

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (3): 20250279.doi: 10.7503/cjcu20250279
刘浩阳1, 潘博2, 刘治刚3, 张志会1( ), 高文秀1, 杨颜如1, 杨苗苗1
), 高文秀1, 杨颜如1, 杨苗苗1
收稿日期:2025-09-28
出版日期:2026-03-10
发布日期:2025-11-21
通讯作者:
张志会
E-mail:zhangzh@jlict.edu.cn
基金资助:
LIU Haoyang1, PAN Bo2, LIU Zhigang3, ZHANG Zhihui1( ), GAO Wenxiu1, YANG Yanru1, YANG Miaomiao1
), GAO Wenxiu1, YANG Yanru1, YANG Miaomiao1
Received:2025-09-28
Online:2026-03-10
Published:2025-11-21
Contact:
ZHANG Zhihui
E-mail:zhangzh@jlict.edu.cn
Supported by:摘要:
在贵金属催化体系中实现贵金属纳米颗粒的高度均匀分散, 同时构建强金属-载体相互作用是抑制活性组分迁移与流失、 提升催化剂本征活性及稳定性的关键因素. 本文以USY分子筛为载体、 PEG为还原与稳定剂, 利用聚乙二醇(PEG)辅助水热合成法, 通过调整PEG分子量与银负载量, 制备了一系列Ag NPs/USY催化剂, 并将其用于4-硝基苯酚(4-NP)的催化加氢反应. 采用X射线衍射(XRD)、 扫描电子显微镜(SEM)、 X射线光电子能谱(XPS)和N2气吸附-脱附曲线(BET)等手段对其结构进行了表征. 结果表明, 利用PEG的空间位阻效应及其与分子筛表面官能团的协同作用, 实现了Ag NPs在USY介孔通道内的高度分散和有效锚定, 显著抑制了Ag NPs的聚集和流失. 在常温常压条件下, PEG-400辅助合成的5%Ag NPs/USY对高浓度4-NP(500 mg/L)表现出优异的催化活性, 在8 min内转化率超过99.9%, 表观速率常数高达0.817 min⁻¹, 7次循环后仍保持90%以上活性, 其稳定性显著优于Ag NPs/HY体系. 表征分析结果进一步证实, 孔道内限域的Ag NPs具有更高的抗氧化与抗流失能力; XPS表征结果显示, 循环后Ag NPs/USY中单质银的保留量为Ag NPs/HY的2.07倍.
中图分类号:
TrendMD:
刘浩阳, 潘博, 刘治刚, 张志会, 高文秀, 杨颜如, 杨苗苗. PEG辅助合成高分散Ag NPs/USY催化剂及其对4-硝基苯酚的高效催化还原. 高等学校化学学报, 2026, 47(3): 20250279.
LIU Haoyang, PAN Bo, LIU Zhigang, ZHANG Zhihui, GAO Wenxiu, YANG Yanru, YANG Miaomiao. PEG-assisted Synthesis of Highly Dispersed Ag NPs/USY Catalysts and Their Efficient Catalytic Reduction of 4-Nitrophenol. Chem. J. Chinese Universities, 2026, 47(3): 20250279.

