

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (3): 709.doi: 10.7503/cjcu20200475
收稿日期:2020-07-20
出版日期:2021-03-10
发布日期:2021-03-08
通讯作者:
刘俊良
E-mail:liujliang5@mail.sysu.edu.cn
基金资助:
RUAN Zeyu, DU Shannan, HUANG Guozhang, TONG Mingliang, LIU Junliang( )
)
Received:2020-07-20
Online:2021-03-10
Published:2021-03-08
Contact:
LIU Junliang
E-mail:liujliang5@mail.sysu.edu.cn
摘要:
阻转异构体是一类特殊的手性化合物. 利用轴手性配体制得的具有阻转异构行为的配合物在不对称催化等领域有广泛的应用. 相比之下, 通过原位反应由非手性底物制备这类手性配合物的报道较少, 而利用非轴手性化合物原位组装得到非轴手性阻转异构体的报道则更加罕见. 乙腈是一种廉价易得的基本化工原料, 在金属离子存在下活化乙腈分子是向有机分子中引入氰甲基的一种有效手段. 本文利用乙腈分子进攻2-二吡啶基酮获得了一例含有氰甲基官能团的配体dpkMeCN-H[dpkMeCN=cyanomethyl-di(pyridin-2-yl)methanol]. 在NiII和KI的配位作用下, 该体系发生原位组装生成了一对阻转异构体. 磁性拟合结果表明镍离子之间主要为铁磁耦合, 且它们由于八面体畸变产生一定的零场分裂, 使手性镍配合物产生磁各向异性.
中图分类号:
TrendMD:
阮泽宇, 杜杉楠, 黄国璋, 童明良, 刘俊良. 原位合成手性镍配合物中的阻转异构及磁性研究. 高等学校化学学报, 2021, 42(3): 709.
RUAN Zeyu, DU Shannan, HUANG Guozhang, TONG Mingliang, LIU Junliang. Atropisomerism and Magnetic Properties of an in situ Synthesized Chiral Nickel Complex. Chem. J. Chinese Universities, 2021, 42(3): 709.
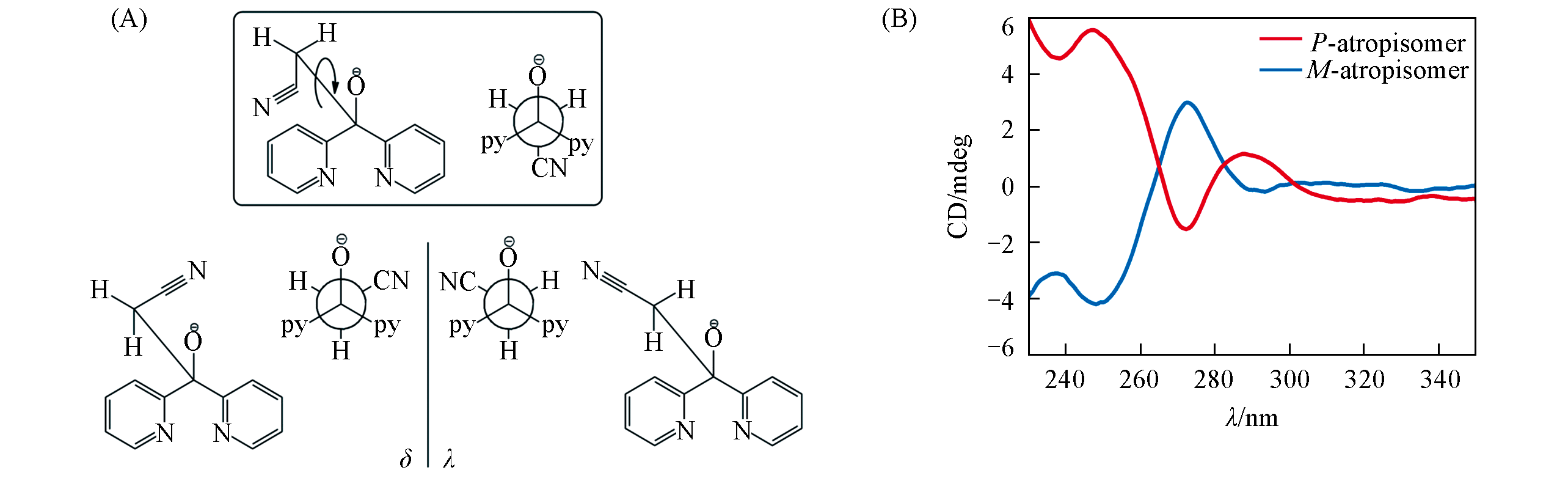
Fig.1 Conformational isomerism of dpkMeCN?H(A) and solid?state circular dichroism(CD) spectra of the powder sample for each [Ni2K] single crystal(B)(A) The conformer in the box is achiral, which is not the case in the [Ni2K] crystal structure in this work. Bottom left: the δ conformer; bottom right: the λ conformer.
