91—0.86(m, 18H, 6CH3) | 173.71(NHC=O), 173.57(NHC=O), 172.93(C=O), 172.69(C=O), 96.08(C1'),80.08(C2'), 79.10(C2), 77.38(C3'), 73.65(C4'), 70.55(C5'), 61.67(C3), 57.26(C1), 48.38(C6'), 36.14, 32.44, 30.07, 29.98, 29.84, 29.74, 29.70, 26.53, 26.33, 23.27(CH2),18.16, 18.13 15.10(CH3) |
| 32 | 8.29—8.24(m, 2H, 2NH), 5.63—5.62(m, 1H, 1'-H), 5.38—5.36(m, 1H, 2-H), 5.29—5.28(m, 1H, 3'-OH), 5.24(s, 1H, 6'-OH), 5.16(s, 1H, 2'-OH), 5.07(s, 1H, 4'-OH), 4.70—4.42(m, 2H,1-H), 4.20(s, 1H, 4'-H), 4.17—4.15(m, 1H, 3a-H), 4.11—4.09(m, 1H, 2'-H), 3.95—3.87(m, 1H, 3b-H), 3.80—3.75(m, 1H, 3'-H), 3.49—3.47(m, 2H, 2COCHNH), 3.29—3.25(m, 1H, 5'-H), 3.18—3.16(m, 2H, 6'-H), 2.26—2.15(m, 6H, 2CH, 2CH2), 1.60—1.62(m, 4H, 2CH2), 1.28—1.25(m, 32H, 16CH2), 0.92—0.87(m, 18H, 6CH3) | 173.72(NHC=O), 173.58(NHC=O), 172.82(C=O), 172.59(C=O), 95.97(C1'),80.09(C2'), 78.92(C2), 77.25(C3'), 73.48(C4'), 70.51(C5'), 61.60(C3), 57.16(C1), 48.20(C6'), 36.10, 32.34, 30.07, 30.06, 29.98, 29.84, 29.76, 29.58, 26.24, 23.14, 19.50, 18.02, 14.99 |
| 33 | 8.24—8.19(m, 2H, 2NH), 5.81—5.62(m, 1H, 1'-H), 5.33—5.30(m, 1H, 2-H), 5.25—5.24(m, 1H, 3'-OH), 5.21—5.20(m, 1H, 6'-OH), 5.12—5.10(m, 1H, 2'-OH), 4.67—4.65(m, 1H, 4'-OH), 4.57—4.56(m, 2H, 1-H), 4.30—4.28(m, 1H, 1H, 4'-H), 4.10—4.04(m, 2H, 3-H), 4.01—3.83(m, 2H, 2COCHNH), 3.65—3.63(m, 1H, 2'-H), 3.45—3.43(m, 1H, 5'-H), 3.21—3.20(m, 1H, 3'-H), 3.12—3.11(m, 2H, 6'-H), 2.16—2.06(m, 6H, 2CH, 2CH2), 1.54—1.56(m, 4H, 2CH2), 1.25—1.22(m, 40H, 20CH2), 0.84—0.79(m, 18H, 6CH3) | 173.62(NHC=O), 173.45(NHC=O), 173.05(C=O), 172.78(C=O), 96.23(C1'),81.25(C2'), 79.24(C2), 77.56(C3'), 73.89(C4'), 70.50(C5'), 61.84(C3), 57.33(C1), 48.55(C6'), 36.35, 36.26, 32.59, 30.33, 30.24, 30.11, 30.00, 29.87, 26.47, 23.38, 19.82, 18.36, 18.32, 14.73(CH3) |
