

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (4): 20220636.doi: 10.7503/cjcu20220636
收稿日期:2022-09-24
出版日期:2023-04-10
发布日期:2022-12-02
通讯作者:
石淑先
E-mail:shisx@mail.buct.edu.cn
基金资助:
YAN Yutian, WU Si, CHANG Kangkang, XIA Yuzheng, CHEN Xiaonong, SHI Shuxian( )
)
Received:2022-09-24
Online:2023-04-10
Published:2022-12-02
Contact:
SHI Shuxian
E-mail:shisx@mail.buct.edu.cn
Supported by:摘要:
金纳米粒子(AuNPs)表面能高, 在水中极易团聚, 使其应用受限. 本文采用物理共混法, 将带有正电荷的温敏聚(N-异丙基丙烯酰胺)(PNIPAM)微凝胶与带负电荷的AuNPs混合, 经自组装制备了微凝胶表面覆盖AuNPs的有机-金属复合粒子AuNPs@PNIPAM. 该复合粒子不仅具有很好的分散稳定性, 而且其粒子的分散液具有温度比色性, 在25 ℃→50 ℃→25 ℃的温度变化过程中呈现“红→紫→红”的可逆颜色变化. 通过对硝基苯酚(4-NP)的还原反应, 研究了复合粒子的催化性能. 结果表明, 复合粒子具有受温度调控的催化能力, 随温度升高催化性能呈现先降后升的趋势. 与文献报道的类似材料相比, AuNPs@PNIPAM复合粒子同时具有温度比色性和催化性能.
中图分类号:
TrendMD:
闫雨甜, 吴思, 常康康, 夏宇正, 陈晓农, 石淑先. 表面覆盖型AuNPs@PNIPAM复合粒子的制备及催化性能. 高等学校化学学报, 2023, 44(4): 20220636.
YAN Yutian, WU Si, CHANG Kangkang, XIA Yuzheng, CHEN Xiaonong, SHI Shuxian. Preparation and Catalytic Performance of Surface-covered AuNPs@PNIPAM Composite Particles. Chem. J. Chinese Universities, 2023, 44(4): 20220636.
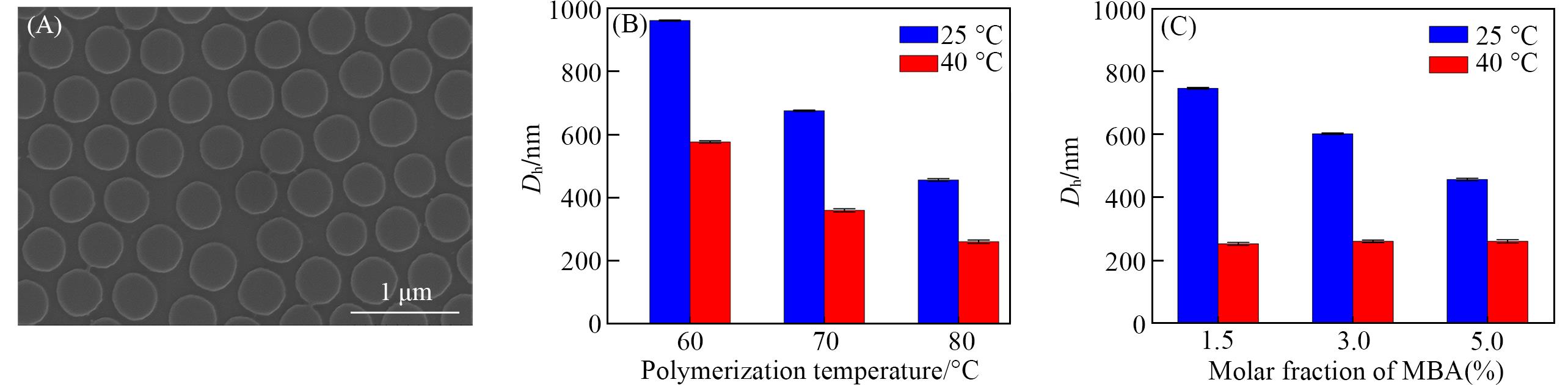
Fig.1 SEM image of PNIPAM microgels prepared at 70 ℃ with 5%(molar fraction) crosslinking agent(A) and Dh of PNIPAM microgels prepared at different temperatures(B) and different molar fractions of crosslinking agent(C)(B) Molar fraction of crosslinking agent is 5%; (C) polymerization temperature is 80 ℃.
