

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (10): 2191.doi: 10.7503/cjcu20140310
• Physical Chemistry • Previous Articles Next Articles
XU Qiong1,*( ), WANG Rui1, ZHANG Tianlei1, ZHANG Haolin1, WANG Zhiyin1, WANG Zhuqing2
), WANG Rui1, ZHANG Tianlei1, ZHANG Haolin1, WANG Zhiyin1, WANG Zhuqing2
Received:2014-04-04
Online:2014-10-10
Published:2014-09-30
Contact:
XU Qiong
E-mail:xuq@snut.edu.cn
Supported by:CLC Number:
TrendMD:
XU Qiong, WANG Rui, ZHANG Tianlei, ZHANG Haolin, WANG Zhiyin, WANG Zhuqing. Theoretical Studies on the Single Water Molecule’s Effect on the Main Channel of H2S+HO2 Reaction†[J]. Chem. J. Chinese Universities, 2014, 35(10): 2191.
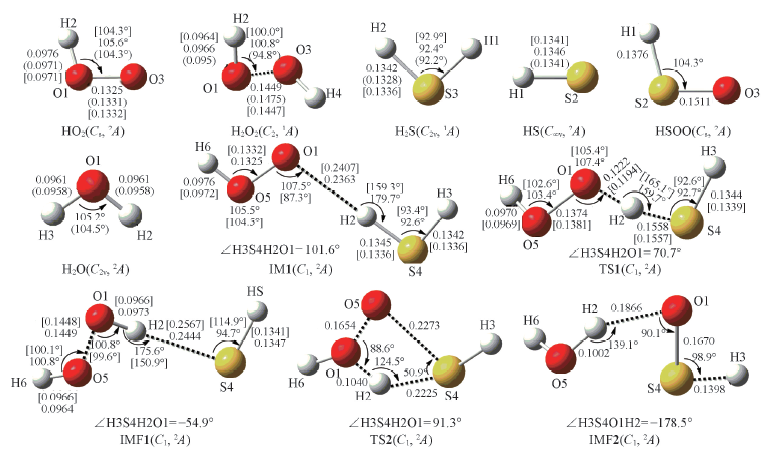
Fig.1 Optimized geometries of all the species involved in HO2+H2S reaction at B3LYP/6-311+G(2df,2p) level Bond lengths are in nm. The values with parentheses were the experimental data from the NIST chemistry webbook[27]; the values with bracket were obtained at the CCSD/aug-cc-pVTZ level of theory.
| Species | ZPE/ (kJ·mol-1) | S/(J·mol-1· K-1) | ΔE/ (kJ·mol-1) | Δ(E+ZPE)/ (kJ·mol-1) | ΔH(298)/ (kJ·mol-1) | ΔG(298)/ (kJ·mol-1) |
|---|---|---|---|---|---|---|
| HO2+H2S | 76.49 | 439.65 | 0 | 0 | 0b | 0 |
| IM1 | 80.84 | 360.23 | -8.69 | -4.35 | -3.22 | 20.48 |
| TS1 | 75.07 | 306.52 | 63.87 | 62.45 | 58.65 | 98.31 |
| IMF1 | 89.20 | 348.53 | -10.12 | 2.59 | 2.51 | 29.68 |
| H2O2+HS | 85.61 | 418.79 | 4.85 | 13.96 | 13.63, 12.96±2.93b | 19.86 |
| TS2 | 88.74 | 293.44 | 154.58 | 166.82 | 161.77 | 205.36 |
| IMF2 | 90.83 | 325.75 | -267.94 | -253.60 | -254.94 | -221.00 |
| H2O+HSO | 82.93 | 429.70 | -241.40 | -235.00 | -234.92 | -231.95 |
Table 1 Zero-point energy(ZPE), entropies(S), relative energies[ΔE and Δ(E+ ZPE)], enthalpies[ΔH(298)], and free energies [ΔG(298)] for the HO2+H2S reaction a