| 34 | 8.23—8.20(m, 2H, 2NH), 5.57—5.49(m, 1H, 1'-H), 5.30—5.24(m, 1H, 2-H), 5.22—5.21(m, 1H, 3'-OH), 5.11—5.02(m, 1H, 6'-OH), 4.68—4.66(m, 1H, 2'-OH), 4.58—4.56(m, 1H, 4'-OH), 4.38—4.30(m, 1H, 2-H), 4.12—4.05(m, 2H, 1-H), 3.98—3.82(m, 2H, 2CH), 3.65—3.64(m, 1H, 4'-H), 3.46—3.44(m, 1H, 2'-H), 3.22—3.21(m, 1H, 5'-H), 3.19—3.16(m, 1H, 3'-H), 3.12—3.09(m, 2H, 6'-H), 2.23—2.19(m, 2H, 2CH, 2.19—2.12(m, 4H, 2CH2), 1.61—1.60(m, 4H, 2CH2), 1.28—1.20(m, 48H, 24CH2), 0.82—0.93(m, 18H, 6CH3) | 173.56(NHC=O), 173.45(NHC=O), 172.58(C=O), 172.36(C=O), 95.70(C1'),81.32(C2'), 78.62(C2), 76.96(C3'), 73.27(C4'), 70.24(C5'), 61.84(C3), 56.95(C1), 48.08(C6'), 35.85, 35.78, 32.10, 29.85, 29.76, 29.62, 29.51, 29.39, 29.35, 25.99, 22.90, 19.19, 17.71 14.70 |
Table 5 1H NMR and 13C NMR data of compounds 27—34
| Compd. | 1H NMR(500 MHz, DMSO), δ | 13C NMR(125 MHz, DMSO), δ |
|---|---|---|
| 27 | 8.21—8.23(m, 2H, 2NH), 5.12—5.05(m, 1H, 1'-H),5.04—4.88(m, 1H, 2-H), 4.88—4.78(m, 2H, 1-H), 4.69—4.62(m, 2H, 1COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.38—4.36(m, 1H, 2'-H), 4.25—4.06(m, 2H, 1COCH2NH), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.16—3.07(m, 2H, 6'-H), 2.11—2.09(m, 4H, 2NHC=OCH2), 1.48—1.46(m, 4H, CH2), 1.23—1.21(m, 24H, 12CH2), 0.85—0.82(m, 6H, 2CH3) | 173.22(NHC=O), 173.19(NHC=O),169.47(C=O), 169.45(C=O), 95.15(C1'), 79.26(C2'), 78.31(C2), 76.59(C3'), 72.83(C4'), 69.72(C5'), 67.74(C3), 64.95(C1), 60.80(C6'), 35.46, 31.67, 29.42, 29.33, 29.20, 29.08, 28.96, 25.56, 22.46, 14.30 |
| 28 | 8.23—8.21(m, 2H, 2NH), 5.72—5.71(m, 1H, 2-H), 5.36—5.34(m, 1H, 1'-H), 5.27—5.26(m, 1H, 3'-OH), 5.11—5.10(m, 1H, 4'-OH), 5.02—5.01(m, 1H, 2'-OH), 4.63—4.60(m, 1H, 6'-OH), 4.39—4.37(m, 2H, 1-H), 4.25—4.21(m, 2H, 1COCH2NH), 4.20—4.18(m, 1H, 4'-H), 3.90—3.87(m, 2H, 1COCH2NH), 3.85—3.83(m, lH, 3-H), 3.81—3.73(m, 1H, 2'-H), 3.65—3.62(m, 1H, 5'-H), 3.47—3.43(m, 1H, 3'-H), 3.22—3.21(m, 2H, 6'-H), 2.24—2.13(m, 4H, 2NHC=OCH2), 1.67—1.59(m, 4H, NHC=OCH2CH2), 1.26—1.25(m, 32H, 16CH2), 0.89—0.86(m, 6H, 2CH3) | 173.53(NHC=O), 173.42(NHC=O),171.53(C=O), 171.52(C=O), 95.67(C1'), 78.64(C2'), 78.59(C2), 76.94(C3'), 73.30(C4'), 73.19(C5'), 70.16(C3), 61.25(C1), 56.92(C6'), 39.48, 35.83, 35.76, 32.08, 29.82, 29.73, 29.60, 29.48, 29.36, 29.33, 25.97, 22.87(CH2), 17.69, 14.68(CH3) |
