

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (11): 2163.doi: 10.7503/cjcu20150491
收稿日期:2015-06-24
出版日期:2015-11-10
发布日期:2015-10-12
作者简介:联系人简介: 刘亚军, 男, 博士, 教授, 主要从事理论与计算光化学的研究. E-mail:基金资助:Received:2015-06-24
Online:2015-11-10
Published:2015-10-12
Contact:
LIU Yajun
E-mail:yajun.liu@bnu.edu.cn
Supported by:摘要:
基于CH2BrI光解离的最新实验报道, 采用多态完全活化空间二阶微扰理论和完全活化空间态相互作用-旋轨耦合作用(MS-CASPT2/CASSI-SO)方法, 计算了CH2BrI在乙腈、 2-丁醇和环己烷3种溶剂中, 在266 nm的光激发下的旋轨耦合态的势能曲线, 指认了实验观测到的解离产物I(2P3/2)和I*(2P1/2) 的具体解离途径. 为了理解不同溶剂对光产物重新结合成CH2Br—I和CH2I—Br的影响, 对CH2BrI在溶剂中光异构化过程的反应机理进行了探讨.
中图分类号:
TrendMD:
宋艳丽, 刘亚军. 旋轨耦合从头算方法研究CH2BrI光解离机理. 高等学校化学学报, 2015, 36(11): 2163.
SONG Yanli, LIU Yajun. Spin-orbit Coupling ab initio Investigation on the Photolysis Mechnism of CH2BrI†. Chem. J. Chinese Universities, 2015, 36(11): 2163.
| Solvent | CH2BrI | ||||||
|---|---|---|---|---|---|---|---|
| d(C—I)/nm | d(C—Br)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—Br/(°) | ∠I—C—Br—H/(°) | ||
| Gas phase[ | 0.2130 | 0.1935 | 0.1091 | 112.3 | 113.2 | 119.3 | |
| Acetonitrile | 0.2130 | 0.1936 | 0.1094 | 112.6 | 112.8 | 119.0 | |
| 2-Butanol | 0.2130 | 0.1937 | 0.1094 | 112.6 | 112.6 | 119.0 | |
| Cyclohexane | 0.2129 | 0.1935 | 0.1092 | 112.2 | 113.0 | 119.2 | |
| Solvent | CH2Br—I | ||||||
| d(C—Br)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠Br—C—H/(°) | ∠C—Br—I/(°) | ∠I—Br—C—H/(°) | |
| Gas phase[ | 0.1822 | 0.2924 | 0.1101 | 121.1 | 116.7 | 121.1 | 77.1 |
| Acetonitrile | 0.1803 | 0.2895 | 0.1090 | 122.5 | 117.0 | 121.8 | 79.6 |
| 2-Butanol | 0.1800 | 0.2898 | 0.1090 | 122.2 | 117.1 | 121.7 | 79.6 |
| Cyclohexane | 0.1807 | 0.2888 | 0.1090 | 121.4 | 117.0 | 122.0 | 78.1 |
| Solvent | CH2I—Br | ||||||
| d(C—I)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—H/(°) | ∠C—I—Br/(°) | ∠Br—I—C—H/(°) | |
| Gas phase[ | 0.2009 | 0.2753 | 0.1101 | 119.7 | 117.0 | 121.9 | 76.1 |
| Acetonitrile | 0.1972 | 0.2831 | 0.1092 | 120.9 | 118.2 | 119.6 | 80.9 |
| 2-Butanol | 0.1967 | 0.2793 | 0.1091 | 121.7 | 117.7 | 117.5 | 80.7 |
| Cyclohexane | 0.1972 | 0.2753 | 0.1091 | 120.0 | 117.5 | 119.6 | 77.5 |
Table 1 CASPT2 optimized geometries of the CH2BrI, CH2Br—I, and CH2I—Br in acetonitrile, 2-butanol and cyclohexane in comparison with the results in the gas phase
| Solvent | CH2BrI | ||||||
|---|---|---|---|---|---|---|---|
| d(C—I)/nm | d(C—Br)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—Br/(°) | ∠I—C—Br—H/(°) | ||