| Species | ZPE/ (kJ·mol-1) | S/(J·mol-1· K-1) | ΔE/ (kJ·mol-1) | Δ(E+ZPE)/ (kJ·mol-1) | ΔH(298)/ (kJ·mol-1) | ΔG(298)/ (kJ·mol-1) |
|---|---|---|---|---|---|---|
| HO2+H2S | 76.49 | 439.65 | 0 | 0 | 0b | 0 |
| IM1 | 80.84 | 360.23 | -8.69 | -4.35 | -3.22 | 20.48 |
| TS1 | 75.07 | 306.52 | 63.87 | 62.45 | 58.65 | 98.31 |
| IMF1 | 89.20 | 348.53 | -10.12 | 2.59 | 2.51 | 29.68 |
| H2O2+HS | 85.61 | 418.79 | 4.85 | 13.96 | 13.63, 12.96±2.93b | 19.86 |
| TS2 | 88.74 | 293.44 | 154.58 | 166.82 | 161.77 | 205.36 |
| IMF2 | 90.83 | 325.75 | -267.94 | -253.60 | -254.94 | -221.00 |
| H2O+HSO | 82.93 | 429.70 | -241.40 | -235.00 | -234.92 | -231.95 |
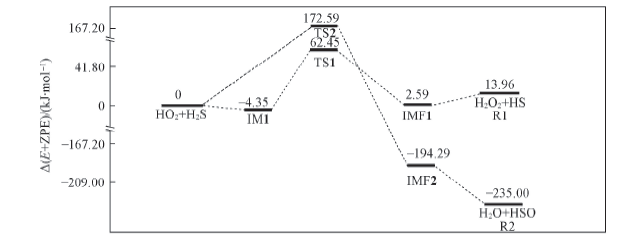
Fig.2 Energy diagrams in the naked reaction of HO2+H2S Energies were computed at the CCSD(T)/aug-cc-pVTZ//B3LYP/6-311+G(2df,2p) level, including the zero-point energy correction.
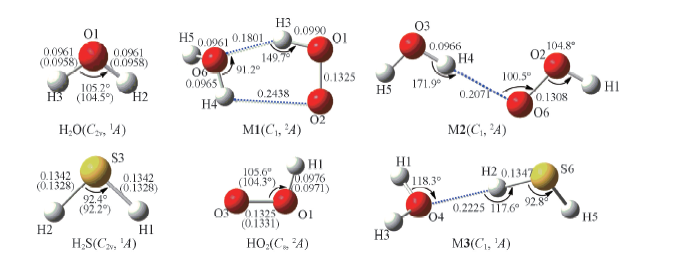
Fig.3 Geometrical parameters for HO2, H2O, H2S and the M1 to M3 complexes optimized at the B3LYP/6-311+G(2df,2p) level Bond lengths are in nm. The values with parentheses were the experimental data from the NIST chemistry webbook[27].
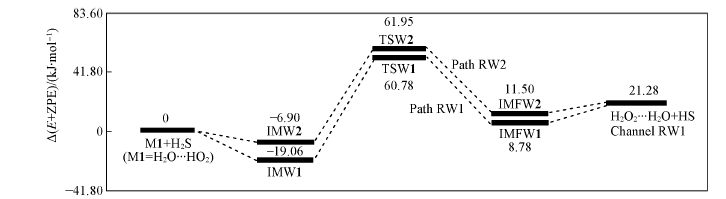
Fig.5 Energy diagrams in the reaction of M1+H2S Energies were computed at the CCSD(T)/aug-cc-pVTZ//B3LYP/6-311+G(2df,2p) level, including the zero-point energy correction.
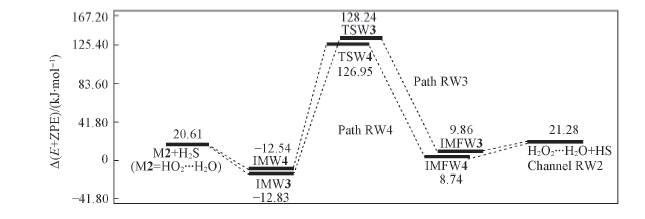
Fig.7 Energy diagrams in the reaction of M2+H2S Energies were computed at the CCSD(T)/aug-cc-pVTZ//B3LYP/6-311+G(2df,2p) level, including the zero-point energy correction.
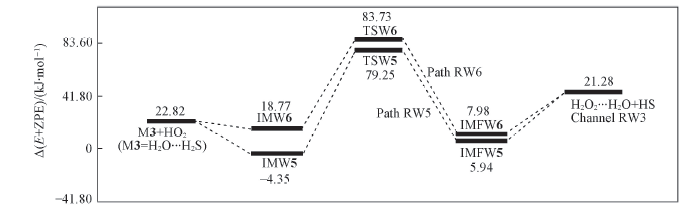
Fig.9 Energy diagrams in the reaction of M3+HO2 Energies were computed at the CCSD(T)/aug-cc-pVTZ//B3LYP/6-311+G(2df,2p) level, including the zero-point energy correction.