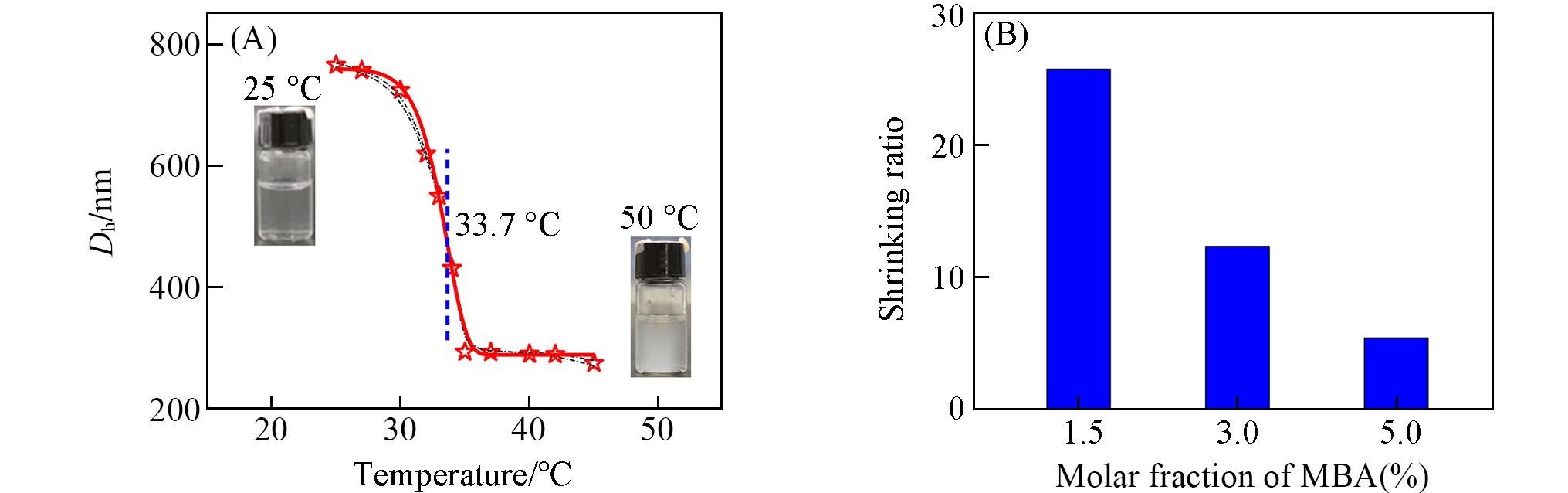
Fig.2 Dh of PNIPAM microgels prepared at 80 ℃ with 1.5% crosslinking agent and its dispersion system at different temperatures(A) and shrinking ratio of PNIPAM microgels prepared at 80 ℃ with different amounts of crosslinking agent(B)Insets of (A): optical photos of PNIPAM microgels dispersion at 25 °C(left) and 50 °C(right).
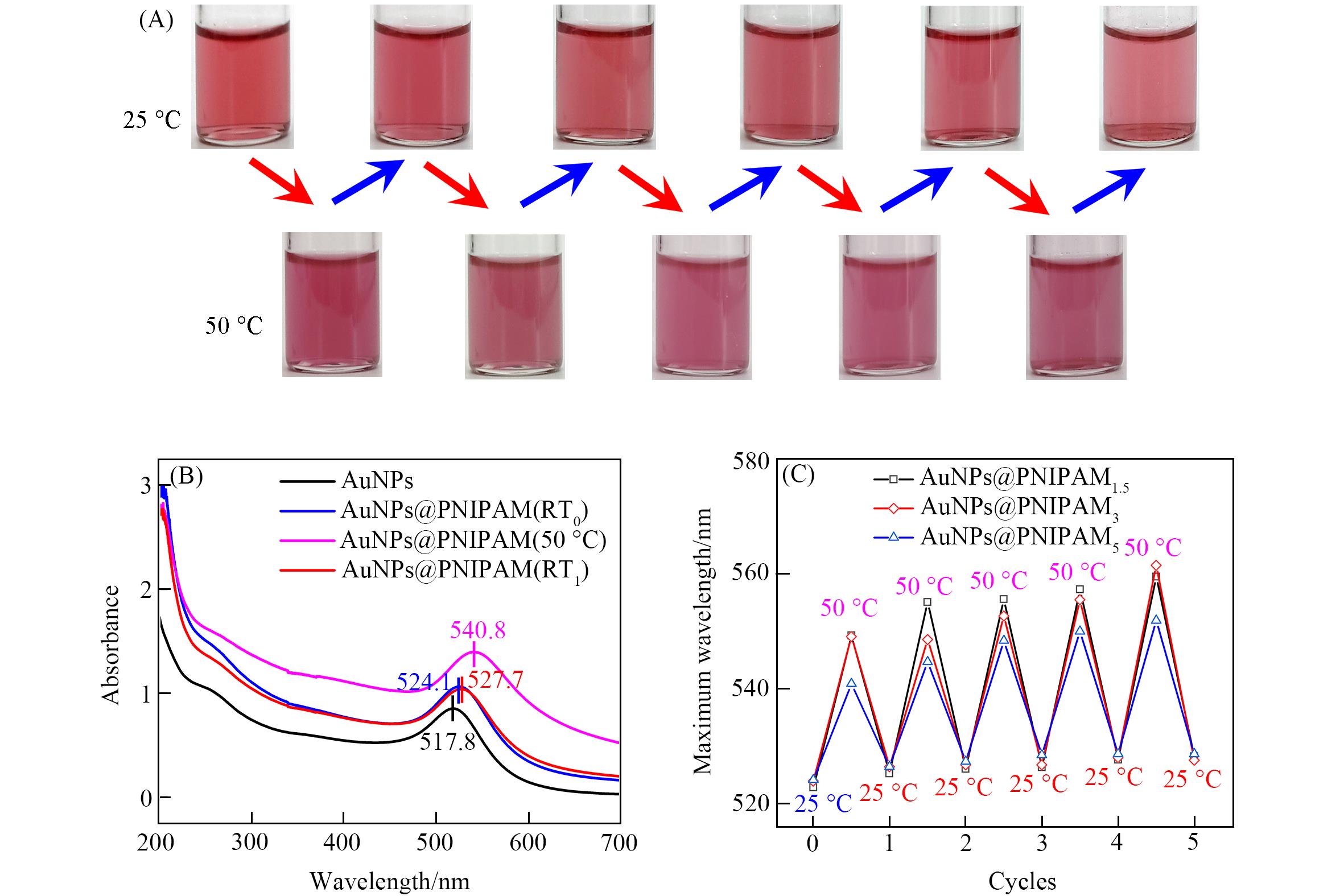
Fig.4 Color changes of AuNPs@PNIPAM dispersion during five heating⁃cooling cycles(A) and UV⁃Vis