

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (4): 701.doi: 10.7503/cjcu20170392
• Organic Chemistry • Previous Articles Next Articles
SHI Yanli1, LIU Yubo1, WU Sijin2, LIU Yajun1, ZHANG Jianing1, LI Wenli1,*( )
)
Received:2017-06-20
Online:2018-04-10
Published:2018-03-16
Contact:
LI Wenli
E-mail:biolwl@dlut.edu.cn
Supported by:CLC Number:
TrendMD:
SHI Yanli, LIU Yubo, WU Sijin, LIU Yajun, ZHANG Jianing, LI Wenli. Virtual Screening and Activity Verification of Novel Inhibitors of ApIV 3C Protease†[J]. Chem. J. Chinese Universities, 2018, 39(4): 701.
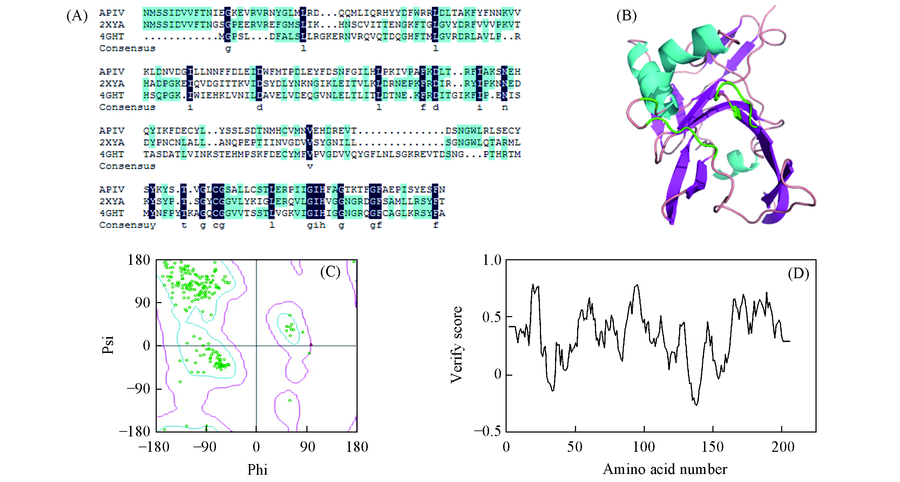
Fig.1 Homology modeling and model validation of ApIV 3C protease(A) Multiple alignment of query sequence and templates(PDB: 2XYA and 4GHT) protein sequence. Navy blue represents conserved residues; (B) Homology model(3D structure) of ApIV 3C protease after molecular dynamics(MD) simulation. The active pocket is constituted by green amino acid residues; (C) the romachandram plot of ApIV 3C protease model; (D) the profile 3D model of ApIV 3C protease.
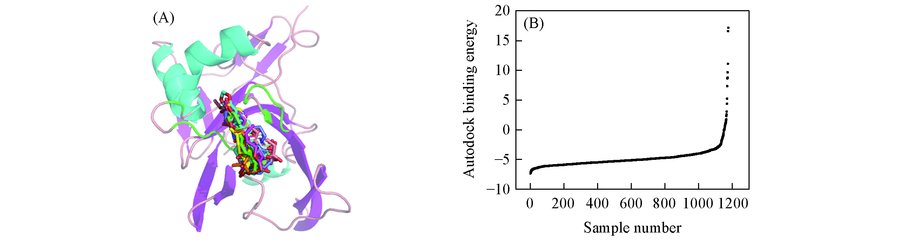
Fig.2 Virtual screening and molecular docking(A) Binding energy distribution of the docking results about liginds; (B) binding mode of fifteen hit compounds in the ApIV 3C protease model by docking studies using AutoDock.
