

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (11): 2477.doi: 10.7503/cjcu20180530
收稿日期:2018-07-27
出版日期:2018-11-10
发布日期:2018-09-29
作者简介:联系人简介: 王宝山, 男, 博士, 教授, 博士生导师, 主要从事量子化学与分子设计研究. E-mail: 基金资助:
HOU Hua1, YU Xiaojuan1, ZHOU Wenjun2, LUO Yunbai1, WANG Baoshan1,*( )
)
Received:2018-07-27
Online:2018-11-10
Published:2018-09-29
Contact:
WANG Baoshan
E-mail:baoshan@whu.edu.cn
Supported by:摘要:
提出了一种全新的构效关系模型, 基于中性分子的静电势参数, 包括总表面积、 正负静电势的分离度、 局域极性、 分子密度及约化正静电势面积等具有明确物理意义的描述符, 直接获得各种类型气体的介电强度. 研究结果表明, 理论值与实验值的平均绝对偏差仅为0.06, 且相关系数达到0.993. 基于构效关系模型, 分析了气体的宏观介电强度受微观电子结构影响的规律, 发现了提高气体介电强度的分子设计思路.
中图分类号:
TrendMD:
侯华, 余小娟, 周文俊, 罗运柏, 王宝山. 绝缘气体介电强度的构效关系. 高等学校化学学报, 2018, 39(11): 2477.
HOU Hua, YU Xiaojuan, ZHOU Wenjun, LUO Yunbai, WANG Baoshan. Theoretical Investigations on the Structure-activity Relationship to the Dielectric Strength of the Insulation Gases†. Chem. J. Chinese Universities, 2018, 39(11): 2477.
| Species | CAS registry | As/nm2 | ν | Π/eV | ρ/(g·cm-3) | Er,cal | ||
|---|---|---|---|---|---|---|---|---|
| H2 | 1333-74-0 | 0.336 | 6.635 | 0.131 | 0.180 | 0.208 | 0.16 | 0.22 |
| O2 | 7782-44-7 | 0.506 | 1.400 | 0.091 | 1.624 | 0.296 | 0.34 | 0.33 |
| N2 | 7727-37-9 | 0.542 | 44.973 | 0.173 | 1.265 | 0.350 | 0.35 | 0.38 |
| N2O | 10024-97-2 | 0.666 | 182.831 | 0.430 | 1.541 | 0.342 | 0.40 | 0.46 |
| CO | 630-08-0 | 0.545 | 94.462 | 0.261 | 1.254 | 0.317 | 0.34 | 0.40 |
| CO2 | 124-38-9 | 0.647 | 207.724 | 0.532 | 1.591 | 0.301 | 0.27 | 0.35 |
| OCS | 463-58-1 | 0.819 | 215.830 | 0.277 | 1.546 | 0.518 | 0.93 | 0.90 |
| SF6 | 2551-62-4 | 1.034 | 0.140 | 0.087 | 2.777 | 0.733 | 0.99 | 1.00 |
| CH4 | 74-82-8 | 0.602 | 9.208 | 0.126 | 0.624 | 0.346 | 0.37 | 0.43 |
| CH3Cl | 74-87-3 | 0.816 | 192.564 | 0.558 | 1.299 | 0.410 | 0.33 | 0.32 |
| CH3Br | 74-83-9 | 0.882 | 196.293 | 0.525 | 2.181 | 0.429 | 0.49 | 0.45 |
| CH2F2 | 75-10-5 | 0.712 | 313.110 | 0.701 | 1.676 | 0.320 | 0.33 | 0.27 |
| CH2Cl2 | 75-09-2 | 1.014 | 194.823 | 0.481 | 1.634 | 0.402 | 0.69 | 0.68 |
