

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (7): 1369.doi: 10.7503/cjcu20140198
• Articles: Inorganic Chemistry • Previous Articles Next Articles
HUANG Song1, DING Songdong1,*( ), JIN Yongdong1, MA Lijian1, XIA Chuanqin1, LI Shoujian1, WU Yuxuan1,2, HUANG Huang1,2
), JIN Yongdong1, MA Lijian1, XIA Chuanqin1, LI Shoujian1, WU Yuxuan1,2, HUANG Huang1,2
Received:2014-03-11
Online:2014-07-10
Published:2014-05-19
Contact:
DING Songdong
E-mail:dsd68@163.com
Supported by:CLC Number:
TrendMD:
HUANG Song, DING Songdong, JIN Yongdong, MA Lijian, XIA Chuanqin, LI Shoujian, WU Yuxuan, HUANG Huang. Extraction of Eu(Ⅲ) and Am(Ⅲ) from Nitric Acid Solution with Bisdiglycolamides†[J]. Chem. J. Chinese Universities, 2014, 35(7): 1369.
| Extractant | DAm | DEu | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Kerosene | Toluene | n-Octanol | Chloroform | Kerosene | Toluene | n-Octanol | Chloroform | ||
| TOE-BisDGA | 1100b | 91 | 8.2 | 0.30 | 3459b | 406 | 37 | 1.1 | |
| TOX-BisDGA | 411b | 98 | 16 | 0.22 | 592b | 273 | 112 | 1.4 | |
Table 1 Effect of diluents on the distribution ratios(D) of Eu(Ⅲ) and Am(Ⅲ)a
| Extractant | DAm | DEu | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Kerosene | Toluene | n-Octanol | Chloroform | Kerosene | Toluene | n-Octanol | Chloroform | ||
| TOE-BisDGA | 1100b | 91 | 8.2 | 0.30 | 3459b | 406 | 37 | 1.1 | |
| TOX-BisDGA | 411b | 98 | 16 | 0.22 | 592b | 273 | 112 | 1.4 | |
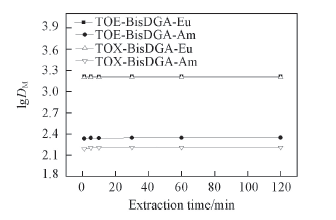
Fig.1 Effect of extraction time on the distribution ratios of Eu(Ⅲ) and Am(Ⅲ) Organic phase: 0.1 mol/L TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10); aqueous phase: 3.0 mol/L HNO3; 1.3 mmol/L Eu(Ⅲ) or tracer amount of Am(Ⅲ).
| cini(HNO3)/(mol·L-1) | TOX-BisDGA | TOE-BisDGA | ||||
|---|---|---|---|---|---|---|
| [HNO3]org/(mol·L-1) | DH | [HNO3]org/(mol·L-1) | DH | |||
| 0.5 | 1.37×10-2 | 2.8×10-2 | 2.06×10-2 | 4.3×10-2 | ||
| 1.0 | 3.66×10-2 | 3.8×10-2 | 4.58×10-2 | 4.8×10-2 | ||
| 2.0 | 9.16×10-2 | 4.8×10-2 | 1.05×10-1 | 5.5×10-2 | ||
| 3.0 | 1.60×10-1 | 5.6×10-2 | 1.69×10-1 | 6.0×10-2 | ||
| 4.0 | 2.29×10-1 | 6.0×10-2 | 2.34×10-1 | 6.2×10-2 | ||
Table 2 Extraction of nitric acid by 0.1 mol/L TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol (volume ratio 90∶10) at different initial nitric acid concentration
| cini(HNO3)/(mol·L-1) | TOX-BisDGA | TOE-BisDGA | ||||
|---|---|---|---|---|---|---|
| [HNO3]org/(mol·L-1) | DH | [HNO3]org/(mol·L-1) | DH | |||
| 0.5 | 1.37×10-2 | 2.8×10-2 | 2.06×10-2 | 4.3×10-2 | ||
| 1.0 | 3.66×10-2 | 3.8×10-2 | 4.58×10-2 | 4.8×10-2 | ||
| 2.0 | 9.16×10-2 | 4.8×10-2 | 1.05×10-1 | 5.5×10-2 | ||
| 3.0 | 1.60×10-1 | 5.6×10-2 | 1.69×10-1 | 6.0×10-2 | ||
| 4.0 | 2.29×10-1 | 6.0×10-2 | 2.34×10-1 | 6.2×10-2 | ||
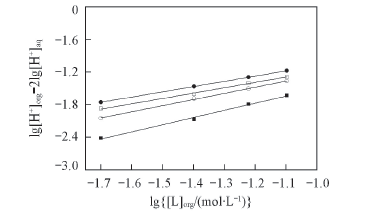
Fig.2 Extraction of nitric acid by TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10) Aqueous phase: 0.5 or 1.0 mol/L HNO3. ■ 1.0 mol/L HNO3+TOX-BisDGA, slope=1.3, intercept=-0.19; ○ 1.0 mol/L HNO3+TOE-BisDGA, slope=1.1, intercept=-0.11; □ 0.5 mol/L HNO3+TOX-BisDGA, slope=1.0, intercept=-0.20; ● 0.5 mol/L HNO3+TOE-BisDGA, slope=1.0, intercept=-0.12.