| No. | Compd. | Biological activity | Dock-Score | Interaction residues | ||
|---|---|---|---|---|---|---|
| AutoDock | LibDock | HBond | π-π Bond | |||
| 1 | Orobol | Hypotensivel, enzyme inhibitor | -6.4 | 114.345 | Gly167, Ser164 | Tyr163 |
| 2 | Tanshinlactone | Anti-tumor | -6.1 | 99.7829 | - | Tyr163 |
| 3 | Maackiain | Antifungal | -5.6 | 81.7844 | Cys123 | Cys123 |
| 4 | (-)-Epiafzelechin | Antioxidant, enzyme inhibitor, anti-inflammatory | -5.4 | 83.1081 | - | Val166 |
| 5 | 2-α,3-α,23-Trihyd-roxy- olean-12-en-28-oic acid | Anti-inflammatory | -5.1 | 78.322 | Cys123 | Tyr163 |
| 6 | (+)-Conocarpan | Analgesic, antifungal | -5.3 | 80.6224 | - | Glu112, Tyr163 |
| 7 | Sakuranetin | Phytoalexin | -5.3 | 80.6224 | - | Tyr163 |
| 8 | Dehydro-α-lapachone | Antivascular, antifungal | -5.3 | 84.7844 | - | - |
| 9 | Ilicic acid | Anti-inflammatory | -5.2 | 82.089 | - | - |
| 10 | Naringenin | Anti-tumor, anti-cancer, antiulcer, antiatherogenic | -5.2 | 75.367 | - | Tyr163 |
| 11 | Luteolin | Anti-inflammatory, antispasmodic, antitussive, antioxidant, antiallergic | -5.2 | 78.959 | Cys123 | Tyr163 |
| 12 | Carabrone | Antifungal | -5.1 | 72.633 | - | - |
| 13 | Dehydrocostus lactone | Antibacterial, antimicrobial | -5.1 | 73.830 | - | - |
| 14 | Apigenin | Anti-tumor, anti-cancer, anti-inflammatory | -5.1 | 70.493 | - | Tyr163 |
| 15 | 1-Hydroxyrutaecarpine | Antiplatelet, aggregation | -5.1 | 75.472 | - | Tyr163 |
Table 1 Summary of virtual screening results
| No. | Compd. | Biological activity | Dock-Score | Interaction residues | ||
|---|---|---|---|---|---|---|
| AutoDock | LibDock | HBond | π-π Bond | |||
| 1 | Orobol | Hypotensivel, enzyme inhibitor | -6.4 | 114.345 | Gly167, Ser164 | Tyr163 |
| 2 | Tanshinlactone | Anti-tumor | -6.1 | 99.7829 | - | Tyr163 |
| 3 | Maackiain | Antifungal | -5.6 | 81.7844 | Cys123 | Cys123 |
| 4 | (-)-Epiafzelechin | Antioxidant, enzyme inhibitor, anti-inflammatory | -5.4 | 83.1081 | - | Val166 |
| 5 | 2-α,3-α,23-Trihyd-roxy- olean-12-en-28-oic acid | Anti-inflammatory | -5.1 | 78.322 | Cys123 | Tyr163 |
