

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (12): 2289.doi: 10.7503/cjcu20170191
• Physical Chemistry • Previous Articles Next Articles
ZHONG Yubo1, KANG Chunli1, WANG Yueqi1, XU Xiaolei1, BAO Siqi1, XUE Honghai2, TIAN Tao1,*( )
)
Received:2017-03-29
Online:2017-12-10
Published:2017-11-13
Contact:
TIAN Tao
E-mail:tiantao@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
ZHONG Yubo, KANG Chunli, WANG Yueqi, XU Xiaolei, BAO Siqi, XUE Honghai, TIAN Tao. Influences of Freezing on the Reduction of Cr(Ⅵ) by Typical Components of Dissolved Organic Matter†[J]. Chem. J. Chinese Universities, 2017, 38(12): 2289.
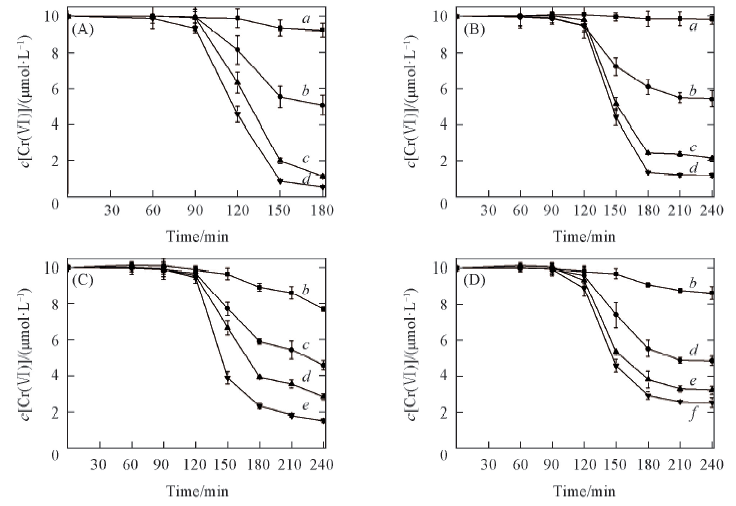
Fig.1 Effects of concentrations of the organic acids on the removal of Cr(Ⅵ) in ice c(Cr(Ⅵ)]=10 μmol/L. (A) Oxalic acid; (B) tartaric acid; (C) malic acid; (D) citric acid.Concentration of organic acid/(μmol·L-1): a. 10; b. 20; c. 30; d. 40; e. 50; f. 60.
| Acid | Concentration/(μmol·L-1) | First order kinetics equation | k/h-1 | Coefficient of determination, R2 |
|---|---|---|---|---|
| Oxalic acid | 10 | y=-0.0007x+0.0543 | 0.0007 | 0.8520 |
| 20 | y=-0.0065x+0.4814 | 0.0065 | 0.9109 | |
| 30 | y=-0.0200x+2.1408 | 0.0200 | 0.9039 | |
| 40 | y=-0.0273x+2.0323 | 0.0273 | 0.9107 | |
| Tartaric acid | 10 | y=-0.0002x+0.0333 | 0.0002 | 0.9000 |
| 20 | y=-0.0054x+0.5112 | 0.0054 | 0.9609 | |
| 30 | y=-0.0142x+1.4327 | 0.0142 | 0.9097 | |
| 40 | y=-0.0207x+2.0973 | 0.0207 | 0.9133 | |
| Malic acid | 20 | y=-0.0014x+0.1477 | 0.0014 | 0.9380 |
| 30 | y=-0.0056x+0.5608 | 0.0056 | 0.9406 | |
| 40 | y=-0.0098x+0.9895 | 0.0098 | 0.9276 | |
| 50 | y=-0.0159x+1.5606 | 0.0159 | 0.9372 | |
| Citric acid | 20 | y=-0.0011x+0.1103 | 0.0011 | 0.9145 |
| 40 | y=-0.0066x+0.6651 | 0.0066 | 0.9543 | |
| 50 | y=-0.0104x+1.0119 | 0.0104 | 0.9469 | |
| 60 | y=-0.0127x+1.2155 | 0.0127 | 0.9445 |
Table 1 Simulation of the first order kinetics of the reduction of Cr(Ⅵ) in ice
| Acid | Concentration/(μmol·L-1) | First order kinetics equation | k/h-1 | Coefficient of determination, R2 |
|---|---|---|---|---|