| 29 | 8.25—8.24(m, 2H, 2NH), 5.12—5.05(m, 1H, 1'-H), 4.96—4.94(m, 1H, 2-H), 4.89—4.87(m, 2H, 1-H), 4.78—4.70(m, 2H, 1COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.44—4.39(m, 1H, 2'-H), 4.25—4.06(m, 1H, 1COCH2NH), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.15—3.05(m, 2H, 6'-H), 2.09—2.06(m, | 173.29(NHC=O), 173.24(NHC=O), 169.55(C=O), 169.50(C=O), 95.24(C1'),79.55(C2'), 78.41(C2), 76.70(C3'), 72.93(C4'), 70.25(C5'), 68.82(C3), 60.90(C1), 56.96(C6'), 35.55, 31.76, |
| 29 | 4H, 2NHC=OCH2), 1.46—1.45(m, 4H, CH2), 1.28—1.25(m, 40H, 20CH2), 0.84—0.82(m, 6H, 2CH3) | 29.52, 29.47, 29.41, 29.29, 29.16, 29.06, 25.65, 22.55, 14.41 |
| 30 | 8.21—8.23(m, 2H, 2NH), 5.11—5.03(m, 1H, 1'-H), 4.95—4.93(m, 1H, 2-H), 4.82—4.70(m, 2H, 1-H), 4.69—4.62(m, 4H, 2COCH2NH), 4.60—4.51(m, 1H, 4'-H), 4.50—4.47(m, lH, 3-H), 4.38—4.36(m, 1H, 2'-H), 3.81—3.80(m, 1H, 5'-H), 3.59—3.58(m, 1H, 3'-H), 3.16—3.07(m, 2H, 6'-H), 2.11—2.09(m, 4H, 2CH2), 1.48—1.46(m, 4H, CH2), 1.23—1.21(m, 48H, 24CH2), 0.85—0.82(m, 6H, 2CH3) | 173.29(NHC=O), 173.24(NHC=O), 169.54(C=O), 169.22(C=O), 95.24(C1'),80.01(C2'), 78.40(C2), 76.68(C3'), 72.92(C4'), 69.82(C5'), 68.57(C3), 60.89(C1), 56.77(C6'), 35.55, 31.76, 29.52, 29.47, 29.41, 29.29, 29.16, 29.06, 25.65, 22.55, 14.41 |
| 31 | 8.25—8.20(m, 2H, 2NH), 5.58—5.51(m, 1H, 1'-H), 5.34—5.30(m, 1H, 2-H), 5.25—5.22(m, 1H, 3'-OH), 5.21—5.12(m, 1H, 6'-OH), 5.03—5.02(m, 1H, 2'-OH), 4.68—4.66(m, 1H, 4'-OH), 4.58—4.41(m, 2H, 1-H), 3.91—4.06(m, 2H, 2COCHN), 3.65—3.63(m, 1H, 4'-H), 3.46—3.44(m, 1H, 2'-H), 3.19—3.17(m, 1H, 5'-H), 3.16—3.14(m, 1H, 3'-H), 3.12—3.09(m, 2H, 6'-H), 2.23—2.12(m, 6H, 2CH, 2CH2), 1.60—1.61(m, 4H, CH), 1.28—1.21(m, 24H, 12CH2), 0.91—0.86(m, 18H, 6CH3) | 173.71(NHC=O), 173.57(NHC=O), 172.93(C=O), 172.69(C=O), 96.08(C1'),80.08(C2'), 79.10(C2), 77.38(C3'), 73.65(C4'), 70.55(C5'), 61.67(C3), 57.26(C1), 48.38(C6'), 36.14, 32.44, 30.07, 29.98, 29.84, 29.74, 29.70, 26.53, 26.33, 23.27(CH2),18.16, 18.13 15.10(CH3) |