| 6 | (+)-Conocarpan | Analgesic, antifungal | -5.3 | 80.6224 | - | Glu112, Tyr163 |
| 7 | Sakuranetin | Phytoalexin | -5.3 | 80.6224 | - | Tyr163 |
| 8 | Dehydro-α-lapachone | Antivascular, antifungal | -5.3 | 84.7844 | - | - |
| 9 | Ilicic acid | Anti-inflammatory | -5.2 | 82.089 | - | - |
| 10 | Naringenin | Anti-tumor, anti-cancer, antiulcer, antiatherogenic | -5.2 | 75.367 | - | Tyr163 |
| 11 | Luteolin | Anti-inflammatory, antispasmodic, antitussive, antioxidant, antiallergic | -5.2 | 78.959 | Cys123 | Tyr163 |
| 12 | Carabrone | Antifungal | -5.1 | 72.633 | - | - |
| 13 | Dehydrocostus lactone | Antibacterial, antimicrobial | -5.1 | 73.830 | - | - |
| 14 | Apigenin | Anti-tumor, anti-cancer, anti-inflammatory | -5.1 | 70.493 | - | Tyr163 |
| 15 | 1-Hydroxyrutaecarpine | Antiplatelet, aggregation | -5.1 | 75.472 | - | Tyr163 |
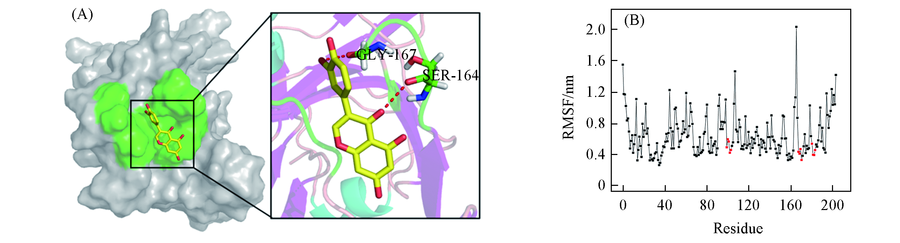
Fig.3 Analysis of binding mode between orobol and ApIV 3C protease(A) Binding mode of orobol with ApIV 3C protease by molecular docking studies using AutoDock. In the solid surface, the pocket(green) is labeled. Local amplify docking interaction structure of orobol along with hydrogen bonds; (B) the root mean square fluctuation(RMSF) of ApIV 3C protease model over 20 ns molecular dynamics(MD) simulation. The red dot represents the amino acid residue in the active site procket.
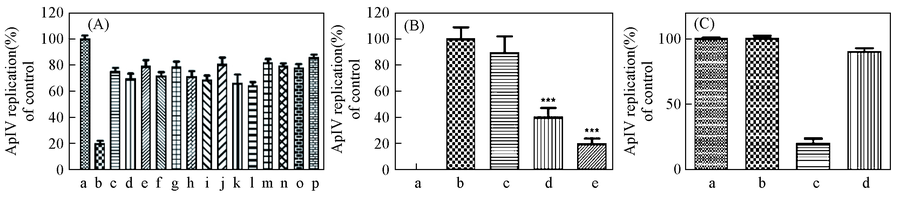
Fig.4 Assays of antiviral activity and cell cytotoxicity in vitro(A) q-PCR analysis of the effect of 15 compounds at 1 μg/mL concentration for ApIV replication. a. Control, b. 1, c. 2, d. 3, e. 4, f. 5, g. 6, h. 7, i. 8, j. 9, k. 10, l. 11, m. 12, n. 13, o. 14, p. 15; (B) q-PCR analysis of the effect of Orobol for ApIV replication. a. negative control, b. positive control, c(orobal)/(μg·mL-1): c. 0.1, d. 1, e. 10; (C) the analysis of cell cytotoxicity for Orobol. a. blank; b. 10 μg/mL orobol; c. ApIV; d. ApIV+10 μg/mL orobol.
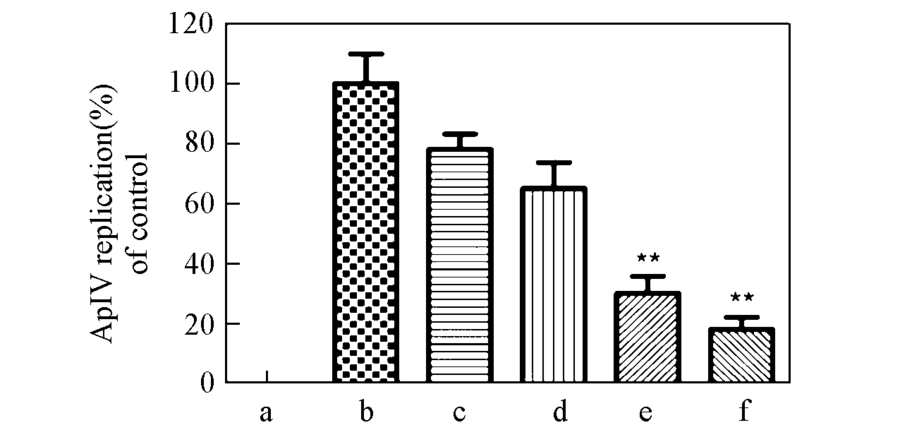
Fig.5 q-PCR analysis of the effect of orobol for ApIV replicationa. negative control; b. positive control; c(orobal)/(μg·mL-1): c. 0.1; d. 1; e. 10; F. 100.
