

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (9): 1988.doi: 10.7503/cjcu20190030
收稿日期:2019-01-14
出版日期:2019-09-10
发布日期:2019-09-09
通讯作者:
马永钧
E-mail:mayj@nwnu.edu.cn
基金资助:
LIU Fen,ZHOU Min,WANG Suxia,WANG Rong,YANG Ning,MA Yongjun( )
)
Received:2019-01-14
Online:2019-09-10
Published:2019-09-09
Contact:
MA Yongjun
E-mail:mayj@nwnu.edu.cn
Supported by:中图分类号:
TrendMD:
刘芬, 周敏, 王苏霞, 王荣, 杨宁, 马永钧. 用化学需氧量指数法研究亚甲基蓝的可见光光电催化脱色反应机理. 高等学校化学学报, 2019, 40(9): 1988.
LIU Fen, ZHOU Min, WANG Suxia, WANG Rong, YANG Ning, MA Yongjun. Study on Photoelectrocatalytic Decolorization Mechanism of Methylene Blue Under the Visible-light Irradiation by Measuring Chemical Oxygen Demand Index†. Chem. J. Chinese Universities, 2019, 40(9): 1988.

Fig.1 SEM images of modified photoelectrodes CuInSe2/GC(A), CuInSe2/Ag3PO4@AgIO4/GC(B) and TEM images of CuInSe2/Ag3PO4@AgIO4 granular catalyst material(C, D)
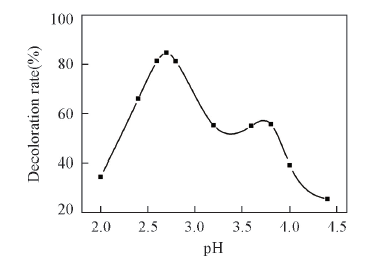
Fig.3 Effect of pH value of supporting electrolyte on the decoloration efficiency of MB dye in the visible-light photoelectrocatalytic reaction H2O2 initial concentation: 0.42 mol/L; reaction temperature: 50 ℃; irradiation time: 45 min.
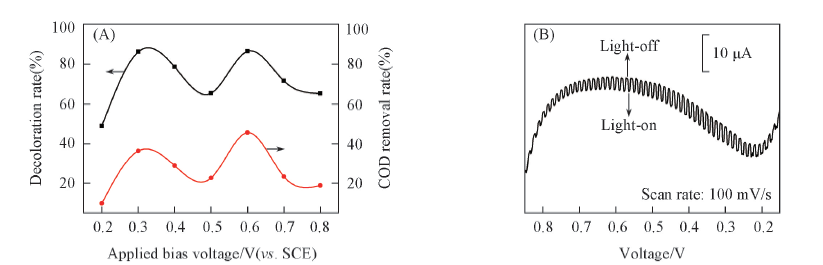
Fig.4 Influence of applied bias voltage on the degradation efficiency of MB dye in the visible-light photoelectrocatalytic reaction(A) and the voltammogram of MB testing solution using linear sweep voltammetry(LSV) under the visible-light irradiation condition(B)
| pH | 2.7 | 3.7 |
|---|---|---|
| Decoloration rate, D(%) | 86.0 | 54.0 |
| η*(%) | 45.0 | 20.8 |
| η**(%) | 16.3 | 6.8 |
| MB Mineralization rate, Ma (%) | 10.1 | 3.7 |
| D/M ratio | 8.5 | 14.6 |
Table 1 Comparison between the decoloration rate and COD removal rate of MB dye at the different acidity conditions of MB+H2O2 system
| pH | 2.7 | 3.7 |
|---|---|---|
| Decoloration rate, D(%) | 86.0 | 54.0 |
| η*(%) | 45.0 | 20.8 |
| η**(%) | 16.3 | 6.8 |
| MB Mineralization rate, Ma (%) | 10.1 | 3.7 |
| D/M ratio | 8.5 | 14.6 |
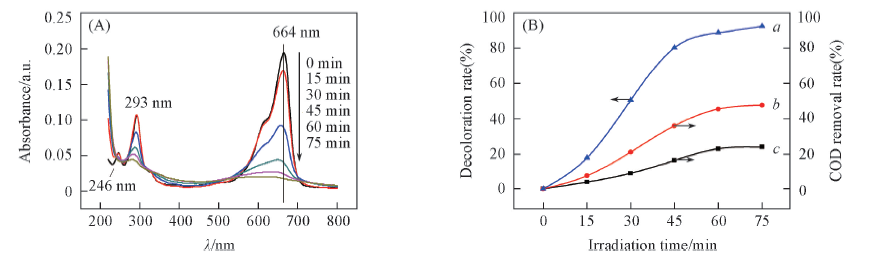
Fig.5 Influence of irradiation time on ultraviolet-visible absorption spectra(A) and the degradation efficiency of MB dye(B) Curves a—c represen the decoloration rate(D), COD removal rate of the supernatant fraction(η*) and total COD removal rate(η**), respectively.