| 32 | 8.29—8.24(m, 2H, 2NH), 5.63—5.62(m, 1H, 1'-H), 5.38—5.36(m, 1H, 2-H), 5.29—5.28(m, 1H, 3'-OH), 5.24(s, 1H, 6'-OH), 5.16(s, 1H, 2'-OH), 5.07(s, 1H, 4'-OH), 4.70—4.42(m, 2H,1-H), 4.20(s, 1H, 4'-H), 4.17—4.15(m, 1H, 3a-H), 4.11—4.09(m, 1H, 2'-H), 3.95—3.87(m, 1H, 3b-H), 3.80—3.75(m, 1H, 3'-H), 3.49—3.47(m, 2H, 2COCHNH), 3.29—3.25(m, 1H, 5'-H), 3.18—3.16(m, 2H, 6'-H), 2.26—2.15(m, 6H, 2CH, 2CH2), 1.60—1.62(m, 4H, 2CH2), 1.28—1.25(m, 32H, 16CH2), 0.92—0.87(m, 18H, 6CH3) | 173.72(NHC=O), 173.58(NHC=O), 172.82(C=O), 172.59(C=O), 95.97(C1'),80.09(C2'), 78.92(C2), 77.25(C3'), 73.48(C4'), 70.51(C5'), 61.60(C3), 57.16(C1), 48.20(C6'), 36.10, 32.34, 30.07, 30.06, 29.98, 29.84, 29.76, 29.58, 26.24, 23.14, 19.50, 18.02, 14.99 |
| 33 | 8.24—8.19(m, 2H, 2NH), 5.81—5.62(m, 1H, 1'-H), 5.33—5.30(m, 1H, 2-H), 5.25—5.24(m, 1H, 3'-OH), 5.21—5.20(m, 1H, 6'-OH), 5.12—5.10(m, 1H, 2'-OH), 4.67—4.65(m, 1H, 4'-OH), 4.57—4.56(m, 2H, 1-H), 4.30—4.28(m, 1H, 1H, 4'-H), 4.10—4.04(m, 2H, 3-H), 4.01—3.83(m, 2H, 2COCHNH), 3.65—3.63(m, 1H, 2'-H), 3.45—3.43(m, 1H, 5'-H), 3.21—3.20(m, 1H, 3'-H), 3.12—3.11(m, 2H, 6'-H), 2.16—2.06(m, 6H, 2CH, 2CH2), 1.54—1.56(m, 4H, 2CH2), 1.25—1.22(m, 40H, 20CH2), 0.84—0.79(m, 18H, 6CH3) | 173.62(NHC=O), 173.45(NHC=O), 173.05(C=O), 172.78(C=O), 96.23(C1'),81.25(C2'), 79.24(C2), 77.56(C3'), 73.89(C4'), 70.50(C5'), 61.84(C3), 57.33(C1), 48.55(C6'), 36.35, 36.26, 32.59, 30.33, 30.24, 30.11, 30.00, 29.87, 26.47, 23.38, 19.82, 18.36, 18.32, 14.73(CH3) |
| 34 | 8.23—8.20(m, 2H, 2NH), 5.57—5.49(m, 1H, 1'-H), 5.30—5.24(m, 1H, 2-H), 5.22—5.21(m, 1H, 3'-OH), 5.11—5.02(m, 1H, 6'-OH), 4.68—4.66(m, 1H, 2'-OH), 4.58—4.56(m, 1H, 4'-OH), 4.38—4.30(m, 1H, 2-H), 4.12—4.05(m, 2H, 1-H), 3.98—3.82(m, 2H, 2CH), 3.65—3.64(m, 1H, 4'-H), 3.46—3.44(m, 1H, 2'-H), 3.22—3.21(m, 1H, 5'-H), 3.19—3.16(m, 1H, 3'-H), 3.12—3.09(m, 2H, 6'-H), 2.23—2.19(m, 2H, 2CH, 2.19—2.12(m, 4H, 2CH2), 1.61—1.60(m, 4H, 2CH2), 1.28—1.20(m, 48H, 24CH2), 0.82—0.93(m, 18H, 6CH3) | 173.56(NHC=O), 173.45(NHC=O), 172.58(C=O), 172.36(C=O), 95.70(C1'),81.32(C2'), 78.62(C2), 76.96(C3'), 73.27(C4'), 70.24(C5'), 61.84(C3), 56.95(C1), 48.08(C6'), 35.85, 35.78, 32.10, 29.85, 29.76, 29.62, 29.51, 29.39, 29.35, 25.99, 22.90, 19.19, 17.71 14.70 |