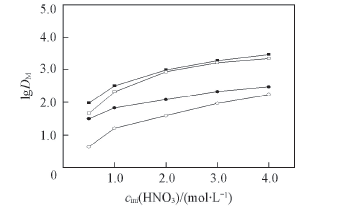
Fig.3 Effect of initial nitric acid concentration on distribution ratios of Eu(Ⅲ) and Am(Ⅲ) Organic phase: 0.1 mol/L TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10); aqueous phase: 3.0 mol/L HNO3; 1.3 mmol/L Eu(Ⅲ) or tracer amount of Am(Ⅲ). ■ TOE-BisDGA-Eu; □ TOX-BisDGA-Eu; ● TOE-BisDGA-Am; ○ TOX-BisDGA-Am.
| cini(HNO3)/(mol·L-1) | SFEu/Am | cini(HNO3)/(mol·L-1) | SFEu/Am | ||
|---|---|---|---|---|---|
| TOE-BisDGA | TOX-BisDGA | TOE-BisDGA | TOX-BisDGA | ||
| 0.5 | 3.1 | 10 | 3.0 | 9.0 | 17 |
| 1.0 | 4.7 | 13 | 4.0 | 9.8 | 12 |
| 2.0 | 8.2 | 22 | |||
Table 3 Effect of initial nitric acid concentration on the separation factor SFEu/Am
| cini(HNO3)/(mol·L-1) | SFEu/Am | cini(HNO3)/(mol·L-1) | SFEu/Am | ||
|---|---|---|---|---|---|
| TOE-BisDGA | TOX-BisDGA | TOE-BisDGA | TOX-BisDGA | ||
| 0.5 | 3.1 | 10 | 3.0 | 9.0 | 17 |
| 1.0 | 4.7 | 13 | 4.0 | 9.8 | 12 |
| 2.0 | 8.2 | 22 | |||
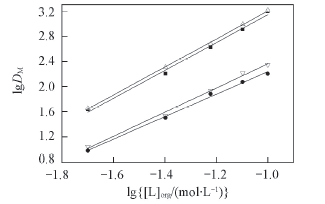
Fig.4 Effect of extractant concentration on the distribution ratios of Eu(Ⅲ) and Am(Ⅲ) Organic phase:TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10); aqueous phase: 3.0 mol/L HNO3; 1.3 mmol/L Eu(Ⅲ) or tracer amount of Am(Ⅲ). △ TOE-BisDGA-Eu, slope=1.8; ■ TOX-BisDGA-Eu, slope=1.9; ▽ TOE-BisDGA-Am, slope=2.2; ● TOX-BisDGA-Am, slope=2.2.