Fig.3 SEM image(A), TEM image(B), EDX mappings(C—F) and HRTEM image(G) of Ag NPs/USY and lattice spacing of Ag NPs of Ag NPs/USY after eight reaction cycles(H)
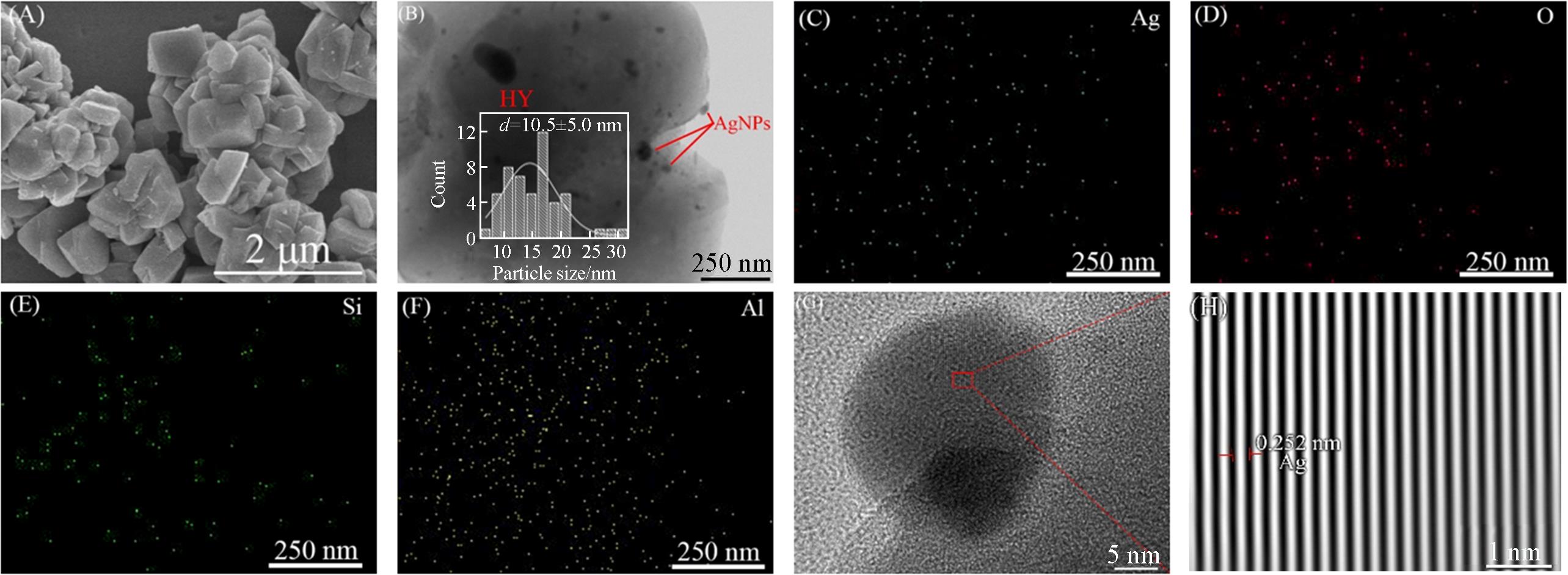
Fig.4 SEM image(A), TEM image(B), EDX mappings(C—F) and HRTEM image(G) of Ag NPs/HY and lattice spacing of Ag NPs of Ag NPs/HY after five reaction cycles(H)
| System | Micropore area/ (m²·g-1) | Micropore volume/ (cm³·g-1) | Average pore diameter/nm | Total pore volume/(cm³·g-1) | Specific surface area/ (m2·g-1) |
|---|---|---|---|---|---|
| USY | 675.88 | 0.2679 | 1.8688 | 0.3152 | 674.60 |
| 2%Ag NPs/USY | 373.27 | 0.1477 | 2.1504 | 0.2018 | 375.31 |
| 5%Ag NPs/USY | 320.85 | 0.1262 | 2.3102 | 0.1773 | 306.97 |
| 10%Ag NPs/USY | 240.26 | 0.0929 | 2.4644 | 0.1480 | 246.30 |
| HY | 780.30 | 0.3082 | 1.7952 | 0.3447 | 768.06 |
| 5%Ag NPs/HY | 315.06 | 0.1537 | 2.5574 | 0.2208 | 411.01 |
Table 1 Pore area, pore volume and pore diameter of USY, Ag NPs/USY, HY and Ag NPs/HY
| System | Micropore area/ (m²·g-1) | Micropore volume/ (cm³·g-1) | Average pore diameter/nm | Total pore volume/(cm³·g-1) | Specific surface area/ (m2·g-1) |
|---|---|---|---|---|---|
| USY | 675.88 | 0.2679 | 1.8688 | 0.3152 | 674.60 |
| 2%Ag NPs/USY | 373.27 | 0.1477 | 2.1504 | 0.2018 | 375.31 |
| 5%Ag NPs/USY | 320.85 | 0.1262 | 2.3102 | 0.1773 | 306.97 |
| 10%Ag NPs/USY | 240.26 | 0.0929 | 2.4644 | 0.1480 | 246.30 |
| HY | 780.30 | 0.3082 | 1.7952 | 0.3447 | 768.06 |
| 5%Ag NPs/HY | 315.06 | 0.1537 | 2.5574 | 0.2208 | 411.01 |
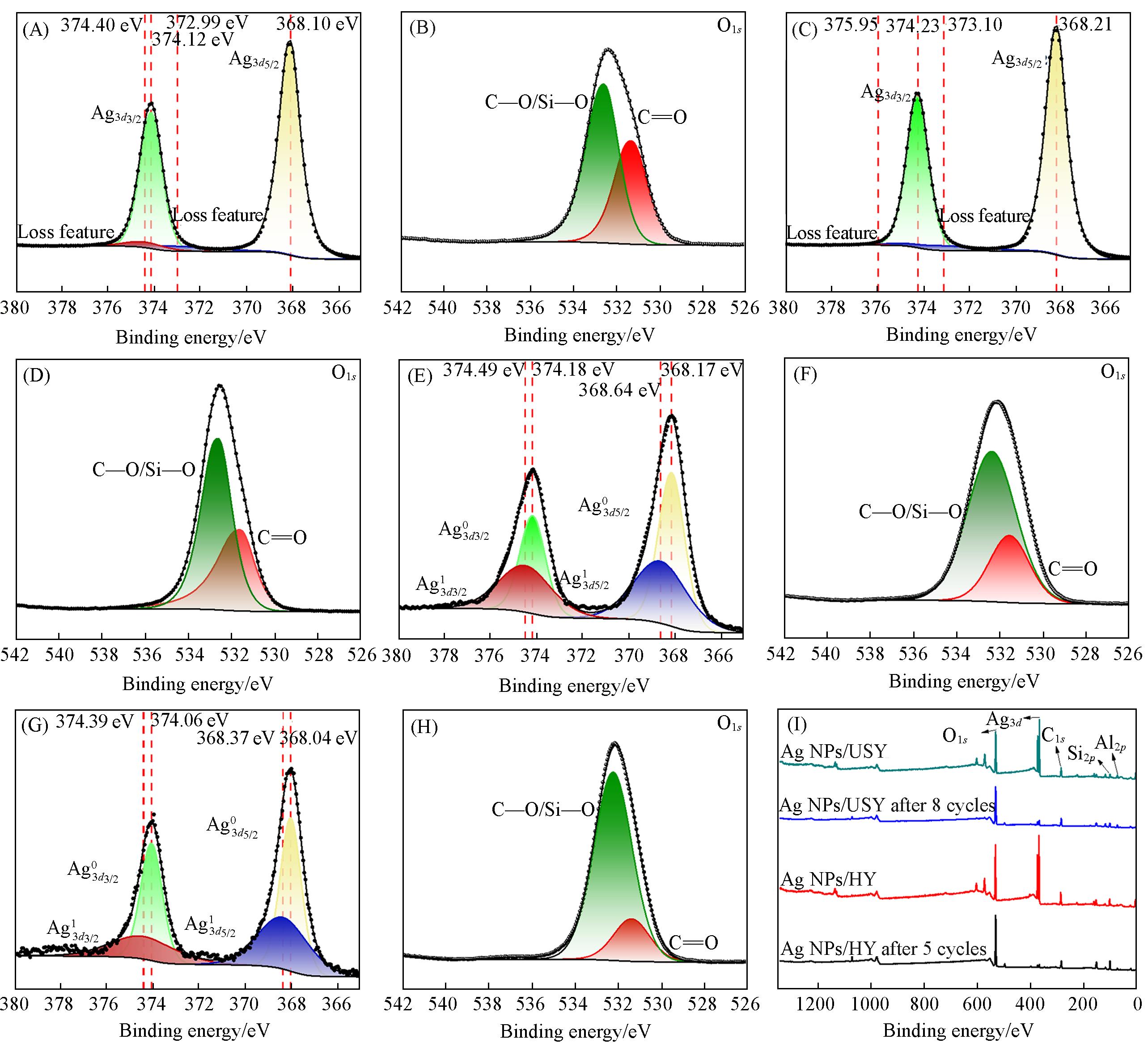
Fig.7 Ag3d XPS spectra of Ag NPs/USY(A), Ag NPs/HY(C), Ag NPs/USY after 8 cycles(E), and Ag NPs/HY after 5 cycles(G), O1s XPS spectra of Ag NPs/USY(B), Ag NPs/HY(D), Ag NPs/USY after 8 cycles(F), and Ag NPs/HY after 5 cycles(H), full XPS spectra of Ag NPs/USY, Ag NPs/HY, Ag NPs/USY after 8 cycles, and Ag NPs/HY after 5 cycles(I)
