

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (2): 310.doi: 10.7503/cjcu20170349
• Physical Chemistry • Previous Articles Next Articles
ZHANG Baohai1, LUO Min1,*( ), YANG Shun1, FU Rongrong1, MA Jinfu2
), YANG Shun1, FU Rongrong1, MA Jinfu2
Received:2017-06-06
Online:2018-02-10
Published:2017-12-05
Contact:
LUO Min
E-mail:martinluomin@163.com
Supported by:CLC Number:
TrendMD:
ZHANG Baohai, LUO Min, YANG Shun, FU Rongrong, MA Jinfu. Preparation and Electrochemical Properties of Hierarchically Porous Carbon Microspheres Derived from Metal Phenolic Precursor†[J]. Chem. J. Chinese Universities, 2018, 39(2): 310.
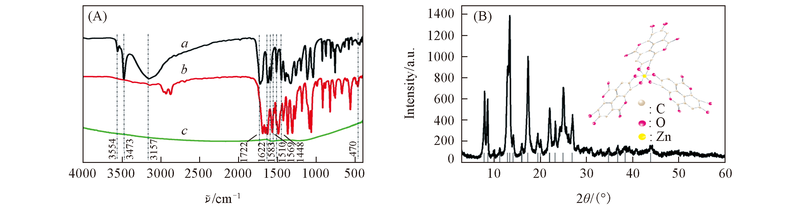
Fig.4 FTTR spectra of the EA(a), ZnEA(b) and C-ZnEA-1000(c) samples(A) and XRD pattern of ZnEA(B) Inset of (B) shows the perspective view of the ZnEA molecular structure.
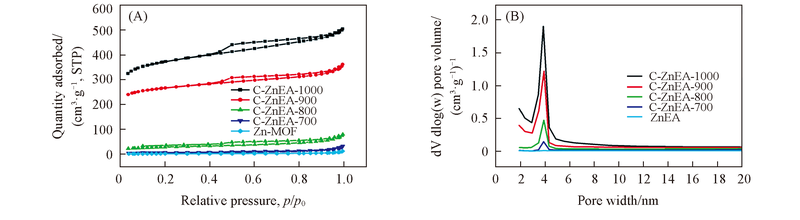
Fig.8 N2 adsorption-desorption isotherms(A) and the pore size distribution curves(B) of the C-ZnEA-700/800/900/1000 hierarchically porous carbon microsphere samples
| Sample | BET surface area/(m2·g-1) | Porevolum/(m3·g-1) | Average pore width/nm |
|---|---|---|---|
| ZnEA | 5.32 | 0.02 | 14.04 |
| C-ZnEA-700 | 20.25 | 0.05 | 9.69 |
| C-ZnEA-800 | 95.40 | 0.12 | 5.11 |
| C-ZnEA-900 | 887.29 | 0.56 | 2.55 |
| C-ZnEA-1000 | 1238.13 | 0.78 | 2.52 |
Table 1 Porosity and properties of C-ZnEA-700/800/900/1000 porous carbon microsphere samples
| Sample | BET surface area/(m2·g-1) | Porevolum/(m3·g-1) | Average pore width/nm |
|---|---|---|---|
| ZnEA | 5.32 | 0.02 | 14.04 |
| C-ZnEA-700 | 20.25 | 0.05 | 9.69 |
| C-ZnEA-800 | 95.40 | 0.12 | 5.11 |
| C-ZnEA-900 | 887.29 | 0.56 | 2.55 |
| C-ZnEA-1000 | 1238.13 | 0.78 | 2.52 |
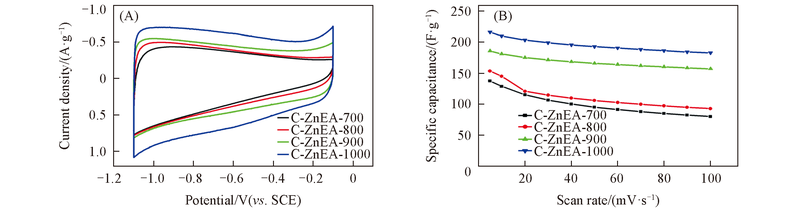
Fig.9 Electrochemical performance of C-ZnEA-700/800/900/1000 samples(A) Cyclic voltammograms curves at a scan rate of 5 mV/s; (B) the specific capacitance variations at different scan rates.