| [1] | Wu P. Y., Wei C. G.,Liaoning Agricultural Sciences, 1995, (5), 12-17 |
| (吴佩玉, 魏成贵. 辽宁农业科学, 1995, (5), 12-17) | |
| [2] | Zhang X. Z., Sun F. Y.,Science of Sericulture, 1980, (6), 187-191 |
| (张秀珍, 孙芳义. 蚕业科学, 1980, (6), 187-191) | |
| [3] | Ma L. F., Hu F. Z.,Liaoning Agricultural Sciences, 2012, (5), 39-40 |
| (马连福, 胡杏枝. 辽宁农业科学, 2012, (5), 39-40) | |
| [4] | Geng P., Li W., Lin L., de Miranda J. R., Emrich S., An L., Terenius O., PLoS One, 2014, 9, 92-107 |
| [5] | Wang X., Zhang J., Liu C., Yu H., Hu Y., Virologica Sinica, 2003, 19(1), 39-42 |
| [6] | Ongus J. R., Peters D., Bonmatin J. M., Bengsch E., Vlak J. M., van Oers M. M., The Journal of General Virology,2004, 85(12), 3747-3755 |
| [7] | Chu L., Choy W. Y., Tsai S. N., Rao Z. H., Ngai S. M., Protein Science, 2006, 15, 699-709 |
| [8] | Liljas L., Tate J., Lin T., Christian P., Johnson J. E., Archives of Virology, 2002, 147(1), 59-84 |
| [9] | Kim B. K., Ko H., Jeon E. S., Ju E. S., Jeong L. S., Kim Y. C., Euro. J. Med. Chem., 2016, 120, 202-216 |
| [10] | Ye S., Xia H., Dong C., Cheng Z., Xia X., Zhang J., Zhou X., Hu Y., Virology,2012, 428(2), 136-145 |
| [11] | Kim B. K., Cho J. H., Jeong P., Lee Y., Lim J. J., Park K. R., Eom S. H., Kim Y. C., Kim B. K., FEBS Letters, 2015, 589(15), 1795-1801 |
| [12] | Weerawarna P. M., Kim Y., Kankanamalage A. C. G., Damalanka V. C., Lushington G. H., Alliston K. R., Mehzabeen N., Battaile K. P., Lovell S., Chang K., Groutas W. C., Euro. J. Med. Chem., 2016, 119, 300-318 |
| [13] | Ma G., Ye Y., Zhang D., Xu X., Si P., Peng J., Xiao Y., Cao R., Yin Y., Chen J., Zhao L. , Zhou Y. , Zhong W. , Liu H. , Luo X. , Chen L. , Shen X. , Euro. J. Med. Chem., 2016, 124, 981-991 |
| [14] | Kim B. K., Ko H., Jeon E. S., Ju E. S., Jeong L. S., Kim Y. C., Euro. J. Med. Chem., 2016, 120, 202-216 |
| [15] | Zhai Y., Ma Y., Ma F., Nie Q., Ren X., Wang Y., Shang L., Yin Z., Euro. J. Med. Chem., 2016, 124, 559-573 |
| [16] | Zeng D., Ma Y., Zhang R., Nie Q., Cui Z., Wang Y., Shang L., Yin Z., Bioorg. Med. Chem. Lett., 2016, 26(7), 1762-1766 |
| [17] | Kawatkar S. P., Gagnon M., Hoesch V., Tiong-Yip C., Johnson K., Ek M., Nilsson E., Lister T., Olsson L., Patel J., Yu Q., Med. Chem. Lett., 2016, 26(14), 3248-3252 |
| [18] | Kuo C., Shie J., Fang J., Yen G., Hsu J. T., Liu H., Liang P., Bioorg. Med. Chem., 2008, 16(15), 7388-7398 |
| [19] | Konno H., Wakabayashi M., Takanuma D., Saito Y., Akaji K., Bioorg. Med. Chem., 2016, 24(13), 3035-3042 |
| [20] | Kumar V., Tan K. P., Wang Y. M., Lin S. W., Liang P. H., Bioorg. Med. Chem., 2016, 24(6), 1241-1254 |