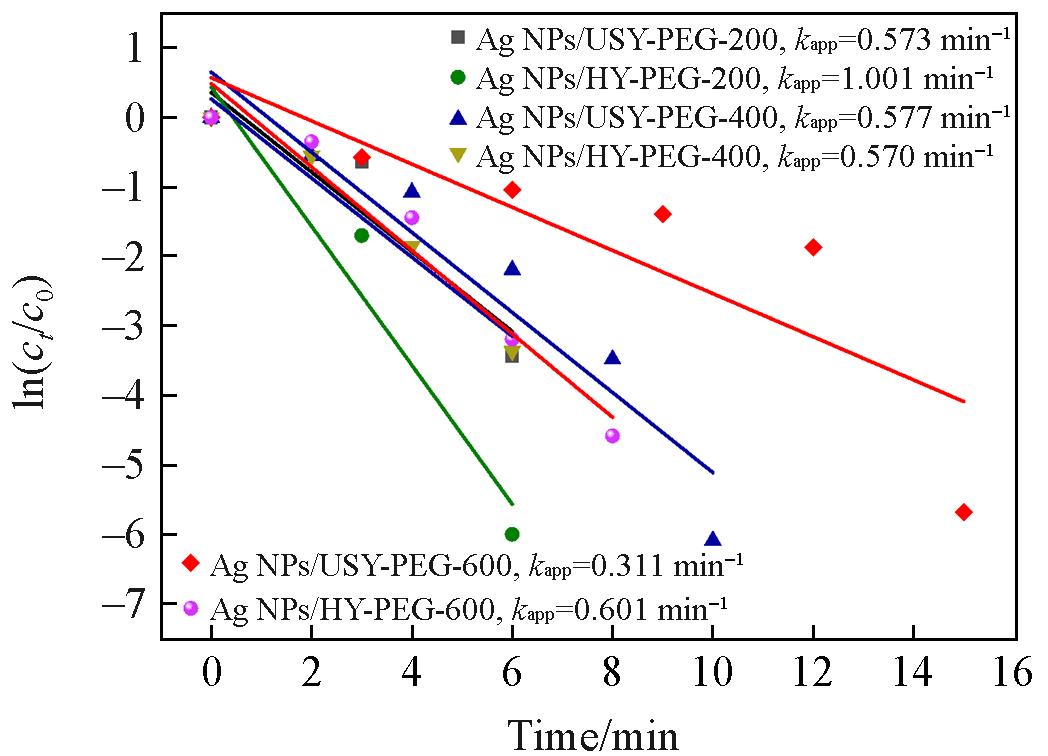
Fig.8 Linear fitting of ln(ct /c0) vs. reaction time for the catalytic degradation of 4⁃NP by Ag NPs/HY and Ag NPs/USY synthesized using PEGs with different molecular weights as reducing agents
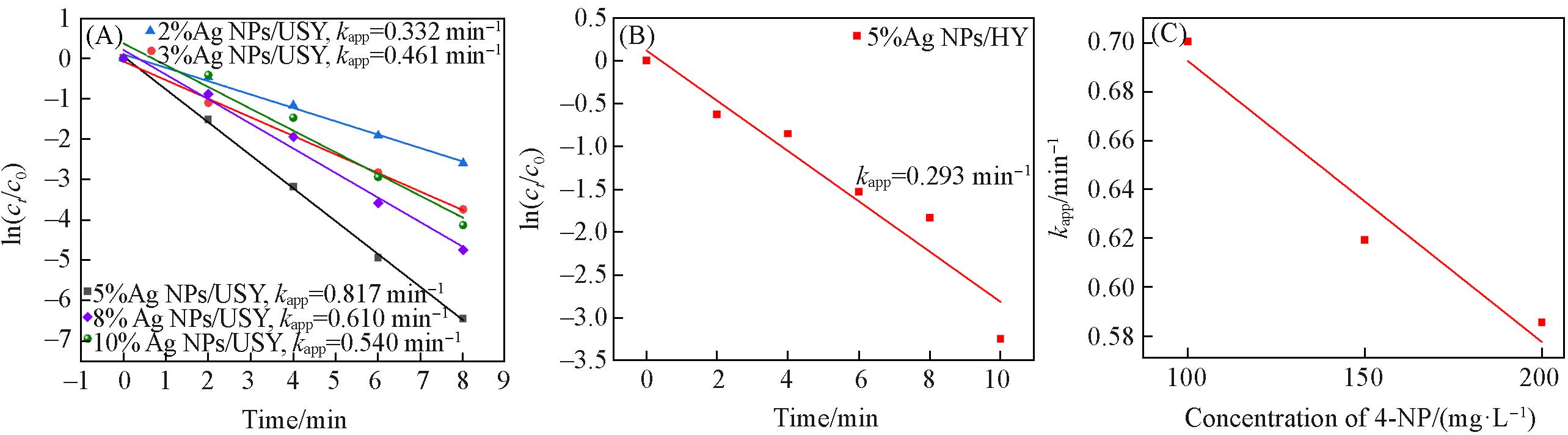
Fig.11 Linear fits of ln(ct /c0) vs. reaction time for Ag NPs/USY(A), Ag NPs/HY(B) and linear fit of kapp of 2%Ag NPs/USY to reactant 4⁃NP concentration(C)
| Entry | Catalyst | kapp/min-1 | Concentration of 4⁃NP solution/(mg·L-1) | Silver concentration(%) | Cycle(act. a >50%) | Ref. |
|---|---|---|---|---|---|---|
| 1 | rGO/Ag | 0.49 | 41.7 | 18 | 8 | [ |
| 2 | rGO/Fe3O4/Ag | 0.37 | 13.9 | 23.1 | 3 | [ |
| 3 | Ag dendrites | 0.34 | 14.3 | ca. 100 | N/A b | [ |
| 4 | Ag/MR⁃Photo | 0.126 | 17.4 | 1 | 4 | [ |
| 5 | PG/Ag | 0.33 | 16.7 | 55.3 | N/A b | [ |
| 7 | Fe3O4/SiO2@Ag | 0.24 | 0.0278 | 1.44 | 4 | [ |
| 8 | Ag NPs⁃loaded⁃zeolite | 0.18 | 9.2 | 4 | N/A b | [ |
| 9 | Ag NPs/USY | 0.817 | 500 | 5 | 8 | This work |
Table 2 Comparison of rate constants, silver mass concentrations, and cycle numbers for different silver-based catalysts
| Entry | Catalyst | kapp/min-1 | Concentration of 4⁃NP solution/(mg·L-1) | Silver concentration(%) | Cycle(act. a >50%) | Ref. |
|---|---|---|---|---|---|---|
| 1 | rGO/Ag | 0.49 | 41.7 | 18 | 8 | [ |
| 2 | rGO/Fe3O4/Ag | 0.37 | 13.9 | 23.1 | 3 | [ |
| 3 | Ag dendrites | 0.34 | 14.3 | ca. 100 | N/A b | [ |
| 4 | Ag/MR⁃Photo | 0.126 | 17.4 | 1 | 4 | [ |
| 5 | PG/Ag | 0.33 | 16.7 | 55.3 | N/A b | [ |
| 7 | Fe3O4/SiO2@Ag | 0.24 | 0.0278 | 1.44 | 4 | [ |
| 8 | Ag NPs⁃loaded⁃zeolite | 0.18 | 9.2 | 4 | N/A b | [ |
