

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (6): 1128.doi: 10.7503/cjcu20190006
收稿日期:2019-01-04
出版日期:2019-06-10
发布日期:2019-05-11
作者简介:联系人简介: 黄池宝, 男, 博士, 教授, 主要从事双光子荧光探针方面的研究. E-mail:
基金资助:
HUANG Chibao( ), PAN Qi, CHEN Huashi, LIANG Xing, LÜ Guoling
), PAN Qi, CHEN Huashi, LIANG Xing, LÜ Guoling
Received:2019-01-04
Online:2019-06-10
Published:2019-05-11
Supported by:摘要:
构建了一个衍生于双氰基二苯代乙烯的具有推-拉电子结构的环境敏感探针(SP), 该探针可作为将溶剂生色与分子转子特性结合起来的一个典型范例. SP探针的最大发射波长随溶剂极性的增加而显著增大, 归根于激发态分子转化为一个或多个扭转分子内电荷迁移(TICT)态, 极性溶剂更有利于电荷分离态分子的稳定. 在TICT态, 分子的荧光量子产率强烈地依赖于溶剂的极性、 黏度与温度. SP探针显示了很宽的溶剂生色范围, 在环己烷和二甲亚砜中的最大发射波长(λem)分别为445和641 nm, 相差196 nm, 能用于溶剂极性、 溶剂种类、 黏度与温度的检测识别. SP探针在环己烷和N,N-二甲基甲酰胺中的双光子吸收截面分别为5560和130 GM, 远高于同类双光子荧光探针; 其超大的斯托克斯位移(232 nm)可显著降低吸收谱对荧光的干扰, 显示出优良的检测成像性能, 可用于细胞黏度实时动态显微成像.
中图分类号:
TrendMD:
黄池宝, 潘淇, 陈华仕, 梁兴, 吕国岭. 双氰基二苯代乙烯型双光子荧光环境敏感探针. 高等学校化学学报, 2019, 40(6): 1128.
HUANG Chibao,PAN Qi,CHEN Huashi,LIANG Xing,LÜ Guoling. Dicyanostilbene-derived Two-photon Fluorescence Environment-sensitive Probe†. Chem. J. Chinese Universities, 2019, 40(6): 1128.
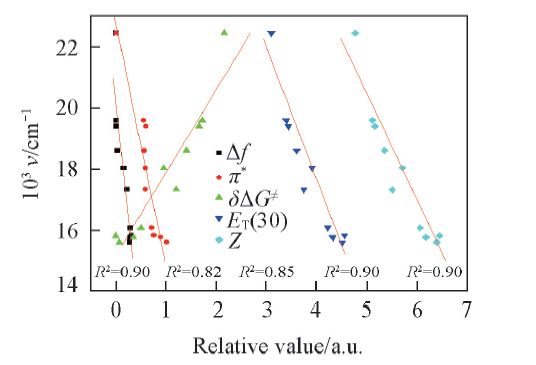
Fig.1 Correlations between the emission maxima of SP in various solvents and selected solvent parameters such as Δf, π*, δΔG≠, ET(30) and Z c(SP)=1 μmol/L, λex(OP)=410 nm.
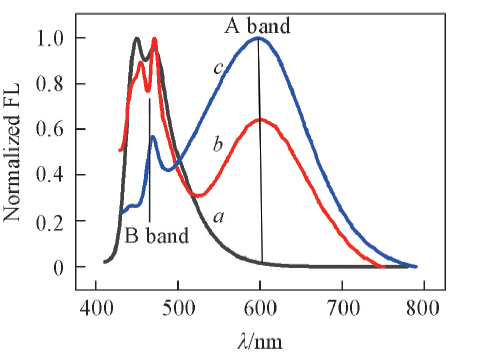
Fig.4 Normalized emission spectra of SP in ethanol at different temperatures c(SP)=3.6 μmol/L, λex(OP)=410 nm, a. 77 K; b. 77—294 K; c. 294 K. No control of the temperature was available when samples at 77 K were warmed up. The exact temperature of spectrum B was unknown, visual inspection showed that was mostly a solution.