| Gas phase[ | 0.2130 | 0.1935 | 0.1091 | 112.3 | 113.2 | 119.3 | |
| Acetonitrile | 0.2130 | 0.1936 | 0.1094 | 112.6 | 112.8 | 119.0 | |
| 2-Butanol | 0.2130 | 0.1937 | 0.1094 | 112.6 | 112.6 | 119.0 | |
| Cyclohexane | 0.2129 | 0.1935 | 0.1092 | 112.2 | 113.0 | 119.2 | |
| Solvent | CH2Br—I | ||||||
| d(C—Br)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠Br—C—H/(°) | ∠C—Br—I/(°) | ∠I—Br—C—H/(°) | |
| Gas phase[ | 0.1822 | 0.2924 | 0.1101 | 121.1 | 116.7 | 121.1 | 77.1 |
| Acetonitrile | 0.1803 | 0.2895 | 0.1090 | 122.5 | 117.0 | 121.8 | 79.6 |
| 2-Butanol | 0.1800 | 0.2898 | 0.1090 | 122.2 | 117.1 | 121.7 | 79.6 |
| Cyclohexane | 0.1807 | 0.2888 | 0.1090 | 121.4 | 117.0 | 122.0 | 78.1 |
| Solvent | CH2I—Br | ||||||
| d(C—I)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—H/(°) | ∠C—I—Br/(°) | ∠Br—I—C—H/(°) | |
| Gas phase[ | 0.2009 | 0.2753 | 0.1101 | 119.7 | 117.0 | 121.9 | 76.1 |
| Acetonitrile | 0.1972 | 0.2831 | 0.1092 | 120.9 | 118.2 | 119.6 | 80.9 |
| 2-Butanol | 0.1967 | 0.2793 | 0.1091 | 121.7 | 117.7 | 117.5 | 80.7 |
| Cyclohexane | 0.1972 | 0.2753 | 0.1091 | 120.0 | 117.5 | 119.6 | 77.5 |
| Isomer | Symmetry | Description | Gas phase[ | Acetonitrile | 2-Butanol | Cyclohexane | Experiment |
|---|---|---|---|---|---|---|---|
| CH2BrI | A' | CH2 sym.str. | 3122 | 3040 | 3055 | 3076 | 2978[ |
| CH2 def. | 1424 | 1439 | 1412 | 1421 | 1374[ | ||
| CH2 wag | 1221 | 1187 | 1192 | 1196 | 1150[ | ||
| C—Br str. | 647 | 577 | 644 | 652 | 616[ | ||
| C—I str. | 550 | 418 | 537 | 538 | 517[ | ||
| C—I—Br bend | 143 | 133 | 144 | 145 | 144[ | ||
| A″ | CH2 asym.str. | 3182 | 3077 | 3076 | 3102 | 3053[ | |
| CH2 twist | 1075 | 1005 | 1076 | 1085 | 1065[ | ||
| CH2 rock | 787 | 750 | 782 | 789 | 754[ | ||
| CH2Br—I | A' | CH2 sym.str. | 3118 | 3107 | 3107 | 3115 | |
| CH2 def. | 1399 | 1384 | 1385 | 1397 | |||
| CH2 wag | 785 | 795 | 793 | 787 | 839[ | ||
| C—Br str. | 661 | 660 | 666 | 661 | 628[ | ||
| C—I str. | 144 | 152 | 150 | 146 | 159[ | ||
| C—I—Br bend | 116 | 116 | 97 | 113 | 111[ | ||
| A″ | CH2 asym.str. | 3230 | 3200 | 3196 | 3181 | ||
| CH2 twist | 949 | 946 | 947 | 945 | |||
| CH2 rock | 380 | 422 | 409 | 417 | |||
| CH2I—Br | A' | CH2 sym.str. | 3115 | 3092 | 3112 | 3106 | |
| CH2 def. | 1387 | 1369 | 1388 | 1367 | |||
| CH2 wag | 741 | 769 | 756 | 755 | 730[ | ||
| C—Br str. | 641 | 666 | 656 | 668 | |||
| C—I str. | 174 | 158 | 177 | 167 | 173[ | ||
| C—I—Br bend | 109 | 94 | 107 | 100 | 118[ | ||