| m(H2O)/m(DMF) | Yield(%) | |||
|---|---|---|---|---|
| 90 ℃ | 110 ℃ | 120 ℃ | 130 ℃ | |
| 0.01 | 58.9 | 64.5 | 61.7 | 54.4 |
| 0.02 | 69.8 | 71.3 | 64.2 | 63.1 |
| 0.03 | 70.3 | 73.7 | 67.6 | 66.2 |
| 0.04 | 68.3 | 73.2 | 63.2 | 62.9 |
| 0.05 | 63.1 | 70.9 | 61.8 | 55.5 |
| 0.06 | 60.8 | 70.3 | 61.5 | 58.7 |
Table 6 Yield of compound 18 in the solvent H2O/DMF at different temperatures
| m(H2O)/m(DMF) | Yield(%) | |||
|---|---|---|---|---|
| 90 ℃ | 110 ℃ | 120 ℃ | 130 ℃ | |
| 0.01 | 58.9 | 64.5 | 61.7 | 54.4 |
| 0.02 | 69.8 | 71.3 | 64.2 | 63.1 |
| 0.03 | 70.3 | 73.7 | 67.6 | 66.2 |
| 0.04 | 68.3 | 73.2 | 63.2 | 62.9 |
| 0.05 | 63.1 | 70.9 | 61.8 | 55.5 |
| 0.06 | 60.8 | 70.3 | 61.5 | 58.7 |
| Compd. | IC50/(μmol·L-1) | ||||||
|---|---|---|---|---|---|---|---|
| MCF-7 | HcpG-2 | K562 | HL7702 | H9C2 | EVC304 | ||
| 27 | 3.53 | 4.71 | 4.13 | >100 | >100 | >100 | |
| 28 | 4.35 | 4.57 | 5.79 | >100 | NT | >100 | |
| 29 | 12.98 | 25.17 | 28.93 | NT | >100 | >100 | |
| 30 | 27.81 | 34.73 | 41.32 | >100 | >100 | NT | |
| 31 | 3.91 | 3.73 | 4.54 | >100 | NT | >100 | |
| 32 | 5.17 | 5.52 | 5.93 | NT | >100 | NT | |
| 33 | 17.79 | 41.39 | 58.31 | NT | >100 | NT | |
| 34 | 37.57 | >60 | >60 | >100 | NT | NT | |
Table 7 Anti-tumor activities of compounds 27—34 against cancer and normal cell lines*
| Compd. | IC50/(μmol·L-1) | ||||||
|---|---|---|---|---|---|---|---|
| MCF-7 | HcpG-2 | K562 | HL7702 | H9C2 | EVC304 | ||
| 27 | 3.53 | 4.71 | 4.13 | >100 | >100 | >100 | |
| 28 | 4.35 | 4.57 | 5.79 | >100 | NT | >100 | |
| 29 | 12.98 | 25.17 | 28.93 | NT | >100 | >100 | |
| 30 | 27.81 | 34.73 | 41.32 | >100 | >100 | NT | |
| 31 | 3.91 | 3.73 | 4.54 | >100 | NT | >100 | |
| 32 | 5.17 | 5.52 | 5.93 | NT | >100 | NT | |
| 33 | 17.79 | 41.39 | 58.31 | NT | >100 | NT | |
| 34 | 37.57 | >60 | >60 | >100 | NT | NT | |