| [1] | Garcia-Segura S., Brillas E., ,. J. Photochem. Photobiol. C 2017, 31, 1— 35 |
| [2] | Brillas E., Martínez-Huitle C. A., ,. Appl. Catal. B 2015, 166/ 167, 603— 643 |
| [3] | Cardoso J. C., Bessegato G. G., Zanoni M. V. B., ,Water Res., 2016, 98, 39— 46 |
| [4] | Li M. Y., Xiong L., Chen Y. Y., Zhang N., Zhang Y. M., Yi H ., Sci. China, Ser. B: Chem., 2005,35( 2), 144— 150 |
| (李明玉, 熊林, 陈芸芸, 张娜, 张渊明, 尹华.中国科学,2005,35(2), 144— 150) | |
| [5] | Yang J., Dai J., Zhao J. C., Miao J., ,Chin. Sci. Bull., 2009,54( 15), 2196— 2204 |
| (杨娟, 戴俊, 赵进才, 缪娟.科学通报,2009,54(15), 2196— 2204) | |
| [6] | He C., Xiong Y., Zha C. H., Wang X. M., Zhu X. H., ,. J. Chem. Technol. Biotechnol. 2003, 78, 717— 723 |
| [7] | Li P. Q., Zhao G. H., Zhao K. J., Gao J. X., Wu T., ,. Dyes Pigm. 2012, 92, 923— 928 |
| [8] | Daghrir R., Drogui P., Robert D., ,. Ind. Eng. Chem. Res. 2013, 52, 3581— 3599 |
| [9] | Karthikeyan N., Sivaranjani T., Dhanavel S., Gupta V. K., Narayanan V., Stephen A., ,. J. Mol. Liq. 2017, 227, 194— 201 |
| [10] | Bermudez V., ,. Solar Energy 2018, 175, 2— 8 |
| [11] | Guo H., Zhou R., Huang Y. Z., Wan L., Gan W., Niu H. H., Xu J. Z., ,. Ceram. Int. 2018, 44, 16092— 16098 |
| [12] | Kim J. S., Baek S. K., Kim Y. B., Do H. W., Kwon Y. H., Cho S. W., Yun Y. D., Yoon J. H., Lee H. B. R., Kim S. W., Cho H. K., ,. Nano Energy 2018, 46, 1— 10 |
| [13] | Yi Z. G., Ye J. H., Kikugawa N., Kako T., Ouyang S. X., Stuart-Williams H., Yang H., Cao J. Y., Luo W. J., Li Z. S., Liu Y., Withers R. L., ,. Nat. Mater. 2010, 9( 7), 559— 564 |
| [14] | Tang J T., Li D. T., Feng Z. X., Tan Z., Ou B. L ., Rsc Adv., 2013, 4, 2151— 2154 |
| [15] | Katsumata H., Taniguchi M., Kaneco S., Suzuki T., ,. Catal. Commun. 2013, 34, 30— 34 |
| [16] | Vu T. A., Dao C. D., Hoang T. T. T., Nguyen K. T., Le G. H., Dang P. T., Tran H. T. K., Nguyen T. V., ,. Mater. Lett. 2013, 92, 57— 60 |
| [17] | Wang X. L., Li C., ,. J. Phys. Chem. C 2018, 122, 21083— 21096 |
| [18] | Cao F. P., Ding C. H., Liu K. C., Kang B. Y., Liu W. M., ,. Cryst. Res. Technol. 2014, 49( 12), 933— 938 |
| [19] | Antoniadou M., Daskalaki V. M., Balis N., Kondarides D. I., Kordulis C., Lianos P., ,. Appl. Catal. B 2011, 107, 188— 196 |
| [20] | Li M. Y., Liu H., Song Y. H., Gao J. J., ,. Int. J. Energy Res. 2018, 42, 4625— 4641 |
| [21] | Lv Y. H., Huang K., Zhang W., Yang B., Chi F. L., Ran S. L., Liu X. G., ,Ceram. Int., 2014, 40, 8087— 8092 |
| [22] | Gao B., Wang T., Fan X. L., Gong H., Meng X. G., Li P., Feng Y. Y., Huang X. L., He J. P., Ye J. H., ,. Sol. RRL 2018, 2, 1800102— 1800111 |