| A″ | CH2 asym.str. | 3224 | 3190 | 3194 | 3214 | ||
| CH2 twist | 886 | 866 | 877 | 873 | |||
| CH2 rock | 432 | 439 | 451 | 447 |
Table 2 CASPT2 calculated harmonic vibrational frequencies(cm-1) of the CH2BrI, CH2Br—I and CH2I—Br in solvents and gas phase in comparison with the experimental results
| Isomer | Symmetry | Description | Gas phase[ | Acetonitrile | 2-Butanol | Cyclohexane | Experiment |
|---|---|---|---|---|---|---|---|
| CH2BrI | A' | CH2 sym.str. | 3122 | 3040 | 3055 | 3076 | 2978[ |
| CH2 def. | 1424 | 1439 | 1412 | 1421 | 1374[ | ||
| CH2 wag | 1221 | 1187 | 1192 | 1196 | 1150[ | ||
| C—Br str. | 647 | 577 | 644 | 652 | 616[ | ||
| C—I str. | 550 | 418 | 537 | 538 | 517[ | ||
| C—I—Br bend | 143 | 133 | 144 | 145 | 144[ | ||
| A″ | CH2 asym.str. | 3182 | 3077 | 3076 | 3102 | 3053[ | |
| CH2 twist | 1075 | 1005 | 1076 | 1085 | 1065[ | ||
| CH2 rock | 787 | 750 | 782 | 789 | 754[ | ||
| CH2Br—I | A' | CH2 sym.str. | 3118 | 3107 | 3107 | 3115 | |
| CH2 def. | 1399 | 1384 | 1385 | 1397 | |||
| CH2 wag | 785 | 795 | 793 | 787 | 839[ | ||
| C—Br str. | 661 | 660 | 666 | 661 | 628[ | ||
| C—I str. | 144 | 152 | 150 | 146 | 159[ | ||
| C—I—Br bend | 116 | 116 | 97 | 113 | 111[ | ||
| A″ | CH2 asym.str. | 3230 | 3200 | 3196 | 3181 | ||
| CH2 twist | 949 | 946 | 947 | 945 | |||
| CH2 rock | 380 | 422 | 409 | 417 | |||
| CH2I—Br | A' | CH2 sym.str. | 3115 | 3092 | 3112 | 3106 | |
| CH2 def. | 1387 | 1369 | 1388 | 1367 | |||
| CH2 wag | 741 | 769 | 756 | 755 | 730[ | ||
| C—Br str. | 641 | 666 | 656 | 668 | |||
| C—I str. | 174 | 158 | 177 | 167 | 173[ | ||
| C—I—Br bend | 109 | 94 | 107 | 100 | 118[ | ||
| A″ | CH2 asym.str. | 3224 | 3190 | 3194 | 3214 | ||
| CH2 twist | 886 | 866 | 877 | 873 | |||
| CH2 rock | 432 | 439 | 451 | 447 |
| Solvent | CH2BrI | ||||||
|---|---|---|---|---|---|---|---|
| d(C—I)/nm | d(C—Br)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—Br/(°) | ∠I—C—Br—H/(°) | ||
| Gas phase[ | 0.4074 | 0.1865 | 0.1085 | 122.9 | 89.3 | 79.1 | |
| Acetonitrile | 0.4051 | 0.1872 | 0.1088 | 123.7 | 92.5 | 79.2 | |
| 2-Butanol | 0.4041 | 0.1872 | 0.1088 | 123.7 | 95.5 | 79.2 | |
| Cyclohexane | 0.4104 | 0.1867 | 0.1086 | 123.3 | 88.4 | 79.6 | |
| Solvent | CH2Br—I | ||||||
| d(C—Br)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠Br—C—H/(°) | ∠C—Br—I/(°) | ∠I—Br—C—H/(°) | |
| Gas phase[ | 0.1864 | 0.3929 | 0.1085 | 123.5 | 117.0 | 179.2 | 98.5 |