spectra of AuNPs and AuNPs@PNIPAM dispersion(B) and UV⁃Vis maximum absorption peaks of AuNPs@PNIPAM dispersion in five heating⁃cooling cycles(C)
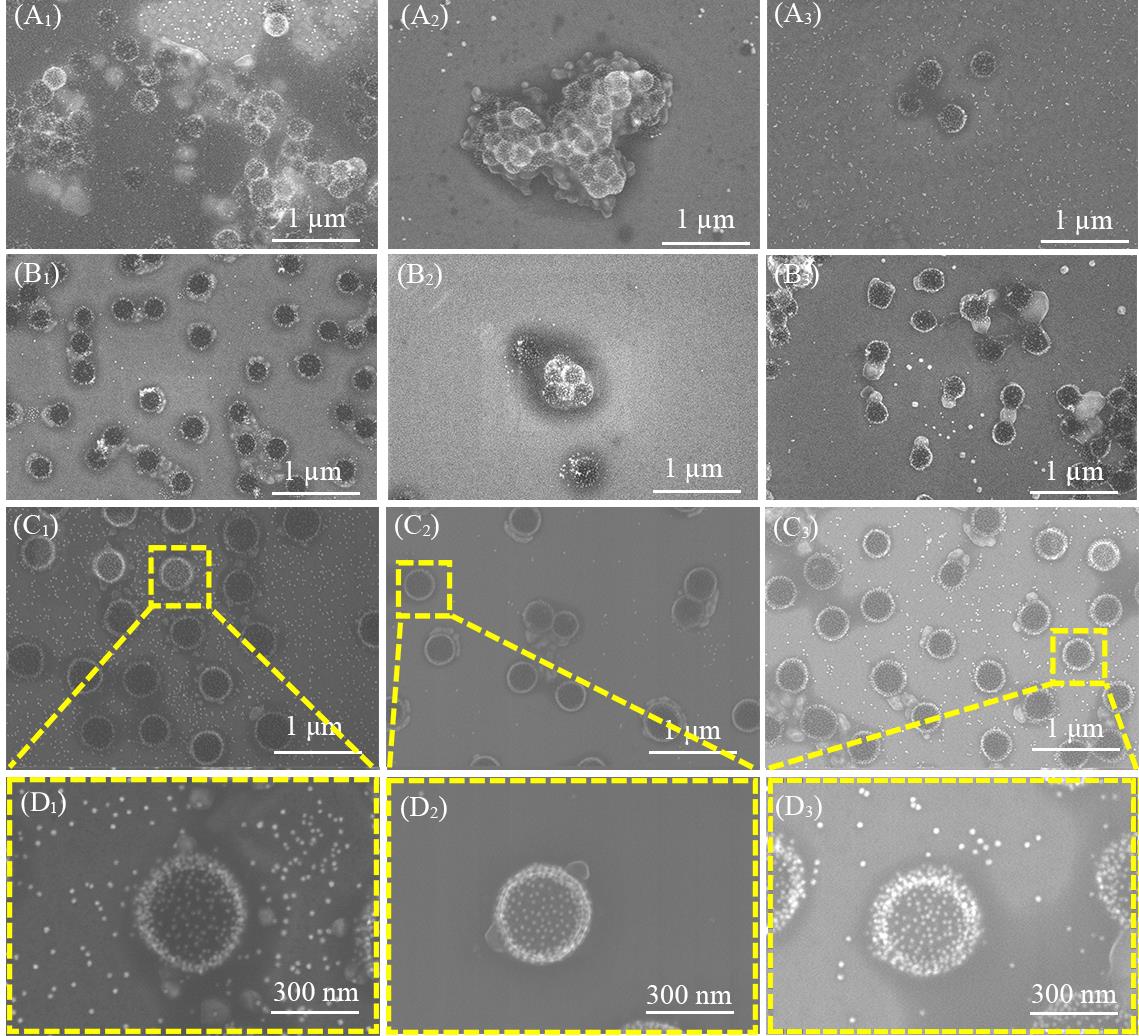
Fig.5 SEM images of AuNPs@PNIPAM composite particles prepared by different dosage of crosslinking agent at initially 25 ℃(A1—D1), after heated to 50 ℃(A2—D2) and after cooled back to 25 ℃(A3—D3)Dosage of crossling agent(%, molar fraction): (A1—A3) 1.5%; (B1—B3) 3.0%; (C1—C3) 5.0%.(D1—D3) The magnified images of (C1—C3).