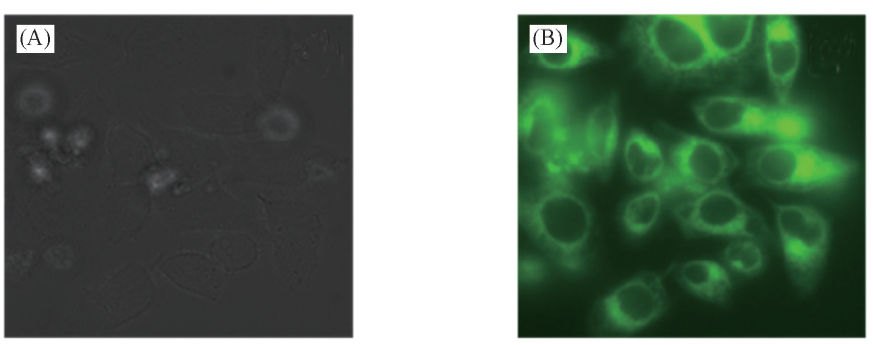
Fig.7 Bright-field(A) and two-photon microscopy(TPM, B) image 1 μmol/L SP-labeled mouse fibroblast two-photon fluorescent images were collected at 550—650 nm, λex=790 nm, replicate experiments 5 times.
| [1] | Luby-Phelps K., Intern. Rev. Cytol., 2000, 192(4), 189-221 |
| [2] | Rohrbach D.H., Timpl R., Molecular and Cellular Aspects of Basement Membranes, Academic Press, San Diego, 1993, 1-448 |
| [3] | Yeagle P.L., The Membranes of Cells, Academic Press, San Diego, 1993, 1-286 |
| [4] | Ohnishi S.T., Ohnishi T., Membrane Abnormalities in Sickle Cell Disease and in Other Red Blood Cell Disorders, CRC Press, Boca Raton, 1994, 1-356 |
| [5] | Reinhart W. H., Biorheology, 2001, 38(2/3), 203-212 |
| [6] | Moriarty P. M., Gibson C. A., Cardiovasc. Rev. Rep.,2003, 24, 321-325 |
| [7] | Uchimura I., Numano F., Diabetes Frontier,1997, 8(1), 33-37 |
| [8] | Simon A., Gariepy J., Chironi G., Megnien J. L., Levenson J., J. Hypertens.,2002, 20(2), 159-169 |
| [9] | Oppenheimer N., Diamant H., Biophys.J.,2009, 96(8), 3041-3049 |
| [10] | Owen D. M., Williamson D., Rentero C., Gaus K., Traffic,2009, 10(8), 962-971 |
| [11] | Frick M., Schmidt K., Nichols B. J., Curr. Biol.,2007, 17(5/6), 462-467 |
| [12] | Luneva O. G., Brazhe N. A., Maksimova N. V., Rodnenkov O. V., Parsina E. Y., Bryzgalova N. Y., Maksimov G. V., Rubin A. B., Orlov S. N., Chazov E. I., Pathophysiology,2007, 14(1), 41-46 |
| [13] | Goodwin J. S., Drake K. R., Remment C. L., Kenworthy A. K., Biophys.J.,2005, 89(2), 1398-1410 |
| [14] | Dibner M. D., Ireland K. A., Koerner L. A., Dexter D. L., Cancer Res.,1985, 45(10), 4998-5003 |
| [15] | Aleardi A. M., Benard G., Augereau O., Malgat M., Talbot J. C., Mazat J. P., Letellier T., Dachary-Prigent J., Solaini G. C., Rossignol R., J. Bioenerg. Biomembr.,2005, 37(4), 207-225 |
| [16] | Hou X., Richardson S. J., Aguilar M. I., Small D. H., Biochemistry,2005, 44(34), 11618-11627 |
| [17] | Ahn J. H., Kim T. Y., Kim Y. J., Han M. W., Yoon T. H., Chung J. W., Diabet.Med.,2006, 23(12), 1339-1343 |
| [18] | Salazar V. B. Y., Salazar V. M. A., Venzor V. C., Negrete A. C., Cabrales P., Diaz J. S., Intaglietta M., Clin. Hemorheol. Microcirc.,2008, 38(2), 67-74 |
| [19] | Kearney-Schwartz A., Virion J. M., Stoltz J. F., Drouin P., Zannad F., Fund. Clin.Pharmacol.,2007, 21, 387-396 |
| [20] | Velcheva I., Antonova N., Dimitrova V., Dimitrov N., Ivanov I., Clin. Hemorheol.Microcirc.,2006, 35(1/2), 155-157 |
| [21] | Bosman G. J. C. G. M.,Bartholomeus I. G. P., de Grip W. J., Gerontology,1991, 37, 95-112 |