| [21] | Mohd H. B., Shazi S., Asad U. K., Medicinal Chemistry Research, 2012, 21, 2229-2237 |
| [22] | Xie Y. F., Jiang Y. R., Pan Y. F., Li C. J., Journal of Central South University of Technology,2012, 19, 3033-3039 |
| [23] | Vivek K., Manjunath G., Kinjal P., Gulamnizami Q., Srumil S., Biomedicine & Pharmacotherapy, 2005, 74, 42-48 |
| [24] | Temml V., Kaserer T., Kutil Z., Landa P., Vanek T., Schuster D., Future Medicinal Chemistry, 2005, 6(17), 1869-1881 |
| [25] | Surekha K., Nachiappan M., Prabhu D., Choubey S. K., Biswal J., Jeyakanthan J., Journal of Molecular Structure, 2017, 1127, 675-688 |
| [26] | Sandeep G., Nagasree K. P., Hanisha M., Kumar M. M., BMC Research Notes, 2011, 4(1), 445-445 |
| [27] | Zhang R. B., Lu J. R., Liu J. B., Mu J. B., Yang X. Y., Wang H. Y., Wang M. J., Zhang H., Zhang M., Chem. J. Chinese Universities, 2015, 36(8), 1521-1529 |
| (张瑞波, 卢俊瑞, 刘金彪, 穆江蓓, 杨旭云, 王宏韫, 王美君, 张贺, 张玫. 高等学校化学学报, 2015, 36(8), 1521-1529) | |
| [28] | Zhang L., Li H. F., Chen P., Sun W. B., Yan P. F., Chem. Res. Chinese Universities, 2016, 32(4), 534-538 |
| [29] | Chen C., Wang P., Zou L. W., Yang L., Fan Y. M., Hu W. Z., Ge G. B., Chem. Res. Chinese Universities, 2017, 33(2), 194-199 |
| [30] | Wu C., Cai Q., Chen C., Li N., Peng X., Cai Y., Yin K., Chen X., Wang X., Zhang R., Liu L., Chen S., Li J., Lin T., Acta Crystallographica Section D-Biological Crystallography,2013, 69(Pt 5), 866-871 |
| [31] | Hirai M., Terenius O., Li W., Faye I., Insect Molecular Biology, 2004, 13(4), 399-405 |
| [1] | HUAN Xinyu, LAI Ganqiang, HUANG Yue, YANG Caiguang. Research Progress on Chemical Intervention of N6-methyladenosine Modification [J]. Chem. J. Chinese Universities, 2022, 43(Album-4): 20220340. |
| [2] | YE Chenghao, LIANG Heng, LI Enmin, XU Liyan, LI Peng, CHEN Guanghui. High-throughput Virtual Screening of CDK2/Cyclin A2 Target Inhibitors [J]. Chem. J. Chinese Universities, 2021, 42(10): 3135. |
| [3] | YOU Yipeng, NIE Lin, LIU Jinbiao, FENG Yahui, LU Gui. Design, Synthesis and Anti-influenza Activities of Novel Neuraminidase Inhibitors† [J]. Chem. J. Chinese Universities, 2020, 41(10): 2279. |
| [4] | GAO Yang, LI Daixi, LIU Baolin, GUO Baisong, WEI Dongqing. Inhibitory Mechanism of Glycerol on the Growth of Ice Crystals by Molecular Dynamics† [J]. Chem. J. Chinese Universities, 2019, 40(4): 763. |
| [5] | ZHANG Juanrong,YOU Huimei,JING Yuxing,ZHAO Jiaowen,WANG Wei,LIU Wenxing,ZHOU Min,JIANG Zhiyong. Three New Phenolic Compounds from Salacia cochinchinensis Lour and Their α-Glucosidase Inhibitory Activities† [J]. Chem. J. Chinese Universities, 2019, 40(3): 456. |
