

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (2): 316.doi: 10.7503/cjcu20140802
• Physical Chemistry • Previous Articles Next Articles
WANG Jiachen1, TONG Minman1, SHAN Chao2, XIAO Gang3,*, LIU Dahuan1, YANG Qingyuan1,*( ), ZHONG Chongli1
), ZHONG Chongli1
Received:2014-09-04
Online:2015-02-10
Published:2015-01-20
Contact:
XIAO Gang,YANG Qingyuan
E-mail:qyyang@mail.buct.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Jiachen, TONG Minman, SHAN Chao, XIAO Gang, LIU Dahuan, YANG Qingyuan, ZHONG Chongli. Molecular Simulation of Effects of Impurities on Flue Gas Separation in Metal-Organic Frameworks†[J]. Chem. J. Chinese Universities, 2015, 36(2): 316.
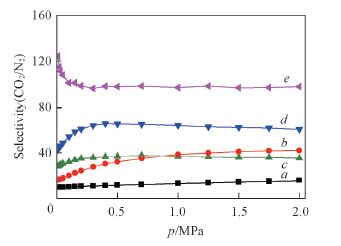
Fig.1 Simulated selectivity of CO2 over N2 in the CO2/N2 binary mixture at 303 K a. ZIF-8; b. NOTT-300; c. UiO-66(Zr); d. UiO-66-NH2; e. UiO-66-2COOH.
| Material | |||||
|---|---|---|---|---|---|
| CO2 | N2 | H2O | O2 | SO2 | |
| ZIF-8 | -19.04 | -11.10 | -11.47 | -12.98 | -25.91 |
| NOTT-300 | -24.43 | -14.50 | -27.90 | -14.84 | -34.66 |
| UiO-66(Zr) | -25.93 | -14.95 | -42.15 | -16.03 | -34.21 |
| UiO-66-NH2 | -28.86 | -15.77 | -46.05 | -16.72 | -37.13 |
| UiO-66-2COOH | -37.95 | -19.97 | -57.80 | -19.80 | -50.62 |
Table 1 Isosteric heat of adsorption of CO2, N2, H2O, O2 and SO2 at infinite dilution in ZIF-8,NOTT-300, UiO-66(Zr), UiO-66-NH2 and UiO-66-2COOH at 303 K
| Material | |||||
|---|---|---|---|---|---|
| CO2 | N2 | H2O | O2 | SO2 | |
| ZIF-8 | -19.04 | -11.10 | -11.47 | -12.98 | -25.91 |
| NOTT-300 | -24.43 | -14.50 | -27.90 | -14.84 | -34.66 |
| UiO-66(Zr) | -25.93 | -14.95 | -42.15 | -16.03 | -34.21 |
| UiO-66-NH2 | -28.86 | -15.77 | -46.05 | -16.72 | -37.13 |
| UiO-66-2COOH | -37.95 | -19.97 | -57.80 | -19.80 | -50.62 |
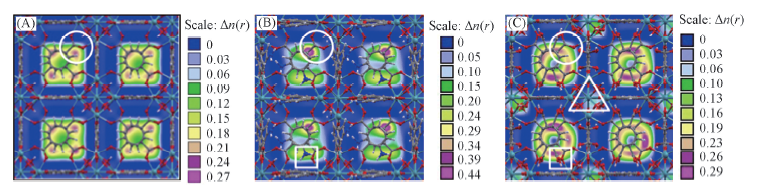
Fig.2 Contour plots of the COM probability density distribution of CO2 in their mixture adsorbed in three MOFs at 0.1 MPa (A) UiO-66(Zr); (B) UiO-66-NH2; (C) UiO-66-2COOH. The bulk composition is V(CO2)∶V(N2)=15∶85. The framework of the material is represented by a ball-stick style(Zr, dark cyan; C, gray; O, red; N, blue; H, white).