| Oxalic acid | 10 | y=-0.0007x+0.0543 | 0.0007 | 0.8520 |
| 20 | y=-0.0065x+0.4814 | 0.0065 | 0.9109 | |
| 30 | y=-0.0200x+2.1408 | 0.0200 | 0.9039 | |
| 40 | y=-0.0273x+2.0323 | 0.0273 | 0.9107 | |
| Tartaric acid | 10 | y=-0.0002x+0.0333 | 0.0002 | 0.9000 |
| 20 | y=-0.0054x+0.5112 | 0.0054 | 0.9609 | |
| 30 | y=-0.0142x+1.4327 | 0.0142 | 0.9097 | |
| 40 | y=-0.0207x+2.0973 | 0.0207 | 0.9133 | |
| Malic acid | 20 | y=-0.0014x+0.1477 | 0.0014 | 0.9380 |
| 30 | y=-0.0056x+0.5608 | 0.0056 | 0.9406 | |
| 40 | y=-0.0098x+0.9895 | 0.0098 | 0.9276 | |
| 50 | y=-0.0159x+1.5606 | 0.0159 | 0.9372 | |
| Citric acid | 20 | y=-0.0011x+0.1103 | 0.0011 | 0.9145 |
| 40 | y=-0.0066x+0.6651 | 0.0066 | 0.9543 | |
| 50 | y=-0.0104x+1.0119 | 0.0104 | 0.9469 | |
| 60 | y=-0.0127x+1.2155 | 0.0127 | 0.9445 |
| Concentration/ (μmol·L-1) | Initial pH value | Concentration/ (μmol·L-1) | Initial pH value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Oxalic acid | Tartaric acid | Malic acid | Citric acid | Oxalic acid | Tartaric acid | Malic acid | Citric acid | ||
| 10 | 5.19 | 5.27 | 40 | 4.60 | 4.64 | 4.75 | 4.68 | ||
| 20 | 4.98 | 4.98 | 5.04 | 5.02 | 50 | 4.60 | 4.46 | ||
| 30 | 4.86 | 4.86 | 4.95 | 60 | 4.39 | ||||
Table 2 Initial pH values of Cr(Ⅵ)-organic acid system under different concentrations of the organic acids
| Concentration/ (μmol·L-1) | Initial pH value | Concentration/ (μmol·L-1) | Initial pH value | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Oxalic acid | Tartaric acid | Malic acid | Citric acid | Oxalic acid | Tartaric acid | Malic acid | Citric acid | ||
| 10 | 5.19 | 5.27 | 40 | 4.60 | 4.64 | 4.75 | 4.68 | ||
| 20 | 4.98 | 4.98 | 5.04 | 5.02 | 50 | 4.60 | 4.46 | ||
| 30 | 4.86 | 4.86 | 4.95 | 60 | 4.39 | ||||
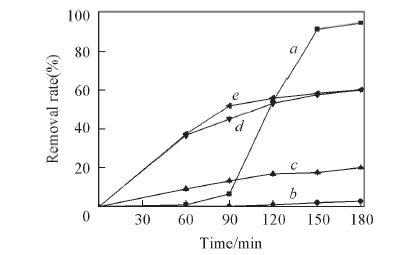
Fig.4 Removal of Cr(Ⅵ) by oxalic acid in watera. c[Cr(Ⅵ)]=10 μmol/L, c(oxalic acid)=40 μmol/L(ice); b. c[Cr(Ⅵ)]=2 mmol/L, c(oxalic acid)=8 mmol/L(water); c. c[Cr(Ⅵ)]=6 mmol/L, c(oxalic acid)=24 mmol/L(water); d. c(Cr(Ⅵ)]=12 mmol/L, c(oxalic acid)=48 mmol/L(water); e. c[Cr(Ⅵ)]=20 mmol/L, c(oxalic acid)=80 mmol/L(water).