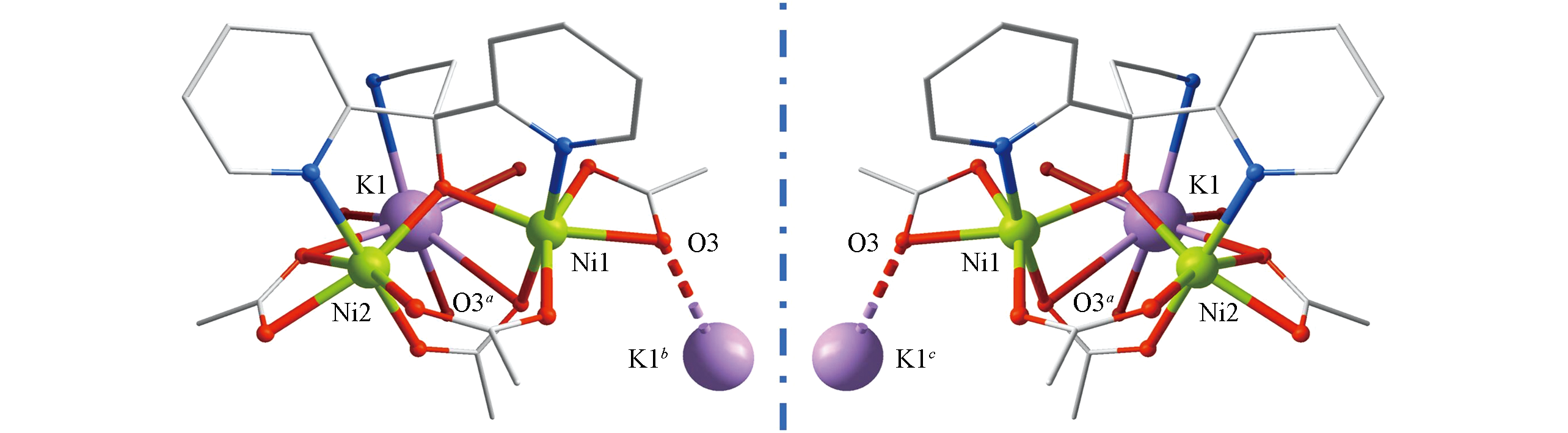
Fig.2 Molecular structure of [Ni2K]The solvent molecules and hydrogen atoms are omitted for clarity. Color code: purple(K), green(Ni), grey(C), red(O), light blue(N). Left: P-atropisomer; right: M-atropisomer. The red-purple dotted line indicates the presence of electrostatic interaction between the oxygen atom and the potassium atom from the adjacent {Ni2K} motif with short distance.Symmetry codes: a. ?1/2+x, 1/2?y, ?z; b. ?3/2+x, 1/2?y, ?z; c. 1/2+x, 1/2?y, ?z.