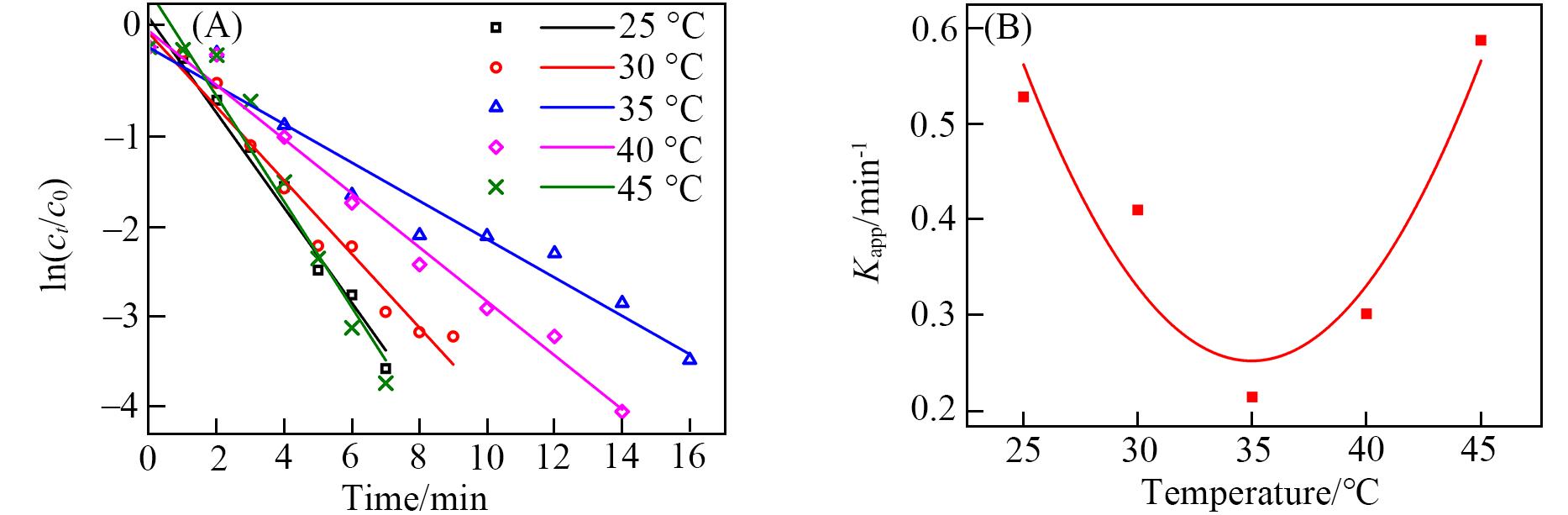
Fig.6 Effect of reaction temperature on the catalytic activity of AuNPs@PNIPAM composite particles(A) Plots of ln(ct /c0) versus reaction time in the presence of AuNPs@PNIPAM; (B) plot of Kappversus temperature in the presence of AuNPs@PNIPAM.