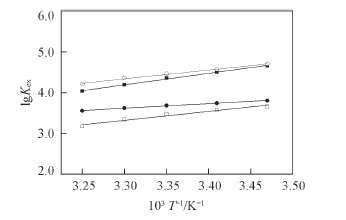
Fig.5 Effect of temperature on the apparent extraction equilibrium constant lgKex Organic phase: 0.1 mol/L TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10); Aqueous phase: 3.0 mol/L HNO3; 1.3 mmol/L Eu(Ⅲ) or tracer amount of Am(Ⅲ). ○ TOE-BisDGA-Eu, slope=2.16, intercept=-2.78; ■ TOX-BisDGA-Eu, slope=2.81, intercept=-5.08; ● TOE-BisDGA-Am, slope=1.12, intercept=-0.09; □ TOX-BisDGA-Am, slope=2.18, inter-cept=-3.88.
| Extraction system | ΔG/ (kJ·mol-1) | ΔH/ (kJ·mol-1) | ΔS/ (J·mol-1·K-1) |
|---|---|---|---|
| TOE-BisDGA-Eu | -26.8 | -43.1 | -54.6 |
| TOE-BisDGA-Am | -21.6 | -22.8 | -4.0 |
| TOX-BisDGA-Eu | -26.6 | -56.3 | -99.6 |
| TOX-BisDGA-Am | -20.3 | -42.7 | -75.1 |
Table 4 Thermodynamic parameters for the extraction of Eu(Ⅲ) and Am(Ⅲ) at 25 ℃*
| Extraction system | ΔG/ (kJ·mol-1) | ΔH/ (kJ·mol-1) | ΔS/ (J·mol-1·K-1) |
|---|---|---|---|
| TOE-BisDGA-Eu | -26.8 | -43.1 | -54.6 |
| TOE-BisDGA-Am | -21.6 | -22.8 | -4.0 |
| TOX-BisDGA-Eu | -26.6 | -56.3 | -99.6 |
| TOX-BisDGA-Am | -20.3 | -42.7 | -75.1 |
| Strip stage | M | TOE-BisDGA | TOX-BisDGA | |||
|---|---|---|---|---|---|---|
| Deionized water | 0.01 mol/L HNO3 | Deionized water | 0.01 mol/L HNO3 | |||
| 1 | Eu | 7 | 6 | 17 | 19 | |
| Am | 14 | 15 | 39 | 36 | ||
| 2 | Eu | 72 | 63 | 94 | 93 | |
| Am | 33 | 42 | 88 | 62 | ||
| 3 | Eu | 80 | 78 | 97 | 99 | |
| Am | 51 | 77 | 91 | 82 | ||
| 4 | Eu | 87 | 91 | 99 | 100 | |
| Am | 84 | 87 | 93 | 89 | ||
| 5 | Eu | 94 | 96 | 100 | ||
| Am | 92 | 93 | 97 | 98 | ||
| 6 | Eu | 99 | 99 | |||
| Am | 99 | 99 | 99 | 99 | ||
Table 5 Percentage of strippingextraction of Eu(Ⅲ) and Am(Ⅲ) from the loaded organic phase of 0.1 mol/L TOE-BisDGA or TOX-BisDGA in kerosene/n-octanol(volume ratio 90∶10) with deionized water or 0.01 mol/L HNO3 as the stripping agents
| Strip stage | M | TOE-BisDGA | TOX-BisDGA | |||
|---|---|---|---|---|---|---|
| Deionized water | 0.01 mol/L HNO3 | Deionized water | 0.01 mol/L HNO3 | |||
| 1 | Eu | 7 | 6 | 17 | 19 | |
| Am | 14 | 15 | 39 | 36 | ||
| 2 | Eu | 72 | 63 | 94 | 93 | |
| Am | 33 | 42 | 88 | 62 | ||
| 3 | Eu | 80 | 78 | 97 | 99 | |
| Am | 51 | 77 | 91 | 82 | ||
| 4 | Eu | 87 | 91 | 99 | 100 | |
| Am | 84 | 87 | 93 | 89 | ||
| 5 | Eu | 94 | 96 | 100 | ||
| Am | 92 | 93 | 97 | 98 | ||
| 6 | Eu | 99 | 99 | |||
| Am | 99 | 99 | 99 | 99 | ||
| Compound | c/(mol·L-1) | λmax/nm | εmax/(L·mol-1·cm-1) |
|---|---|---|---|
| TOX-BisDGA-Eu | 1.0×10-5 | 206 | 9.0×104 |
| TOX-BisDGA | 1.0×10-5 | 205 | 2.8×104 |
| TOE-BisDGA-Eu | 1.0×10-5 | 206 | 4.6×104 |
| TOE-BisDGA | 1.0×10-5 | 206 | 1.7×104 |
Table 6 Relevant data of UV spectra of free ligands and their Eu(Ⅲ) complexes
| Compound | c/(mol·L-1) | λmax/nm | εmax/(L·mol-1·cm-1) |
|---|---|---|---|
