

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (9): 1888.doi: 10.7503/cjcu20190185
收稿日期:2019-03-27
出版日期:2019-09-10
发布日期:2019-07-29
通讯作者:
苏二正
E-mail:ezhsu@njfu.edu.cn
基金资助:
WU Rong1,2,DONG Qihui1,2,SUN Yiyi1,SU Erzheng1,2,*( )
)
Received:2019-03-27
Online:2019-09-10
Published:2019-07-29
Contact:
SU Erzheng
E-mail:ezhsu@njfu.edu.cn
Supported by:摘要:
以食品工业中常用的木瓜蛋白酶为模式酶, 建立了吸附-纤维素覆膜联合固定化酶方法. 通过对吸附载体类别、 纤维素种类及溶剂、 保护剂种类及其浓度、 干燥方式及时间等的优化, 得到最佳的吸附-纤维素覆膜联合固定化酶工艺. 以硅藻土或HPD-417(大孔树脂)作为吸附载体, 甲基纤维素(分子量40000~50000)丙酮溶液作为覆膜溶液, 加入6%(质量分数)的聚乙二醇或麦芽糖作为覆膜保护剂, 于4 ℃干燥9 h, 制得固定化木瓜蛋白酶, 硅藻土吸附-纤维素覆膜固定化酶酶活回收率达到96.50%, HPD-417吸附-纤维素覆膜固定化酶酶活回收率达到93.92%. 对吸附-纤维素覆膜固定化酶的性质进行了研究, 发现纤维素覆膜后固定化酶具有良好的热稳定性, 于80 ℃下保存12 h后, 固定化酶活残余率仍然能保持90%左右; 在pH=4.5~9.5的范围内, 固定化酶的稳定性较好; 连续使用9次后, 固定化酶活残余率仍能保持95%左右.
中图分类号:
TrendMD:
吴蓉, 董其惠, 孙伊伊, 苏二正. 吸附-纤维素覆膜联合固定化酶. 高等学校化学学报, 2019, 40(9): 1888.
WU Rong, DONG Qihui, SUN Yiyi, SU Erzheng. Efficient Enzyme Immobilization by Combining Adsorption and Cellulose Membrane Coating †. Chem. J. Chinese Universities, 2019, 40(9): 1888.
| Adsorption material | Protein immobilization yield(%) | Immobilized enzyme activity/(U·g-1) | Enzyme activity recovery rate(%) |
|---|---|---|---|
| D130 | 94.91±0.07 | 11.68±0.040 | 41.91±1.57 |
| HPD-450 | 99.01±0.06 | 12.18±0.016 | 40.49±0.27 |
| ADS-17 | 95.95±0.18 | 25.68±0.054 | 66.51±0.59 |
| HPD-417 | 95.11±0.07 | 28.57±0.032 | 60.33±0.46 |
| D101 | 96.21±0.33 | 9.38±0.042 | 30.38±0.34 |
| AB-8 | 95.65±0.03 | 10.34±0.12 | 39.63±0.90 |
| Activated carbon(30 mesh) | 34.12±0.49 | 6.01±0.012 | 43.29±1.34 |
| Activated carbon(40 mesh) | 35.49±0.33 | 6.09±0.022 | 37.64±0.46 |
| Zeolite | 38.69±0.25 | 9.44±0.026 | 42.96±0.61 |
| Kaolin | 55.55±0.25 | 29.07±0.016 | 98.13±1.12 |
| Imvite | 100.00 | 32.76±0.026 | 99.47±1.28 |
| Attapulgite | 58.92±0.40 | 13.05±0.032 | 53.08±1.76 |
| Diatomite | 51.47±0.09 | 30.61±0.022 | 98.38±2.62 |
Table 1 Immobilization of papain on different carriers by adsorbing method
| Adsorption material | Protein immobilization yield(%) | Immobilized enzyme activity/(U·g-1) | Enzyme activity recovery rate(%) |
|---|---|---|---|
| D130 | 94.91±0.07 | 11.68±0.040 | 41.91±1.57 |
| HPD-450 | 99.01±0.06 | 12.18±0.016 | 40.49±0.27 |
| ADS-17 | 95.95±0.18 | 25.68±0.054 | 66.51±0.59 |
| HPD-417 | 95.11±0.07 | 28.57±0.032 | 60.33±0.46 |
| D101 | 96.21±0.33 | 9.38±0.042 | 30.38±0.34 |
| AB-8 | 95.65±0.03 | 10.34±0.12 | 39.63±0.90 |