| CHF2Cl | 75-45-6 | 0.914 | 152.353 | 0.450 | 1.945 | 0.345 | 0.51 | 0.42 |
| CHFCl2 | 75-43-4 | 1.057 | 146.752 | 0.382 | 1.881 | 0.424 | 0.78 | 0.92 |
| CF4 | 75-73-0 | 0.811 | 4.219 | 0.175 | 2.388 | 0.382 | 0.49 | 0.42 |
| CF3Cl | 75-72-9 | 0.957 | 10.906 | 0.172 | 2.208 | 0.550 | 0.68 | 0.58 |
| CF2Cl2 | 75-71-8 | 1.095 | 38.548 | 0.176 | 2.108 | 0.576 | 0.91 | 0.99 |
| CF3Br | 75-63-8 | 1.023 | 20.324 | 0.187 | 2.864 | 0.517 | 0.78 | 0.75 |
| CH3CF3 | 420-46-2 | 0.977 | 170.332 | 0.564 | 1.745 | 0.425 | 0.43 | 0.41 |
| CH3CHCl2 | 75-34-3 | 1.204 | 214.009 | 0.461 | 1.497 | 0.544 | 1.01 | 1.01 |
| C2F6 | 76-16-4 | 1.118 | 3.519 | 0.144 | 2.427 | 0.638 | 0.92 | 0.80 |
| CF3CF2Cl | 76-15-3 | 1.241 | 9.646 | 0.158 | 2.314 | 0.633 | 1.05 | 1.04 |
| F2C=CFCl | 79-38-9 | 1.103 | 76.028 | 0.251 | 2.089 | 0.557 | 0.89 | 0.72 |
| CF3CH=CH2 | 677-21-4 | 1.114 | 223.217 | 0.494 | 1.674 | 0.501 | 0.87 | 0.80 |
| CF3CF=CF2 | 116-15-4 | 1.261 | 55.424 | 0.296 | 2.291 | 0.609 | 0.95 | 0.94 |
| CF2=CF—CF=CF2 | 685-63-2 | 1.401 | 89.157 | 0.315 | 2.184 | 0.586 | 1.18 | 1.20 |
| c-C6F10 | 355-75-9 | 1.786 | 14.075 | 0.220 | 2.428 | 0.892 | 1.80 | 1.90 |
| c-C4F8 | 115-25-3 | 1.472 | 5.602 | 0.241 | 2.437 | 0.605 | 1.18 | 1.25 |
| c-C6F12 | 355-68-0 | 1.856 | 0.910 | 0.107 | 2.553 | 1.304 | 2.33 | 2.35 |
| c-CF3(C4F6)CF3 | 1583-97-7 | 1.935 | 7.545 | 0.176 | 2.512 | 1.063 | 2.19 | 2.30 |
| CF3OCF3 | 1479-49-8 | 1.214 | 8.963 | 0.163 | 2.442 | 0.601 | 1.01 | 1.00 |
| c-CF3(C2F2O)CF3 | 117642-58-7 | 1.590 | 33.769 | 0.221 | 2.406 | 0.793 | 1.53 | 1.60 |
| HC≡CH | 74-86-2 | 0.672 | 304.742 | 0.534 | 0.858 | 0.306 | 0.57 | 0.60 |
| SO2F2 | 2699-79-8 | 0.922 | 186.140 | 0.383 | 2.303 | 0.543 | 0.79 | 0.73 |
| CF3SO2F | 335-05-7 | 1.230 | 216.863 | 0.346 | 2.349 | 0.782 | 1.36 | 1.45 |
| CH3CN | 75-05-8 | 0.851 | 532.756 | 0.892 | 1.021 | 0.470 | 0.77 | 0.80 |
| CF3CN | 353-85-5 | 1.013 | 366.065 | 0.334 | 1.931 | 0.706 | 1.53 | 1.50 |
| C2F5CN | 422-04-8 | 1.304 | 297.109 | 0.257 | 2.110 | 0.940 | 1.88 | 2.00 |