| [23] | Chavaco L. C., Arcos C. A., Prato-Garcia D., ,. J. Environ. Manage. 2017, 198, 203— 212 |
| [24] | Riga A., Soutsas K., Ntampegliotis K., Karayannis V., Papapolymerou G ., Desalination 2007, 211, 72— 86 |
| [25] | Tammina S. K., Mandal B. K., Kadiyala N. K., ,. Environ. Nanotechnol. Monit. Manage. 2018, 10, 339— 350 |
| [26] | Ghaly M. Y., Farah J. Y., Fathy A. M ., Desalination 2007, 217, 74— 84 |
| [27] | Hu E. L., Wu X. B., Shang S. M., Tao X., Jiang S., Gan L., ,. J. Clean. Prod. 2016, 112, 4710— 4718 |
| [28] | Tayade R. J., Natarajan T. S., Bajaj H. C., ,. Ind. Eng. Chem. Res. 2009, 48, 10262— 10267 |
| [29] | Yao Y. W., Zhao C. M., Zhao M. M., Wang X., ,. J. Hazard. Mater. 2013, 263, 726— 734 |
| [30] | Alaoui A., Kacemi K. K. E., Ass K. E., Kitane S., Bouzidi S. E., ,. Sep. Purif. Technol. 2015, 154, 281— 289 |
| [31] | Geerdink R. B., Sebastiaan V. D. H. R., Epema O. J., ,. Anal. Chim. Acta 2017, 961, 1— 11 |
| [32] | Li J. Q., Zheng L., Li L. P., Shi G. Y., Xian Y. Z., Jin L. T ., Electroanalysis 2006, 18( 10), 1014— 1018 |
| [33] | Qu X., Tian M., Chen S., Liao B. Q., Chen A. C ., Electroanalysis 2011, 23( 5), 1267— 1275 |
| [34] | Houas A., Lachheb H., Ksibi M., Elaloui E., Guillard C., Herrmann J. M., ,. Appl. Catal. B 2001, 31, 145— 157 |
| [35] | Magalhães P., Ângelo J., Nunes O. C., Mendes A., ,. Appl. Surf. Sci. 2018, 458, 597— 602 |
| [36] | Zhou X., Zhang J., Ma Y., Cheng H. Q., Fu S. Z., Zhou D. D., Dong S. S., ,. Photochem. Photobiol. 2017, 93, 1170— 1177 |
| [37] | Xu C., Rangaiah G. P., Zhao X. S., ,. Ind. Eng. Chem. Res. 2014, 53, 14641— 14649 |
| [38] | Shanmugam M., Alsalme A., Alghamdi A., Jayavel R., ,. ACS Appl. Mater. Interfaces 2015, 7, 14905— 14911 |
| [39] | Wu C. H., Chern J. M., ,. Ind. Eng. Chem. Res. 2006, 45, 6450— 6457 |
| [40] | Adeleke J. T., Theivasanthi T., Thiruppathi M., Swaminathan M., Akomolafe T., Alabi A. B., ,. Appl. Surf. Sci. 2018, 455, 195— 200 |
| [41] | Zhang W., Ji Y. Y., Peng H., Dai S. Y., Liu Y., Zhang J. M., Wang D. M., ,Chem. J. Chinese Universities, 2018,39( 9), 1985— 1992 |
| (张武, 纪妍妍, 彭涵, 戴少英, 刘莹, 张纪梅, 王冬梅. 高等学校化学学报, 2018, 39(9), 1985— 1992) | |
| [42] | Zhang J. W., Wang S., Liu F. S., Fu X. J., Ma G. Q., Hou M. S., Tang Z., ,Acta Phys.-Chem. Sin., 2019,35( 8), 885— 895 |
| (章家伟, 王晟, 刘福生, 付小杰, 马国权, 侯美顺, 唐卓.物理化学学报,2019,35(8), 885— 895) | |
| [43] | Fu L., Sokiransky M. M., Wang J. M., Lai G. S., Yu A. M., ,. Physica E 2016, 83, 146— 150 |