| TOX-BisDGA-Eu | 1.0×10-5 | 206 | 9.0×104 |
| TOX-BisDGA | 1.0×10-5 | 205 | 2.8×104 |
| TOE-BisDGA-Eu | 1.0×10-5 | 206 | 4.6×104 |
| TOE-BisDGA | 1.0×10-5 | 206 | 1.7×104 |
| [1] | Law D., Brewer K. N., Herbst R. S., Todd T. A., Wood D. J., Waste Manage., 1999, 19(1), 27—37 |
| [2] | Schulz W. W., Bray L. A., Sep. Sci. Technol., 1987, 22(2/3), 191—214 |
| [3] | Mathur J. N., Murali M. S., Nash K. L., Solvent Extr. Ion Exch., 2001, 19(3), 357—390 |
| [4] | Ravi J., Venkatesan K. A., Antony M. P., Srinivasan T. G., Vasudeva R. P. R., J. Radioanal. Nucl. Chem., 2013, 295(2), 1283—1292 |
| [5] | Morita Y., Glatz J. P., Kubota M., Koch L., Pagliosa G., Römer K., Nicholl A., Solvent Extr. Ion Exch., 1996, 14(3), 385—400 |
| [6] | Horwitz E. P., Kalina D. G., Diamond H. D., Vandergrift G. F., Schultz W. W., Solvent Extr. Ion. Exch., 1985, 3(1/2), 75—109 |
| [7] | Schultz W. W., Horwitz E. P., Sep. Sci. Technol., 1988, 23(12/13), 1191—1210 |
| [8] | Courson O., Lebrun M., Malmbeck R., Pagliosa G., Römer K., Sätmark B., Glatz J. P., Radiochim. Acta, 2000, 88(12), 857—863 |
| [9] | Malmbeck R., Courson O., Pagliosa G., Römer K., Sätmark B., Glatz J. P., Baron P., Radiochim. Acta, 2000, 88(12), 865—871 |
| [10] | Chen W. J., Zhu L., Ding S. D., Liu Z. Q., Chen S. J., Jin Y. D., Chem. J. Chinese Universities, 1998, 19(11),1724—1726(陈文浚, 祝霖, 丁颂东, 刘忠群, 陈绍金, 金永东. 高等学校化学学报, 1998, 19(11),1724—1726, 1724—1726) |
| [11] | Zhang P., Chen J., Tian G. X., The Method for Stripping and Separating Radioactive Elements from Organic Phase by Water-soluble Oxa-amide Compounds, ZL 03146394.0, 2004-02-25(张平, 陈靖, 田国兴. 用水溶性氧杂酰胺从有机相反萃分离放射性元素的方法, ZL 03146394.0, 2004-02-25) |
| [12] | Sasaki Y., Sugo Y., Suzuki S., Tachimori S., Solvent Extr. Ion Exch., 2001, 19(1), 91—103 |
| [13] | Sasaki Y., Tachimori S., Solvent Extr. Ion Exch., 2002, 20(1), 21—34 |
| [14] | Tachimori S., Sasaki Y., Suzuki S., Solvent Extr. Ion Exch., 2002, 20(6), 687—699 |
| [15] | Mowafy E. A., Aly H. F., Solvent Extr. Ion Exch., 2007, 25(2), 205—224 |
| [16] | Gujar R. B., Ansari S. A., Prabhu D. R., Raut D. R., Solvent Extr. Ion Exch., 2010, 28(6), 764—777 |
| [17] | Ansari S. A., Pathak P. N., Manchanda V. K., Hussain M., Prasad A. K., Parmar V. S., Solvent Extr. Ion Exch., 2005, 23(4), 350—366 |
| [18] | Gujar R. B., Ansari S. A., Mohapatra P. K., Manchanda V. K., Solvent Extr. Ion Exch., 2010, 28(3), 350—366 |
| [19] | Dam H. H., Reinhoudt D. N., Verboom W., Chem. Soc. Rev., 2007, 36(2) , 367—377 |
| [20] | Iqbala M., Mohapatrab P. K., Ansarib S. A., Huskensa J., Verbooma W., Tetrahedron, 2012, 68(38), 7840—7847 |
| [21] | Matloka K., Gelis A., Regalbuto M., Vandegrift G., Scott M.J.,Dalton Trans., 2005, 3719—3721 |