| C3F7CN | 375-00-8 | 1.565 | 280.934 | 0.230 | 2.219 | 1.142 | 2.32 | 2.40 |
| i-C3F7CN | 42532-60-5 | 1.548 | 245.345 | 0.218 | 2.227 | 1.138 | 2.22 | 2.20 |
| i-C3F7COCF3 | 756-12-7 | 1.812 | 165.640 | 0.210 | 2.428 | 1.045 | 2.38 | 2.10 |
| i-C3F7COC2F5 | 756-13-8 | 2.090 | 125.727 | 0.184 | 2.449 | 1.201 | 2.85 | 2.80 |
Table 1 GIPF parameters and dielectric strengths for a total of 43 insulation gaseous molecules
| Species | CAS registry | As/nm2 | ν | Π/eV | ρ/(g·cm-3) | Er,cal | ||
|---|---|---|---|---|---|---|---|---|
| H2 | 1333-74-0 | 0.336 | 6.635 | 0.131 | 0.180 | 0.208 | 0.16 | 0.22 |
| O2 | 7782-44-7 | 0.506 | 1.400 | 0.091 | 1.624 | 0.296 | 0.34 | 0.33 |
| N2 | 7727-37-9 | 0.542 | 44.973 | 0.173 | 1.265 | 0.350 | 0.35 | 0.38 |
| N2O | 10024-97-2 | 0.666 | 182.831 | 0.430 | 1.541 | 0.342 | 0.40 | 0.46 |
| CO | 630-08-0 | 0.545 | 94.462 | 0.261 | 1.254 | 0.317 | 0.34 | 0.40 |
| CO2 | 124-38-9 | 0.647 | 207.724 | 0.532 | 1.591 | 0.301 | 0.27 | 0.35 |
| OCS | 463-58-1 | 0.819 | 215.830 | 0.277 | 1.546 | 0.518 | 0.93 | 0.90 |
| SF6 | 2551-62-4 | 1.034 | 0.140 | 0.087 | 2.777 | 0.733 | 0.99 | 1.00 |
| CH4 | 74-82-8 | 0.602 | 9.208 | 0.126 | 0.624 | 0.346 | 0.37 | 0.43 |
| CH3Cl | 74-87-3 | 0.816 | 192.564 | 0.558 | 1.299 | 0.410 | 0.33 | 0.32 |
| CH3Br | 74-83-9 | 0.882 | 196.293 | 0.525 | 2.181 | 0.429 | 0.49 | 0.45 |
| CH2F2 | 75-10-5 | 0.712 | 313.110 | 0.701 | 1.676 | 0.320 | 0.33 | 0.27 |
| CH2Cl2 | 75-09-2 | 1.014 | 194.823 | 0.481 | 1.634 | 0.402 | 0.69 | 0.68 |
| CHF2Cl | 75-45-6 | 0.914 | 152.353 | 0.450 | 1.945 | 0.345 | 0.51 | 0.42 |
| CHFCl2 | 75-43-4 | 1.057 | 146.752 | 0.382 | 1.881 | 0.424 | 0.78 | 0.92 |
| CF4 | 75-73-0 | 0.811 | 4.219 | 0.175 | 2.388 | 0.382 | 0.49 | 0.42 |
| CF3Cl | 75-72-9 | 0.957 | 10.906 | 0.172 | 2.208 | 0.550 | 0.68 | 0.58 |
| CF2Cl2 | 75-71-8 | 1.095 | 38.548 | 0.176 | 2.108 | 0.576 | 0.91 | 0.99 |
| CF3Br | 75-63-8 | 1.023 | 20.324 | 0.187 | 2.864 | 0.517 | 0.78 | 0.75 |
| CH3CF3 | 420-46-2 | 0.977 | 170.332 | 0.564 | 1.745 | 0.425 | 0.43 | 0.41 |
