

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (5): 909.doi: 10.7503/cjcu20180828
李普, 陈英, 夏榕娇, 郭涛, 张敏, 仕春, 汤旭, 贺鸣, 薛伟( )
)
收稿日期:2018-12-10
出版日期:2019-04-12
发布日期:2019-04-12
作者简介:联系人简介: 薛 伟, 男, 博士, 教授, 博士生导师, 主要从事新农药创制方面的研究. E-mail:
基金资助:
LI Pu, CHEN Ying, XIA Rongjiao, GUO Tao, ZHANG Min, JIANG Shichun, ANG Xu, HE Ming, XUE Wei*( )
)
Received:2018-12-10
Online:2019-04-12
Published:2019-04-12
Contact:
XUE Wei
E-mail:wxue@gzu.edu.cn
Supported by:摘要:
利用活性拼接原理, 将喹喔啉引入到杨梅素结构中, 合成了一系列含喹喔啉基团的杨梅素新型衍生物. 采用浊度法测试了目标化合物的体外抑菌活性, 结果表明, 目标化合物对柑橘溃疡病菌(X. Citri)和水稻白叶枯病菌(X. Oryzae)均表现出较好的抑制活性. 目标化合物对柑橘溃疡病菌的抑制活性(EC50)均优于对照药叶枯唑和噻菌铜(EC50分别为54.85和61.13 μg/mL), 其中化合物4o抑制活性(EC50=11.17 μg/mL)最优; 目标化合物对水稻白叶枯病菌的抑制活性EC50均优于对照药叶枯唑和噻菌铜(EC50分别为148.20和175.47 μg/mL), 其中化合物4f抑制活性(EC50=34.49 μg/mL)最优. 采用半叶枯斑法测试了目标化合物的抗烟草花叶病毒(TMV)活性, 结果表明, 所有目标化合物在浓度为500 mg/L时均有一定的抑制作用.
中图分类号:
TrendMD:
李普, 陈英, 夏榕娇, 郭涛, 张敏, 仕春, 汤旭, 贺鸣, 薛伟. 含喹喔啉杨梅素衍生物的合成及生物活性. 高等学校化学学报, 2019, 40(5): 909.
LI Pu,CHEN Ying,XIA Rongjiao,GUO Tao,ZHANG Min,JIANG Shichun,ANG Xu,HE Ming,XUE Wei. Synthesis and Biological Activities of Myricetin Derivatives Containing Quinoxaline†. Chem. J. Chinese Universities, 2019, 40(5): 909.
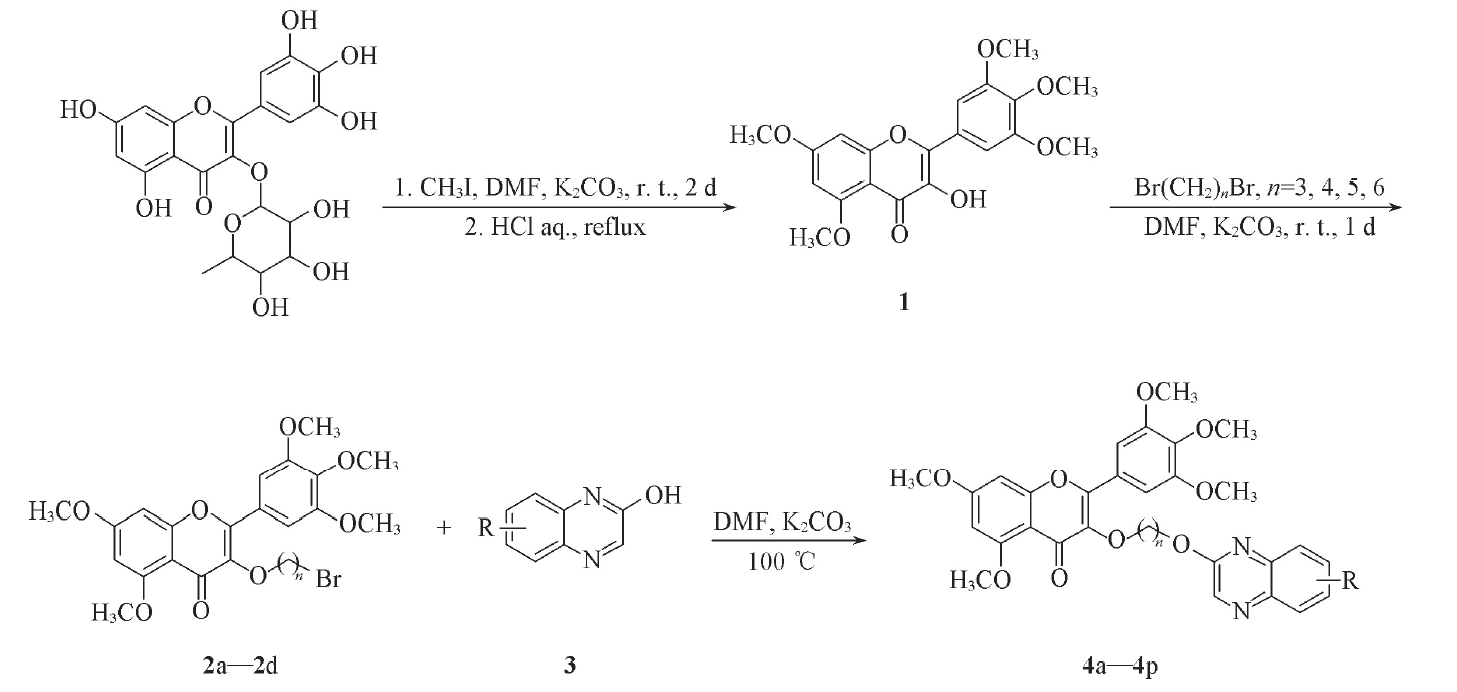
Scheme 1 Synthetic routes of target compounds 4a─4p2a: n=3; 2b: n=4; 2c: n=5; 2d: n=6; 4a: n=3, R=H; 4b: n=3, R=3-CH3; 4c: n=3, R=6-Cl; 4d: n=3, R=3-OH; 4e: n=4, R=H; 4f: n=4, R=3-CH3; 4g: n=4, R=6-Cl; 4h: n=4, R=3-OH; 4i: n=5, R=H; 4j: n=5, R=3-CH3; 4k: n=5, R=6-Cl; 4l: n=5, R=3-OH; 4m: n=6, R=H; 4n: n=6, R=3-CH3; 4o: n=6, R=6-Cl; 4p: n=6, R=3-OH
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(calcd. ), m/z[M+H]+ |
|---|---|---|---|---|
| 1 | Yellow solid | 48.8 | 228—230(227—229[ | |