| No. | Composite | Temperature range/°C | Color change | Δλ | Note | Ref. |
|---|---|---|---|---|---|---|
| 1 | AuNPs@PNIPAM in PAAm hydrogel | 24—50 | Red↔grayish violet | 176 | Eight times concentration of AuNPs@PNIPAM in hydrogel | [ |
| 2 | PNIPAM/AuNP | 25—50 | Wine↔violet | 152 | Low AuNPs loadings(surface covered) | [ |
| 3 | PNIPAM⁃Au a | 20—50 | — b | 40 | In situ reduction | [ |
| 4 | Au@NH2⁃PNIPAM | 25—45 | — b | 30 | PNIPAM⁃NH2 | [ |
| 5 | Au@PNIPAM⁃co⁃AA | 25—50 | Wine↔violet | 29 | Equilibrium time was 30 min | [ |
| 6 | AuNPs@PNIPAM a | 25—50 | Red↔purple | 16.7 | Equilibrium time was 10 min | This work |
| 7 | AuAg@pNIPAM@Ag a | 25—50 | — b | 11 | Core⁃satellite | [ |
| 8 | Au@pNIPAM | 10—50 | — b | 10 | Core⁃shell | [ |
| 9 | PNIPAMs⁃AuNP a | 25—50 | Remained red and transparent | 1.2 | Core⁃shell | [ |
| 10 | SiO2@PMBA@Au@PNIPAM a | 20—40 | — b | No shift | Yolk⁃shell | [ |
| 11 | MNP@SiO2⁃PNIPAm⁃AuNPs a | 25—40 | Small variation in transparency | No date d | Core⁃satellite | [ |
| 12 | Au⁃PNIPAM | 25—50 | — b | No date d | Yolk⁃shell | [ |
| 13 | Core⁃satellite nanoassemblies | 25—55 | — b | No date d | Core⁃satellite | [ |
| 14 | PNIPAM⁃Au a | 20—45 | Small variation in transparency | — b | High AuNPs loadings(random filled) | [ |
| 15 | Gold@polymer core⁃shell nanoparticles | 25—50 | Small variation in transparency | — b | Core⁃shell | [ |
| 16 | PNIPAM/Au@meso⁃SiO2a | 25—50 | Light purple(transparent to cloudy) | — b | Hollow microsphere | [ |
Table 1 Comparison of color change and absorption peak shifts in different references
| No. | Composite | Temperature range/°C | Color change | Δλ | Note | Ref. |
|---|---|---|---|---|---|---|
| 1 | AuNPs@PNIPAM in PAAm hydrogel | 24—50 | Red↔grayish violet | 176 | Eight times concentration of AuNPs@PNIPAM in hydrogel | [ |
| 2 | PNIPAM/AuNP | 25—50 | Wine↔violet | 152 | Low AuNPs loadings(surface covered) | [ |
| 3 | PNIPAM⁃Au a | 20—50 | — b | 40 | In situ reduction | [ |
| 4 | Au@NH2⁃PNIPAM | 25—45 | — b | 30 | PNIPAM⁃NH2 | [ |
| 5 | Au@PNIPAM⁃co⁃AA | 25—50 | Wine↔violet | 29 | Equilibrium time was 30 min | [ |
| 6 | AuNPs@PNIPAM a | 25—50 | Red↔purple | 16.7 | Equilibrium time was 10 min | This work |
| 7 | AuAg@pNIPAM@Ag a | 25—50 | — b | 11 | Core⁃satellite | [ |
| 8 | Au@pNIPAM | 10—50 | — b | 10 | Core⁃shell | [ |
| 9 | PNIPAMs⁃AuNP a | 25—50 | Remained red and transparent | 1.2 | Core⁃shell | [ |
| 10 | SiO2@PMBA@Au@PNIPAM a | 20—40 | — b | No shift | Yolk⁃shell | [ |
| 11 | MNP@SiO2⁃PNIPAm⁃AuNPs a | 25—40 | Small variation in transparency | No date d | Core⁃satellite | [ |
| 12 | Au⁃PNIPAM | 25—50 | — b | No date d | Yolk⁃shell | [ |
| 13 | Core⁃satellite nanoassemblies | 25—55 | — b | No date d | Core⁃satellite | [ |
| 14 | PNIPAM⁃Au a | 20—45 | Small variation in transparency | — b | High AuNPs loadings(random filled) | [ |
| 15 | Gold@polymer core⁃shell nanoparticles | 25—50 | Small variation in transparency | — b | Core⁃shell | [ |
| 16 | PNIPAM/Au@meso⁃SiO2a | 25—50 | Light purple(transparent to cloudy) | — b | Hollow microsphere | [ |