| Activated carbon(30 mesh) | 34.12±0.49 | 6.01±0.012 | 43.29±1.34 |
| Activated carbon(40 mesh) | 35.49±0.33 | 6.09±0.022 | 37.64±0.46 |
| Zeolite | 38.69±0.25 | 9.44±0.026 | 42.96±0.61 |
| Kaolin | 55.55±0.25 | 29.07±0.016 | 98.13±1.12 |
| Imvite | 100.00 | 32.76±0.026 | 99.47±1.28 |
| Attapulgite | 58.92±0.40 | 13.05±0.032 | 53.08±1.76 |
| Diatomite | 51.47±0.09 | 30.61±0.022 | 98.38±2.62 |
| Cellulose | Adsorption material | Immobilized enzyme activity by adsorption/(U·g-1) | Immobilized enzyme activity after coating/(U·g-1) | Residual enzyme activity after coating(%) |
|---|---|---|---|---|
| Cellulose acetate | Diatomite | 30.94±0.27 | 18.68±0.88 | 60.37±0.73 |
| ADS-17 | 25.56±1.92 | 17.60±0.93 | 68.88±0.39 | |
| HPD-417 | 27.66±0.83 | 19.91±1.21 | 71.99±1.38 | |
| Diacetate cellulose | Diatomite | 30.94±0.27 | 20.90±0.96 | 67.54±1.91 |
| ADS-17 | 25.56±1.92 | 18.76±0.77 | 73.41±1.28 | |
| HPD-417 | 27.66±0.83 | 20.78±0.35 | 75.11±0.93 | |
| Methyl cellulose | Diatomite | 30.94±0.27 | 21.03±0.93 | 67.97±1.36 |
| 40000—50000 | ADS-17 | 25.56±1.92 | 19.26±1.24 | 75.34±1.21 |
| HPD-417 | 27.66±0.83 | 21.55±0.82 | 77.91±0.63 | |
| Methyl cellulose | Diatomite | 30.94±0.27 | 21.47±0.69 | 69.38±1.91 |
| 12000—13000 | ADS-17 | 25.56±1.92 | 20.36±0.79 | 79.67±0.86 |
| HPD-417 | 27.66±0.83 | 21.18±0.83 | 76.56±1.22 |
Table 2 Immobilization of papain by adsorbing-cellulose membrane coating method with cellulose dissolved in acetone
| Cellulose | Adsorption material | Immobilized enzyme activity by adsorption/(U·g-1) | Immobilized enzyme activity after coating/(U·g-1) | Residual enzyme activity after coating(%) |
|---|---|---|---|---|
| Cellulose acetate | Diatomite | 30.94±0.27 | 18.68±0.88 | 60.37±0.73 |
| ADS-17 | 25.56±1.92 | 17.60±0.93 | 68.88±0.39 | |
| HPD-417 | 27.66±0.83 | 19.91±1.21 | 71.99±1.38 | |
| Diacetate cellulose | Diatomite | 30.94±0.27 | 20.90±0.96 | 67.54±1.91 |
| ADS-17 | 25.56±1.92 | 18.76±0.77 | 73.41±1.28 | |
| HPD-417 | 27.66±0.83 | 20.78±0.35 | 75.11±0.93 | |
| Methyl cellulose | Diatomite | 30.94±0.27 | 21.03±0.93 | 67.97±1.36 |
| 40000—50000 | ADS-17 | 25.56±1.92 | 19.26±1.24 | 75.34±1.21 |
| HPD-417 | 27.66±0.83 | 21.55±0.82 | 77.91±0.63 | |
| Methyl cellulose | Diatomite | 30.94±0.27 | 21.47±0.69 | 69.38±1.91 |
| 12000—13000 | ADS-17 | 25.56±1.92 | 20.36±0.79 | 79.67±0.86 |
| HPD-417 | 27.66±0.83 | 21.18±0.83 | 76.56±1.22 |
