

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (9): 1674.doi: 10.7503/cjcu20150184
• Analytical Chemistry • Previous Articles Next Articles
ZHUANG Qianfen1, WANG Yong1, NI Yongnian1,2,*( )
)
Received:2015-03-09
Online:2015-09-10
Published:2015-08-17
Contact:
NI Yongnian
E-mail:ynni@ncu.edu.cn
Supported by:CLC Number:
TrendMD:
ZHUANG Qianfen, WANG Yong, NI Yongnian. Electrochemical Sensor for the Detection of Riboflavin Based on Nanocomposite Film of Polydeoxyadenylic Acid/Reduced Graphene Oxide†[J]. Chem. J. Chinese Universities, 2015, 36(9): 1674.
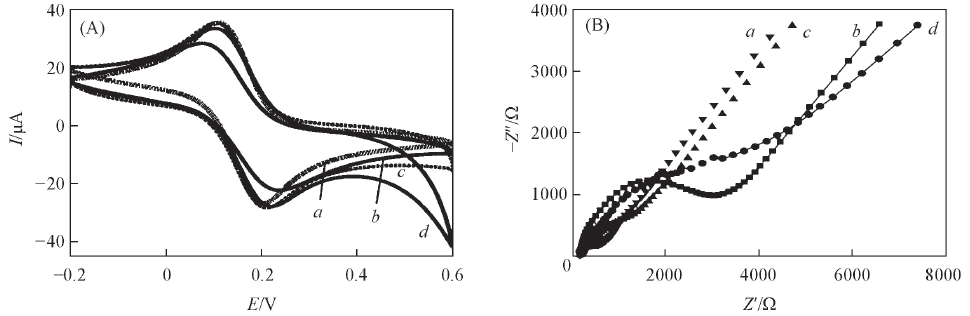
Fig.3 CV curves(A) and EIS responses(B) of different electrodes in 5.0 mmol/L [Fe(CN)6]3-/[Fe(CN)6]4-(1∶1) solution containing 0.1 mol/L KCla. Bare Au electrode; b. A32/Au electrode; c. RGO/A32/Au electrode; d. rMoS2-graphene/A32/Au electrode.
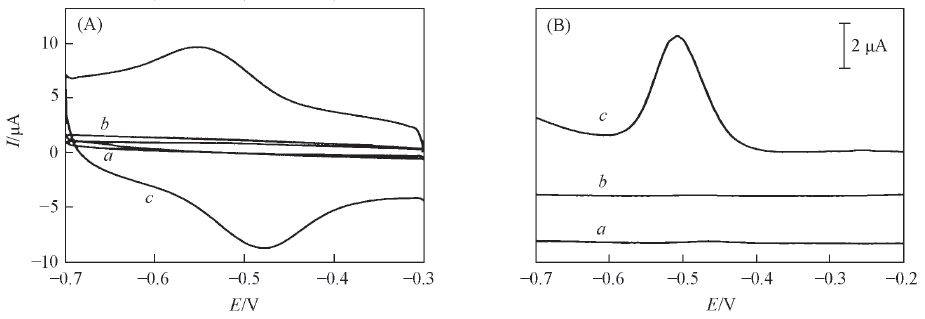
Fig.4 CV(A) and DPV(B) curves of different electrodes in the N2-saturated electrochemistry buffer solution containing 2.0 μmol/L riboflavina. Bare Au electrode; b. A32/Au electrode; c. RGO/A32/Au electrode.