| No. | Catalyst | Configuration | Au(composite)/4⁃NP a | T/℃ | t1/2/min | Kapp/min-1 | Con.(%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | MNP@SiO2⁃PNIPAm⁃AuNPs | Core⁃satellite | 7.5 | 25 | 0.99 | 0.70 | >97 | [ |
| 2 | AuNPs@GFDP | Core⁃shell | 0.43 | 25 | 1.05 | 0.66 | >97 | [ |
| 3 | AuNPs@PNIPAM | Surface covered | 0.17 | 25 | 1.31 | 0.53 | >97 | This work |
| 4 | PNIPAM⁃Au | Random filled | 0.00068 | 20 | 1.47 | 0.47 | >85 | [ |
| 5 | SiO2@PMBA@Au@PNIPAM | Yolk⁃shell | 0.059 | 20 | 3.85 | 0.18 | >97 | [ |
| 6 | PNIPAM⁃AuNPs | Core⁃shell | 0.23 | 25 | 5.33 | 0.16 | >93 | [ |
| 7 | Au/micelle composite | Random filled | 0.018 | 25 | 91.69 | 7.56×10-3 | >80 | [ |
| 8 | Au@P1 | Core⁃shell | 0.017 | 25 | 1.44×104 | 4.80×10-5 | >95 | [ |
| 9 | Au@PNIPAm/PEI | Surface covered | 13.5 | 29 | 0.47 | 1.46 | >30 | [ |
| 10 | Au@mesoporous⁃SiO2 | Hollow microsphere | 0.024 | 30 | 1.16 | 0.60 | >90 | [ |
| 11 | AuAg@pNIPAM@Ag | Core⁃satellite | 4.5 | 25 | 1.39 | 0.50 | >97 | [ |
| 12 | RGO/PVEIM⁃b⁃PNIPAM/GNP | Surface covered(on sheets) | 0.33 | 30 | 1.51 | 0.46 | >85 | [ |
| 13 | Fe3O4/Au/SiO2/NIPAM | Yolk⁃shell | 0.0035 | 25 | 3.47 | 0.20 | >90 | [ |
| 14 | PNIPAM⁃catechol@Au | Random filled | 7.0 | 25 | 10.50 | 6.60×10-2 | >60 | [ |
| 15 | AuNR@PNV@AuNP | Core⁃satellite | 0.05 mg b | 25±5 | 2.77 | 0.25 | >97 | [ |
| 16 | PNIPAM⁃Au | Random filled | 0.03 mg b | 30 | 4.08 | 0.17 | >95 | [ |
Table 2 Comparison of the reaction time and Kapp in different references
| No. | Catalyst | Configuration | Au(composite)/4⁃NP a | T/℃ | t1/2/min | Kapp/min-1 | Con.(%) | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | MNP@SiO2⁃PNIPAm⁃AuNPs | Core⁃satellite | 7.5 | 25 | 0.99 | 0.70 | >97 | [ |
| 2 | AuNPs@GFDP | Core⁃shell | 0.43 | 25 | 1.05 | 0.66 | >97 | [ |
| 3 | AuNPs@PNIPAM | Surface covered | 0.17 | 25 | 1.31 | 0.53 | >97 | This work |
| 4 | PNIPAM⁃Au | Random filled | 0.00068 | 20 | 1.47 | 0.47 | >85 | [ |
| 5 | SiO2@PMBA@Au@PNIPAM | Yolk⁃shell | 0.059 | 20 | 3.85 | 0.18 | >97 | [ |
| 6 | PNIPAM⁃AuNPs | Core⁃shell | 0.23 | 25 | 5.33 | 0.16 | >93 | [ |
| 7 | Au/micelle composite | Random filled | 0.018 | 25 | 91.69 | 7.56×10-3 | >80 | [ |
| 8 | Au@P1 | Core⁃shell | 0.017 | 25 | 1.44×104 | 4.80×10-5 | >95 | [ |
| 9 | Au@PNIPAm/PEI | Surface covered | 13.5 | 29 | 0.47 | 1.46 | >30 | [ |
| 10 | Au@mesoporous⁃SiO2 | Hollow microsphere | 0.024 | 30 | 1.16 | 0.60 | >90 | [ |
| 11 | AuAg@pNIPAM@Ag | Core⁃satellite | 4.5 | 25 | 1.39 | 0.50 | >97 | [ |
| 12 | RGO/PVEIM⁃b⁃PNIPAM/GNP | Surface covered(on sheets) | 0.33 | 30 | 1.51 | 0.46 | >85 | [ |
| 13 | Fe3O4/Au/SiO2/NIPAM | Yolk⁃shell | 0.0035 | 25 | 3.47 | 0.20 | >90 | [ |
| 14 | PNIPAM⁃catechol@Au | Random filled | 7.0 | 25 | 10.50 | 6.60×10-2 | >60 | [ |
| 15 | AuNR@PNV@AuNP | Core⁃satellite | 0.05 mg b | 25±5 | 2.77 | 0.25 | >97 | [ |
| 16 | PNIPAM⁃Au | Random filled | 0.03 mg b | 30 | 4.08 | 0.17 | >95 | [ |
| 1 | Sarfraz N., Khan I., Chem. Asian J., 2021, 16, 720—742 |
| 2 | Hua Z., Yu T., Liu D., Xianyu Y., Biosens. Bioelectron., 2021, 179, 113076—113089 |
| 3 | Wang Y. Q., Zhang Y. Y., Wu X. G., He X. W., Li W. Y., Mater. Lett., 2015, 143, 326—329 |