| CH3CHCl2 | 75-34-3 | 1.204 | 214.009 | 0.461 | 1.497 | 0.544 | 1.01 | 1.01 |
| C2F6 | 76-16-4 | 1.118 | 3.519 | 0.144 | 2.427 | 0.638 | 0.92 | 0.80 |
| CF3CF2Cl | 76-15-3 | 1.241 | 9.646 | 0.158 | 2.314 | 0.633 | 1.05 | 1.04 |
| F2C=CFCl | 79-38-9 | 1.103 | 76.028 | 0.251 | 2.089 | 0.557 | 0.89 | 0.72 |
| CF3CH=CH2 | 677-21-4 | 1.114 | 223.217 | 0.494 | 1.674 | 0.501 | 0.87 | 0.80 |
| CF3CF=CF2 | 116-15-4 | 1.261 | 55.424 | 0.296 | 2.291 | 0.609 | 0.95 | 0.94 |
| CF2=CF—CF=CF2 | 685-63-2 | 1.401 | 89.157 | 0.315 | 2.184 | 0.586 | 1.18 | 1.20 |
| c-C6F10 | 355-75-9 | 1.786 | 14.075 | 0.220 | 2.428 | 0.892 | 1.80 | 1.90 |
| c-C4F8 | 115-25-3 | 1.472 | 5.602 | 0.241 | 2.437 | 0.605 | 1.18 | 1.25 |
| c-C6F12 | 355-68-0 | 1.856 | 0.910 | 0.107 | 2.553 | 1.304 | 2.33 | 2.35 |
| c-CF3(C4F6)CF3 | 1583-97-7 | 1.935 | 7.545 | 0.176 | 2.512 | 1.063 | 2.19 | 2.30 |
| CF3OCF3 | 1479-49-8 | 1.214 | 8.963 | 0.163 | 2.442 | 0.601 | 1.01 | 1.00 |
| c-CF3(C2F2O)CF3 | 117642-58-7 | 1.590 | 33.769 | 0.221 | 2.406 | 0.793 | 1.53 | 1.60 |
| HC≡CH | 74-86-2 | 0.672 | 304.742 | 0.534 | 0.858 | 0.306 | 0.57 | 0.60 |
| SO2F2 | 2699-79-8 | 0.922 | 186.140 | 0.383 | 2.303 | 0.543 | 0.79 | 0.73 |
| CF3SO2F | 335-05-7 | 1.230 | 216.863 | 0.346 | 2.349 | 0.782 | 1.36 | 1.45 |
| CH3CN | 75-05-8 | 0.851 | 532.756 | 0.892 | 1.021 | 0.470 | 0.77 | 0.80 |
| CF3CN | 353-85-5 | 1.013 | 366.065 | 0.334 | 1.931 | 0.706 | 1.53 | 1.50 |
| C2F5CN | 422-04-8 | 1.304 | 297.109 | 0.257 | 2.110 | 0.940 | 1.88 | 2.00 |
| C3F7CN | 375-00-8 | 1.565 | 280.934 | 0.230 | 2.219 | 1.142 | 2.32 | 2.40 |
| i-C3F7CN | 42532-60-5 | 1.548 | 245.345 | 0.218 | 2.227 | 1.138 | 2.22 | 2.20 |
| i-C3F7COCF3 | 756-12-7 | 1.812 | 165.640 | 0.210 | 2.428 | 1.045 | 2.38 | 2.10 |
| i-C3F7COC2F5 | 756-13-8 | 2.090 | 125.727 | 0.184 | 2.449 | 1.201 | 2.85 | 2.80 |
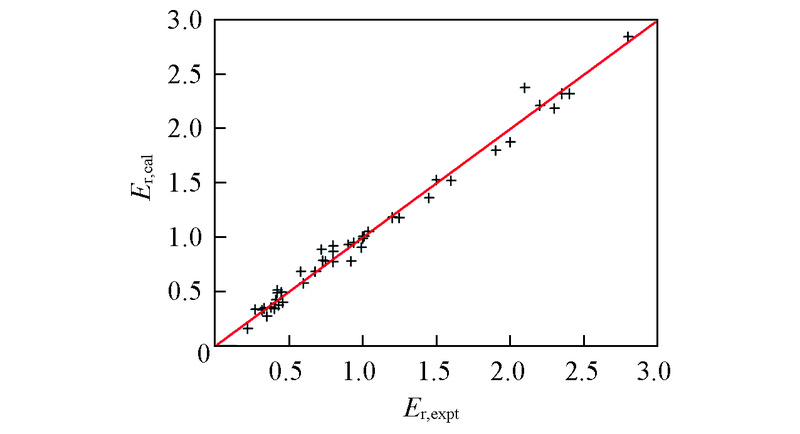
Fig.4 Theoretical dielectric strength predicted by the structure-activity relationship model S4 versus the experimental dataLine: diagonal line with y=x.