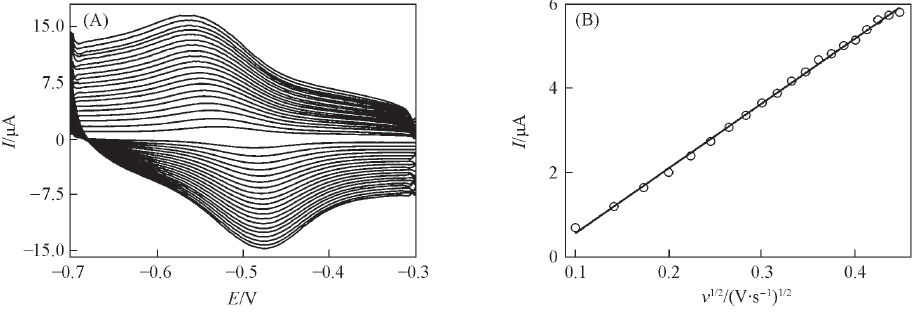
Fig.5 CVs curves of RGO/A32/Au electrode with different scan rates in the N2-saturated electrochemistry buffer solution containing 2.0 μmol/L riboflavin(A), and the linear relationship between the anodic peak current and the square root of scan rate(B)Scan rates from inner to outer curves/(V·s-1): 0.01, 0.02, 0.03, 0.04, 0.05, 0.06, 0.07, 0.08, 0.09, 0.1, 0.11, 0.12, 0.13, 0.14, 0.15, 0.16, 0.17, 0.18, 0.19, 0.2.
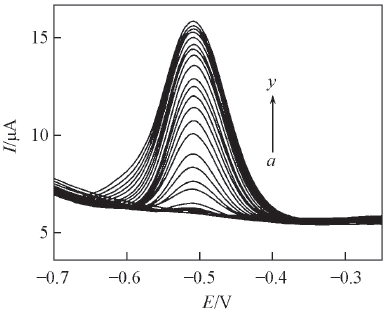
Fig.6 DPV curves of the RGO/ A32/Au electrode with different concentrations of riboflavin in the N2-saturated electrochemistry buffer solutionConcentrations of riboflavin from a to y/(μmol·L-1): 0, 0.025, 0.05, 0.1, 0.25, 0.5, 0.75, 1.0, 1.25, 1.5, 1.75, 2.0, 2.25, 2.5, 2.75, 3.0, 3.5, 4.0, 4.5, 5.0, 6.0, 7.0, 8.0, 9.0, 10.0.
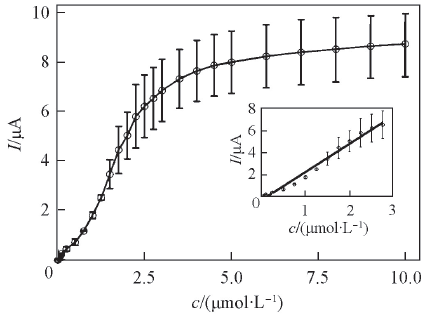
Fig.7 Plot of cathodic peak current for the RGO/ A32/Au electrode against riboflavin concentration in the N2-saturated electrochemistry buffer solutionInset: calibration curve of the electrode for riboflavin. The error bar on each datum point represents mean value plus or minus one standard deviation.
| Method | Linear range/(μmol·L-1) | LOD/(μmol·L-1) | Reference |
|---|---|---|---|
| Electrochemistry | 0.1—200 | 0.05 | [ |
| Electrochemistry | 1.33—186.0 | 0.903 | [ |
| Electrochemistry | 0.323—40 | 0.187 | [ |
| Electrochemistry | 0.2—100 | 0.107 | [ |
| Electrochemistry | 0.009—55.9 | 0.001 | [ |
| Electrochemistry | 0.3—0.8 | 0.10 | [ |
| Electrochemistry | 0.025—2.25 | 0.02 | [ |
| Fluorescence | 0.58—20 | 0.18 | [ |
| Fluorescence | 0—265.7 | 1.59 | [ |
| Electrochemistry | 0.025—2.75 | 0.015 | This work |
Table 1 Comparison of determination of riboflavin using various sensors
| Method | Linear range/(μmol·L-1) | LOD/(μmol·L-1) | Reference |
|---|---|---|---|
| Electrochemistry | 0.1—200 | 0.05 | [ |
| Electrochemistry | 1.33—186.0 | 0.903 | [ |
| Electrochemistry | 0.323—40 | 0.187 | [ |
| Electrochemistry | 0.2—100 | 0.107 | [ |
| Electrochemistry | 0.009—55.9 | 0.001 | [ |
| Electrochemistry | 0.3—0.8 | 0.10 | [ |
| Electrochemistry | 0.025—2.25 | 0.02 | [ |
| Fluorescence | 0.58—20 | 0.18 | [ |
| Fluorescence | 0—265.7 | 1.59 | [ |
| Electrochemistry | 0.025—2.75 | 0.015 | This work |
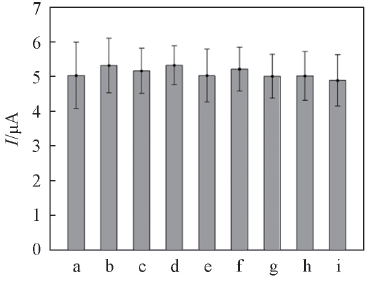
Fig.8 Plot of cathodic peak current for the RGO/ A32/Au electrode against riboflavin(2.0 μmol/L) with and without other bioactive substance(100.0 μmol/L)a. Riboflavin; b. riboflavin+ascorbic acid; c. riboflavin+vitamin B6; d. riboflavin+nicotinamide; e. riboflavin+thiamine; f. riboflavin+glucose; g. riboflavin+lysine; h. riboflavin+folic acid; i. riboflavin+uric acid.