| 4 | Falahati M., Attar F., Sharifi M., Saboury A. A., Salihi A., Aziz F. M., Kostova I., Burda C., Priecel P., Lopez⁃Sanchez J. A., Laurent S., Hooshmand N., El⁃Sayed M. A., Biochim. Biophys. Acta. Gen. Subj., 2020, 1864, 129435—129461 |
| 5 | Zhao P., Li N., Astruc D., Coord. Chem. Rev., 2013, 257, 638—665 |
| 6 | Chatterjee S., Lou X. Y., Liang F., Yang Y. W., Coord. Chem. Rev., 2022, 459, 214461—214495 |
| 7 | Siirilä J., Karesoja M., Pulkkinen P., Malho J. M., Tenhu H., Eur. Polym. J., 2019, 115, 59—69 |
| 8 | Zheng P., Jiang X., Zhang X., Zhang W., Shi L., Langmuir, 2006, 22, 9393—9396 |
| 9 | Zhang C. L., Cao F. H., Wang J. L., Yu Z. L., Ge J., Lu Y., Wang Z. H., Yu S. H., ACS Appl. Mater. Interfaces, 2017, 9, 24857—24863 |
| 10 | Tanaka T., Phys. Rev. Lett., 1978, 40, 820—823 |
| 11 | Yee M. M., Tsubone M., Morita T., Yusa S. I., Nakashima K., J. Lumin., 2016, 176, 318—323 |
| 12 | Ngadaonye J. I., Cloonan M. O., Geever L. M., Higginbotham C. L., J. Polym. Res., 2011, 18, 2307—2324 |
| 13 | Chen Z., Cui Z. M., Cao C. Y., He W. D., Jiang L., Song W. G., Langmuir, 2012, 28, 13452—13458 |
| 14 | Wang D., Duan H., Lü J., Lü C., J. Mater. Chem. A, 2017, 5, 5088—5097 |
| 15 | Chen L. Y., Ou C. M., Chen W. Y., Huang C. C., Chang H. T., ACS Appl. Mater. Interfaces, 2013, 5, 4383—4388 |
| 16 | Marcelo G., López⁃González M., Mendicuti F., Tarazona M. P., Valiente M., Macromolecules, 2014, 47, 6028—6036 |
| 17 | Tzounis L., Dona M., Lopez⁃Romero J. M., Fery A., Contreras⁃Caceres R., ACS Appl. Mater. Interfaces, 2019, 11, 29360—29372 |
| 18 | Murphy S., Jaber S., Ritchie C., Karg M., Mulvaney P., Langmuir, 2016, 32, 12497—12503 |
| 19 | Magnozzi M., Brasse Y., König T. A. F., Bisio F., Bittrich E., Fery A., Canepa M., ACS Appl. Nano Mater., 2020, 3, 1674—1682 |
| 20 | Tian J., Huang B., Zhang W., Langmuir, 2019, 35, 266—275 |
| 21 | Wu L., Glebe U., Böker A., Adv. Mater. Interfaces, 2017, 4, 1700092—1700101 |
| 22 | Song J. E., Cho E. C., Sci. Rep., 2016, 6, 34622—34631 |
| 23 | Contreras⁃Cáceres R., Pacifico J., Pastoriza⁃Santos I., Pérez⁃Juste J., Fernández⁃Barbero A., Liz⁃Marzán L. M., Adv. Funct. Mater., 2009, 19, 3070—3076 |
| 24 | Goodall A. R., Wilkinson M. C., Hearn J., J. Polym. Sci. A: Polym. Chem., 1977, 15, 2193—2218 |
| 25 | Gu T., The Study on the Poly(N⁃isopropylacrylamide) Hydrogel Microspheres with Narrow Phase Transition Temperature Range, Donghua University, Shanghai, 2007 |
| 顾婷. 相转变温度范围窄的聚(N⁃异丙基丙烯酰胺)水凝胶微球的研究, 上海: 东华大学, 2007 | |
| 26 | Wu Y. M., Liu Y. T., Xu J., Zhang N. N., E⁃Polymers, 2010, 10, 78—86 |
| 27 | Varga I., Gilányi T., Mészáros R., Filipcsei G., Zrínyi M., J. Phys. Chem. B, 2001, 105, 9071—9076 |
| 28 | Zhang Q. S., Synthesis and Characterization of Novel Temperature⁃sensitive Poly(N⁃isopropylacrylamide) Based Microgels, Donghua University, Shanghai, 2008 |
| 张青松. 新型聚(N⁃异丙基丙烯酰胺)类温敏性微凝胶的合成与表征, 上海: 东华大学, 2008 | |
| 29 | Contreras⁃Caceres R., Schellkopf L., Fernandez⁃Lopez C., Pastoriza⁃Santos I., Perez⁃Juste J., Stamm M., Langmuir, 2015, 31, 1142—1149 |
| 30 | Lim S., Song J. E., La J. A., Cho E. C., Chem. Mater., 2014, 26, 3272—3279 |
| 31 | Choe A., Yeom J., Shanker R., Kim M. P., Kang S., Ko H., NPG Asia Mater., 2018, 10, 912—922 |