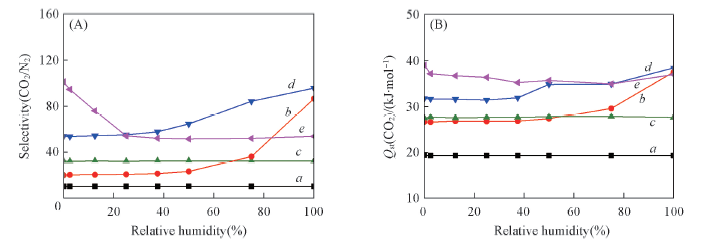
Fig.3 Simulated selectivity for CO2 over N2(A) and calculated adsorption enthalpies of CO2(B) in the CO2/N2/H2O mixture at 303 K and 0.1 MPa as a function of the relative humidity a. ZIF-8; b. NOTT-300; c. UiO-66(Zr); d. UiO-66-NH2; e. UiO-66-2COOH.
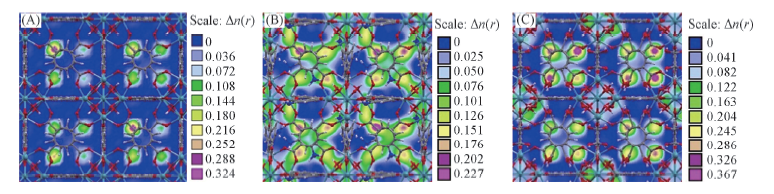
Fig.4 Contour plots of the COM probability density distribution of H2O in their mixture adsorbed in three MOFs at 0.1 MPa (A) UiO-66(Zr); (B) UiO-66-NH2; (C) UiO-66-2COOH. The bulk composition n(CO2)∶n(N2)∶n(H2O)=15∶81∶4. The framework of the material is represented by a ball-stick style(Zr, dark cyan; C, gray; O, red; N, blue; H, white).
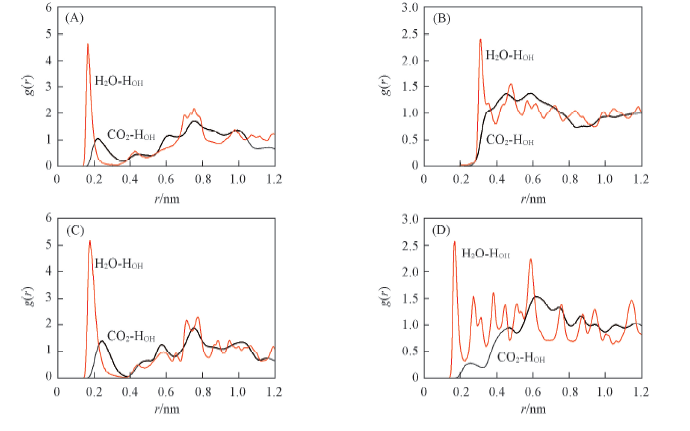
Fig.5 Radial distribution functions of CO2 and H2O molecules around the functionalized groups in two materials UiO-66-NH2(A, B) and UiO-66-2COOH(C, D) at 303 K n(CO2)∶n(N2)∶n(H2O)=15∶81∶4. (A) —OH; (B) —NH2; (C) —OH; (D) —COOH.
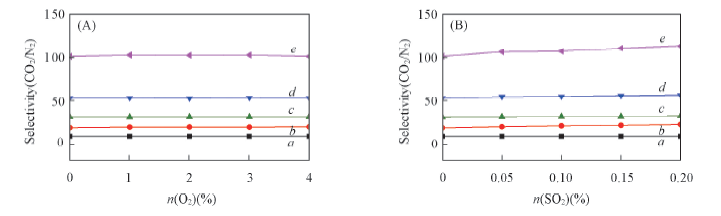
Fig.6 Simulated selectivity for CO2 over N2 in the different mixture at 0.1 MPa and 303 K in five MOFs (A) Effect of O2 concentration; (B) effect of SO2 concentration. a. ZIF-8; b. NOTT-300; c. UiO-66(Zr); d. UiO-66-NH2; e. UiO-66-2COOH.