| 2a | White solid | 83.4 | 138—140(141—142[ | |
| 2b | White solid | 71.2 | 152—153(154—156[ | |
| 2c | White solid | 65.5 | 149—151 | |
| 2d | White solid | 58.1 | 150—152 | |
| 4a | White solid | 45.3 | 79—81 | 575.20241(575.20215) |
| 4b | Yellow solid | 63.2 | 98—100 | 589.21806(589.21686) |
| 4c | Yellow solid | 48.7 | 164—166 | 609.16343(609.16229) |
| 4d | Yellow solid | 52.2 | 113—115 | 613.17927(613.17822) |
| 4e | Yellow solid | 47.6 | 58—60 | 589.21806(589.21747) |
| 4f | Yellow solid | 49.4 | 103—105 | 603.23371(603.23242) |
| 4g | Yellow solid | 57.3 | 124—126 | 623.17908(623.17780) |
| 4h | Yellow solid | 54.5 | 84—86 | 605.21297(605.21216) |
| 4i | Yellow solid | 41.7 | 152—154 | 603.23371(603.23236) |
| 4j | Yellow solid | 51.6 | 89—91 | 617.24936(617.24792) |
| 4k | Yellow solid | 60.3 | 131—133 | 659.17668(659.17493) |
| 4l | Yellow solid | 56.0 | 88—90 | 605.21297(605.21216) |
| 4m | White solid | 44.1 | 96—98 | 655.20524(655.20654) |
| 4n | White solid | 53.2 | 127—129 | 631.26501(631.26349) |
| 4o | Yellow solid | 57.4 | 83—85 | 651.21038(651.21038) |
| 4p | White solid | 48.6 | 76—78 | 633.24427(633.24274) |
Table 1 Appearance, yields, melting points and HRMS data of compounds 1, 2a—2d and 4a—4p
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(calcd. ), m/z[M+H]+ |
|---|---|---|---|---|
| 1 | Yellow solid | 48.8 | 228—230(227—229[ | |
| 2a | White solid | 83.4 | 138—140(141—142[ | |
| 2b | White solid | 71.2 | 152—153(154—156[ | |
| 2c | White solid | 65.5 | 149—151 | |
| 2d | White solid | 58.1 | 150—152 | |
| 4a | White solid | 45.3 | 79—81 | 575.20241(575.20215) |
| 4b | Yellow solid | 63.2 | 98—100 | 589.21806(589.21686) |
| 4c | Yellow solid | 48.7 | 164—166 | 609.16343(609.16229) |