| Acetonitrile | 0.1868 | 0.3949 | 0.1087 | 125.4 | 117.3 | 180.0 | 90.6 |
| 2-Butanol | 0.1869 | 0.3941 | 0.1087 | 125.4 | 117.3 | 180.0 | 90.7 |
| Cyclohexane | 0.1864 | 0.3928 | 0.1085 | 124.8 | 117.5 | 179.6 | 92.7 |
| Solvent | CH2I—Br | ||||||
| d(C—I)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—H/(°) | ∠C—I—Br/(°) | ∠Br—I—C—H/(°) | |
| Gas phase[ | 0.2044 | 0.3899 | 0.1086 | 123.1 | 118.4 | 179.2 | 90.0 |
| Acetonitrile | 0.2051 | 0.3937 | 0.1088 | 124.1 | 117.9 | 180.0 | 90.6 |
| 2-Butanol | 0.2051 | 0.3926 | 0.1088 | 124.0 | 118.0 | 179.7 | 89.2 |
| Cyclohexane | 0.2048 | 0.3920 | 0.1087 | 123.6 | 118.2 | 180.0 | 89.7 |
Table 3 CASPT2 optimized geometries of the 11A″ excited state of CH2BrI, CH2Br—I and CH2I—Br in acetonitrile, 2-butanol and cyclohexane in comparison with gas-phase results
| Solvent | CH2BrI | ||||||
|---|---|---|---|---|---|---|---|
| d(C—I)/nm | d(C—Br)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—Br/(°) | ∠I—C—Br—H/(°) | ||
| Gas phase[ | 0.4074 | 0.1865 | 0.1085 | 122.9 | 89.3 | 79.1 | |
| Acetonitrile | 0.4051 | 0.1872 | 0.1088 | 123.7 | 92.5 | 79.2 | |
| 2-Butanol | 0.4041 | 0.1872 | 0.1088 | 123.7 | 95.5 | 79.2 | |
| Cyclohexane | 0.4104 | 0.1867 | 0.1086 | 123.3 | 88.4 | 79.6 | |
| Solvent | CH2Br—I | ||||||
| d(C—Br)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠Br—C—H/(°) | ∠C—Br—I/(°) | ∠I—Br—C—H/(°) | |
| Gas phase[ | 0.1864 | 0.3929 | 0.1085 | 123.5 | 117.0 | 179.2 | 98.5 |
| Acetonitrile | 0.1868 | 0.3949 | 0.1087 | 125.4 | 117.3 | 180.0 | 90.6 |
| 2-Butanol | 0.1869 | 0.3941 | 0.1087 | 125.4 | 117.3 | 180.0 | 90.7 |
| Cyclohexane | 0.1864 | 0.3928 | 0.1085 | 124.8 | 117.5 | 179.6 | 92.7 |
| Solvent | CH2I—Br | ||||||
| d(C—I)/nm | d(Br—I)/nm | d(C—H)/nm | ∠H—C—H/(°) | ∠I—C—H/(°) | ∠C—I—Br/(°) | ∠Br—I—C—H/(°) | |
| Gas phase[ | 0.2044 | 0.3899 | 0.1086 | 123.1 | 118.4 | 179.2 | 90.0 |
| Acetonitrile | 0.2051 | 0.3937 | 0.1088 | 124.1 | 117.9 | 180.0 | 90.6 |
| 2-Butanol | 0.2051 | 0.3926 | 0.1088 | 124.0 | 118.0 | 179.7 | 89.2 |
| Cyclohexane | 0.2048 | 0.3920 | 0.1087 | 123.6 | 118.2 | 180.0 | 89.7 |
| Isoner | State | Acetonitrile | 2-Butanol | ||
|---|---|---|---|---|---|
| Ev/cm-1(eV) | f | Ev/cm-1(eV) | f | ||
| CH2BrI | X11A' | 0(0) | 0(0) | ||
| 11A″ | 38213(4.74) | 6.78×10-3 | 38236(4.74) | 6.77×10-3 | |
| 21A' | 38615(4.79) | 1.24×10-2 | 38582(4.78) | 1.26×10-2 | |
| 31A' | 48043(5.96) | 2.22×10-2 | 47974(5.95) | 2.13×10-2 | |