| Cellulose | Adsorption material | Immobilized enzyme activity by adsorption/(U·g-1) | Immobilized enzyme activity after coating/(U·g-1) | Residual enzyme activity after coating(%) |
|---|---|---|---|---|
| Cellulose acetate | Diatomite | 30.94±0.27 | 19.64±0.91 | 63.47±0.72 |
| ADS-17 | 25.56±1.92 | 17.24±1.21 | 67.47±0.69 | |
| HPD-417 | 27.66±0.83 | 19.29±0.66 | 69.74±1.35 | |
| Diacetate cellulose | Diatomite | 30.94±0.27 | 21.02±0.71 | 67.93±1.22 |
| ADS-17 | 25.56±1.92 | 17.30±1.92 | 67.69±0.82 | |
| HPD-417 | 27.66±0.83 | 19.89±1.25 | 71.89±0.69 | |
| Methylcellulose | Diatomite | 30.94±0.27 | 21.44±0.52 | 69.35±0.77 |
| 40000—50000 | ADS-17 | 25.56±1.92 | 18.79±1.12 | 73.51±0.96 |
| HPD-417 | 27.66±0.83 | 20.31±0.57 | 73.42±1.02 | |
| Methylcellulose | Diatomite | 30.94±0.27 | 21.60±0.82 | 69.81±1.28 |
| 12000—13000 | ADS-17 | 25.56±1.92 | 18.56±0.72 | 72.61±0.65 |
| HPD-417 | 27.66±0.83 | 21.04±0.91 | 76.07±0.74 |
Table 3 Immobilization of papain by adsorbing-cellulose membrane coating method with cellulose dissolved in ethyl acetate
| Cellulose | Adsorption material | Immobilized enzyme activity by adsorption/(U·g-1) | Immobilized enzyme activity after coating/(U·g-1) | Residual enzyme activity after coating(%) |
|---|---|---|---|---|
| Cellulose acetate | Diatomite | 30.94±0.27 | 19.64±0.91 | 63.47±0.72 |
| ADS-17 | 25.56±1.92 | 17.24±1.21 | 67.47±0.69 | |
| HPD-417 | 27.66±0.83 | 19.29±0.66 | 69.74±1.35 | |
| Diacetate cellulose | Diatomite | 30.94±0.27 | 21.02±0.71 | 67.93±1.22 |
| ADS-17 | 25.56±1.92 | 17.30±1.92 | 67.69±0.82 | |
| HPD-417 | 27.66±0.83 | 19.89±1.25 | 71.89±0.69 | |
| Methylcellulose | Diatomite | 30.94±0.27 | 21.44±0.52 | 69.35±0.77 |
| 40000—50000 | ADS-17 | 25.56±1.92 | 18.79±1.12 | 73.51±0.96 |
| HPD-417 | 27.66±0.83 | 20.31±0.57 | 73.42±1.02 | |
| Methylcellulose | Diatomite | 30.94±0.27 | 21.60±0.82 | 69.81±1.28 |
| 12000—13000 | ADS-17 | 25.56±1.92 | 18.56±0.72 | 72.61±0.65 |
| HPD-417 | 27.66±0.83 | 21.04±0.91 | 76.07±0.74 |
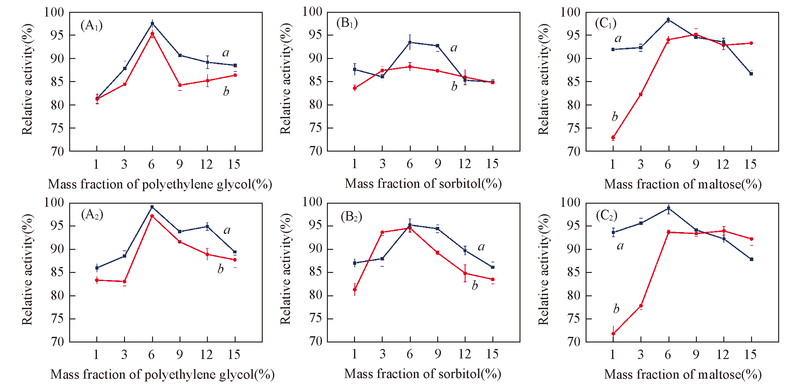
Fig.1 Effects of polyethylene glycol(A1, A2), sorbitol(B1, B2) and maltose(C1, C2) as protectants on immobilized papain with methyl cellulose(12000—13000)(A1—C1) and methyl cellulose(40000—50000)(A2—C2) dissolved in acetone as coating solution a. Diatomite; b. HPD-417.
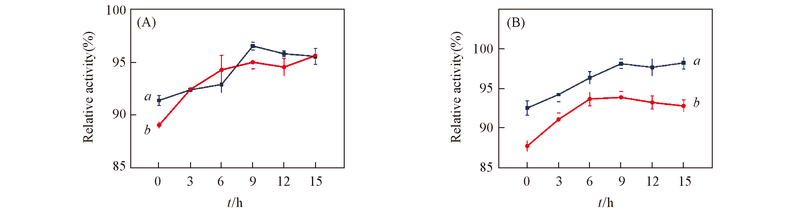
Fig.3 Effect of drying time on the immobilization of papain (A) Polyethylene glycol as the protectant; (B) maltose as the protectant. a. Diatomite; b. HPD-417.