| 9 | Ag NPs/USY | 0.817 | 500 | 5 | 8 | This work |
| [44] | Chen L., Wang Y. Q., Wang X. X., Wang Q. L., Li B. L., Li S. J., Zhang S. H., Li W., J. Hazard. Mater., 2022, 424, 127334 |
| [45] | Malfait W. J., Halter W. E., Phys. Rev. B, 2008, 77(1), 014201 |
| [46] | Peng P., Gao X. H., Yan Z. F., Mintova S., Natl. Sci. Rev., 2020, 7(11), 1726—1742 |
| [47] | Hoflund G. B., Hazos Z. F., Salaita G. N., Phys. Rev. B, 2000, 62(16), 11126—11133 |
| [48] | Deckman I., Moshonov M., Obuchovsky S., Brener R., Frey G. L., J. Mater. Chem. A, 2014, 2(39), 16746—16754 |
| [49] | Ye L., Dong X. M., Miao P. R., Xu Y. X., Wu P., Zhao J. L., Zhang H. M., Zhou B. J., Zhang H., Lin Y. Y., Zhou J. L., Chem. Eng. J., 2025, 508, 160878 |
| [50] | Abebe A., Kaushik R., Kumar S., Mondal I. C., Ghosh S., Halder A., ACS Appl. Nano Mater., 2025, 8(4), 1882—1892 |
| [51] | Fang S., Wu F. L., Zarrabeitia M., Kuenzel M., Roscher D., Gao X., Kim J., Kim G., Passerini S., Batteries Supercaps, 2022, 5(10), e202200286 |
| [52] | He Z. Q., Cheng H. N., Olanya O. M., Uknalis J., Zhang X. D., Koplitz B. D., He J. B., JMSR, 2017, 7(1), 28 |
| [53] | Li Y. Z., Cao Y. L., Xie J., Jia D. Z., Qin H. Y., Liang Z. T., Catal. Commun., 2015, 58, 21—25 |
| [54] | Thu T. V., Ko P. J., Nguyen T. V., Vinh N. T., Khai D. M., Lu L. T., Appl. Organomet. Chem., 2017, 31(11), e3781 |
| [55] | Zhang W., Tan F. T., Wang W., Qiu X. L., Qiao X. L., Chen J. G., J. Hazard. Mater., 2012, 217218, 36—42 |
| [56] | Jiang S., Wang L., Duan Y. D., An J., Luo Q. Z., Zhang Y. M., Tang Y. F., Huang J. Y., Zhang B. K., Liu J., Wang D. S., Appl. Catal. B: Environ., 2021, 283, 119592 |
| [57] | Baruah B., Gabriel G. J., Akbashev M. J., Booher M. E., Langmuir, 2013, 29(13), 4225—4234 |