| Sample | Added/(μmol·L-1) | Founda/(μmol·L-1) | Mean recoveryb(%) |
|---|---|---|---|
| 1 | 0.25 | 0.26±0.02 | 104 |
| 2 | 1.0 | 1.07±0.08 | 107 |
| 3 | 2.0 | 1.96±0.04 | 98 |
Table 2 Determination of riboflavin spiked in urine samples(n=3)
| Sample | Added/(μmol·L-1) | Founda/(μmol·L-1) | Mean recoveryb(%) |
|---|---|---|---|
| 1 | 0.25 | 0.26±0.02 | 104 |
| 2 | 1.0 | 1.07±0.08 | 107 |
| 3 | 2.0 | 1.96±0.04 | 98 |
| [1] | Foraker A. B., Khantwal C. M., Swaan P. W., Adv. Drug Deliv. Rev., 2003, 55(11), 1467—1483 |
| [2] | Gastaldi G., Ferrari G., Verri A., Casirola D., Orsenigo M. N., Laforenza U., J. Nutr., 2000, 130(10), 2556—2561 |
| [3] | Bishop A. M., Fernadndez C., Whitehead R. D., Morales P., Barr D. B., Wilder L. C., Baker S. E., J. Chromatogr. Technol. Biomed. Life Sci., 2011, 879(20), 1823—1826 |
| [4] | Caelen I., Kalman A., Wahlstrom L., Anal. Chem., 2004, 76(1), 137—143 |
| [5] | Aranda M., Morlock G., J. Chromatogr. A, 2006, 1131(1/2), 253—260 |
| [6] | Kundu A., Nandi S., Layek R. K., Nandi A. K., ACS Appl. Mater. Interfaces, 2013, 5(15), 7392—7399 |
| [7] | Yan Y., Xu J. G., Lin J., Chen G. Z., Chem. J. Chinese Universities, 1997, 18(6), 877—879 |
| (鄢远, 许金钩, 林江, 陈国珍. 高等学校化学学报, 1997, 18(6), 877—879) | |
| [8] | Geim A. K., Science, 2009, 324(5934), 1530—1534 |
| [9] | Wang Z. T., Xiao C. F., Zhao J., Hu X., Xu N. K., Chem. J. Chinese Universities, 2014, 35(11), 2410—2417 |
| (王子涛, 肖长发, 赵健, 胡霄, 徐乃库. 高等学校化学学报, 2014, 35(11), 2410—2417) | |
| [10] | Chen Z. X., Lu H. B., Chem. J. Chinese Universities, 2013, 34(9), 2020—2033 |
| (陈仲欣, 卢红斌. 高等学校化学学报, 2013, 34(9), 2020—2033) | |
| [11] | Chen Y., Wang J., Liu Z. M., Chin. J. Anal. Chem., 2012, 40(11), 1772—1779 |
| (陈钰, 王婕, 刘仲明. 分析化学, 2012, 40(11), 1772—1779) | |
| [12] | Lawal A. T., Talanta, 2015, 131, 424—443 |
| [13] | Ping J. F., Zhou Y. B., Wu Y. Y., Papper V., Boujday S., Marks R. S., Steele T. W. J., Biosens. Bioelectron., 2015, 64, 373—385 |
| [14] | Liu J. Q., Liu Z., Barrow C. J., Yang W. R., Anal. Chim. Acta, 2015, 859, 1—19 |
| [15] | Wang H. J., Cheng N. N., Yang X. Y., Li X. M., Zhu L. D., Chem. Res. Chinese Universities, 2013, 29(1), 132—138 |
| [16] | An J., Li J. P., Chen W. X., Yang C. X., Hu F. D., Wang C. M., Chem. Res. Chinese Universities, 2013, 29(4), 798—805 |
| [17] | Teller C., Willner I., Curr. Opin. Biotechnol., 2010, 21(4), 376—391 |
| [18] | Tang Y. T., Ge B. X., Sen D., Yu H. Z., Chem. Soc. Rev., 2014, 43(2), 518—529 |
| [19] | Opdahl A., Petrovykh D. Y., Kimura-Suda H., Tarlov M. J., Whitman L. J., Proc. Natl. Acad. Sci. USA, 2007, 104(1), 9—14 |
| [20] | Liu J. W., Phys. Chem. Chem. Phys., 2012, 14(30), 10485—10496 |