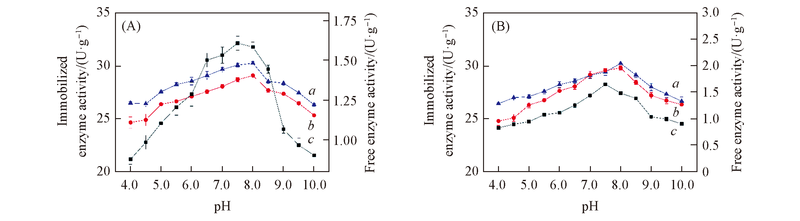
Fig.4 Optimal pH(A) and pH stability(B) of free and immobilized papain by adsorbing-cellulose membrane coating method a. Diatomite; b. HPD-417; c. free enzyme.
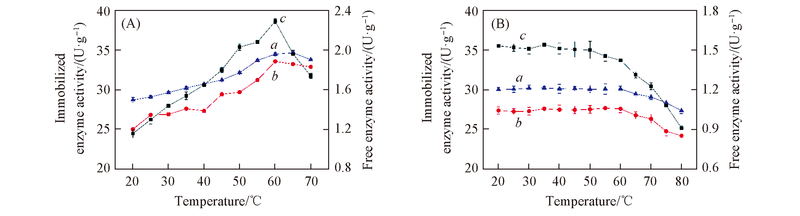
Fig.5 Optimum temperature(A) and thermal stability(B) of free and immobilized papain by adsorbing-cellulose membrane coating method a. Diatomite; b. HPD-417; c. free enzyme.
| [35] |
(雷青娟, 高保娇, 张正国.物理化学学报, 2011, 27( 11), 2697— 2704)
doi: 10.3866/PKU.WHXB20111122 |
| [36] | Raviyan P., Tang J., Rasco B. A ., J. Agric. Food Chem 2003, 51( 18), 5462— 5466 |
| [37] | Xiang X. R., Wan X. M., Suo H. B., Hu Y., ., Acta Phys.-Chim. Sin, 2018, 34( 1), 99— 107 |
| (相欣然, 万晓梅, 索红波, 胡燚.物理化学学报,2018,34(1), 99— 107) | |
| [1] | Hu Y., Yang J., Tang S. S., Chu X. M., Zou B., Huang H., ., Chem. J. Chinese Universities 2013, 34( 5), 1195— 1202 |
| (胡燚, 杨姣, 唐苏苏, 初旭明, 邹彬, 黄和. 高等学校化学学报, 2013, 34(5), 1195— 1202) | |
| [2] | Zheng G. W., Xu J. H ., Curr. Opin Biotechnol, 2011, 22( 6), 784— 792 |
| [3] | Sheldon R. A ., Adv. Synth Catal, 2007, 349( 8/9), 1289— 1307 |
| [4] | Datta S., Christena L. R., Rajaram Y. R. S., 3 Biotech , 2013, 3( 1), 1— 9 |
| [5] | Mateo C., Palomo J. M., Fernandez-Lorente G., Guisan J. M., Fernandez-Lafuente R ., Enzyme Microb Technol, 2007, 40( 4), 1451— 1463 |
| [6] | Rodrigues R. C., Ortiz C., Berenguer-Murcia Á., Torres R., Fernández-Lafuente R ., Chem. Soc Rev, 2013, 42( 15), 6290— 6307 |
| [7] | Wang M. F., Qi W., Su R. X., He Z. M., ., Chem. J. Chinese Universities 2010, 31( 9), 1774— 1779 |
| (王梦凡, 齐崴, 苏荣欣, 何志敏.高等学校化学学报, 2010, 31(9), 1774— 1779) | |
| [8] | Jesionowski T., Zdarta J., Krajewska B ., Adsorption, 2014, 20( 5/6), 801— 821 |
| [9] | Brady D., Jordaan J ., Biotechnol Lett, 2009, 31( 5), 1639 |
| [10] | Hanefeld U., Gardossi L., Magner E ., Chem. Soc Rev, 2009, 38( 2), 453— 468 |
| [11] | Sheldon R. A., Pelt S. V ., Chem. Soc Rev, 2013, 42( 15), 6223— 6235 |
| [12] | Ke C. X., Fan Y. L., Su F., Xu L., Yan Y. J., ., Chin. J. Biotech 2018, 34, 188— 203 |
| ( 柯彩霞, 范艳利, 苏枫, 徐莉, 闫云君 . 生物工程学报, 2018, 34, 188— 203) | |
| [13] | Su E. Z., Yang M., Ning C. X., Ma X. Q., Deng S. W ., J Biotechnol, 2018, 271( 1— 7 |