| Species | As/nm2 | ν | Π/eV | ρ/(g·cm-3) | Er,cal | ||
|---|---|---|---|---|---|---|---|
| SF5CF3 | 1.308 | 17.628 | 0.167 | 2.716 | 0.746 | 1.19 | 1.2—1.6 |
| NSF3 | 0.942 | 350.030 | 0.378 | 2.252 | 0.704 | 1.34 | 1.4 |
| SO2 | 0.754 | 462.050 | 0.762 | 1.849 | 0.361 | 0.71 | 0.52—1.0 |
| SeF6 | 1.101 | 0.858 | 0.131 | 3.413 | 0.687 | 1.00 | 1.03—1.14 |
| C6F6 | 1.551 | 35.222 | 0.380 | 2.150 | 0.738 | 1.11 | 1.05—1.15 |
| C4F10 | 1.639 | 2.661 | 0.129 | 2.502 | 1.125 | 1.76 | 1.0—1.58 |
Table 2 M06-2X/6-31++G(d,p) calculated GIPF parameters and dielectric strengths for the prototypical insulation gaseous molecules
| Species | As/nm2 | ν | Π/eV | ρ/(g·cm-3) | Er,cal | ||
|---|---|---|---|---|---|---|---|
| SF5CF3 | 1.308 | 17.628 | 0.167 | 2.716 | 0.746 | 1.19 | 1.2—1.6 |
| NSF3 | 0.942 | 350.030 | 0.378 | 2.252 | 0.704 | 1.34 | 1.4 |
| SO2 | 0.754 | 462.050 | 0.762 | 1.849 | 0.361 | 0.71 | 0.52—1.0 |
| SeF6 | 1.101 | 0.858 | 0.131 | 3.413 | 0.687 | 1.00 | 1.03—1.14 |
| C6F6 | 1.551 | 35.222 | 0.380 | 2.150 | 0.738 | 1.11 | 1.05—1.15 |
| C4F10 | 1.639 | 2.661 | 0.129 | 2.502 | 1.125 | 1.76 | 1.0—1.58 |
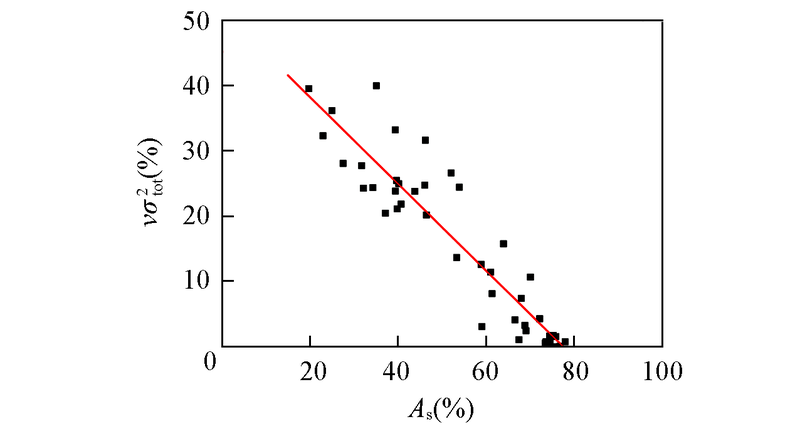
Fig.5 Dependence of the contribution percentages of νσtot2 to the dielectric strength on those of As predicted by the structure-activity relationship model S5
| [1] | Okubo H., Beroual A., IEEE Electr. Insul. Mag., 2011, 27(2), 34—42 |
| [2] | Du J. J., Xu H. C., Electr. Power Environ. Protect, 2009, 25(5), 58—60 |
| (杜建军, 徐慧超. 电力环境保护, 2009, 25(5), 58—60) | |
| [3] | Ko M., Sze N., Wang W. C., Shia G., Goldman A., Murcray F., Murcray D., Rinsland C., J. Geophys. Res., 1993, 98(D6), 10499—10507 |
| [4] | Fang X., Hu X., Greet J. M., Wu J., Han J., Su S., Zhang J., Hu J., Environ. Sci. Technol., 2013, 47(8), 3848—3855 |
| [5] | Franck C. M., Dahl D. A., Rabie M., Haefliger P., Koch M., Contrib. Plasma Phys., 2014, 54(1), 3—13 |
| [6] | Kieffel Y., Irwin T., Ponchon P., Owens J., IEEE Power Energy Mag., 2016, 14(2), 32—39 |
| [7] | Beroual A., Haddad A., Energies, 2017, 10(8), 1216 |
| [8] | Li Z. Y., Sci. China Ser. A, 1992, 35(4), 442—448 |
| (李正瀛. 中国科学A辑, 1992, 35(4), 442—448) | |
| [9] | Paschen F., Ann. Phys., 1889, 273(5), 69—75 |
| [10] | Boggs S. A., IEEE Electr. Insul. Mag., 1989, 5(6), 16—21 |
| [11] | Rothhardt L., Mastovsky J., Blaha J., J. Phys. D, Appl. Phys., 1985, 18(10), 155—157 |
| [12] | Zhang L. C., Xiao D. M., Zhang D., Wu B. T., Trans. China Electrotech. Soc., 2008, 23(6), 14—18 |
| (张刘春, 肖登明, 张栋, 吴变桃. 电工技术学报, 2008, 23(6), 14—18) | |
| [13] | Xing W. J., Zhang G. Q., Li K., Niu W. H., Wang X., Wang Y. Y., Proc. CSEE, 2011, 31(7), 119—124 |