| [58] | Shin K. S., Cho Y. K., Choi J. Y., Kim K., Appl. Catal. A: Gen., 2012, 413/414, 170—175 |
| [59] | Guerrini L., Alvarez⁃Puebla R. A., Pazos-Perez N., Materials, 2018, 11(7), 1154 |
| [60] | Tian L., Gao X. P., Zhu M. Z., Huang Z. X., Wu B., Chen C., Ma X. H., Ruan Y. E., Guo W. X., Meng X. M., Wang H. J., Du W. B., He S. N., Pan H. G., Zheng X. S., Wu Z. J., Zhou H., Xia J., Wu Y., Adv. Mater., 2025, 37(12), 2417095 |
| [61] | Song C., Guo S., Chen L., Catalysts, 2020, 11(1), 43 |
| [62] | Liu Y. Y., Zhao Y. H., Zhou Y., Guo X. L., Chen Z. T., Zhang W. J., Zhang Y., Chen J., Wang Z. M., Sun L. T., Zhang T., Nanotechnology, 2018, 29(31), 315702 |
| [63] | Varshney S., Bar‐Ziv R., Zidki T., ChemCatChem, 2020, 12(18), 4680—4688 |
| [1] | Dsikowitzky L., Schwarzbauer J., Environ. Chem. Lett., 2014, 12(3), 371—386 |
| [2] | Benmaati A., Boukoussa B., Hadjadj Aoul R., Hachemaoui M., Kerbadou R. M., Habib Zahmani H., Hacini S., Silicon, 2022, 14(14), 8831—8843 |
| [3] | Pradhan A. A., Gogate P. R., J. Hazard. Mater., 2010, 173(1/3), 517—522 |
| [4] | Yu S. Q., Hu J., Wang J. L., Radiat. Phys. Chem., 2010, 79(10), 1039—1046 |
| [5] | Bhatti Z. I., Toda H., Furukawa K., Water Res., 2002, 36(5), 1135—1142 |
| [6] | Tiwari J., Tarale P., Sivanesan S., Bafana A., Environ. Sci. Pollut. Res., 2019, 26(28), 28650—28667 |
| [7] | Gautam G. J., Chaube R., Joy K., Endocrine Disruptors, 2015, 3(1), e981442 |
| [8] | Kadam V. V., Shanmugam S. D., Ettiyappan J. P., Balakrishnan R. M., Environ. Sci. Pollut. Res., 2021, 28(10), 12119—12130 |
| [9] | Zhou L., Zhou M. H., Zhang C., Jiang Y. H., Bi Z. H., Yang J., Chem. Eng. J., 2013, 233, 185—192 |
| [10] | Lai T. L., Yong K. F., Yu J. W., Chen J. H., Shu Y. Y., Wang C. B., J. Hazard. Mater., 2011, 185(1), 366—372 |