| h/km | T/K | kR1/(cm3· molecule-1·s-1) | kRW1 | [H2O][ | Keq(A) | k | ktotal | k |
|---|---|---|---|---|---|---|---|---|
| 0 | 298.2 | 6.01×10-23 | 6.82×10-18 | 2.59×1017 | 4.02×10-23 | 7.1×10-23 | 1.31×10-22 | 54.2 |
| 0 | 288.2 | 3.55×10-23 | 5.48×10-18 | 2.59×1017 | 4.24×10-23 | 6.02×10-23 | 9.57×10-23 | 62.9 |
| 2 | 275.2 | 1.76×10-23 | 4.04×10-18 | 1.11×1017 | 4.59×10-23 | 2.06×10-23 | 3.82×10-23 | 53.9 |
| 4 | 262.2 | 8.69×10-24 | 3.10×10-18 | 5.38×1016 | 5.04×10-23 | 8.4×10-24 | 1.71×10-23 | 49.1 |
| 6 | 249.3 | 4.25×10-24 | 2.44×10-18 | 1.16×1016 | 5.61×10-23 | 1.59×10-24 | 5.84×10-24 | 27.2 |
| 8 | 236.3 | 2.08×10-24 | 2.03×10-18 | 3.50×1015 | 6.38×10-23 | 4.53×10-25 | 2.53×10-24 | 17.9 |
| 10 | 223.3 | 1.02×10-24 | 1.78×10-18 | 9.23×1014 | 7.42×10-23 | 1.22×10-25 | 1.14×10-24 | 10.7 |
| 12 | 216.7 | 7.08×10-25 | 1.71×10-18 | 2.15×1014 | 8.10×10-23 | 2.97×10-26 | 7.38×10-25 | 4 |
Table 2 Kinetic results for formation process of H2O2+HS occurring through channels R1 and RW1*
| h/km | T/K | kR1/(cm3· molecule-1·s-1) | kRW1 | [H2O][ | Keq(A) | k | ktotal | k |
|---|---|---|---|---|---|---|---|---|
| 0 | 298.2 | 6.01×10-23 | 6.82×10-18 | 2.59×1017 | 4.02×10-23 | 7.1×10-23 | 1.31×10-22 | 54.2 |
| 0 | 288.2 | 3.55×10-23 | 5.48×10-18 | 2.59×1017 | 4.24×10-23 | 6.02×10-23 | 9.57×10-23 | 62.9 |
| 2 | 275.2 | 1.76×10-23 | 4.04×10-18 | 1.11×1017 | 4.59×10-23 | 2.06×10-23 | 3.82×10-23 | 53.9 |
| 4 | 262.2 | 8.69×10-24 | 3.10×10-18 | 5.38×1016 | 5.04×10-23 | 8.4×10-24 | 1.71×10-23 | 49.1 |
| 6 | 249.3 | 4.25×10-24 | 2.44×10-18 | 1.16×1016 | 5.61×10-23 | 1.59×10-24 | 5.84×10-24 | 27.2 |
| 8 | 236.3 | 2.08×10-24 | 2.03×10-18 | 3.50×1015 | 6.38×10-23 | 4.53×10-25 | 2.53×10-24 | 17.9 |
| 10 | 223.3 | 1.02×10-24 | 1.78×10-18 | 9.23×1014 | 7.42×10-23 | 1.22×10-25 | 1.14×10-24 | 10.7 |
| 12 | 216.7 | 7.08×10-25 | 1.71×10-18 | 2.15×1014 | 8.10×10-23 | 2.97×10-26 | 7.38×10-25 | 4 |
| [1] | Wallington T. J., Dagaut P., Kurylo M. J., Chem. Rev., 1992, 92(4), 667—710 |
| [2] | Zhu R. S., Lin M. C., Phys. Chem. Comm., 2001, 4(23), 106 |
| [3] | von Ahsen S., Willner H., Argüello G. A., J. Fluorine. Chem., 2004, 125(7), 1057—1070 |
| [4] | Zhou Y. Z., Zhang S. W., Li Q. S., Chem. J. Chinese Universities,2006, 27(8), 1496—1499 |
| (周玉芝, 张绍文, 李前树. 高等学校化学学报, 2006, 27(8), 1496—1499) | |
| [5] | Lightfoot P. D., Cox R. A., Crowley J. N., Destriau M., Hayman G. D., Jenkin M. E., Moortgat G. K., Zabel F., Atmos. Environ. Part. A,1992, 26(10), 1805—1961 |
| [6] | Bates T. S., Lamb B. K., Guenther A., Dignon J., Stoiber R. E., J. Atmos. Chem., 1992, 14(1—4), 315—337 |
| [7] | Lomans B. P., van der Drift C., Pol A., Op den Camp H. J. M., CMLS Cell. Mol. Life. Sci., 2002, 59(4), 575—588 |
| [8] | Mellouki A., Ravishankara A. R., Int. J. Chem. Kinet., 1994, 26(3), 355—365 |
| [9] | Gonzalez J., Anglada J. M., Buszek R. J., Francisco J. S., J. Am. Chem. Soc., 2011, 133(10), 3345—3353 |
| [10] | Aloisio S., Francisco J. S., J. Phys. Chem. A,1998, 102(11), 1899—1902 |