| (邢卫军, 张国强, 李康, 牛文豪, 王新, 王迎迎. 中国电机工程学报, 2011, 31(7), 119—124) | |
| [14] | Pinheiro M. J., Loureiro J., J. Phys. D, Appl. Phys., 2002, 35(23), 3077—3084 |
| [15] | Wu B. T., Xiao D. M., Liu Z. S., Liu X. L., J. Phys. D., Appl. Phys., 2006, 39(19), 4204—4207 |
| [16] | Zhao H., Li X. W., Jia S. L., J. Xi’an Jiaotong University, 2013, 47(2), 109—115 |
| (赵虎, 李兴文, 贾申利. 西安交通大学学报, 2013, 47(2), 109—115) | |
| [17] | Hamilton J. R., Tennyson J., Huang S., Kushner M. J., Plasma Sources Sci. Technol., 2017, 26(6), 065010 |
| [18] | Vijh A. K., IEEE Trans. Electr. Insul., 1982, 17(4), 84—87 |
| [19] | Brand K. P., IEEE Trans. Electr. Insul., 1982, 17(5), 451—456 |
| [20] | Meurice N., Sandre E., Aslanides A., Vercauteren D. P., IEEE Trans. Dielectr. Electr. Insul., 2004, 11(6), 946—948 |
| [21] | Rabie M., Dahl D. A., Donald S. M. A., Reiher M., Franck C. M., IEEE Trans. Dielectr. Electr. Insul., 2013, 20(3), 856—863 |
| [22] | Jiao J., Xiao D., Zhao X., Deng Y., Plasma Sci. Technol., 2016, 18(5), 554—559 |
| [23] | Zhang C., Shi H., Cheng L., Zhao K., Xie X., Ma H., IEEE Trans. Dielect. Electr. Insul., 2016, 23(5), 2572—2578 |
| [24] | Yu X., Hou H., Wang B., J. Comput. Chem., 2017, 38(10), 721—729 |
| [25] | Murray J. S., Brinck T., Politzer P., Chem. Phys., 1996, 204(2/3), 289—299 |
| [26] | Murray J. S., Brinck T., Lane P., Paulsen K., Politzer P., J. Mol. Struct.(Theochem.), 1994, 307(1), 55—64 |
| [27] | Politzer P., Murray J. S., Theor. Chem. Acc., 2002, 108(3), 134—142 |
| [28] | Pearson R. G., Inorg. Chem., 1988, 27(4), 734—740 |
| [29] | Nie X. Q., Xu W. G., Lu S. X., Chem. J. Chinese Universities, 2008, 29(8), 1629—1634 |
| (聂小琴, 徐文国, 卢士香. 高等学校化学学报, 2008, 29(8), 1629—1634) | |
| [30] | Yu X., Hou H., Wang B., J. Phys. Chem. A, 2018, 122(13), 3462—3469 |
| [31] | Jonathan C. R., Gregory S. T., Henry F. S., Sreela N., Ellison G. B., Chem. Rev., 2002, 102(1), 231—282 |
| [32] | Anderson L. N., Oviedo M. B., Wong B. M., J. Chem. Theory Comput., 2017, 13(4), 1656—1666 |
| [33] | Bozkaya U., Ünal A., J. Phys. Chem. A, 2018, 122(17), 4375—4380 |
| [34] | Bozkaya U., J. Chem. Theory Comput., 2014, 10(5), 2041—2048 |
| [35] | Soydas E., Bozkaya U., J. Chem. Theory Comput., 2015, 11(4), 1564—1573 |
| [36] | Curtiss L. A., Redfern P. C., J. Chem. Phys., 1998, 109(1), 42—55 |
| [37] | Eisfeld W., J. Chem. Phys., 2011, 134(5), 054303 |
| [38] | Troe J., Miller T. M., Viggiano A. A., J. Chem. Phys., 2012, 136(12), 121102 |
| [39] | Menk S., Das S., Blaum K., Froese M. W., Lange M., Mukherjee M., Repnow R., Schwalm D., Hahn R., Wolf A., Phys. Rev. A, 2014, 89(2), 022502 |
| [40] | Olivet A., Duque D., Vega L. F., J. Appl. Phys., 2007, 101(2), 023308 |