| [11] | Xu R. H., J. Chem. Ind. & Eng., 2007, 28(3), 31—34 |
| 徐荣华. 化学工业与工程技术, 2007, 28(3), 31—34 | |
| [12] | Gao F., Zhang W. B., Qin R., Zhang X. T., Zou S. L., Chem. Res. Appl., 2021, 33(10), 1991—1998 |
| 高飞, 张文斌, 秦容, 张潇天, 邹素兰. 化学研究与应用, 2021, 33(10), 1991—1998 | |
| [13] | Vu A. N., Le H. N. T., Phan T. B., Le H. V., Polymers, 2023, 15(16), 3373 |
| [14] | Pandey S., Mishra S. B., Carbohyd. Polym., 2014, 113, 525—531 |
| [15] | Bogireddy N. K. R., Sahare P., Pal U., Méndez S. F. O., Gomez L. M., Agarwal V., Chemical Engineering Journal, 2020, 388, 124237 |
| [16] | Vaidya M. J., Kulkarni S. M., Chaudhari R. V., Org. Process Res. Dev., 2003, 7(2), 202—208 |
| [17] | Kharlamova T. S., Salina M. V., Svetlichnyi V. A., Salaev M. A., Stadnichenko A. I., Mamontov G. V., Catalysis Today, 2022, 384/386, 12—24 |
| [18] | Duan M. Y., Li J., Li M., Zhang Z. Q., Wang C., Appl. Surf. Sci., 2012, 258(14), 5499—5504 |
| [19] | Bashir M. S., Jiang X., Kong X. Z., Eur. Polym. J., 2020, 129, 109652 |
| [20] | Sati A., Ranade T. N., Mali S. N., Ahmad Yasin H. K., Pratap A., ACS Omega, 2025, 10(8), 7549—7582 |
| [21] | Ouyang C. L., Liu S., Guo Y. J., Yi S. M., Li Q., Appl. Surf. Sci., 2024, 652, 159281 |
| [22] | Guo D. L., Li J., Wang J. X., Zeng M. F., Fan S. T., Song M. M., Qi X. W., Zhang Y. T., Li B. J., Zhang S., Macromol. Chem. Phys., 2025, 226(15), e00149 |
| [23] | Sudhakar P., Soni H., J. Environ. Chem. Eng., 2018, 6(1), 28—36 |
| [24] | Chishti A. N., Ni L., Guo F., Lin X., Liu Y., Wu H., Chen M., Diao G. W., J. Environ. Chem. Eng., 2021, 9(2), 104948 |
| [25] | Alhokbany N., Ahama T., Ruksana, Naushad M., Alshehri S. M., Compos. Part B: Eng., 2019, 173, 106950 |
| [26] | Hunge Y. M., Yadav A. A., Kang S. W., Kim H., J. Alloy. Compd., 2022, 928, 167133 |