| 21A″ | 48229(5.98) | 1.48×10-3 | 48179(5.97) | 1.79×10-3 | |
| CH2Br—I | X11A' | 0(0) | 0(0) | ||
| 11A″ | 15591(1.93) | 4.04×10-5 | 15412(1.91) | 3.94×10-5 | |
| 21A' | 16956(2.10) | 4.21×10-4 | 16753(2.08) | 4.02×10-4 | |
| 31A' | 26368(3.27) | 6.50×10-1 | 26427(3.28) | 6.50×10-1 | |
| 21A″ | 36305(4.50) | 3.59×10-7 | 36416(4.52) | 2.84×10-7 | |
| CH2I—Br | X11A' | 0(0) | 0(0) | ||
| 11A″ | 20205(2.51) | 1.75×10-6 | 21188(2.63) | 1.93×10-6 | |
| 21A' | 22896(2.84) | 7.50×10-4 | 24144(2.99) | 9.38×10-4 | |
| 31A' | 29302(3.63) | 5.80×10-1 | 30567(3.79) | 6.00×10-1 | |
| 21A″ | 32852(4.07) | 5.55×10-4 | 34168(4.24) | 5.69×10-4 | |
Table 4 MS-CASPT2 calculated vertical excitation energies(Ev) and oscillator strength(f) of the five lowest-lying spin-orbital-free states of CH2BrI, CH2Br—I and CH2I—Br in acetonitrile and 2-butanol
| Isoner | State | Acetonitrile | 2-Butanol | ||
|---|---|---|---|---|---|
| Ev/cm-1(eV) | f | Ev/cm-1(eV) | f | ||
| CH2BrI | X11A' | 0(0) | 0(0) | ||
| 11A″ | 38213(4.74) | 6.78×10-3 | 38236(4.74) | 6.77×10-3 | |
| 21A' | 38615(4.79) | 1.24×10-2 | 38582(4.78) | 1.26×10-2 | |
| 31A' | 48043(5.96) | 2.22×10-2 | 47974(5.95) | 2.13×10-2 | |
| 21A″ | 48229(5.98) | 1.48×10-3 | 48179(5.97) | 1.79×10-3 | |
| CH2Br—I | X11A' | 0(0) | 0(0) | ||
| 11A″ | 15591(1.93) | 4.04×10-5 | 15412(1.91) | 3.94×10-5 | |
| 21A' | 16956(2.10) | 4.21×10-4 | 16753(2.08) | 4.02×10-4 | |
| 31A' | 26368(3.27) | 6.50×10-1 | 26427(3.28) | 6.50×10-1 | |
| 21A″ | 36305(4.50) | 3.59×10-7 | 36416(4.52) | 2.84×10-7 | |
| CH2I—Br | X11A' | 0(0) | 0(0) | ||
| 11A″ | 20205(2.51) | 1.75×10-6 | 21188(2.63) | 1.93×10-6 | |
| 21A' | 22896(2.84) | 7.50×10-4 | 24144(2.99) | 9.38×10-4 | |
| 31A' | 29302(3.63) | 5.80×10-1 | 30567(3.79) | 6.00×10-1 | |
| 21A″ | 32852(4.07) | 5.55×10-4 | 34168(4.24) | 5.69×10-4 | |
| Isoner | State | Cyclohexane | Gas phase[ | ||
|---|---|---|---|---|---|
| Ev/cm-1(eV) | f | Ev/cm-1(eV) | f | ||
| CH2BrI | X11A' | 0(0) | 0(0) | ||
| 11A″ | 37879(4.70) | 6.81×10-3 | 38283(4.75) | 8.10×10-3 | |
| 21A' | 38163(4.73) | 1.23×10-2 | 38570(4.78) | 6.00×10-3 | |
| 31A' | 47584(5.90) | 2.14×10-2 | 46622(5.78) | 1.40×10-3 | |
| 21A″ | 47873(5.94) | 1.60×10-3 | 47184(5.85) | 1.80×10-2 | |
| CH2Br—I | X11A' | 0(0) | 0(0) | ||
| 11A″ | 13031(1.62) | 3.26×10-5 | 13229(1.64) | 3.70×10-5 | |
| 21A' | 14265(1.77) | 1.54×10-4 | 14363(1.78) | 1.70×10-4 | |