| [14] | Bradford M. M ., Anal Biochem, 1976, 72( 248— 254 |
| [15] | Li M. L., Su E. Z., You P. Y., Gong X. Y., Sun M. Xu D. S., Wei D. Z ., Plos One, 2010, 5( 12), e15168 |
| [16] | Singhania T., Sinha H., Das P., Mukherjee A. K ., Food Technol Biotechnol, 2015, 53( 1), 91— 95 |
| [17] | Qu Y., Sun L., Li X., Zhou S., Zhang Q., Sun L., Yu D., Jiang L., Tian B ., Food Sci Technol, 2016, 73( 290— 295 |
| [18] | Jesionowski T., Zdarta J., Krajewska B ., Adsorption, 2014, 20( 5/6), 801— 821 |
| [19] | Sheldon R. A., Van P. S ., ChemInform, 2013, 44( 38), 6223— 6235 |
| [20] | Lin K., Li S. X., Xu J. H., Pan J ., Food and Drug, 2011, 13( 5), 153— 156 |
| (林康, 李素霞, 许建和, 潘江.食品与药品,2011,13(5), 153— 156) | |
| [21] | Mateo C., Palomo J. M., Fernandez-Lorente G., Guisan J. M., Fernandez-Lafuente R ., Enzyme Microb Technol, 2007, 40( 4), 1451— 1463 |
| [22] | Sassolas A., Blum L. J., Leca-Bouvier B. D ., Biotechnol Adv, 2012, 30( 3), 489— 511 |
| [23] | Mislovi c ˙ ová D. , Masárová J., Vikartovská A., Gemeiner P., Michalková E ., J Biotechnol, 2004, 110( 1), 11— 19 |
| [24] | Drozd R., Rakoczy R., Wasak A., Junka A., Fija-Kowski K ., Int. J. Biol Macromol, 2018, 108( 462— 470 |
| [25] | Yan C Y., . Microporous Structure Control and Mechanical Performance Study of Micro/nanofibrillated cellulose Films, South China University of Technology, Guangzhou, 2018 |
| ( 闫长媛 . 微/纳米纤维素膜的微孔结构调控及力学性能研究, 广州: 华南理工大学, 2018) | |
| [26] | Hu Y. H., Dai R. J., Liu X. J., Chen Y. L., An J., Wang H. H., Deng Y. L., ., Fine Chem, 2015, 32( 7), 736— 740 |
| (胡英慧, 戴荣继, 刘秀洁, 陈彦丽, 安静, 王慧亨, 邓玉林.精细化工,2015,32(7), 736— 740) | |
| [27] | He P., Huang J. L., Shi Q. C., Wu G. H., Chu Z. Z., Huang Z. L., ., Journal of Northwest A&F University( Nat. Sci. Ed), 2009, 37(3),223—229, 234 |
| ( 何平, 黄家乐, 史清翠, 巫光宏, 初志战, 黄卓烈 . 西北农林科技大学学报(自然科学版), 2009, 37(3),223—229, 234) | |
| [28] | Yang J. C., Dong S., Yang X. M., ., J. Chem. Eng, 2000, 51( 2), 193— 197 |
| (杨基础, 董燊, 杨小民.化工学报,2000, 51( 2), 193— 197) | |
| [29] | Wang H. Y., Gao J., Wang H. O., ., Chin. J.Process Eng, 2008, 8( 6), 1190— 1194 |
| (王红玉, 高静, 王海鸥.过程工程学报, 2008, 8( 6), 1190— 1194) | |
| [30] | Yu M., Zhou H. Y., Ren L. W., Jiang Z. H., Zhou H., Wei P., ., Chin. J. Bioprocess Eng, 2015, 6, 18— 23 |