| [27] | Chen F. J., Yu J. H., Acc. Chem. Res., 2025, 58(15), 2402—2414 |
| [28] | Kwakye⁃Awuah B., Williams C., Kenward M. A., Radecka I., J. Appl. Microbiol., 2008, 104(5), 1516—1524 |
| [29] | Severance M., Dutta P. K., J. Phys. Chem. C, 2014, 118(49), 28580—28591 |
| [30] | Zhou F. J., He D. F., Ren G. J., Yarahmadi H., Sci. Rep., 2024, 14(1), 1143 |
| [31] | Belhadri A., Boukoussa B., Benali F., Mekki A., Mokhtar A., Hachemaoui M., Ismail I., Iqbal J., Patole S. P., Taha I., Hamacha R., Abboud M., J. Water Process Eng., 2025, 70, 106954 |
| [32] | Hamciuc C., Hamciuc E., Popovici D., Danaila A. I., Butnaru M., Rimbu C., Carp⁃Carare C., Kalvachev Y., Mater. Lett., 2018, 212, 339—342 |
| [33] | Wang L., Dong F., Meng Y., Kang Y. H., Zhang H. T., Tang Z. C., J. Mater. Chem. A, 2025, 13 (24), 18209—18252 |
| [34] | Aggour Y. A., Kenawy E. R., Magdy M., Elbayoumy E., RSC Adv., 2024, 14(41), 30127—30139 |
| [35] | Lutz W., Shutilov R. A., Gavrilov V. Y., Z. Anorg. Allg. Chem., 2014, 640(3/4), 577—581 |
| [36] | Lutz W., Kurzhals R., Kryukova G., Enke D., Weber M., Heidemann D., Z. Anorg. Allg. Chem., 2010, 636(8), 1497—1505 |
| [37] | Bai Y. R., Wang H. L., Jin C., Gao C., Xiong S. C., Gong Z. J., Peng Y., Li J. H., Appl. Catal. B: Environ. Energy, 2025, 378, 125604 |
| [38] | Zhou L. P., Shi M. T., Cai Q. Y., Wu L., Hu X. P., Yang X. M., Chen C., Xu J., Micropor. Mesopor. Mater., 2013, 169, 54—59 |
| [39] | Sharma R. K., Yadav S., Dutta S., Kale H. B., Warkad I. R., Zbořil R., Varma R. S., Gawande M. B., Chem. Soc. Rev., 2021, 50(20), 11293—11380 |
| [40] | Li J., Lin Y., Zhao B., J. Nanopart. Res., 2002, 4(4), 345—349 |
| [41] | Medasani B., Park Y. H., Vasiliev I., Phys. Rev. B, 2007, 75(23), 235436 |