| 31A' | 28438(3.53) | 5.70×10-1 | 28771(3.57) | 5.60×10-1 | |
| 21A″ | 38295(4.75) | 2.96×10-6 | 38146(4.73) | 1.10×10-6 | |
| CH2I—Br | X11A' | 0(0) | 0(0) | ||
| 11A″ | 20249(2.51) | 3.85×10-6 | 19619(2.43) | 4.20×10-7 | |
| 21A' | 22757(2.82) | 6.92×10-4 | 22269(2.76) | 6.20×10-4 | |
| 31A' | 31533(3.91) | 5.50×10-1 | 31630(3.92) | 4.80×10-1 | |
| 21A″ | 35127(4.36) | 8.56×10-4 | 35054(4.35) | 9.30×10-4 | |
Table 5 MS-CASPT2 calculated vertical excitation energies(Ev) and oscillator strength(f) of the five lowest-lying spin-orbital-free states of CH2BrI, CH2Br—I and CH2I—Br in cyclohexane and gas phase
| Isoner | State | Cyclohexane | Gas phase[ | ||
|---|---|---|---|---|---|
| Ev/cm-1(eV) | f | Ev/cm-1(eV) | f | ||
| CH2BrI | X11A' | 0(0) | 0(0) | ||
| 11A″ | 37879(4.70) | 6.81×10-3 | 38283(4.75) | 8.10×10-3 | |
| 21A' | 38163(4.73) | 1.23×10-2 | 38570(4.78) | 6.00×10-3 | |
| 31A' | 47584(5.90) | 2.14×10-2 | 46622(5.78) | 1.40×10-3 | |
| 21A″ | 47873(5.94) | 1.60×10-3 | 47184(5.85) | 1.80×10-2 | |
| CH2Br—I | X11A' | 0(0) | 0(0) | ||
| 11A″ | 13031(1.62) | 3.26×10-5 | 13229(1.64) | 3.70×10-5 | |
| 21A' | 14265(1.77) | 1.54×10-4 | 14363(1.78) | 1.70×10-4 | |
| 31A' | 28438(3.53) | 5.70×10-1 | 28771(3.57) | 5.60×10-1 | |
| 21A″ | 38295(4.75) | 2.96×10-6 | 38146(4.73) | 1.10×10-6 | |
| CH2I—Br | X11A' | 0(0) | 0(0) | ||
| 11A″ | 20249(2.51) | 3.85×10-6 | 19619(2.43) | 4.20×10-7 | |
| 21A' | 22757(2.82) | 6.92×10-4 | 22269(2.76) | 6.20×10-4 | |
| 31A' | 31533(3.91) | 5.50×10-1 | 31630(3.92) | 4.80×10-1 | |
| 21A″ | 35127(4.36) | 8.56×10-4 | 35054(4.35) | 9.30×10-4 | |
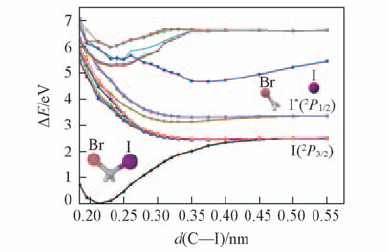
Fig.2 MS-CASPT2//CASPT2 calculated potential energy curevs of the spin-orbit-coupledstates with respect to the C—I bond coordinate of CH2BrI in cyclohexane
| [1] | Carpenter L. J., Wevill D. J., Palmer C. J., Michels J., Mar. Chem., 2007, 103(3/4), 227—236 |
| [2] | Class T. H., Ballschmiter K., J. Atmos. Chem., 1988, 6(1/2), 35—46 |
| [3] | Blomstro D., Herbig K., Simmons H. E., J. Org. Chem., 1965, 30(4), 959—964 |
| [4] | Pienta N. J., Kropp P. J., J. Am. Chem. Soc., 1978, 100(2), 655—656 |
| [5] | Kropp P. J., Pienta N. J., Sawyer J. A., Polniaszek R. P., Tetrahedron, 1981, 37(19), 3229—3236 |
| [6] | Kropp P. J., Acc. Chem. Res., 1984, 17(4), 131—137 |
| [7] | Phillips D. L., Fang W. H., Zheng X. M., J. Am. Chem. Soc., 2001, 123(18), 4197—4203 |
| [8] | Liu Y. J., Xiao H. Y., Sun M. T., Fang W. H., J. Comput. Chem., 2008, 29(15), 2513—2519 |