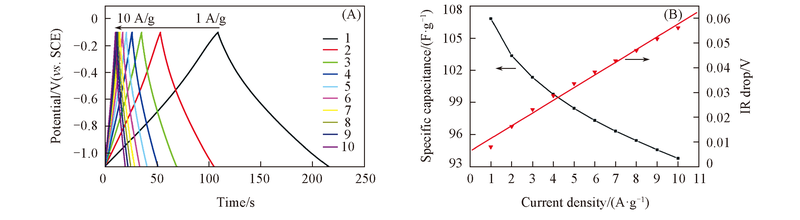
Fig.10 Glavanostatic charge-discharge curves of C-ZnEA-1000 at different current density and specific capacitance variation and IR drop of C-ZnEA-1000 with discharge current density(B)
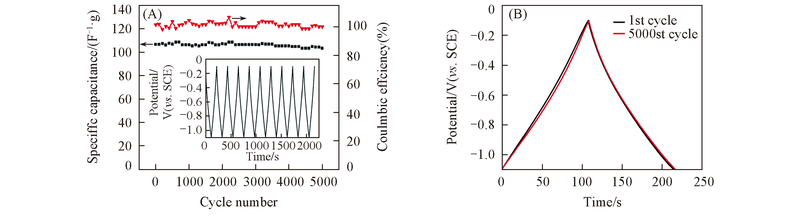
Fig.12 Cycle performance and Coulombic efficiency of C-ZnEA-1000(A) at 1 A/g and constant current charge-discharge curve of the Its cycle and 5000th cycle(B) The inset is the constant current charge-discharge diagram for the last 20 cycles of the cycle.
| Material | Electrolyte(potential window/V) | Rate capability(%) (scan rate or current density) | Capacitance/(F·g-1)(scan rate or current density) | Cycle life(%) (Cycling No.) | Ref. |
|---|---|---|---|---|---|
| Zn-bdc derived porous carbon | 6 mol/L KOH (-1—0) | 65(1—100 mV/s) | 170 (1 mV/s) | 98(5000) | [ |
| MOF-5 derived 3D porous carbon | 6 mol/L KOH (-0.9—0.1) | 82.9(0.05—20 A/g) | 212(0.05 A/g) | 95.9(1000) | [ |
| Al-PCP derived carbons | 30% KOH (-1—0) | 75(0.1—1 A/g) | 232.8(0.1 A/g) | [ | |
| ZIF-8 derived porous carbon | 1 mol/L H2SO4 (0—0.6) | 60(5—200 mV/s) | 251(15 mV/s) | 92(2000) | [ |
| HPC microsphere derived from ZnEA | 6 mol/L KOH (-1.1—-0.1) | 84.67(5—100 mV/s) | 216(5 mV/s) | 97(5000) | This work |
Table 2 Comparison of capacitance of MOFs derived porous carbon supercapacitor*
| Material | Electrolyte(potential window/V) | Rate capability(%) (scan rate or current density) | Capacitance/(F·g-1)(scan rate or current density) | Cycle life(%) (Cycling No.) | Ref. |
|---|---|---|---|---|---|
| Zn-bdc derived porous carbon | 6 mol/L KOH (-1—0) | 65(1—100 mV/s) | 170 (1 mV/s) | 98(5000) | [ |
| MOF-5 derived 3D porous carbon | 6 mol/L KOH (-0.9—0.1) | 82.9(0.05—20 A/g) | 212(0.05 A/g) | 95.9(1000) | [ |
| Al-PCP derived carbons | 30% KOH (-1—0) | 75(0.1—1 A/g) | 232.8(0.1 A/g) | [ | |
| ZIF-8 derived porous carbon | 1 mol/L H2SO4 (0—0.6) | 60(5—200 mV/s) | 251(15 mV/s) | 92(2000) | [ |
| HPC microsphere derived from ZnEA | 6 mol/L KOH (-1.1—-0.1) | 84.67(5—100 mV/s) | 216(5 mV/s) | 97(5000) | This work |
| [1] | Conway B.E., Electrochemical Supercapacitors. Scientific Fundamentals and Technological Applications, Plenum Press, New York, 1999 |
| [2] | Wang G., Zhang L., Zhang J., Chem. Soc. Rev., 2012, 41(2), 797—828 |
| [3] | Fu R. R., Luo M., Ma Y. H., Yang S., Chem. J. Chinese Universities, 2016, 37(8), 1485—1490) |
| (付蓉蓉,罗民,马永华,杨顺. 高等学校化学学报, 2016,37(8), 1485—1490) | |
| [4] | Sarangapani S., Tilak B. V., Chen C. P., J. Electrochem. Soc., 1996, 143(11), 3791—3799 |
| [5] | Luo M., Ding X. Y., Dou Y. Y., Zhao L., Inorg. Chem., 2015, 31(1), 54—60 |
| (罗民, 丁肖怡, 窦元运, 赵亮. 无机化学学报, 2015,31(1), 54—60) | |
| [6] | Zhu Y., Murali S., Stoller M. D., Ganesh K. J., Cai W., Ferreira P. J., Pirkle A., Wallace R. M., Cychosz K. A., Thommes M., Su D., Stach E. A., Ruoff R. S., Science,2011, 332(6037), 1537—1541 |