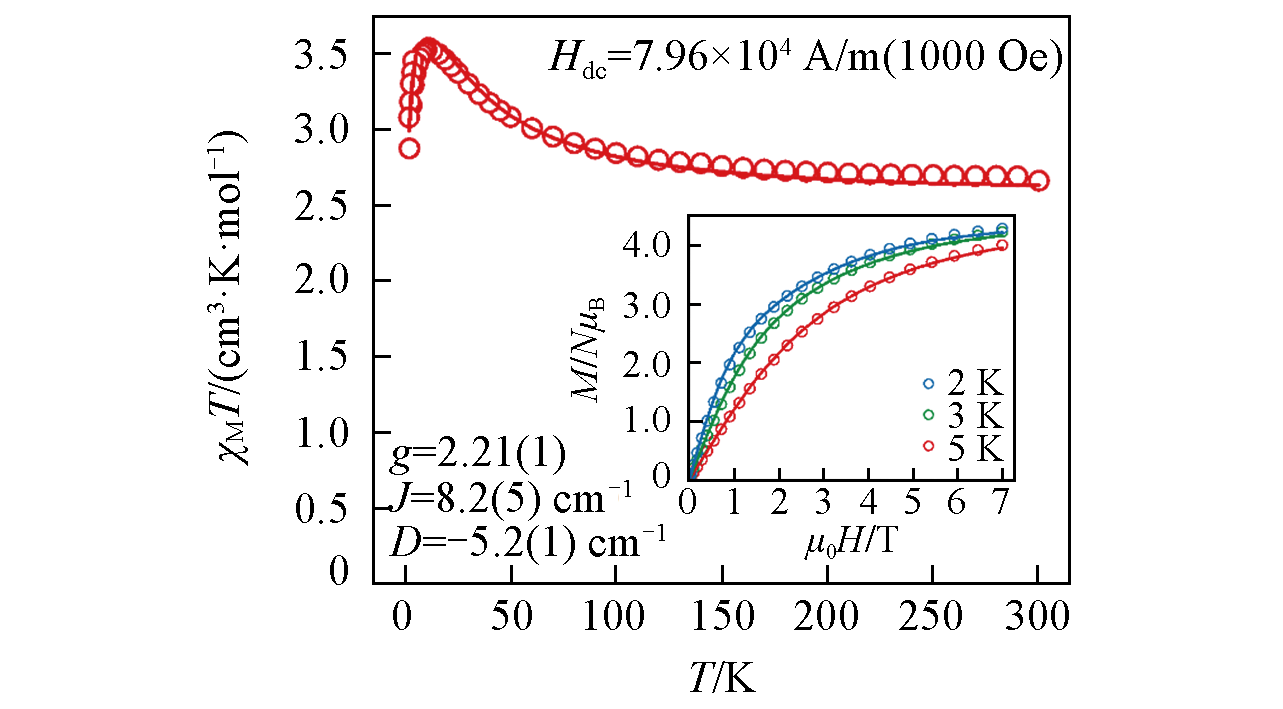
Fig.3 Temperature dependent direct?current(DC) molar magnetic susceptibilities of [Ni2K]Inset is the filed-dependent magnetization(M vs. H) at 2 K, 3 K and 5 K. The hollow circles refer to the experimental data while the solid lines correspond to the best fits from PHI.
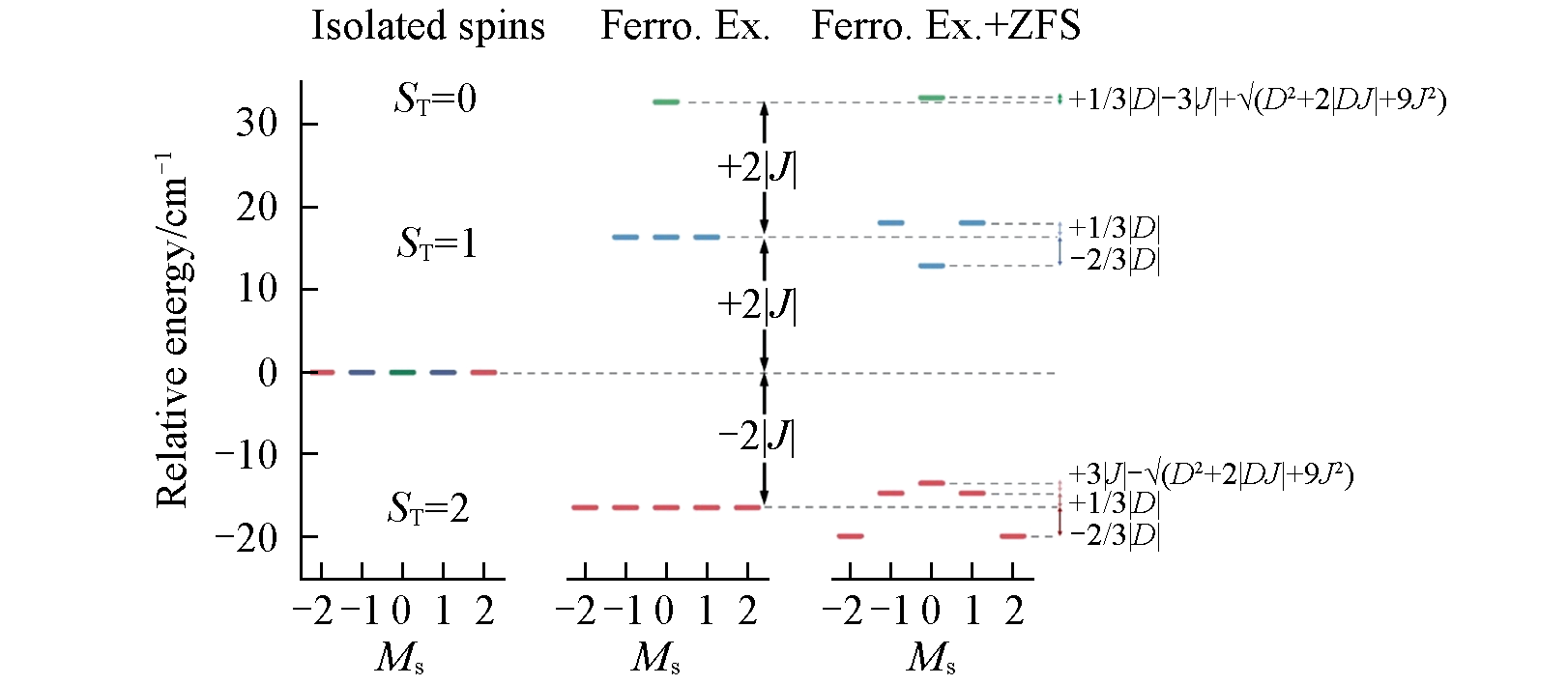
Fig.4 Energy level diagrams of [Ni2K]The relative energy in the figure refers to the eigenvalues obtained by the diagonalization of spin Hamiltonian. The figure shows the relative energy changes from pure spins(left) to only ferromagnetic exchange(middle), to ferromagnetic exchange and zero-field splitting(right).
| 1 | Guijarro A., Yus M., the Origin of Chirality in the Molecules of Life: A Revision from Awareness to the Current Theories and Perspectives of this Unsolved Problem, Royal Society of Chemistry, Cambridge, 2008, Chapter 3 |
| 2 | Ruffolo R. R., Spradlin T. A., Pollock G. D., Waddell J. E., Murphy P. J., J. Pharmacol. Exp. Ther.,1981, 219(2), 447 |
| 3 | Baker R., Rao V. B., J. Chem. Soc., Perkin Trans. 1,1982, 69—71 |
| 4 | Ashburn T. T., Thor K. B., Nat. Rev. Drug Discovery,2004, 3(8), 673—683 |
| 5 | Xing Q. Y., Pei W. W., Xu R. Q., Pei J., Basic Organic Chemistry, Peking University Press, Beijing, 2016, 106—109(邢其毅, 裴伟伟, 徐瑞秋, 裴坚. 基础有机化学, 北京: 北京大学出版社, 2016, 106—109) |
| 6 | Weng G. G., Zheng L. M., Sci. China Chem.,2020, 63(5), 619—636 |
| 7 | Han Z., Shi W., Cheng P., Chin. Chem. Lett.,2018, 29(6), 819—822 |
| 8 | Yan J., Su H., Yang H., Hu C., Malola S., Lin S., Teo B. K., Häkkinen H., Zheng N., J. Am. Chem. Soc.,2016, 138(39), 12751—12754 |
| 9 | Barton J. K., Science,1986, 233(4765), 727 |
| 10 | Aggeli A., Nyrkova I. A., Bell M., Harding R., Carrick L., McLeish T. C. B., Semenov A. N., Boden N., Proc. Natl. Acad. Sci. USA,2001, 98(21), 11857 |