| [22] | Mohapatra P. K., Iqbal M., Raut D. R., Huskens J., Verboom W., J. Membr. Sci., 2012, 411412, 64—72 |
| [23] | Mohapatra P. K., Sengupta A., Iqbal M., Huskens J., Verboom W., Inorg. Chem., 2013, 52(5), 2533—2541 |
| [24] | Sharma J. N., Ruhela R., Manohar S., Wattal P. K., Suri A. K., Dialkyldiaza-tetraalkyloctane Diamide Derivatives Useful for The Separation of Trivalent Actinides from Lanthanides and Process for the Preparation Thereof, US 0172626A1,2012-07-05 |
| [25] | Mowafy E. A., Aly H. F., Solvent Extr. Ion Exch., 2007, 25(2), 205—224 |
| [26] | Zhang P., Wang H. R., Wang J. C., Chinese J. Org. Chem., 2008, 28(2),335—338(张平, 王海荣, 王建晨. 有机化学, 2008, 28(2), 335—338) |
| [27] | Wu Y. X., Ding S. D., Liu N., Huang S., Huang H., Su D. P., Chem. J. Chinese Universities, 2014, 35(2),257—261(吴宇轩, 丁颂东, 刘宁, 黄松, 黄璜, 苏冬萍. 高等学校化学学报, 2014, 35(2), 257—261) |
| [28] | Djedovic N., Ferdani R., Harder E., Pajewska J., Pajewski R., Weber M. E., Schlesinger P. H., Gokel G. W., New J. Chem., 2005, 29(2), 291—305 |
| [29] | Sasaki Y., Choppin G. R., Anal. Sci., 1996, 12(2), 225—230 |
| [30] | Nave S., Modolo G., Madic C., Testard F., Solvent Extr. Ion Exch., 2004, 22(4), 527—551 |
| [31] | Cui Y., Sun G. X., Bao M., Zhang Z. W., Sun S. X., Chinese J. Inorg. Chem., 2003, 19(11), 1175—1178(崔玉, 孙国新, 鲍猛, 张振伟, 孙思修. 无机化学学报, 2003, 19(11), 1175—1178) |
| [32] | Geist A., Solvent Extr. Ion Exch., 2010, 28(5), 596—607 |
| [33] | Horwitz E. P., Muscatello A. C., Kalina D. G., Kaplan L., Sep. Sci. Technol., 1981, 16(4), 417—437 |
| [34] | Ansari S. A., Pathak P. N., Husain M., Prasad A. K., Parmar V. S., Manchanda V. K., Radiochim. Acta, 2006, 94(6/7), 307—312 |
| [35] | Lever A. B. P., Mantovani E., Ramaswamy B. S., Can. J. Chem., 1971, 49(11), 1957—1964 |
| [36] | Curtis N. F., Curtis Y. M., Inorg. Chem., 1965, 4(6), 804—809 |
| [37] | Nakamoto K., Infrared and Raman Spectra of Inorganic and Coordination Compounds, 4th, John Wiley and Sons Ltd., New York, 1978, 221—227 |
| [38] | Sun T., Synthesis, Characterization and Photoelectric Study of Functional Polynuclear Carbazole Carboxylate Complexes, Anhui University, Hefei, 2007 |
| (孙涛. 功能性多核咔唑羧酸配合物的合成及光电性质研究, 合肥: 安徽大学, 2007) | |
| (Ed.: F, K, M |
| [1] | TANG Quanjun, LIU Yingxin, MENG Rongwei, ZHANG Ruotian, LING Guowei, ZHANG Chen. Application of Single-atom Catalysis in Marine Energy [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220324. |
| [2] | SONG Yingying, HUANG Lin, LI Qingsen, CHEN Limiao. Preparation of CuO/BiVO4 Photocatalyst and Research on Carbon Dioxide Reduction [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220126. |
| [3] | BI Gening, XIAO Xiaohua, LI Gongke. Development and Validation of Multiple Physical Fields Coupling Model for Microwave-assisted Extraction [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210739. |