| [11] | Long B., Tan X. F., Long Z. W., Wang Y. B., Ren D. S., Zhang W. J., J. Phys. Chem. A,2011, 115(24), 6559—6567 |
| [12] | Frost G., Vaida V., J. Geophys. Res., 1995, 100(D9), 18803—18809 |
| [13] | Tao F. M., Higgins K., Klemperer W., Nelson D. D., Geophys. Res. Lett., 1996, 23(14), 1797—1800 |
| [14] | Stone D., Rowley D. M., Phys. Chem. Chem. Phys., 2005, 7(10), 2156—2163 |
| [15] | Gonzalez J., Anglada J. M., J. Phys. Chem. A.2010, 114(34), 9151—9162 |
| [16] | Song G. Y, Su Y., Roy A. P., Han K. L, Robert H. C., Zhang H. J., Li X. W., Angew. Chem. Int. Ed., 2010, 49, 912—917 |
| [17] | Anglada J. M., Gonzalez J., Chem. Phys. Chem., 2009, 10(17), 3034—3045 |
| [18] | Luo Y. M., Ohno K. S., Chem. Phys. Lett., 2009, 469(1—3), 57—61 |
| [19] | Jorgensen S., Kjaergaard H. G., J. Phys. Chem. A,2010, 114(14), 4857—4863 |
| [20] | Zhang T. L., Wang W. L., Zhang P., Lu J., Zhang Y., Phys. Chem. Chem. Phys., 2011, 13(46), 20794—20805 |
| [21] | Gao C. G., Long Z. W., Tan X. F., Long B., Long C. Y., Qin S. J., Zhang W. J., Acta Chim. Sinica,2013, 71, 849—856 |
| (高成贵, 隆正文, 谭兴凤, 龙波, 龙超云, 秦水介, 张为俊. 化学学报, 2013, 71, 849—856) | |
| [22] | Yung Y.L., Demore W. B., Photochemistry of Planetary Atomspheres, Oxford Univ., Oxford, 1999 |
| [23] | Raghavachari K., Trucks G. W., Pople J. A., Headgordon M., Chem. Phys. Lett.1989, 157(6), 479—483 |
| [24] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Montgomery J. A. Jr., Vreven T., Kudin K. N., Burant J. C., Millam J. M., Iyengar S. S., Tomasi J., Barone V., Mennucci B., Cossi M., Scalmani G., Rega N., Petersson G. A., Nakatsuji H., Hada M. Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Klene M., Li X., Knox J. E., Hratchian H. P., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Ayala P. Y., Morokuma K., Voth G. A., Salvador P., Dannenberg J. J., Zakrzewski V. G., Dapprich S., Daniels A. D., Strain M. C., Farkas O., Malick D. K., Rabuck A. D., Raghavachari K., Foresman J. B., Ortiz J. V., Cui Q., Baboul A. G., Clifford S., Cioslowski J., Stefanov B. B., Liu G., Liashenko A., Piskorz P., Komaromi I., Martin R. L. Fox D. J., Keith T., Al-Laham M. A., Peng C. Y., Nanayakkara A., Challacombe M., Gill P. M. W., Johnson B., Chen W., Wong M. W., Gonzalez C., Pople J. A., Gaussian 09, Revision A. 01, Gaussian Inc., Pittsburgh PA, 2009 |
| [25] | Zhang S.W., Truong T. N., VKLab Version 1.0, University of Utah, Salt Lake City, 2001 |
| [26] | Lu Y. X, Wang W. L., Wang W. N., Liu Y. Y., Zhang Y., Acta. Chim. Sinica.2010, 68, 1253—1260 |
| (卢彦霞, 王文亮, 王渭娜, 刘英英, 张越. 化学学报, 2010, 68, 1253—1260) | |
| [27] | Computational Chemistry Comparison and Benchmark Database, |
| [28] | Zhang T., Wang W., Li C., Du Y., Lü J., RSC Advances,2013, 3(20), 7381—7391 |
| [29] | Anglada J. M., Gonzalez J., Chem. Phys. Chem., 2009, 10(17), 3034—3045 |
| [1] | ZHOU Zixuan, YANG Haiyan, SUN Yuhan, GAO Peng. Recent Progress in Heterogeneous Catalysts for the Hydrogenation of Carbon Dioxide to Methanol [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220235. |