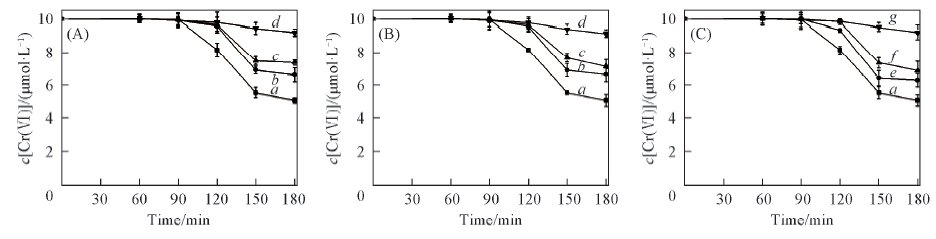
Fig.5 Effects of the concentrations of inorganic salts on the removal of Cr(Ⅵ) in ice c[Cr(Ⅵ)]=10 μmol/L, c(oxalic acid)=20 μmol/L. (A) NaNO3; (B) NaCl; (C) Na2SO4.Concentration of inorganic salt/(μmol·L-1): a. 0; b. 15; c. 150; d. 1.5; e. 10; f. 100; g. 1.
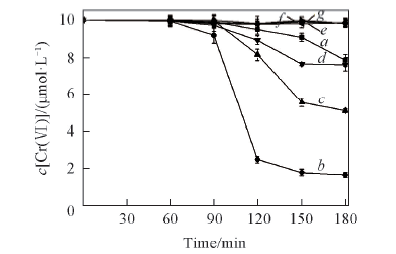
Fig.6 Effects of initial pH values on the removal of Cr(Ⅵ) in Cr(Ⅵ)-oxalic acid system in ice c[Cr(Ⅵ)]=10 μmol/L, c(oxalic acid)=20 μmol/L; pH: a. 1.58; b. 3.58; c. 4.98; d. 5.52; e. 6.55; f. 7.56; g. 8.53.
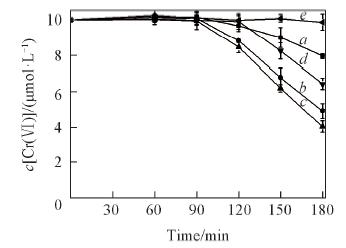
Fig.7 Effects of initial pH values on the removal of Cr(Ⅵ) in Cr(Ⅵ)-H2SO4 system in ice c[Cr(Ⅵ)]=10 μmol/L; pH: a. 1.53; b. 3.58; c. 4.62; d. 5.20; e. 6.55.
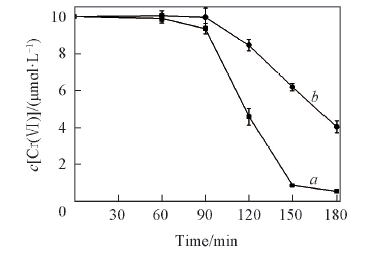
Fig.8 Removal of Cr(Ⅵ) in Cr(Ⅵ)-oxalic acid system and Cr(Ⅵ)-H2SO4 system at the same initial pH value in icec[Cr(Ⅵ)]=10 μmol/L; a. oxalic acid, pH=4.60; b. H2SO4, pH=4.62.