| ( 于敏, 周海燕, 任立伟, 蒋振华, 周华, 韦萍 . 生物加工过程, 2015, 6, 18— 23) | |
| [31] | Reetz M. T., Zonta A., Vijayakrishnan V., Schimossek K ., J. Mol. Catal A-Chem, 1998, 134( s1—3), 251— 258 |
| [32] | Levin Y., Pecht M., Goldstein L., Katchalski K ., Biochemistry, 1964, 3, 1905— 1913 |
| [33] | Sun X, Wang X ., Biosens Bioelectron, 2010, 25( 11), 2611— 2614 |
| [34] | Shao J J., . Studies on the Methods of Phytoesterase Immobilization and Its Application, Jilin Agricultural University, Changchun, 2008 |
| ( 邵佳甲 . 大豆酯酶的固定化及应用研究, 长春: 吉林农业大学, 2008) | |
| [35] |
Lei Q. J., Gao B. J., Zhang Z. G., ., Acta Phys.-Chim. Sin, 2011, 27( 11), 2697— 2704
doi: 10.3866/PKU.WHXB20111122 |
| [1] | 姜宏斌, 代文臣, 张娆, 徐晓晨, 陈捷, 杨光, 杨凤林. Co3O4/UiO-66@α-Al2O3陶瓷膜对VOCs废气的分离催化性能[J]. 高等学校化学学报, 2022, 43(6): 20220025. |
| [2] | 戴卫, 侯华, 王宝山. 七氟异丁腈负离子结构与反应活性的理论研究[J]. 高等学校化学学报, 2022, 43(6): 20220044. |
| [3] | 郝宏蕾, 孟繁雨, 李若钰, 李迎秋, 贾明君, 张文祥, 袁晓玲. 生物质基氮掺杂多孔炭材料的制备及对水中亚甲基蓝的吸附性能[J]. 高等学校化学学报, 2022, 43(6): 20220055. |
| [4] | 王红宁, 黄丽, 清江, 马腾洲, 蒋伟, 黄维秋, 陈若愚. 香蒲基生物炭的活化及对VOCs吸附的应用[J]. 高等学校化学学报, 2022, 43(4): 20210824. |
| [5] | 陈潇禄, 袁珍闫, 仲迎春, 任浩. 机械球磨制备三苯胺基PAF-106s及C2烃吸附性质[J]. 高等学校化学学报, 2022, 43(3): 20210771. |
| [6] | 孟祥龙, 杨歌, 郭海玲, 刘晨光, 柴永明, 王纯正, 郭永梅. 纳米分子筛的合成及硫化氢吸附性能[J]. 高等学校化学学报, 2022, 43(3): 20210687. |
| [7] | 靳科研, 白璞, 李小龙, 张佳楠, 闫文付. 新型Mg-Al吸附剂去除压水堆核电厂废水中高浓度硼[J]. 高等学校化学学报, 2022, 43(2): 20210516. |
| [8] | 谭乐见, 仲宣树, 王锦, 刘宗建, 张爱英, 叶霖, 冯增国. β-环糊精的低临界溶解温度现象及其在有序纳米孔道片晶制备中的应用[J]. 高等学校化学学报, 2022, 43(11): 20220405. |
| [9] | 郑美琪, 毛方琪, 孔祥贵, 段雪. 类水滑石材料在核废水处理领域的应用[J]. 高等学校化学学报, 2022, 43(10): 20220456. |
| [10] | 田晓康, 张青松, 杨舒淋, 白洁, 陈冰洁, 潘杰, 陈莉, 危岩. 微生物发酵诱导多孔材料: 制备方法和应用[J]. 高等学校化学学报, 2022, 43(10): 20220216. |
| [11] | 张弛, 孙福兴, 朱广山. 双金属同构金属-有机框架材料CAU-21-Al/M的合成、 氮气吸附及复合膜性能[J]. 高等学校化学学报, 2022, 43(1): 20210578. |
| [12] | 马鉴新, 刘晓东, 徐娜, 刘国成, 王秀丽. 一种具有发光传感、 安培传感和染料吸附性能的多功能Zn(II)配位聚合物[J]. 高等学校化学学报, 2022, 43(1): 20210585. |
| [13] | 刘昌辉, 梁国俊, 李妍璐, 程秀凤, 赵显. NH3在硼纳米管表面吸附的密度泛函理论研究[J]. 高等学校化学学报, 2021, 42(7): 2263. |
| [14] | 刘云鸿, 彭新艳. 新型蛋白结合类毒素血液灌流吸附剂的制备及吸附性能[J]. 高等学校化学学报, 2021, 42(6): 1952. |
| [15] | 王红宁, 黄丽, 宋夫交, 朱婷, 黄维秋, 钟璟, 陈若愚. 中空碳纳米球的制备及VOCs吸附性能[J]. 高等学校化学学报, 2021, 42(6): 1704. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||