| 32 | Du J.T., Qiao M., Pu Y., Wang J. X., Chen J. F., Appl. Catal. A⁃Gen., 2021, 624, 118323—118331 |
| 33 | Qian Z., Guye K. N., Masiello D. J., Ginger D. S., J. Phys. Chem. B, 2017, 121, 1092—1099 |
| 34 | Samai S., Qian Z., Ling J., Guye K. N., Ginger D. S., ACS Appl. Mater. Interfaces, 2018, 10, 8976—8984 |
| 35 | Turek V. A., Cormier S., Sierra⁃Martin B., Keyser U. F., Ding T., Baumberg J. J., Adv. Opt. Mater., 2018, 6, 1701270—1701275 |
| 36 | Carregal⁃Romero S., Buurma N. J., Pérez⁃Juste J., Liz⁃Marzán L. M., Hervés P., Chem. Mater., 2010, 22, 3051—3059 |
| 37 | Zhao P., Feng X., Huang D., Yang G., Astruc D., Coord. Chem. Rev., 2015, 287, 114—136 |
| 38 | Pradhan N., Pal A., Pal T., Colloids Surf. A: Physicochem. Eng. Asp., 2002, 196, 247—257 |
| 39 | Wang Y., Wang L., Hao J., Dong S., New J. Chem., 2018, 42, 2149—2157 |
| 40 | Besold D., Risse S., Lu Y., Dzubiella J., Ballauff M., Ind. Eng. Chem. Res., 2021, 60, 3922—3935 |
| 41 | Shi S., Zhang L., Wang T., Wang Q., Gao Y., Wang N., Soft Matter, 2013, 9, 10966—10970 |
| 42 | Liu W., Zhu X., Xu C., Dai Z., Meng Z., Nanomaterials (Basel), 2018, 8, 963—978 |
| 43 | Wu S., Lei L., Xia Y. Z., Oliver S., Chen X. N., Boyer C., Nie Z. Y., Shi S. X., Polym. Chem., 2021, 12, 6903—6913 |
| 44 | Lu J., Yang Y., Gao J., Duan H., Lu C., Langmuir, 2018, 34, 8205—8214 |
| 45 | Xiong D., Li Z., Zou L., He Z., Liu Y., An Y., Ma R., Shi L., J. Colloid Interface Sci., 2010, 341, 273—279 |
| 46 | Tan N. P. B., Lee C. H., Li P., Polymers (Basel), 2016, 8, 105—118 |
| 47 | Chen S., Xiang Y., Peng C., Xu W., Banks M. K., Wu R., Inorg. Chem. Front., 2019, 6, 903—913 |
| 48 | Yang K., Dai Z., Chu Y., Chen G., Micro Nano Lett., 2016, 11, 129—136 |
| [1] | 匡华艺, 陈晨. 贵金属纳米框架设计合成及电催化性能的研究进展[J]. 高等学校化学学报, 2023, 44(1): 20220586. |
| [2] | 张太文, 郭军, 张丹, 袁常梅, 邱双艳. trz-Cl-Cu-PMo12的合成、 表征及催化氧化碘离子性能[J]. 高等学校化学学报, 2022, 43(10): 20220215. |
| [3] | 李荣烨, 倪云霞, 刘丹丹, 李志, 程玉新, 夏明欣, 付小会. 一种新型温度响应性聚氨基酸/聚类肽嵌段共聚物的合成与表征[J]. 高等学校化学学报, 2021, 42(3): 850. |
| [4] | 刘亚冰,李明阳,田戈,阿拉腾沙嘎,裴桐鹤,聂婧思. 基于2-氨基吡啶的两个簇基超分子化合物的合成、 结构及催化性能[J]. 高等学校化学学报, 2020, 41(5): 995. |
| [5] | 张荣慧, 闵灯, 王来来, 谢文健. 气相氟化合成氢氟烯烃催化剂的研究进展[J]. 高等学校化学学报, 2020, 41(10): 2199. |
| [6] | 朱钦富, 胡可珍, 李小杰, 陈明清. pH响应性树枝状聚合物-金纳米粒子复合药物载体的制备[J]. 高等学校化学学报, 2019, 40(5): 1065. |
| [7] | 李颖, 康君君, 赵雪茹, 徐文凯, 齐琦. 金修饰磁性石墨烯基分子印迹复合材料的制备及对水中邻苯二甲酸二正丁酯的电化学传感检测[J]. 高等学校化学学报, 2019, 40(3): 448. |
| [8] | 孙思齐,王影,孙传胤,王润伟,张震东,张宗弢,裘式纶. 碗状双亲型ZSM-5分子筛负载金纳米粒子的制备及催化性能[J]. 高等学校化学学报, 2019, 40(12): 2436. |
| [9] | 马清海,崔放,刘沐斐,徐林煦,张佳佳,崔铁钰. Pd/PdO纳米复合微球的合成及催化性能[J]. 高等学校化学学报, 2019, 40(10): 2041. |
| [10] | 张晶, 董玉明, 刘湘, 李和兴. Z型光催化剂Sb2WO6/g-C3N4的制备及光催化性能[J]. 高等学校化学学报, 2019, 40(1): 123. |
| [11] | 冯微, 王博蔚, 郑艳, 姜洋. 金纳米粒子簇的制备及表面增强拉曼光谱[J]. 高等学校化学学报, 2018, 39(9): 1875. |
| [12] | 张璐涛, 周光明, 罗丹, 陈蓉. 表面增强拉曼光谱快速检测蜂蜜中的金霉素残留[J]. 高等学校化学学报, 2018, 39(8): 1662. |
| [13] | 肖珊珊, 欧阳逸挺, 李小云, 王朝, 吴攀, 邓兆, 陈丽华, 苏宝连. 介孔SiO2包覆Ag/ZIF-8核壳型催化材料的合成[J]. 高等学校化学学报, 2018, 39(6): 1235. |
| [14] | 李娟, 祝林芳, 赵安婷, 雷国明, 高丽, 夏文, 汪丽. 六元瓜环修饰的花团状铜的催化活性[J]. 高等学校化学学报, 2018, 39(3): 422. |
| [15] | 李艾橘, 王玉玺, 卢少勇, 刘堃. 硫醇末端基聚合物对金纳米粒子配体置换[J]. 高等学校化学学报, 2018, 39(3): 552. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||