| [6] | Yanjie LI,Ensi WANG,Xiaowei SHAO,Xingmin ZHANG,Shengxiu NIU,Lijuan YANG,Yi WU. Synthesis and Biological Activity in vitro of Imidazo[4,5-c]quinoline Derivatives † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2502. |
| [7] | Qiuchen DONG,Guanghua ZHANG,Wanbin ZHANG,Xue ZHANG,Jing LIU. Corrosion Inhibition of Q235 Steel by Ionic Liquid Based on the 2-(Dimethylamino)ethyl Methacrylate † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2556. |
| [8] | DONG Qiuchen,ZHANG Guanghua,ZHANG Wanbin,LIU Jing. Experimental and Theoretical Analysis of Quinoline Diquaternary Ammonium Salt as Corrosion Inhibitor † [J]. Chem. J. Chinese Universities, 2019, 40(10): 2195. |
| [9] | ZHANG Guanghua,DONG Qiuchen,ZHANG Wanbin,WANG Shuang. Corrosion Inhibition of Q235 Steel by Octyl Dimethyl Benzyl Quaternary Ammonium Salt Ionic Liquid† [J]. Chem. J. Chinese Universities, 2019, 40(1): 130. |
| [10] | LIU Haichun, LU Shuai, ZHANG Yanmin, ZHOU Weineng, YIN Lingfeng, ZHU Lu, ZHAO Junnan, LU Tao, CHEN Yadong. Molecular Dynamics Simulation of the Selectivity of Fedratinib Complex with JAK2/JAK3† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1540. |
| [11] | DONG Shigang, GAO Yingbo, GUAN Zichao, WANG Haipeng, WANG Xia, DU Ronggui, SONG Guangling. Inhibition Effect of Polyvinylpyrrolidone on Corrosion of Reinforcing Steel in Simulated Concrete Pore Solutions† [J]. Chem. J. Chinese Universities, 2018, 39(6): 1260. |
| [12] | LIU Yubo, ZHANG Nana, CHEN Jinjiao, ZHU Tong, ZHANG Jianing, LI Wenli. Discovery and Activity Verification of a O-GlcNAc Transferase Inhibitor by Structure-based Virtual Screening† [J]. Chem. J. Chinese Universities, 2018, 39(6): 1185. |
| [13] | LU Tong, WANG Chunyang, ZHU Zhihui, JIANG Wei, HUO Mingnan, LI Fei. Polyethyleneimine Functionalized Graphene Oxide Against hIAPP Amyloid Aggregation† [J]. Chem. J. Chinese Universities, 2018, 39(6): 1274. |
| [14] | WANG Lei, ZHENG Guojun, JI Qi, CHEN Bo, GONG Longlong, GAO Congmin, DU Zhenjian, ZHANG Xingmin. Synthesis and Biological Activity of Novel PI3K/mTOR Inhibitors† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1590. |
| [15] | DUAN Yongbin, YIN Yan, MENG Fanli, ZHAO Lianhua, LIU Yukun, YUAN Zhe, FENG Yangbo. Design, Synthesis and Biological Evaluation of Benzothiazoles as Highly Potent ROCK Inhibitors Through Molecular Docking and Free Energy Calculations† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1568. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||