| [2] | YANG Dan, LIU Xu, DAI Yihu, ZHU Yan, YANG Yanhui. Research Progress in Electrocatalytic CO2 Reduction Reaction over Gold Clusters [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220198. |
| [3] | SUN Cuihong, LYU Liqiang, LIU Ying, WANG Yan, YANG Jing, ZHANG Shaowen. Mechanism and Kinetics on the Reaction of Isopropyl Nitrate with Cl, OH and NO3 Radicals [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210591. |
| [4] | CHENG Yuanyuan, XI Biying. Theoretical Study on the Fragmentation Mechanism of CH3SSCH3 Radical Cation Initiated by OH Radical [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220271. |
| [5] | MENG Fanwei, GAO Qi, YE Qing, LI Chenxi. Potassium Poisoning Mechanism of Cu-SAPO-18 Catalyst for Selective Catalytic Reduction of NOx by Ammonia [J]. Chem. J. Chinese Universities, 2021, 42(9): 2832. |
| [6] | YANG Yiying, ZHU Rongxiu, ZHANG Dongju, LIU Chengbu. Theoretical Study on Gold-catalyzed Cyclization of Alkynyl Benzodioxin to 8-Hydroxy-isocoumarin [J]. Chem. J. Chinese Universities, 2021, 42(7): 2299. |
| [7] | LI Xinyi, LIU Yongjun. Theoretical Insights into the Cleavage of β-Hydroxy Ketone Catalyzed by Artificial Retro-aldolase RA95.5-8F [J]. Chem. J. Chinese Universities, 2021, 42(7): 2306. |
| [8] | LI Yiwei, SHENTU Jiangtao, WANG Jingbo, LI Xiangyuan. Combustion Mechanism Construction Based on Minimized Reaction Network: C1⁃Oxygen Combustion [J]. Chem. J. Chinese Universities, 2021, 42(6): 1871. |
| [9] | TIAN Shengqiao, WEI Meiju. Reaction Mechanism for Rh(Ⅱ)-catalyzed [3+3] Cyclization of Indole Derivatives and Propertis of Product [J]. Chem. J. Chinese Universities, 2021, 42(6): 1899. |
| [10] | REN Ying, LI Changhua, WANG Tao, XUE Shanshan, ZHANG Tingting, JIA Jianfeng, WU Haishun. Theoretical Studies on Pd-catalyzed Oxidative N─H Carbonylation to Synthesis of 1,3,4-Oxadiazole-2(3H)-one Heterocyclic Compounds [J]. Chem. J. Chinese Universities, 2021, 42(6): 1793. |
| [11] | QI Guodong, YE Xiaodong, XU Jun, DENG Feng. Progress in NMR Studies of Carbohydrates Conversion on Zeolites [J]. Chem. J. Chinese Universities, 2021, 42(1): 148. |
| [12] | LI Xiangyuan, SHENTU Jiangtao, LI Yiwei, LI Juanqin, WANG Jingbo. Combustion Mechanism Construction Based on Minimized Reaction Network: Hydrogen-Oxygen Combustion † [J]. Chem. J. Chinese Universities, 2020, 41(4): 772. |
| [13] | LI Xiangyuan,YAO Xiaoxia,SHENTU Jiangtao,SUN Xiaohui,LI Juanqin,LIU Mingxia,XU Shimin. Combustion Reaction Mechanism Construction by Two-parameter Rate Constant Method † [J]. Chem. J. Chinese Universities, 2020, 41(3): 512. |
| [14] | ZHANG Lin, ZHANG Wei, YUE Xin, LI Pengjie, YANG Zuoyin, PU Min, LEI Ming. Theoretical Study on Mechanism of CO2 Hydrogenation to Formic Acid Catalyzed by Manganese Complex † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1911. |
| [15] | ZHANG Xiaoying,DU Guifang,ZHU Bo,GUAN Wei. Theoretical Mechanistic Study on Nickel-Catalyzed Cycloaddition of Azetidinone with Butadiene† [J]. Chem. J. Chinese Universities, 2019, 40(4): 770. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||