| [21] | Varghese N., Mogera U., Govindaraj A., Das A., Chem. Phys. Chem., 2009, 10(1), 206—210 |
| [22] | Marcano D. C., Kosynkin D. V., Berlin J. M., Sinitskii A., Sun Z. Z., Slesarev A., Alemany L. B., Lu W., Tour J. M., ACS Nano, 2010, 4(8), 4806—4814 |
| [23] | Stankovich S., Dikin D. A., Piner R. D., Kohlhaas K. A., Kleinhammes A., Jia Y. Y., Wu Y., Nguyen S. T., Ruoff R. S., Carbon, 2007, 45(7), 1558—1565 |
| [24] | Dutta S., Sarkar S., Ray C., Pal T., RSC Adv., 2013, 3(44), 21475—21483 |
| [25] | Some S., Kim Y., Yoon Y., Yoo H., Lee S., Park Y., Lee H., Sci. Rep., 2013,3, 1929 |
| [26] | Wang Y., Zhuang Q. F., Ni Y. N., J. Electroanal. Chem., 2015, 736, 47—54 |
| [27] | Huang G. C., Chen T., Chen W. X., Wang Z., Chang K., Ma L., Huang F. H., Chen D. Y., Lee J. Y., Small, 2013, 9(21), 3693—3703 |
| [28] | Chang K., Chen W. X., ACS Nano, 2011, 5(6), 4750—4728 |
| [29] | Li J. L., Liu X. J., Pan L. K., Qin W., Chen T. Q., Sun Z., RSC Adv., 2014, 4(19), 9647—9651 |
| [30] | Zhang H., Zhao J. S., Liu H. T., Wang H. S., Liu R. M., Liu J. F., Int. J. Electrochem. Sci., 2010, 5(3), 295—301 |
| [31] | Ensafi A. A., Heydari-Bafrooei E., Amini M., Biosens. Bioelectron., 2012, 31(1), 376—381 |
| [32] | Li Y., Hsu P. C., Chen S. M., Sens. Actuators A, 2012, 174, 427—435 |
| [33] | Lavanya N., Radhakrishnan S., Sekar C., Navaneethan M., Hayakawa Y., Analyst, 2013, 138(7), 2061—2067 |
| [34] | Kumar D. R., Manoj D., Santhanalakshmi J., Anal. Methods, 2014, 6(4), 1011—1020 |
| [35] | Sa E. S., Da Silva P. S., Jost C. L., Spinelli A., Sens. Actuators A, 2015, 209, 423—430 |
| [36] | Li P. P., Liu S. P., Wang X. D., Liu Z. Q., He Y. Q., Luminescence, 2013, 28(6), 910—914 |
| [1] | LI Yulong, XIE Fating, GUAN Yan, LIU Jiali, ZHANG Guiqun, YAO Chao, YANG Tong, YANG Yunhui, HU Rong. A Ratiometric Electrochemical Sensor Based on Silver Ion Interaction with DNA for the Detection of Silver Ion [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220202. |
| [2] | WANG Xueli, SONG Xiangwei, XIE Yanning, DU Niyang, WANG Zhenxin. Preparation, Characterization of Partially Reduced Graphene Oxide and Its Killing Effect on Human Cervical Cancer Cells [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210595. |
| [3] | WEI Chuangyu, CHEN Yanli, JIANG Jianzhuang. Fabrication of Electrochemical Sensor for Dopamine and Uric Acid Based on a Novel Dimeric Phthalocyanine-involved Quintuple-decker Modified Indium Tin Oxide Electrode [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210582. |