| [41] | Christophorou L. G., Olthoff J. K., Green D. S., NIST Technical Note 1425, US Government Printing Office, Washington, 1997, 1—35 |
| [42] | Devins J. C., IEEE Trans. Electr. Insul., 1980, 15(2), 81—86 |
| [43] | Christophorou L. G., Sauers I., James D. R., Rodrigo H., Pace M. O., Carter J. G., Hunter S. R., IEEE Trans. Electr. Insul., 1984, 19(6), 550—566 |
| [44] | Wooton R. E., Kegelman M. R., Electric Power Research Institute EL-2620, Westinghouse Research and Development Center, Pennsylvania, 1982, 1—85 |
| [45] | Zhao Y., Truhlar D. G., Theor. Chem. Acc., 2008, 120(1—3), 215—241 |
| [46] | Frisch M. J., Pople J. A., Binkley J. S., J. Chem. Phys., 1984, 80(7), 3265—3269 |
| [47] | Frisch M. J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Montgomery J. A., Vreven T., Kudin K. N., Burant J. C., Millam J. M., Iyengar S., Tomasi S. J., Barone V., Mennucci B., Cossi M., Scalmani G., Rega N., Petersson G. A., Nakatsuji H., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Klene M., Li X., Knox J. E., Hratchian H. P., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., et al., Gaussian 09, Revision E.01, Gaussian Inc., Wallingford CT, 2009 |
| [48] | Geballe R., Linn F. S., J. Appl. Phys., 1950, 21(6), 592—594 |
| [49] | Eibeck R. E., Dielectric Gaseous Mixture of Thiazyltrifluoride and SF6, US3390091, 1968-06-25 |
| [50] | Vijh A. K., IEEE. Trans. Electr. Insul., 1977, 12(4), 313—315 |
| [51] | Banks A. A., Rudge A. J., Nature, 1953, 171(4348), 390—391 |
| [1] | 张咪, 田亚锋, 高克利, 侯华, 王宝山. 三氟甲基磺酰氟绝缘介质理化特性的分子动力学模拟[J]. 高等学校化学学报, 2022, 43(11): 20220424. |
| [2] | 侯华, 王宝山. 六氟化硫替代气体绝缘强度的官能团加和理论方法[J]. 高等学校化学学报, 2021, 42(12): 3709. |
| [3] | 周小平, 张沐新, 王广树, 杨晓虹. 乙型肝炎病毒表面抗原三维结构的同源模建及功能预测[J]. 高等学校化学学报, 2010, 31(4): 742. |
| [4] | 邵琛, 胡冬华, 孙海珠, 颜力楷, 苏忠民, 王荣顺, 朱文圣, 郭建华, 史宁中, 孙晖, 李泽生, 孙家锺. SARS冠状病毒E蛋白的结构研究及功能预测[J]. 高等学校化学学报, 2005, 26(8): 1507. |
| [5] | 赵健伟, 王楠. 导电原子力显微镜对蛋白质在分子水平上的电学表征[J]. 高等学校化学学报, 2005, 26(4): 751. |
| [6] | 邹建卫, 商志才, 俞庆森. 药物溶解性能与溶剂分子三维静电势参数的定量关系[J]. 高等学校化学学报, 2002, 23(4): 644. |
| [7] | 袁满雪, 杨大军, 石静, 卜显和, 赖城明. 有机化合物中二级化学键的理论研究(Ⅳ)——喹啉基氮杂大环铜配合物中的二级化学键[J]. 高等学校化学学报, 1998, 19(4): 583. |
| [8] | 李慧英, 王霞, 黄润秋, 马军安, 邱德文, 袁满雪, 赖城明. 应用分子图形学、分子力学、量子化学及静电势研究农药分子结构与性能关系(XI)──抗植物病毒病药物──胡椒醛肟醚基-4-喹唑啉的结构特征[J]. 高等学校化学学报, 1997, 18(10): 1633. |
| [9] | 王一波, 史鸿运. 从头计算法研究沙蚕毒和杀虫环分子的几何构型与电子结构[J]. 高等学校化学学报, 1996, 17(3): 436. |
| [10] | 赖城明, 袁满雪, 李正名, 贾国锋. 应用分子图形学、分子力学、量子化学及静电势研究农药分子结构与性能的关系(Ⅴ)─磺酰脲类分子中的化学键及电子结构[J]. 高等学校化学学报, 1994, 15(7): 1004. |
| [11] | 刘靖疆, 黄常华, 吕亮德, 王彦中. 环三氢集团的研究[J]. 高等学校化学学报, 1991, 12(4): 475. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||