| 11 | Huang J., Ding H. M., Xu Y., Zeng D., Zhu H., Zang D. M., Bao S. S., Ma Y. Q., Zheng L. M., Nat. Commun.,2017, 8(1), 2131 |
| 12 | Wu X., Han X., Xu Q., Liu Y., Yuan C., Yang S., Liu Y., Jiang J., Cui Y., J. Am. Chem. Soc.,2019, 141(17), 7081—7089 |
| 13 | Liu J. L., Liu W., Huang G. Z., Tong M. L., Sci. Bull.,2015, 60(4), 447—452 |
| 14 | Zhang B., Wang Z. M., Kurmoo M., Gao S., Inoue K., Kobayashi H., Adv. Funct. Mater.,2007, 17(4), 577—584 |
| 15 | Han X., Xia Q., Huang J., Liu Y., Tan C., Cui Y., J. Am. Chem. Soc.,2017, 139(25), 8693—8697 |
| 16 | Zhang S. Y., Yang C. X., Shi W., Yan X. P., Cheng P., Wojtas L., Zaworotko M. J., Chemistry,2017, 3(2), 281—289 |
| 17 | Lin G. Q., Li Y. M., Chan A. S. C., Principles and Applications of Asymmetric Synthesis, Wiley, New York, 2002 |
| 18 | Zhou Q. L., Xie J. H., Acta Chim. Sinica,2014, 72(7), 778—797(周其林, 谢建华. 化学学报, 2014, 72(7), 778—797) |
| 19 | Zhang Z. J., Tao Z. L., Arafate A., Gong L. Z., Acta Chim. Sinica,2017, 75(12), 1196—1201(张子競, 陶忠林, 阿拉法特·阿地力, 龚流柱. 化学学报, 2017, 75(12), 1196—1201) |
| 20 | Zhou Q., Lu P., Acta Chim. Sinica,2018, 76(11), 825—830(周锵, 陆平, 化学学报, 2018, 76(11), 825—830) |
| 21 | LaPlante S. R., Edwards P. J., Fader L. D., Jakalian A., Hucke O., ChemMedChem,2011, 6(3), 505—513 |
| 22 | Gustafson J. L., Lim D., Miller S. J., Science,2010, 328(5983), 1251 |
| 23 | Clayden J., Mitjans D., Youssef L. H., J. Am. Chem. Soc.,2002, 124(19), 5266—5267 |
| 24 | Yang H., Sun J., Gu W., Tang W., J. Am. Chem. Soc.,2020, 142(17), 8036—8043 |
| 25 | Jolliffe J. D., Armstrong R. J., Smith M. D., Nat. Chem.,2017, 9(6), 558—562 |
| 26 | Moss G. P., Pure Appl. Chem.,1996, 68(12), 2193—2222 |
| 27 | Leitner A., Shekhar S., Pouy M. J., Hartwig J. F., J. Am. Chem. Soc.,2005, 127(44), 15506—15514 |
| 28 | Van Veldhuizen J. J., Campbell J. E., Giudici R. E., Hoveyda A. H., J. Am. Chem. Soc.,2005, 127(18), 6877—6882 |