| [7] | Li W., Zhang F., Dou Y., Wu Z., Liu H., Qian X., Gu D., Xia Y., Tu B., Zhao D., Advanced Energy Materials, 2011, 1(3), 382—386 |
| [8] | Li Z., Xu Z., Tan X., Wang H., Holt C. M. B., Stephenson T., Olsen B. C., Mitlin D., Energy & Environmental Science, 2013, 6(3), 871—878 |
| [9] | Tamai H., Kunihiro M., Morita M., Yasuda H., J. Mater. Sci., 2005, 40(14), 3703—3707 |
| [10] | Vivekchand S. R. C., Rout C. S., Subrahmanyam K. S., Govindaraj A., Rao C. N. R., J. Chem. Sci., 2008, 120(1), 9—13 |
| [11] | Zhang L. L., Gu Y., Zhao X. S., J. Mater. Chem. A, 2013, 1(33), 9395—9408 |
| [12] | Frackowiak E., Beguin F., Carbon,2001, 39(6), 937—950 |
| [13] | Jiang H. L., Liu B., Lan Y. Q., Kuratani K., Akita T., Shioyama H., Zong F., Xu Q., J. Am. Chem. Soc., 2011, 133(31), 11854—11857 |
| [14] | Liu B., Shioyama H., Akita T., Xu Q., J. Am. Chem. Soc., 2008, 130(16), 5390 |
| [15] | Przewloka S. R., Shearer B. J., Holzforschung,2002, 56(1), 13—19 |
| [16] | Yang S. J., Antonietti M., Fechler N., J. Am. Chem. Soc., 2015, 137(25), 8269 |
| [17] | Nelson K. M., Mahurin S. M., Mayes R. T., Williamson B., Teague C. M., Binder A. J., Baggetto L., Veith G. M., Dai S., Microporous and Mesoporous Materials, 2016, 222, 94—103 |
| [18] | Liu H.Y., HU X., Ming Y. X., Liu Y., Huang J. W., Ji L. N.,Inorg. Chem., 1998, (4), 5—21 |
| (刘海洋, 胡希, 明应晓, 刘义, 黄锦汪, 计亮年. 无机化学学报, 1998, (4), 5—21) | |
| [19] | Zhang L., Hu Y. H., J. Phys. Chem. C, 2010, 114(6), 2566—2572 |
| [20] | Kimitsuka Y., Hosono E., Ueno S., Zhou H., Fujihara S., Inorg. Chem., 2013, 52(24), 14028—14033 |
| [21] | Zhang S.L.,Raman Spectroscopy and Low Dimensional Nanometer Semiconductor, Science Press, Beijing, 2008, 98 |
| (张树霖.拉曼光谱学与低维纳米半导体 , 北京: 科学出版社 2008, 98) | |
| [22] | Yang Y., Hao S., Zhao H., Wang Y., Zhang X., Electrochim. Acta, 2015, 180, 651—657 |
| [23] | Yu M., Zhang L., He X., Yu H., Han J., Wu M., Materials Letters, 2016, 172, 81—84 |
| [24] | Yan X., Li X., Yan Z., Komarneni S., Applied Surface Science, 2014, 308, 306—310 |
| [25] | Salunkhe R. R., Kamachi Y., Torad N. L., Hwang S. M., Sun Z., Dou S. X., Kim J. H., Yamauchi Y., J. Mater. Chem. A, 2014, 2(46), 19848—19854 |
| [1] | HOU Congcong, WANG Huiying, LI Tingting, ZHANG Zhiming, CHANG Chunrui, AN Libao. Preparation and Electrochemical Properties of N-CNTs/NiCo-LDH Composite [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220351. |
| [2] | LIANG Yu, LIU Huan, GONG Lige, WANG Chunxiao, WANG Chunmei, YU Kai, ZHOU Baibin. Synthesis and Supercapacitor Properties of Biimidazole-modified {SiW12O40} Hybrid [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210556. |
| [3] | WEI Yuchen, WU Tingting, YANG Lei, JIN Biyu, LI Hongqiang, HE Xiaojun. Preparation and Supercapacitive Performance of Naphthalene-based Interconnected Porous Carbon Nanocapsules [J]. Chem. J. Chinese Universities, 2021, 42(9): 2852. |