| [1] | Karl T. R., Trenberth K. E., Science, 2003, 302, 1719—1723 |
| [2] | Wu G., Wei D., Zhou Z. D., Tang M. F., Fu X., Acta Ecol. Sinica, 2014, 34, 2812—2820 |
| (吴钢, 魏东, 周政达, 唐明方, 付晓. 生态学报, 2014, 34, 2812—2820) | |
| [3] | D’Alessandro D. M., Smit B., Long J. R., Angew. Chem. Int. Ed., 2010, 49, 6058—6082 |
| [4] | Wells B. A., Chaffee A. L., Adsorption, 2011, 17, 255—264 |
| [5] | Li J. R., Sculley J., Zhou H. C., Chem. Rev., 2012, 112, 869—932 |
| [6] | Banerjee R., Furukawa H., Britt D., Knobler C., O’Keeffe M., Yaghi O. M., J. Am. Chem. Soc., 2009, 131, 3875—3877 |
| [7] | Li B., Zhang Z., Li Y., Yao K., Zhu Y., Deng Z., Yang F., Zhou X., Li G., Wu H., Nijem N., Chabal Y. J., Lai Z., Han Y., Shi Z., Feng S., Li J., Angew. Chem. Int. Ed., 2012, 51, 1412—1415 |
| [8] | Liu J., Tian J., Thallapally P. K., McGrail B. P., J. Phys. Chem. C, 2012, 116, 9575—9581 |
| [9] | Lu W., Yuan D., Makal T. A., Li J. R., Zhou H. C., Angew. Chem. Int. Ed., 2012, 51, 1580—1584 |
| [10] | Lu Z., Xing H., Sun R., Bai J., Zheng B., Li Y., Cryst. Growth Des., 2012, 12, 1081—1084 |
| [11] | Nugent P., Belmabkhout Y., Burd S. D., Cairns A. J., Luebke R., Forrest K., Pham T., Ma S., Space B., Wojtas L., Eddaoudi M., Zaworotko M. J., Nature, 2013, 495, 80—84 |
| [12] | Xiang S., He Y., Zhang Z., Wu H., Zhou W., Krishna R., Chen B., Nat. Commun., 2012, 3, 954 |
| [13] | Yang Q. Y., Liu D. H., Zhong C. L., CIESC J., 2009, 60, 805—819 |
| (阳庆元, 刘大欢, 仲崇立. 化工学报, 2009, 60, 805—819) | |
| [14] | Ma Q. T., Xiao Y. L., Tong M. M., Li Z. J., Yang Q. Y., Liu D. H., Zhong C. L., Scientia Sinica Chimica, 2014, 44, 812—821 |
| (麻沁甜, 肖远龙, 童敏曼, 李正杰, 阳庆元, 刘大欢, 仲崇立. 中国科学: 化学, 2014, 44, 812—821) | |
| [15] | Haldoupis E., Nair S., Sholl D. S., J. Am. Chem. Soc., 2012, 134, 4313—4323 |
| [16] | Krishna R., van Baten J. M., Phys. Chem. Chem. Phys., 2011, 13, 10593—10616 |
| [17] | Liu J., Keskin S., Sholl D. S., Johnson J. K., J. Phys. Chem. C, 2011, 115, 12560—12566 |
| [18] | Wilmer C. E., Farha O. K., Bae Y. S., Hupp J. T., Snurr R. Q., Energy Environ. Sci., 2012, 5, 9849—9856 |
| [19] | Wu D., Yang Q., Zhong C., Liu D., Huang H., Zhang W., Maurin G., Langmuir, 2012, 28, 12094—12099 |
| [20] | Drage T. C., Snape C. E., Stevens L. A., Wood J., Wang J., Cooper A. I., Dawson R., Guo X., Satterley C., Irons R., J. Mater. Chem., 2012, 22, 2815—2823 |
| [21] | Lee K. B., Sircar S., AIChE J., 2008, 54, 2293—2302 |
| [22] | Sumida K., Rogow D. L., Mason J. A., McDonald T. M., Bloch E. D., Herm Z. R., Bae T., Long J. R., Chem. Rev., 2012, 112, 724—781 |
| [23] | Demessence A., D’Alessandro D. M., Foo M. L., Long J. R., J. Am. Chem. Soc., 2009, 131, 8784—8786 |
| [24] | Ding L., Yazaydin A. O., J. Phys. Chem. C, 2012, 116, 22987—22991 |
| [25] | Thallapally P. K., Motkuri R. K., Fernandez C. A., McGrail B. P., Behrooz G. S., Inorg. Chem., 2010, 49, 4909—4915 |
| [26] | Yang S., Sun J., Ramirez-Cuesta A. J., Callear S. K., David W. I. F., Anderson D. P., Newby R., Blake A. J., Parker J. E., Tang C. C., Schröder M., Nature Chem., 2012, 4, 887—894 |
| [27] | Yu J., Ma Y., Balbuena P. B., Langmuir, 2012, 28, 8064—8071 |
| [28] | Yu K., Kiesling K., Schmidt J. R., J. Phys. Chem. C, 2012, 116, 20480—20488 |
| [29] | Garibay S. J., Cohen S. M., Chem. Commun., 2010, 46, 7700—7702 |