| [9] | Fang W. H., J. Am. Chem. Soc., 1999, 121(36), 8376—8384 |
| [10] | He H. Y., Fang W. H., J. Am. Chem. Soc., 2003, 125(51), 16139—16147 |
| [11] | Butler L. J., Hintsa E. J., Shane S. F., Lee Y. T., J. Chem. Phys., 1987, 86(4), 2051—2074 |
| [12] | Butler L. J., Hintsa E. J., Lee Y. T., J. Chem. Phys., 1986, 84(7), 4104—4106 |
| [13] | Lee S. J., Bersohn R., J. Phys. Chem., 1982, 86(5), 728—730 |
| [14] | Liu Y. J., Ajitha D., Krogh J. W., Tarnovsky A. N., Lindh R., Chemphyschem, 2006, 7(4), 955—963 |
| [15] | Tarnovsky A. N., Wall M., Gustafsson M., Lascoux N., Sundstrom V., Akesson E., J. Phys. Chem. A, 2002, 106(25), 5999—6005 |
| [16] | Zheng X. M., Phillips D. L., J. Chem. Phys., 2000, 113(8), 3194—3203 |
| [17] | Kwok W. M., Ma C. S., Phillips D., Parker A. W., Towrie M., Matousek P., Phillips D. L., Chem. Phys. Lett., 2001, 341(3/4), 292—298 |
| [18] | Anderson C. P., Spears K. G., Wilson K. R., Sension R. J., J. Chem. Phys., 2013, 139(19), 194307 |
| [19] | Podsiadlo M., Katrusiak A., Crystengcomm, 2008, 10(10), 1436—1442 |
| [20] | Tang K. C., Peng J., Spears K. G., Sension R. J., J. Chem. Phys., 2010, 132(14), 141102 |
| [21] | Andersson K., Malmqvist P. Å., Roos B. O., J. Chem. Phys., 1992, 96(2), 1218—1226 |
| [22] | Andersson K., Theor. Chim. Acta, 1995, 91(1/2), 31—46 |
| [23] | Malmqvist P. Å., Roos B. O., Chem. Phys. Lett., 1989, 155(2), 189—194 |
| [24] | Roos B.O., Andersson K., Fülscher M. P., Malmqvist P. Å., Serrano Andrés L., Pierloot K., Merchán M., Advances in Chemical Physics: New Methods in Computational Quantum Mechanics, John Wiley & Sons, New York, 1996, 219—331 |
| [25] | Finley J., Malmqvist P. Å., Roos B. O., Serrano-Andrés L., Chem. Phys. Lett., 1998, 288(2—4), 299—306 |
| [26] | Malmqvist P. Å., Roos B. O., Schimmelpfennig B., Chem. Phys. Lett., 2002, 357(3/4), 230—240 |
| [27] | Roos B. O., Malmqvist P. Å., Phys. Chem. Chem. Phys., 2004, 6(11), 2919—2927 |
| [28] | Marian C. M., Wahlgren U., Chem. Phys. Lett., 1996, 251(5/6), 357—364 |
| [29] | Ghigo G., Roos B. O., Malmqvist P. Å., Chem. Phys. Lett., 2004, 396(1—3), 142—149 |
| [30] | Barone V., Cossi M., J. Phys. Chem. A, 1998, 102(11), 1995—2001 |
| [31] | Tarnovsky A. N., Pascher I., Pascher T., J. Phys. Chem. A, 2007, 111(46), 11814—11817 |
| [32] | Man S. Q., Kwok W. M., Phillips D. L., Johnson A. E., J. Chem. Phys., 1996, 105(14), 5842—5857 |