| 29 | Qu Z. R., Chinese J. Inorg. Chem.,2007, (12), 2126—2127(瞿志荣. 无机化学学报, 2007, (12), 2126—2127) |
| 30 | Powell K. J., Han L. C., Sharma P., Moses J. E., Org. Lett.,2014, 16(8), 2158—2161 |
| 31 | Henry F., Nitrile Oxides, Nitrones & Nitronates in Organic Synthesis: Novel Strategies in Synthesis, Wiley, Hoboken, 2008 |
| 32 | Fleming F. F., Wang Q., Chem. Rev.,2003, 103(5), 2035—2078 |
| 33 | Rappoport Z., The Chemistry of the Cyano Group, John Wiley & Sons Ltd., London, 1970 |
| 34 | Anbarasan P., Schareina T., Beller M., Chem. Soc. Rev.,2011, 40(10), 5049—5067 |
| 35 | López R., Palomo C., Angew. Chem. Int. Ed.,2015, 54(45), 13170—13184 |
| 36 | Liu Y., Yang K., Ge H., Chem. Sci.,2016, 7(4), 2804—2808 |
| 37 | Wu Q., Li Y., Wang C., Zhang J., Huang M., Kim J. K., Wu Y., Org. Chem. Front.,2018, 5(16), 2496—2500 |
| 38 | Liu Q. Y., Zhao X. H., Li J. L., Cao S., Acta Chim. Sinica,2018, 76(12), 945—950(刘青雲, 赵祥虎, 李佳录, 曹松. 化学学报, 2018, 76(12), 945—950) |
| 39 | Wu T., Mu X., Liu G., Angew. Chem. Int. Ed.,2011, 50(52), 12578—12581 |
| 40 | Rikken G. L. J. A., Raupach E., Nature,2000, 405(6789), 932—935 |
| 41 | Barron L. D., Nature,2000, 405(6789), 895—896 |
| 42 | Rikken G. L. J. A., Raupach E., Nature,1997, 390(6659), 493—494 |
| 43 | Zhang Y. J., Wu G., Xu H., Wang X., Long L. S., Kong X. J., Zheng L. S., Inorg. Chem.,2020, 59(1), 193—197 |
| 44 | Bogani L., Cavigli L., Bernot K., Sessoli R., Gurioli M., Gatteschi D., J. Mater. Chem.,2006, 16(26), 2587—2592 |
| 45 | Yuan C., Li X., Semin S., Feng Y., Rasing T., Xu J., Nano Lett.,2018, 18(9), 5411—5417 |
| 46 | Zhang W., Xiong R. G., Chem. Rev.,2012, 112(2), 1163—1195 |
| 47 | Xu G. C., Ma X. M., Zhang L., Wang Z. M., Gao S., J. Am. Chem. Soc.,2010, 132(28), 9588—9590 |
| 48 | Gatteschi D., Sessoli R., Villain J., Molecular Nanomagnets, Oxford University Press, New York, 2006 |
| 49 | Carlin R. L., Magnetochemistry, Springer, New York, 1986 |