| [1] | Hernandez M. P., Kanavouras A., Lagaron J. M., Gavara R., J. Agric. Food Chem., 2005, 28(53), 8216—8223 |
| [2] | Sun S. M., Song Y. H., Zheng Q., Food Hydrocolloids,2007, 21, 992—1005 |
| [3] | Lin C. C., Lin H. Y., Chen H. C., Yu M. W., Lee M. H., Food Chem., 2009, 116, 923—928 |
| [4] | Wu Z., Fu X. L., Yang N., Wang Q. A., Chem. Res. Chinese Universities,2013, 29(3), 460—465 |
| [5] | An D., Zhao X. H., Ye Z. W., Carbohyd. Res., 2015, 414(23), 32—38 |
| [6] | Alexandra L. C., Paolo C., Pasquale S., Dominique L., Denis B., Florian A., Peter S., Org. Biomol. Chem., 2014, 12(33), 6281—6534 |
| [7] | Pahlsson P., Spitalnik S. L., Areh. Biochem. Bio. Phys., 2001, 396(2), 187—198 |
| [8] | Hou D. X., Curr. Mol. Med., 2003, 3, 149—159 |
| [9] | Zhao J. Z., Zhang X. Q., Wu X., Xing Z. B., Yue A. Q., Shao H. W., Chem. Res. Chinese Universities,2013, 29(1), 71—75 |
| [10] | Zhang X., Dean K., Hoobin P., Burgar I., Do M. D., Biomacromolecules, 2005, 6, 1661—1671 |
| [11] | Malik M., Zhao C., Schoene N., Guisti M. M., Moyer M. P., Magnuson B. A., Nutr. Cancer,2003, 46, 186—196 |
| [12] | Shosharma F., Stutzman J. D., Kelloff G. J., Steele V. E., Cancer Res., 2004, 54, 5848—5855 |
| [13] | Shosharma F., Vered R., J. Nat. Prod., 2011, 61, 891—895 |
| [14] | Colombo D., Franchini L., Toma L., Ronchetti F., J. Med. Chem., 2011, 41, 1456—1463 |
| [15] | Roelants S. L., Saerens K. M., Derycke T., Biotechnol. Bioeng., 2013, 110(9), 2494—2503 |
| [16] | Van B. N., Holvoet K., Roeilants S. L., Mol. Micmbiol., 2013, 88(3), 501—509 |
| [17] | Qian W. H., Zheng Z. Z., Peng H., Anal. Biochem., 2012, 49(2), 161—166 |
| [18] | Stefanie W., Francesca B., Addolorata S., Cazzato M. K., Wolfgang L., Anal. Bioanal. Chem., 2013, 405, 8011—8026 |
| [19] | Michel G., Christian B., Carl B., Swindell M., Tingol J., Bioorg. Med. Chem. Lett., 2011, 21, 288—293 |
| [20] | María A., Fernández H., Hugo L. M., José M. V., Hernández V., Mario P., Jesús S. R., Eur. J. Med. Chem., 2011, 46, 3877—3886 |
| [21] | Amy S.C., Arthur C., Michael D., Org. Biomol. Chem., 2014, 12, 1919—1934 |