| [4] | MIAO Weijun, WU Feng, WANG Yong, WANG Zongbao. In⁃situ Study of the Epitaxial Crystallization of PCL/RGO at High Shear Rate [J]. Chem. J. Chinese Universities, 2021, 42(3): 910. |
| [5] | HUANG Dongxue, ZHANG Ying, ZENG Ting, ZHANG Yuanyuan, WAN Qijin, YANG Nianjun. Transition Metal Sulfides Hybridized with Reduced Graphene Oxide for High-Performance Supercapacitors [J]. Chem. J. Chinese Universities, 2021, 42(2): 643. |
| [6] | FU Kefei, LIAN Huiting, WEI Xiaofeng, SUN Xiangying, LIU Bin. Construction of Cyclodextrin-based Impedance Sensor for Recognition of L-Cysteine † [J]. Chem. J. Chinese Universities, 2020, 41(4): 706. |
| [7] | GUAN Fanglan,LI Xin,ZHANG Qun,GONG Yan,LIN Ziyu,CHEN Yao,WANG Lejun. Fabrication and Capacitance Performance of Laser-machined RGO/MWCNT/CF In-plane Flexible Micro-supercapacitor † [J]. Chem. J. Chinese Universities, 2020, 41(2): 300. |
| [8] | LI Botian,SHAO Wei,XIAO Da,ZHOU Xue,DONG Junwei,TANG Liming. Polypyrrole Nanowire Gels Based on Templating Fabrication and Their Energy Storage and Electrochemical Sensing Properties † [J]. Chem. J. Chinese Universities, 2020, 41(1): 183. |
| [9] | LI Long,HU Hongli,DING Shujiang. Facile Synthesis of Scale-like CoMn2O4 Nanosheets on Reduced Graphene Oxide for Supercapacitors† [J]. Chem. J. Chinese Universities, 2018, 39(9): 2010. |
| [10] | SUN Bing,JIANG Shang,WANG Runwei,NI Ling,QIU Shilun,ZHANG Zongtao. Preparation and Application of High Performance Lithium Titanate/reduced Graphene Oxide Nanocomposites for Lithium Batteries† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2767. |
| [11] | QIU Lijuan,ZHANG Ying,LIU Shuaizhuo,ZHANG Qian,ZHOU Ying. Preparation and Application of Superhydrophobic and Robust Graphene Composites Oil/Water Separation Material† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2758. |
| [12] | WANG Xueli,WANG Zhenxin. Preparation of a Targeted Tumor Nanocomposites for Combined Photodynamic-photothermal Therapy Based on Partially Reduced Graphene Oxide† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2185. |
| [13] | BULIN Chaoke, GUO Ting, ZHANG Bangwen, DAI Zhian, YU Huitao, XING Ruiguang, ZI Luxiong. Fast Removal of Aqueous Mn(Ⅱ) Using Partially Reduced Graphene Oxide-Fe3O4† [J]. Chem. J. Chinese Universities, 2017, 38(2): 217. |
| [14] | LI Yingying, ZHANG Qi, ZHANG Yiheng, WANG Lei. Preparation of NF/rGO/Co3O4/NiO Electrode and Its Application in Supercapacitor† [J]. Chem. J. Chinese Universities, 2017, 38(11): 2031. |
| [15] | DU Shichao,REN Zhiyu,WU Jun,FU Honggang. Ni-Fe LDH/Reduced Graphene Oxide as Catalyst for Oxygen Evolution Reaction [J]. Chem. J. Chinese Universities, 2016, 37(8): 1415. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||