| 50 | Harriman K. L. M., Errulat D., Murugesu M., Trends Chem.,2019, 1(4), 425—439 |
| 51 | Tong M. L., Monfort M., Juan J. M. C., Chen X. M., Bu X. H., Ohba M., Kitagawa S., Chem. Commun.,2005(2), 233—235 |
| 52 | Rigaku, PROCESS⁃AUTO, Rigaku Corporation, Tokyo, 1998 |
| 53 | Sheldrick M. G., Acta Crystallogr. Sect. A, 2008, 64, 112—122 |
| 54 | Sheldrick M. G., Acta Crystallogr. Sect. C,2015, 71, 3—8 |
| 55 | Dolomanov O. V., Bourhis L. J., Gildea R. J., Howard J. A. K., Puschmann H., J. Appl. Crystallogr.,2009, 42(2), 339—341 |
| 56 | Chen X. D., Du M., Mak T. C. W., Chem. Commun.,2005, 4417—4419 |
| 57 | Neese F., WIREs Computational Molecular Science,2012, 2(1), 73—78 |
| 58 | Neese F., WIREs Computational Molecular Science,2018, 8(1), e1327 |
| 59 | Davoren J. E., Bundesmann M. W., Yan Q. T., Collantes E. M., Mente S., Nason D. M., Gray D. L., ACS Med. Chem. Lett.,2012, 3(5), 433—435 |
| 60 | Clayden J., Chem. Commun.,2004, 127—135 |
| 61 | Liu J. L., Bao X., Leng J. D., Lin Z. J., Tong M. L., Cryst. Growth Des.,2011, 11(6), 2398—2403 |
| 62 | Clayden J., Angew. Chem. Int. Ed. Engl.,1997, 36(9), 949—951 |
| 63 | Paul B., Butterfoss G. L., Boswell M. G., Renfrew P. D., Yeung F. G., Shah N. H., Wolf C., Bonneau R., Kirshenbaum K., J. Am. Chem. Soc.,2011, 133(28), 10910—10919 |
| 64 | Kumarasamy E., Raghunathan R., Sibi M. P., Sivaguru J., Chem. Rev.,2015, 115(20), 11239—11300 |
| 65 | Ruiz⁃Martínez A., Casanova D., Alvarez S., Chem. Eur. J.,2008, 14(4), 1291—1303 |
| 66 | Macrae C. F., Edgington P. R., McCabe P., Pidcock E., Shields G. P., Taylor R., Towler M., van de Streek J., J. Appl. Crystallogr.,2006, 39(3), 453—457 |
| 67 | Vainshtein B. K., Fundamentals of Crystals: Symmetry, and Methods of Structural Crystallography, Springer, Berlin Heidelberg, 1996 |
| 68 | Orchard A. F., Magnetochemistry, 1st Edition, Oxford University Press, Oxford, 2003 |