| [44] | Cao X .J ., Zhou M., Ma Y. J., Liu F., Yang N., Xie Y ., Anal. Methods, 2018,10, 1902— 1910 |
| [45] |
Li T. T., Ma Y. J., Liu J., He C. X., Zhou M., Peng B., ,Chin. J. Appl. Chem., 2012,29( 8), 954— 961
doi: 10.3724/SP.J.1095.2012.00405 |
|
(李婷婷, 马永钧, 刘婧, 何春晓, 周敏, 彭波.应用化学,2012,29(8), 954— 961)
doi: 10.3724/SP.J.1095.2012.00405 |
|
| [46] | Wang W. J., Wang L., Zou L. N., Li G. P., Ye B. X ., Talanta 2016, 150, 346— 354 |
| [47] | Jiang D. W., Zhou T. S., Sun Q., Yu Y. Y., Shi G. Y., Jin L. T., ,. Chin. J. Chem. 2011, 29, 2505— 2510 |
| [48] | Ferreira V. C., Monteiro O. C., ,. Electrochim. Acta 2013, 113, 817— 824 |
| [49] | Sun J. Y., Guo Y. P., Wang Y., Cao D., Tian S.C., Xiao K., Mao R., Zhao X., ,. Chem. Eng. J. 2018, 332, 312— 320 |
| [50] | Tantis L., Stathatos E., Mantzavinos D., Lianos P., ,. J. Chem. Technol. Biotechnol. 2015, 90, 1338— 1344 |
| [51] | Yu Z. Q., Chuang S. S. C., ,. J. Phys. Chem. C 2007, 111, 13813— 13820 |
| [52] | Li R. Z., Zhou A. H., Lu Q., Yang C. Z., Zhang J. D., ,. Colloids Surf. A 2013, 436, 270— 278 |
| [1] | 郝宏蕾, 孟繁雨, 李若钰, 李迎秋, 贾明君, 张文祥, 袁晓玲. 生物质基氮掺杂多孔炭材料的制备及对水中亚甲基蓝的吸附性能[J]. 高等学校化学学报, 2022, 43(6): 20220055. |
| [2] | 柳雪广, 杨晓珊, 马菁菁, 刘伟生. 铕基金属有机框架材料从混合染料中选择性分离亚甲基蓝[J]. 高等学校化学学报, 2022, 43(1): 20210715. |
| [3] | 马莹,王恬,张恒. 氧化石墨烯吸附亚甲基蓝的分子动力学模拟[J]. 高等学校化学学报, 2019, 40(12): 2534. |
| [4] | 索路路, 李生娟, 李应涛, 张莉, 张熙. 三维全碳多孔结构对亚甲基蓝吸附性能的动力学探究[J]. 高等学校化学学报, 2016, 37(11): 2043. |
| [5] | 李锋, 王桂燕, 张岩, 李洪仁. Cu2O微晶的可控制备及可见光催化亚甲基蓝降解性能[J]. 高等学校化学学报, 2015, 36(7): 1351. |
| [6] | 徐鹏, 李佑稷, 刘晨, 李铭, 邓瑞成. 钒掺杂介孔二氧化钛的制备及可见光催化性能[J]. 高等学校化学学报, 2014, 35(9): 1954. |
| [7] | 梁建, 何霞, 董海亮, 刘海瑞, 张华, 许并社. ZnO/ZnSe复合纳米结构的制备及可见光光催化性能[J]. 高等学校化学学报, 2014, 35(3): 455. |
| [8] | 王凯, 齐广宇, 蒋章妹, 林奥雷. 亚甲基蓝溶液中分子二聚现象的压致增强研究[J]. 高等学校化学学报, 2014, 35(11): 2431. |
| [9] | 张雪娜, 钟新文, 刘鸣禹, 李辰, 林海波. 离子交换膜对采用生物膜电极法降解五氯酚的影响[J]. 高等学校化学学报, 2013, 34(12): 2841. |
| [10] | 张振飞, 刘海瑞, 张华, 刘旭光, 贾虎生, 许并社. ZnO/Ag微米球的合成与光催化性能[J]. 高等学校化学学报, 2013, 34(12): 2827. |
| [11] | 赵春玲, 华梅, 赵晓慧, 黄亮亮, 冯亚娟, 牛司朋, 符雪文, 袁聪, 杨云慧. 非标记型血小板衍生生长因子核酸适体传感器[J]. 高等学校化学学报, 2013, 34(1): 61. |
| [12] | 蒋保江, 王鹏飞, 侯宗伟, 乔英杰, 付宏刚. 膨胀石墨层间生长高活性氧化锌及其光催化性能[J]. 高等学校化学学报, 2012, 33(11): 2544. |
| [13] | 艾拜拉·热合曼, 阿斯娅·克里木, 帕提曼·亚森, 帕提曼·尼扎木丁, 阿达来提·阿不都热合曼, 阿布力孜·伊米提. MB-硬脂酸复合薄膜光波导传感器检测氯化氢气体[J]. 高等学校化学学报, 2012, 33(10): 2173. |
| [14] | 刘涛 张崇磊 陈平 汤国庆 林列. 时间分辨光谱技术研究亚甲基蓝与小牛胸腺DNA的相互作用[J]. 高等学校化学学报, 2011, 32(8): 1854. |
| [15] | 刘南, 刘淼, 焦昕倩, 刘歆, 冷粟, 陈力可. Ti-PbO2-La电极处理硝基苯废水[J]. 高等学校化学学报, 2011, 32(6): 1266. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||