| [22] | International Committee for Standardization in Haematology , J. Clin.Pathol., 1984, 37(10), 1147-1152 |
| [23] | Wang S., Boss A. H., Kensey K. R., Rosenson R. S., Clin. Chim.Acta,2003, 332(1/2), 79-82 |
| [24] | Haidekker M. A., Theodorakis E. A., Org. Biomol. Chem.,2007, 5, 1669-1678 |
| [25] | Demchenko P., Mely Y., Duportail G., Klymchenko A. S., Biophys.J.,2009, 96(9), 3461-3470 |
| [26] | Grabowski Z. R., Rotkiewicz K., Rettig W., Chem.Rev.,2003, 103(10), 3899-4031 |
| [27] | Zhong Q. L., Chen Y., Wang Y. H., Chi X. C., Wang Y., Ni M. C., Zhang H. Z., Chem. Res. Chinese Universities,2017, 33(3), 400-405 |
| [28] | Loutfy R. O., Pure Appl. Chem., 1986, 58(9), 1239-1248 |
| [29] | Levitt J. A., Kuimova M. K., Yahioglu G., Chung P. H., Suhling K., Phillips D., J. Phys. Chem. C,2009, 113(27), 11634-11642 |
| [30] | Bohne C., Ihmels H., Waidelich M., Yihwa C., J. Am. Chem.Soc.,2005, 127(49), 17158-17159 |
| [31] | Uchiyama S., Takehira K., Yoshihara T., Tobita S., Ohwada T., Organic Letters,2006, 8(25), 5869-5872 |
| [32] | Sutharsan J., Lichlyter D., Wright N. E., Dakanali M., Haidekker M. A., Theodorakis E. A., Tetrahedron,2010, 66, 2582-2588 |
| [33] | Xu G. P., Tang Y. H., Lin W. Y., Chem. Res. Chinese Universities,2018, 34(4), 523-527 |
| [34] | Li H., Pang M. L., Guo X. F., Meng J. B., Chem. Res. Chinese Universities,2015, 31(3), 357-361 |
| [35] | Droumaguet C. L., Mongin O., Werts M. H. V., Blanchard-Desce M., Chem.Commun.,2005, 41(22), 2802-2804 |
| [36] | Strehmel B., Sarker A. M., Detert H., ChemPhysChem,2003, 4, 249-259 |
| [37] | Kim H. M., Jeong B. H., Hyon J. Y., An M. J., Seo M. S., Hong J. H., Lee K. J., Kim C. H., Joo T., Hong S. C., Cho B. R., J. Am. Chem.Soc.,2008, 130(13), 4246-4247 |
| [38] | Kim H. M., Choo H. J., Jung S. Y., Ko Y. G., Park W. H., Jeon S. J., Kim C. H., Joo T., Cho B. R., ChemBioChem,2007, 8(5), 553-559 |
| [39] | Huang C. B., Qu J., Qi J., Yan M., Xu G., Org.Lett.,2011, 13(6), 1462-1465 |
| [40] | Huang C. B., Chen H. S., Zeng B. P., Chen X. Y., Chinese Journal of Analytical Chemistry,2015, 43(4), 507-511 |
| (黄池宝,陈华仕,曾伯平,陈晓远. 分析化学, 2015, 43(4), 507-511) | |
| [41] | Huang C. B., Pan Q., Chen X. Y., Zhao G. L., Chen H. S., Liang X., Lv G. L., Chem. J. Chinese Universities,2017, 38(10),1751—1756 |
| (黄池宝,潘淇,陈晓远,赵光练,陈华仕,梁兴,吕国岭. 高等学校化学学报, 2017, 38(10), 1751-1756) | |
| [42] | Huang C. B., Ding C., Analytica Chimica Acta,2011, 699(2), 198-205 |
| [43] | Huang H., He Q., Lin H., Bai F., Sun Z., Li Q., Polym. Adv.Technol.,2004, 15(1/2), 84-88 |