| 69 | Kahn O., Molecular Magnetism, Vch Pub, New York, 1993 |
| 70 | Chilton N. F., Anderson R. P., Turner L. D., Soncini A., Murray K. S., J. Comput. Chem.,2013, 34(13), 1164—1175 |
| 71 | Zhang H., Coordination Chemistrty-Principles and Applications, Chemical Industry Press Co., Ltd., Beijing, 2009(章慧. 配位化学—原理与应用, 北京: 化学工业出版社, 2009) |
| 72 | Boča R., Coord. Chem. Rev.,2004, 248(9), 757—815 |
| 73 | Titiš J., Boča R., Inorg. Chem.,2010, 49(9), 3971—3973 |
| [1] | 黄秋红, 李文军, 李鑫. 有机催化靛红衍生酮亚胺与噁唑酮的不对称Mannich型加成反应[J]. 高等学校化学学报, 2022, 43(8): 20220131. |
| [2] | 郭彪, 赵晨灿, 刘芯辛, 于洲, 周丽景, 袁宏明, 赵震. 表面水热碳层对磁性NiFe2O4八面体光催化活性的影响[J]. 高等学校化学学报, 2022, 43(11): 20220472. |
| [3] | 秦该照, 唐明华, 赖亚琳, 袁黎明. 手性金属有机笼MOC-PA作为毛细管电泳手性固定相[J]. 高等学校化学学报, 2022, 43(11): 20220417. |
| [4] | 史歌, 徐茜, 代枭, 张洁, 沈军, 宛新华. 芳香取代基结构对螺旋聚乙炔高效液相色谱手性固定相手性识别性能的影响[J]. 高等学校化学学报, 2021, 42(8): 2673. |
| [5] | 王煦鑫, 朱华结. 手性化合物立体结构鉴定中的若干关键共性科学问题探讨[J]. 高等学校化学学报, 2021, 42(6): 1685. |
| [6] | 刘冬生. 超分子作用构筑具有高光学不对称性的表面等离子纳米粒子手性组装体[J]. 高等学校化学学报, 2021, 42(6): 1619. |
| [7] | 孟妍, 王秀凤, 张莉, 刘鸣华. 不同取代位置的萘衍生两亲分子在气/液界面组装膜中的螺旋结构与圆偏振发光[J]. 高等学校化学学报, 2021, 42(4): 1253. |
| [8] | 郑海娇, 姜丽艳, 贾琼. 精氨酸功能化磁性纳米材料的制备及在磷酸化肽富集中的应用[J]. 高等学校化学学报, 2021, 42(3): 717. |
| [9] | 龚怡然, 陈昌兵, 毛倩楠, 马栋良, 刘国权, 杨国坚, 张宇模. 基于电致酸/碱理论的手性光学切换体系[J]. 高等学校化学学报, 2021, 42(12): 3651. |
| [10] | 李菲, 李小轩, 李一峻, 何锡文, 陈朗星, 张玉奎. 表面定向磁性印迹聚合物的制备及对槲皮素的选择性识别[J]. 高等学校化学学报, 2021, 42(12): 3606. |
| [11] | 孙启睿, 赵楠, 刘树威, 辛华, 张皓, 张乐宁. 用于磁性靶向治疗肺纤维化的聚多巴胺包覆的Fe3O4/甲强龙/环磷酰胺复合超粒子[J]. 高等学校化学学报, 2021, 42(10): 3225. |
| [12] | 宋文尧, 周张浪, 杨鑫莉, 陈岚, 葛广路. 介孔二氧化硅对映选择性吸附的手性印迹调控[J]. 高等学校化学学报, 2021, 42(10): 3144. |
| [13] | 黄明耀, 朱守非. 催化不对称碳硼成键反应研究进展[J]. 高等学校化学学报, 2020, 41(7): 1426. |
| [14] | 张作然, 张利, 张志凌. 基于侧向磁泳的微流控芯片对两种磁性纳米球的同时分选[J]. 高等学校化学学报, 2020, 41(6): 1243. |
| [15] | 赵子一,郑洪芝,徐雁. 晶态纳米纤维素的多色圆偏振荧光性能[J]. 高等学校化学学报, 2020, 41(5): 1120. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||