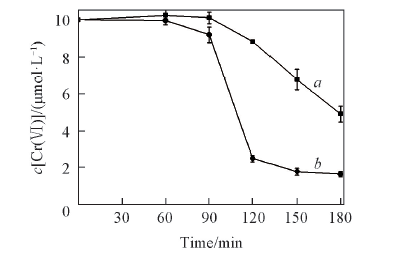
Fig.9 Removal of Cr(Ⅵ) in Cr(Ⅵ)-H2SO4 system and Cr(Ⅵ)-oxalic acid-H2SO4 system at the same initial pH value(pH=3.58) in icec[Cr(Ⅵ)]=10 μmol/L, c(oxalic acid)=20 μmol/L; a. H2SO4; b. oxalic acid+ H2SO4.
| [1] | Bower J. P., Anastasio C., J. Phys. Chem. A, 2013, 117(30), 6612—6621 |
| [2] | Ge L. K., Li J., Na G. S., Chen C. E., Huo C., Zhang P., Yao Z. W., Chemosphere,2016, 155, 375—379 |
| [3] | Fede A., Grannas A. M., Environ. Sci. Technol., 2015, 49(21), 12808—12815 |
| [4] | Bower J. P., Anastasio C., Environ. Sci.: Processes and Impacts,2014, 16(4), 748—756 |
| [5] | Tang X. J., Wang Y. X., Kang C. L., Liu H. F., Chen B. Y., Qiu S. L., Chem. J. Chinese Universities, 2015, 36(9), 1719—1723 |
| (唐晓剑, 王依雪, 康春莉, 刘汉飞, 陈柏言, 裘式纶. 高等学校化学学报, 2015, 36(9), 1719—1723) | |
| [6] | Kahan T. F., Donaldson D. J., Environ. Sci. Technol., 2010, 44(10), 3819—3824 |
| [7] | Takenaka N., Furuya S., Sato K., Bandow H., Maeda Y., Furukawa Y., Inte. J. Chem. Kinet., 2003, 35(5), 198—205 |
| [8] | Bartels-Rausch T., Jacobi H. P., Kuhs W. F., Kuo M. H., Maus S., Moussa S. G., McNeill V. F., Newberg J. T., Pettersson J. B. C., Roeselova, M., Sodeau J. R., Atmos. Chem. Phys., 2014, 14, 1587—1633 |
| [9] | Xue H. H., Tang X. J., Kang C. L., Liu J., Shi L., Wang H. L., Yang T., Water Sci. Technol., 2013, 68(11), 2479—2484 |
| [10] | Bao S. Q., Kang C. L., Zhong Y. B., Zhou L., Yao Z. F., Huang D. M., Wang Y. H., Tian T., Chem. J. Chinese Universities, 2016, 37(12), 2253—2259 |
| (包思琪, 康春莉, 钟宇博, 周林, 姚志富, 黄冬梅, 王宇寒, 田涛. 高等学校化学学报, 2016, 37(12), 2253—2259) | |
| [11] | Adorno W. T., Rezzadori K., Arend G. D., Chaves V. C., Reginatto F. H., Luccio M. D., Petrus J. C. C., Int. J. Food Sci. Technol., 2017, 52, 781—787 |
| [12] | Heger D., Jirkovsky J., Klán P., J. Phys. Chem. A, 2005, 109(30), 6702—6709 |
| [13] | Abida O., Osthoff H. D., Geophys. Res. Lett., 2011, 38, L16808—L16812 |
| [14] | Zhang F., Wang B., Wang J. N., Li X. Y., Li C. J., Chem. J. Chinese Universities, 2016, 37(11), 2117—2124 |
| (张凡, 汪滨, 王娇娜, 李秀艳, 李从举. 高等学校化学学报, 2016, 37(11), 2117—2124) | |
| [15] | Deng B., Stone A., Environ. Sci. Technol., 1996, 30, 2484—2494 |
| [16] | Kim K., Chung H.Y., Ju J. J., Kim J. W.,Science of Total Environment, 2017, 590, 107—113 |
| [17] | Wang Y. H., Chen W. Y., Gu H. B., China Leather, 2005, 34(3), 30—32 |
| (王应红, 陈武勇, 辜海彬. 中国皮革, 2005, 34(3), 30—32) | |
| [18] | Kim K., Choi W., Environ. Sci. Technol., 2011, 45(6), 2202—2208 |
| [19] | Dong X. L., Ma L. Q., Gress J., Harris W., Li Y. C., J. Hazard. Mater., 2014, 267, 62—70 |
| [20] | Grannas A. M., Bausch A. R., Mahanna K. M., J. Phys. Chem. A, 2007, 111(43), 11043—11049 |
| [21] | Kim K., Kim J., Bokare A. D., Choi W., Yoon H., Kim J., Environ. Sci. Technol., 2015, 49(18), 10937—10944 |
| [1] | CHENG Qian, YANG Bolong, WU Wenyi, XIANG Zhonghua. S-doped Fe-N-C as Catalysts for Highly Reactive Oxygen Reduction Reactions [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220341. |