| [42] | Bai Y., Pu C. S., Liu S., Gu X. Y., Liu Y., IOP Conf. Ser.: Earth Environ. Sci., 2021, 859(1), 012017 |
| [43] | Shi Y. C., Wang G. J., Prog. Org. Coat., 2016, 90, 390—398 |
| [1] | 张宗洋, 李玉平, 张若茜, 刘宇峰, 陈泽, 韩丽娜, 韩培德. L沸石晶间转化快速合成SSZ-13沸石及其中空结构形成机理[J]. 高等学校化学学报, 2023, 44(10): 20230034. |
| [2] | 彭奎霖, 李桂林, 江重阳, 曾少娟, 张香平. 电解液调控CO2电催化还原性能微观机制的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220238. |
| [3] | 陈韶云, 张行颖, 刘奔, 田杜, 李奇, 陈芳, 胡成龙, 陈建. Ag纳米粒子在TiO2四棱柱阵列上的可控生长及其SERS效应[J]. 高等学校化学学报, 2021, 42(8): 2381. |
| [4] | 卓孟宁,李飞,蒋浩,陈倩文,李鹏,王立章. SnO2/GDE阴极的制备及电催化还原CO2产甲酸性能[J]. 高等学校化学学报, 2020, 41(3): 530. |
| [5] | 章凌,段宏昌,谭争国,吴勤明,孟祥举,肖丰收. 用于柴油车尾气消除反应(NH3-SCR)的八元环沸石分子筛研究进展[J]. 高等学校化学学报, 2020, 41(1): 19. |
| [6] | 李春晓, 李坚, 梁文俊, 梁全明. Cr负载V2O5-WO3/TiO2催化剂的低温NH3-SCR脱硝活性[J]. 高等学校化学学报, 2019, 40(7): 1447. |
| [7] | 冯微, 王博蔚, 姜洋, 李龙云. 银纳米粒子簇的设计、 制备和表面增强拉曼光谱[J]. 高等学校化学学报, 2019, 40(7): 1345. |
| [8] | 何鹏琛, 周健, 周阿武, 豆义波, 李建荣. MOFs基材料在光催化CO2还原中的应用[J]. 高等学校化学学报, 2019, 40(5): 855. |
| [9] | 王稳, 陶霞芳, 吴赟炎, 赵南, 程晓农, 杨娟, 周亚洲. 三明治结构纳米银/氧化石墨烯基底的制备及SERS性能[J]. 高等学校化学学报, 2019, 40(4): 667. |
| [10] | 阿卜杜黑热木∙阿瓦提, 张得栋, 哈丽丹∙买买提. 基于氨基化煤基碳点的复合光催化剂的制备及对CO2还原的催化性能[J]. 高等学校化学学报, 2019, 40(2): 306. |
| [11] | 朱红太, 宋丽云, 何洪, 尹孟奇, 程杰, 孙炎明, 李世宁, 邱文革. CeTiOx脱硝催化剂的抗硫中毒性能[J]. 高等学校化学学报, 2019, 40(2): 350. |
| [12] | 袁宝明,董晓明,杨帆,彭传刚,王金成,吴丹凯. 载银离子PLGA-PEG-PLGA温度敏感水凝胶的制备及体外抗菌性能[J]. 高等学校化学学报, 2019, 40(10): 2225. |
| [13] | 孔艳, 王立章, 杨胜翔, 赵鹏. Ti(100-δ)Cuδ(δ=0.02, 0.28, 1.39, 5.65)阴极的制备及电催化还原NO3-机理[J]. 高等学校化学学报, 2018, 39(1): 132. |
| [14] | 阿山, 郑家威, 刘聚明, 白杰, 杨桔材, 张前程. 乙酸修饰Ag/TiO2复合光催化剂的制备及协同催化性能[J]. 高等学校化学学报, 2017, 38(8): 1450. |
| [15] | 徐婉珍, 邱春孝, 黄卫红, 刘鸿, 杨文明. 量子点荧光印迹传感器的计算机模拟设计、 制备及检测河水中4-硝基苯酚的应用[J]. 高等学校化学学报, 2017, 38(7): 1155. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||