| [30] | Hu Z., Chen Y., Jiang J., J. Chem. Phys., 2011, 134, 134705 |
| [31] | Wiersum A. D., Soubeyrand-Lenoir E., Yang Q., Moulin B., Guillerm V., Ben Yahia M., Bourrelly S., Vimont A., Miller S., Vagner C., Daturi M., Clet G., Serre C., Maurin G., Llewellyn P. L., Chem. Asian J., 2011, 6, 3270—3280 |
| [32] | Yang Q., Vaesen S., Ragon F., Wiersum A. D., Wu D., Lago A., Devic T., Martineau C., Taulelle F., Llewellyn P. L., Jobic H., Zhong C., Serre C., De Weireld G., Maurin G., Angew. Chem. Int. Ed., 2013, 52, 10316—10320 |
| [33] | Yang Q., Wiersum A. D., Llewellyn P. L., Guillerm V., Serre C., Maurin G., Chem. Commun., 2011, 47, 9603—9605 |
| [34] | Harris J. G., Yung K. H., J. Phys. Chem., 1995, 99, 12021—12024 |
| [35] | Potoff J. J., Siepmann J. I., AIChE J., 2001, 47, 1676—1682 |
| [36] | Zhang L., Siepmann J. I., Theor. Chem. Acc., 2006, 115, 391—397 |
| [37] | Sokolic' F., Guissani Y., Guillot B., Mol. Phys., 1985, 56, 239—253 |
| [38] | Rick S. W., J. Chem. Phys., 2004, 120, 6085—6093 |
| [39] | Rappé A. K., Casewit C. J., Colwell K. S., Goddard III W. A., Skiff W. M., J. Am. Chem. Soc., 1992, 114, 10024—10035 |
| [40] | Babarao R., Jiang J., J. Am. Chem. Soc., 2009, 131, 11417—11425 |
| [41] | Huang H., Zhang W., Liu D., Zhong C., Ind. Eng. Chem. Res., 2012, 51, 10031—10038 |
| [42] | Liu B., Smit B., J. Phys. Chem. C, 2010, 114, 8515—8522 |
| [43] | Peng X., Cao D., AIChE J., 2013, 59, 2928—2942 |
| [44] | Mayo S. L., Olafson B. D., Goddard III W. A., J. Phys. Chem., 1990, 94, 8897—8909 |
| [45] | Cmarik G. E., Kim M., Cohen S. M., Walton K. S., Langmuir, 2012, 28, 15606—15613 |
| [46] | Do D. D., Do H. D., J. Phys. Chem. B, 2006, 110, 17531—17538 |
| [47] | Vlugt T. J. H., García-Pérez E., Dubbeldam D., Ban S., Calero S., J. Chem. Theory Comput., 2008, 4, 1107—1118 |
| [48] | Yang Q., Liu D., Zhong C., Li J., Chem. Rev., 2013, 113, 8261—8323 |
| [49] | Zhong C.L., Liu D. H., Yang Q. Y., Structure-Property Relationship of Metal-Organic Frameworks and Their Design, Science Press, Beijing, 2013, 35—3 |
| 7(仲崇立, 刘大欢, 阳庆元. 金属-有机骨架材料的构效关系及设计, 北京: 科学出版社, 2013, 35—37) | |
| [50] | Zhou Z. E., Xue C. Y., Yang Q. Y., Zhong C. L., Acta Chim. Sinica, 2009, 67, 477—482 |
| (周子娥, 薛春瑜, 阳庆元, 仲崇立. 化学学报, 2009, 67, 477—482) | |
| [51] | Bae Y.S., Farha O. K., Hupp J. T., Snurr R. Q., J. Mater. Chem., 2009, 19, 2131—2134 |
| [52] | Yazaydin A. O., Benin A. I., Faheem S. A., Jakubczak P., Low J. J., Willis R. R., Snurr R. Q., Chem. Mater., 2009, 21, 1425—1430 |
| [53] | Babarao R., Jiang Y., Medhekar N. V., J. Phys. Chem. C, 2013, 117, 26976—26987 |
| [54] | Ding L., Yazaydin A. O., Phys. Chem. Chem. Phys., 2013, 15, 11856—11861 |
| [55] | Martín-Calvo A., García-Pérez E., García-Sánchez A., Bueno-Pérez R., Hamad S., Calero S., Phys. Chem. Chem. Phys., 2011, 13, 11165—11174 |
| [56] | Liu Y., Liu J., Lin Y. S., Chang M., J. Phys. Chem. C, 2014, 118, 6744—6751 |
| [57] | Mahoney M. W., Jorgensen W. L., J. Chem. Phys., 2000, 112, 8910—8922 |
| [58] | Khatri R. A., Chuang S. S. C., Soong Y., Gray M., Energy Fuels, 2006, 20, 1514—1520 |
| (Ed.: Y, Z ) |
| [1] | ZHAO Yingzhe, ZHANG Jianling. Applications of Metal-organic Framework-based Material in Carbon Dioxide Photocatalytic Conversion [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220223. |