| [4] | HUANG Dongxue, ZHANG Ying, ZENG Ting, ZHANG Yuanyuan, WAN Qijin, YANG Nianjun. Transition Metal Sulfides Hybridized with Reduced Graphene Oxide for High-Performance Supercapacitors [J]. Chem. J. Chinese Universities, 2021, 42(2): 643. |
| [5] | SHA Huiwen, MA Weiting, ZHOU Xiaojuan, SONG Weixing. One-step Preparation and Applications of Laser Induced Three-dimensional Reticular Graphene [J]. Chem. J. Chinese Universities, 2021, 42(2): 607. |
| [6] | CHEN Minghua, LI Hongwu, FAN He, LI Yu, LIU Weiduo, XIA Xinhui, CHEN Qingguo. Research Progress of Two-dimensional Transition Metal Dichalcogenides in Supercapacitors [J]. Chem. J. Chinese Universities, 2021, 42(2): 539. |
| [7] | ZHANG Weiguo, FAN Songhua, WANG Hongzhi, YAO Suwei. Synthesis of Self-assembled α-Fe2O3/Graphene Hydrogel for Supercapacitors with Promising Electrochemical Properties [J]. Chem. J. Chinese Universities, 2020, 41(8): 1850. |
| [8] | GUAN Fanglan,LI Xin,ZHANG Qun,GONG Yan,LIN Ziyu,CHEN Yao,WANG Lejun. Fabrication and Capacitance Performance of Laser-machined RGO/MWCNT/CF In-plane Flexible Micro-supercapacitor † [J]. Chem. J. Chinese Universities, 2020, 41(2): 300. |
| [9] | LI Botian,SHAO Wei,XIAO Da,ZHOU Xue,DONG Junwei,TANG Liming. Polypyrrole Nanowire Gels Based on Templating Fabrication and Their Energy Storage and Electrochemical Sensing Properties † [J]. Chem. J. Chinese Universities, 2020, 41(1): 183. |
| [10] | LIU Ben,ZHANG Xingying,CHEN Shaoyun,HU Chenglong. Preparation and Electrochemical Energy Storage Performance of One Dimensional Orderly Polyaniline Nanowires Array† [J]. Chem. J. Chinese Universities, 2019, 40(3): 498. |
| [11] | LIU Hao,ZHAO Dingxuan,GONG Guodong,ZHANG Zhuxin,JIA Tuo,CHEN Hanzhe. Effect of Temperature on Morphology and Supercapacitor Performance of Carbon Nano-spheres† [J]. Chem. J. Chinese Universities, 2019, 40(1): 18. |
| [12] | LI Long,HU Hongli,DING Shujiang. Facile Synthesis of Scale-like CoMn2O4 Nanosheets on Reduced Graphene Oxide for Supercapacitors† [J]. Chem. J. Chinese Universities, 2018, 39(9): 2010. |
| [13] | NIE Guangdi, ZHU Yun, TIAN Di, WANG Ce. Research Progress in the Electrospun Nanofiber-based Supercapacitor Electrode Materials† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1349. |
| [14] | WANG Fengmei, XU Guangwei, JIN Chengchang. Synthesis and Electrochemical Performance for Supercapacitors of Bi-doped α-MnO2 Nanorods [J]. Chem. J. Chinese Universities, 2018, 39(3): 530. |
| [15] | FENG Dongyang,GUO Di,LIU Xiaoxia. Functionalization of Carbon Electrode and Subsequent Electrochemical Deposition of Nanostructured Manganese Oxide† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2280. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||