| [44] | Huang C., Fan J., Peng X., Lin Z., Guo B., Ren A., Cui J., Sun S., J. Photochem. Photobio. A: Chem.,2008, 199(2/3), 144-149 |
| [45] | BakerⅢ T. N., Doherty Jr. W. P., Kelley W. S., Newmeyer W., Rogers Jr J. E., Spalding R. E., Walter R. I., J. Org.Chem.,1965, 30(11), 3714-3718 |
| [46] | Reichardt C., Chem.Rev., 1994, 94(8), 2319-2358 |
| [47] | Kbamlet M. J., Abboud J. L. M., Taft R. W., Prog. Phys. Org.Chem.,1981, 13, 485-630 |
| [48] | Lippert E. Z., Z. Naturforsch.A, 1955, 10(7), 541-545 |
| [49] | Mataga N., Kaifu Y., Koizumi M., Bull. Chem. Soc.Jpn.,1956, 29(4), 465-471 |
| [50] | Platt J. R., J. Chem.Phys., 1949, 17(5), 470-481 |
| [51] | Grabowski Z. R., Rotkiewicz K., Rubaszewska W., Kirlor-Kaminska E., Acta Phys. Pol.A,1978, A54, 767-772 |
| [52] | Huang C. B., Ren A. X., Li H. B., Yang N. F., Chem. J. Chinese Universities,2010, 31(11), 2222-2227 |
| (黄池宝,任安祥,李海渤, 阳年发. 高等学校化学学报, 2010, 31(11), 2222-2227) | |
| [53] | Huang C. B., Ren A. X., Acta Chimica Sinica,2007, 65(23),2765—2770 |
| (黄池宝,任安祥. 化学学报, 2007, 65(23), 2765-2770) |
| [1] | 张勇, 许俊, 鲍雨, 崔树勋. 非极性有机溶剂对分子内氢键弱化程度的单分子力谱定量研究[J]. 高等学校化学学报, 2022, 43(4): 20210863. |
| [2] | 刘苗, 刘瑞波, 刘巴蒂, 钱鹰. 溶酶体靶向吲哚氟硼二吡咯光敏剂的合成、 双光子荧光成像及光动力治疗[J]. 高等学校化学学报, 2022, 43(10): 20220326. |
| [3] | 李海勃, 肖长发, 江龙, 黄云, 淡宜. MCM-41分子筛负载氯化铝催化丙烯酸甲酯与1-辛烯共聚[J]. 高等学校化学学报, 2021, 42(9): 2974. |
| [4] | 赵宝东, 刘亚静, 潘永飞, 刘卫孝, 高福磊, 汪营磊. 含能增塑剂2, 2-偕二硝基丙基三氟丙酸酯的合成及性能[J]. 高等学校化学学报, 2021, 42(9): 2815. |
| [5] | 李安然, 赵冰, 阚伟, 宋天舒, 孔祥东, 卜凡强, 孙立, 殷广明, 王丽艳. 基于菲并咪唑的ON⁃OFF⁃ON双比色荧光探针及细胞成像[J]. 高等学校化学学报, 2021, 42(8): 2403. |
| [6] | 黄池宝, 康帅, 潘淇, 吕国岭. 衍生于咔唑的双氰基二苯代乙烯型双光子荧光脂筏探针[J]. 高等学校化学学报, 2021, 42(8): 2443. |
| [7] | 李梦硕, 张静, 刘丹, 朱亚先, 张勇. 芘与人血清白蛋白和牛血清白蛋白结合位点微环境极性的差异[J]. 高等学校化学学报, 2021, 42(3): 731. |
| [8] | 谌委菊, 陈诗雅, 薛曹叶, 刘波, 郑晶. 缺氧响应荧光探针的成像及治疗应用[J]. 高等学校化学学报, 2021, 42(11): 3433. |
| [9] | 赵宇辉, 李明乐, 龙飒然, 樊江莉, 彭孝军. 极性敏感的BDP分子溶剂化效应的光谱性质[J]. 高等学校化学学报, 2020, 41(9): 2018. |
| [10] | 尹少云, 张露尹, 王政, 潘梅. “眼镜”状碘化亚铜配合物的调光和双光子发光性质[J]. 高等学校化学学报, 2020, 41(4): 646. |
| [11] | 武文博,刘斌. 可双光子激发的聚集诱导发光光敏剂及其生物医学应用[J]. 高等学校化学学报, 2020, 41(2): 191. |
| [12] | 陈秋宏, 叶艳春, 任孟然, 王凯民, 唐怀军, 汪正良, 周强. 含有三苯胺-三唑双极性单元的橙红光阳离子型铱(Ⅲ)配合物的合成及在LEDs中的应用[J]. 高等学校化学学报, 2020, 41(12): 2717. |
| [13] | 沐亚新, 阿丽, 庄欠粉, 王勇, 倪永年. 超小荧光二硫化钨量子点的水热合成及细胞成像应用[J]. 高等学校化学学报, 2019, 40(4): 693. |
| [14] | 喻照川, 马文辉, 吴涛, 问婧, 张永, 王丽艳, 初红涛. B,N,S共掺杂石墨烯量子点的制备及对Fe 3+和H2P |
| [15] | 黄池宝, 潘淇, 陈华仕, 梁兴, 吕国岭. 基于三苯胺的双氰基二苯代乙烯型双光子荧光溶剂生色探针[J]. 高等学校化学学报, 2018, 39(8): 1676. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||