| [4] | SHI Ge, XU Qian, DAI Xiao, ZHANG Jie, SHEN Jun, WAN Xinhua. Effect of Aromatic Substituent on Chiral Recognition of Helical Polyacetylene-based Chiral Stationary Phases for High-Performance Liquid Chromatography [J]. Chem. J. Chinese Universities, 2021, 42(8): 2673. |
| [5] | WANG Gaobo, MA Jing. Binding Selectivity Between Diazobenzene and Different Nucleophilic Reagents: Covalent and Noncovalent Interactions [J]. Chem. J. Chinese Universities, 2021, 42(7): 2238. |
| [6] | TENG Yunyang, QU Zexing, ZHOU Zhongjun, HUANG Xuri. Theoretical Study on Photoinduced Stepwise Dearomatization of Benzenoid Arenes with Different States [J]. Chem. J. Chinese Universities, 2021, 42(3): 752. |
| [7] | ZHENG Haijiao, JIANG Liyan, JIA Qiong. Arginine Functionalized Magnetic Nanoparticles and Its Application in Phosphopeptides Enrichment [J]. Chem. J. Chinese Universities, 2021, 42(3): 717. |
| [8] | ZHAO Shufang, HUANG Jun. Study by Solid-state NMR Spectroscopy on the Acidity and Shape-selectivity of Zeolites [J]. Chem. J. Chinese Universities, 2021, 42(1): 165. |
| [9] | DENG Jiewei, YANG Yunyun, LIN Li, LUAN Tiangang. Rapid Classification of Daphniamagna and Daphnia pulex by Surface-coated Probe Nanoelectrospray Ionization Mass Spectrometry Lipidomics [J]. Chem. J. Chinese Universities, 2020, 41(9): 2011. |
| [10] | LI Xiaoqian, ZHANG Hua, LU Haijian, LIU Chang, LIU Qinglong, MA Xiayu, FANG Yuanping, LIANG Dapeng. Mechanism of Photocatalytic Degradation of Rhodamine B by TiO2 Nanowire Array with Internal Extraction Electrospray Ionization Mass Spectrometry [J]. Chem. J. Chinese Universities, 2020, 41(9): 2003. |
| [11] | WANG Xiaoru,ZHANG Na,XING Jun. Preparation and Application of Melamine Imprinted Material Using Itaconic Acid as Multidentate Functional Monomer [J]. Chem. J. Chinese Universities, 2020, 41(7): 1521. |
| [12] | ZHANG Xiaofei, WU Lie, LI Shanshan, ZHU Manyu, CHENG Xiaowei, JIANG Xiu’e. Effect of Phase Behavior of Phospholipids on Lipid Membrane Damage Induced by Graphene Oxide † [J]. Chem. J. Chinese Universities, 2020, 41(4): 661. |
| [13] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [14] | WANG Tianqi,YU Qiongwei,FENG Yuqi. Analysis of Imidazole Propionic Acid in Serum of Patients with Type 2 Diabetes Based on NiO@SiO2 Solid-phase Extraction Coupled with Liquid Chromatography-Mass Spectrometry † [J]. Chem. J. Chinese Universities, 2020, 41(2): 262. |
| [15] | ZHENG Youwei, TIAN Fei, ZHANG Qian, XU Di, YANG Guohai, QU Lulu. Detection of Nitroreductase in Living Cells Based on Surface Enhanced Raman Scattering Nanoprobes [J]. Chem. J. Chinese Universities, 2020, 41(12): 2742. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||