| [2] | CHU Yuyi, LAN Chang, LUO Ergui, LIU Changpeng, GE Junjie, XING Wei. Single-atom Cerium Sites Designed for Durable Oxygen Reduction Reaction Catalyst with Weak Fenton Effect [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220294. |
| [3] | WU Yu, LI Xuan, YANG Hengpan, HE Chuanxin. Construction of Cobalt Single Atoms via Double-confinement Strategy for High-performance Electrocatalytic Reduction of Carbon Dioxide [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220343. |
| [4] | WANG Xintian, LI Pan, CAO Yue, HONG Wenhao, GENG Zhongxuan, AN Zhiyang, WANG Haoyu, WANG Hua, SUN Bin, ZHU Wenlei, ZHOU Yang. Techno-economic Analysis and Industrial Application Prospects of Single-atom Materials in CO2 Catalysis [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220347. |
| [5] | XIA Wu, REN Yingyi, LIU Jing, WANG Feng. Chitosan Encapsulated CdSe QDs Assemblies for Visible Light-induced CO2 Reduction in an Aqueous Solution [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220192. |
| [6] | SONG Dewen, WANG Mingwang, WANG Yani, JIAO Zhenmei, NING Hui, WU Mingbo. Progress of CO2 Electroreduction to Oxalic Acid [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220248. |
| [7] | ZHAO Runyao, JI Guipeng, LIU Zhimin. Efficient Electrocatalytic CO2 Reduction over Pyrrole Nitrogen-coordinated Single-atom Copper Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220272. |
| [8] | WANG Ruhan, JIA Shunhan, WU Limin, SUN Xiaofu, HAN Buxing. CO2-involved Electrochemical C—N Coupling into Value-added Chemicals [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220395. |
| [9] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [10] | WANG Lijun, LI Xin, HONG Song, ZHAN Xinyu, WANG Di, HAO Leiduan, SUN Zhenyu. Efficient Electrocatalytic CO2 Reduction to CO by Tuning CdO-Carbon Black Interface [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220317. |
| [11] | YANG Dan, LIU Xu, DAI Yihu, ZHU Yan, YANG Yanhui. Research Progress in Electrocatalytic CO2 Reduction Reaction over Gold Clusters [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220198. |
| [12] | SONG Yingying, HUANG Lin, LI Qingsen, CHEN Limiao. Preparation of CuO/BiVO4 Photocatalyst and Research on Carbon Dioxide Reduction [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220126. |
| [13] | CHANG Yunfei, LIAO Mingyi, WEN Jiaming. Reduction Performance and Mechanism of Liquid Terminated-carboxyl Fluoroelastomers Using NaBH4/MCl x Reduction System [J]. Chem. J. Chinese Universities, 2022, 43(6): 20210835. |
| [14] | TAO Yu, OU Honghui, LEI Yongpeng, XIONG Yu. Research Progress of Single-atom Catalysts in Photocatalytic Reduction of Carbon Dioxide [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220143. |
| [15] | GU Yu, XI Baojuan, LI Jiangxiao, XIONG Shenglin. Structure Regulation of Single-atom Catalysts in Oxygen Reduction Reactions [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220036. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||