| [33] | Widmark P. O., Persson B. J., Roos B. O., Theor. Chim. Acta, 1991, 79(6), 419—432 |
| [34] | Widmark P. O., Malmqvist P. A., Roos B. O., Theor. Chim. Acta, 1990, 77(5), 291—306 |
| [35] | Roos B. O., Lindh R., Malmqvist P. Å., Veryazov V., Widmark P. O., J. Phys. Chem. A, 2004, 108(15), 2851—2858 |
| [36] | Karlstrom G., Lindh R., Malmqvist P. Å., Roos B. O., Ryde U., Veryazov V., Widmark P. O., Cossi M., Schimmelpfennig B., Neogrady P., Seijo L., Comput. Mater. Sci., 2003, 28(2), 222—239 |
| [37] | Veryazov V., Widmark P. O., Serrano-Andrés L., Lindh R., Roos B. O., Int. J. Quantum Chem., 2004, 100(4), 626—635 |
| [38] | Fang W. H., Acc. Chem. Res., 2008, 41(3), 452—457 |
| [39] | Fang W. H., J. Am. Chem. Soc., 1998, 120(30), 7568—7576 |
| [40] | El-Sabban M. Z., J. Chem. Phys., 1966, 44(5), 1770—1779 |
| [41] | Liu Y. J., De Vico L., Lindh R., Fang W. H., Chemphyschem, 2007, 8(6), 890—898 |
| [42] | Donovan R.J.,J. Photochem., 1972, 75—78 |
| [1] | 张伶育, 张继龙, 曲泽星. RDX分子内振动能量重分配的动力学研究[J]. 高等学校化学学报, 2022, 43(10): 20220393. |
| [2] | 周敏, 石莹莹, 李树奇, 张凯林, 崔永亮, 张森, 张先燚, 孔祥蕾. 不同电荷态泛素蛋白离子的193 nm紫外光解离质谱[J]. 高等学校化学学报, 2021, 42(8): 2436. |
| [3] | 王建, 张红星. 四配位铂磷光发射体结构与光物理性质关系的理论研究[J]. 高等学校化学学报, 2021, 42(7): 2245. |
| [4] | 徐琰东,尤静林,王建,龚晓晔,丁雅妮,曹培明,郑少波,吴永全,余仲达. Bi4B2O9晶体及其熔体结构的高温原位拉曼光谱研究[J]. 高等学校化学学报, 2019, 40(10): 2143. |
| [5] | 吴志东, 尤静林, 王建, 王敏, 何莹霞, 杨冶金. 基于精细结构对二元钠硅酸盐玻璃核磁共振波谱的研究[J]. 高等学校化学学报, 2019, 40(1): 108. |
| [6] | 杨梦, 张腾烁, 郑旭明, 薛佳丹. 2-硝基萘激发态衰减动力学和光解通道的从头计算研究[J]. 高等学校化学学报, 2018, 39(8): 1734. |
| [7] | 方升, 刘静静, 段雪梅, 陶福明, 刘靖尧. 大气中一元酸催化亚硫酸分解反应的从头算及动力学研究[J]. 高等学校化学学报, 2017, 38(8): 1390. |
| [8] | 陈德利, 杨鹏勇, 武胜男, 何思慧, 王芳芳. 从头算分子动力学模拟Pd团簇负载UiO-66材料结构及稳定性[J]. 高等学校化学学报, 2017, 38(7): 1210. |
| [9] | 于泳博, 刘翠, 宫利东. 从头算和ABEEMσπ/MM对(CH3OH)n(n=3~12)和[Na(CH3OH)n]+(n=3~6)体系的研究[J]. 高等学校化学学报, 2016, 37(8): 1468. |
| [10] | 仓玉萍, 陈东, 杨帆, 杨慧明. 氮化锗多形体的四方、 单斜和正交畸变的理论研究[J]. 高等学校化学学报, 2016, 37(4): 674. |
| [11] | 姜海洋, 冯伟, 孙艳伟, 齐乔芳, 田宏伟, 刘慧玲, 黄旭日. 9-氨基奎宁催化剂催化1-溴代硝基甲烷和亚苄基丙酮的共轭加成反应的理论研究[J]. 高等学校化学学报, 2014, 35(7): 1500. |
| [12] | 孟素慈, 殷秀莲, 马晶, 谢吉民. 有机π共轭配体溶剂化效应与分子间相互作用的理论研究[J]. 高等学校化学学报, 2012, 33(11): 2492. |
| [13] | 仇毅翔, 王曙光. 膦配体对金团簇[Au@Au8(PR3)8]3+(R=Me,OMe,H,F,Cl,CN)稳定化作用的理论研究[J]. 高等学校化学学报, 2012, 33(11): 2549. |
| [14] | 胡彬, 欧阳密, 张玉建, 项文勤, 张诚. 具有双极性端枝的3D结构化合物的合成及光学性能[J]. 高等学校化学学报, 2012, 33(01): 82. |
| [15] | 慈成刚, 段雪梅, 刘靖尧, 孙家钟. 乙醇醛光解离机理的理论研究[J]. 高等学校化学学报, 2011, 32(7): 1588. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