| [22] | Liu C., Zheng Y. Y., Hong F., Hu J. D., Zhao W. N., Yuan Y. F., Shao J. W., Wang W. F., Chem. J. Chinese Universities,2013, 34(6), 1408—1415 |
| (刘诚, 郑艳艳, 洪芳, 胡建达, 赵文娜, 袁耀锋, 邵敬伟, 王文峰. 高等学校化学学报,2013, 34(6), 1408—1415) | |
| [23] | Aoife S., Patricia N., Gary H., Jin W., Helmi Z., Wang Z. Y., Bioorg. Med. Chem. Lett., 2011, 21, 288—293 |
| [24] | Tiago A. D., Radim H., Gilbert K., Ana M. F., Oliveira C., Ligia M., Arkivoc, 2012, 4, 185—193 |
| [1] | 李志光, 齐国栋, 徐君, 邓风. Sn-Al-β分子筛酸性在葡萄糖转化反应中作用的固体NMR研究[J]. 高等学校化学学报, 2022, 43(6): 20220138. |
| [2] | 曾晛阳, 赵熹, 黄旭日. 细胞松弛素B对葡萄糖/质子共转运蛋白GlcPSe的抑制机理[J]. 高等学校化学学报, 2022, 43(4): 20210822. |
| [3] | 田雪琴, 莫争, 丁鑫, 武鹏彦, 王雨, 王健. 方胺功能化荧光金属-有机框架材料的制备及对组氨酸的识别研究[J]. 高等学校化学学报, 2022, 43(2): 20210589. |
| [4] | 袁春玲, 姚晓条, 徐远金, 覃秀, 石睿, 成诗琦, 王益林. 双功能碳点用于葡萄糖的比色/比率荧光测定[J]. 高等学校化学学报, 2021, 42(8): 2428. |
| [5] | 李柳, 孙仕勇, 吕瑞, GOLUBEVYevgeny Aleksandrovich, 王可, 董发勤, 段涛, KOTOVAOlga Borisovna, KOTOVAElena Leonidovna. 铁氨基黏土-葡萄糖氧化酶纳米复合催化剂的构筑及多酶级联反应研究[J]. 高等学校化学学报, 2021, 42(3): 803. |
| [6] | 王柏纯, 袁雨欣, 闫迎华, 丁传凡, 唐科奇. 葡萄糖-6-磷酸功能化亲水磁探针:有效分离富集糖肽/磷酸肽的双用途亲和材料[J]. 高等学校化学学报, 2021, 42(10): 3062. |
| [7] | 丁鑫, 师红东, 刘扬中. 以人血清白蛋白为载体的Ru(Ⅲ)和全反式维甲酸共运输纳米药物的构建及抗肿瘤转移作用[J]. 高等学校化学学报, 2021, 42(10): 3040. |
| [8] | 王欢, 所金泉, 王春艳, 王润伟. 氨基化树枝状介孔二氧化硅固定葡萄糖氧化酶用于检测葡萄糖[J]. 高等学校化学学报, 2020, 41(8): 1731. |
| [9] | 张开翔, 刘军杰, 宋巧丽, 王丹钰, 史进进, 张海悦, 李景虹. 基于DNA纳米花的细胞自噬基因沉默用于增敏抗肿瘤化疗[J]. 高等学校化学学报, 2020, 41(7): 1461. |
| [10] | 叶晓栋, 齐国栋, 徐君, 邓风. Au负载SBA-15分子筛上葡萄糖氧化反应[J]. 高等学校化学学报, 2020, 41(5): 960. |
| [11] | 梁龙琪, 陈彩玲, 于影, 李雨昕, 李春光, 施展. 氨基酸包覆的YVO4∶Eu纳米粒子的合成、 发光及细胞成像性能[J]. 高等学校化学学报, 2020, 41(3): 425. |
| [12] | 白冰,王龙,徐改改,杨鹏飞,张改红,毛多斌. 西柏三烯-4,6-二醇衍生物的合成及抗肿瘤活性[J]. 高等学校化学学报, 2020, 41(3): 481. |
| [13] | 肖艳华, 张广杰, 宗良, 刘国宏, 任丽君, 董俊兴. 开口箭化学成分及抗肿瘤活性[J]. 高等学校化学学报, 2019, 40(9): 1897. |
| [14] | 张岩岩, 郝凯风, 张国强, 赵炜. 2-氰基苄基诱导的葡萄糖醛酸β糖苷键立体选择性[J]. 高等学校化学学报, 2019, 40(9): 1904. |
| [15] | 周海, 陈豪, 郭娅, 康敏. 介孔Co3O4多面体的制备及电化学性能[J]. 高等学校化学学报, 2019, 40(7): 1374. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||