| [2] | SONG Yingying, HUANG Lin, LI Qingsen, CHEN Limiao. Preparation of CuO/BiVO4 Photocatalyst and Research on Carbon Dioxide Reduction [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220126. |
| [3] | LU Cong, LI Zhenhua, LIU Jinlu, HUA Jia, LI Guanghua, SHI Zhan, FENG Shouhua. Synthesis, Structure and Fluorescence Detection Properties of a New Lanthanide Metal-Organic Framework Material [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220037. |
| [4] | TIAN Xueqin, MO Zheng, DING Xin, WU Pengyan, WANG Yu, WANG Jian. A Squaramide-containing Luminescent Metal-organic Framework as a High Selective Sensor for Histidine [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210589. |
| [5] | WANG Shoubai, WU Xiuming, SHU Chen, ZHONG Min, HUANG Wei, YAN Deyue. Gas Separation Performance of Polyimide Homogeneous MembranesContaining tert-Butyl Groups [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220357. |
| [6] | XING Peiqi, LU Tong, LI Guanghua, WANG Liyan. Controllable Syntheses of Two Cd(II) Metal-organic Frameworks Possessing Related Structures [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220218. |
| [7] | LI Shurong, WANG Lin, CHEN Yuzhen, JIANG Hailong. Research Progress of Metal⁃organic Frameworks on Liquid Phase Catalytic Chemical Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210575. |
| [8] | ZHANG Chi, SUN Fuxing, ZHU Guangshan. Synthesis, N2 Adsorption and Mixed-matrix Membrane Performance of Bimetal Isostructural CAU-21 [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210578. |
| [9] | HAN Zongsu, YU Xiaoyong, MIN Hui, SHI Wei, CHENG Peng. A Rare Earth Metal-Organic Framework with H6TTAB Ligand [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210342. |
| [10] | SHI Xiaofan, ZHU Jian, BAI Tianyu, FU Zixuan, ZHANG Jijie, BU Xianhe. Research Status and Progress of MOFs with Application in Photoelectrochemical Water-splitting [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210613. |
| [11] | WU Ji, ZHANG Hao, LUO Yuhui, GENG Wuyue, LAN Yaqian. A Microporous Cationic Ga(III)-MOF with Fluorescence Properties for Selective sensing Fe3+ Ion and Nitroaromatic Compounds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210617. |
| [12] | LI Wen, QIAO Junyi, LIU Xinyao, LIU Yunling. Zirconium-based Metal-Organic Framework with Naphthalene for Fluorescent Detection of Nitroaromatic Explosives in Water [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210654. |
| [13] | WANG Jie, HUO Haiyan, WANG Yang, ZHANG Zhong, LIU Shuxia. General Strategy for In situ Synthesis of NENU-n Series Polyoxometalate-based MOFs on Copper Foil [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210557. |
| [14] | MO Zongwen, ZHANG Xuewen, ZHOU Haolong, ZHOU Dongdong, ZHANG Jiepeng. Guest-responses of A Porous Coordination Polymer Based on Synergistic Hydrogen Bonds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210576. |
| [15] | LIU Xueguang, YANG Xiaoshan, MA Jingjing, LIU Weisheng. Separating Methyl Blue Selectively from the Mixture of Dyes by Europium Metal-organic Frameworks [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210715. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||