| 4d | Yellow solid | 52.2 | 113—115 | 613.17927(613.17822) |
| 4e | Yellow solid | 47.6 | 58—60 | 589.21806(589.21747) |
| 4f | Yellow solid | 49.4 | 103—105 | 603.23371(603.23242) |
| 4g | Yellow solid | 57.3 | 124—126 | 623.17908(623.17780) |
| 4h | Yellow solid | 54.5 | 84—86 | 605.21297(605.21216) |
| 4i | Yellow solid | 41.7 | 152—154 | 603.23371(603.23236) |
| 4j | Yellow solid | 51.6 | 89—91 | 617.24936(617.24792) |
| 4k | Yellow solid | 60.3 | 131—133 | 659.17668(659.17493) |
| 4l | Yellow solid | 56.0 | 88—90 | 605.21297(605.21216) |
| 4m | White solid | 44.1 | 96—98 | 655.20524(655.20654) |
| 4n | White solid | 53.2 | 127—129 | 631.26501(631.26349) |
| 4o | Yellow solid | 57.4 | 83—85 | 651.21038(651.21038) |
| 4p | White solid | 48.6 | 76—78 | 633.24427(633.24274) |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
|---|---|---|
| 2c | 7.37(s, 2H), 6.81(s, 1H), 6.48(s, 1H), 3.94(t, J=5.6 Hz, 2H), 3.90(s, 3H), 3.88(s, 6H), 3.85(s, 3H), 3.77(s, 3H), 3.48(t, J=6.7 Hz, 2H), 1.89—1.72(m, 2H), 1.71—1.62(m, 2H), 1.51—1.40(m, 2H) | |
| 2d | 7.37(s, 2H), 6.84(s, 1H), 6.49(s, 1H), 3.95(t, J=6.0 Hz, 2H), 3.90(s, 3H), 3.87(s, 6H), 3.85(s, 3H), 3.76(s, 3H), 3.58(t, J=6.6 Hz, 2H), 1.67—1.64(m, 2H), 1.58—1.47(m, 2H), 1.43—1.35(m, 2H), 1.29—1.23(m, 2H) | |
| 4a | 8.53(s, 1H), 8.00(d, J=9.2 Hz, 1H), 7.82(d, J=8.6 Hz, 1H), 7.76(t, J=7.6 Hz, 1H), 7.64(t, J=6.8 Hz, 1H), 7.39(s, 1H), 7.38(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.54(t, J=6.6 Hz, 2H), 4.16(t, J=6.1 Hz, 2H), 3.90(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.28—2.07(m, 2H) | 172.6, 164.2, 160.7, 158.6, 157.4, 153.2, 152.2, 140.3, 140.2, 140.1, 139.9, 138.8, 130.9, 129.1, 127.3, 127.2, 126.0, 108.9, 106.2, 96.4, 93.6, 69.0, 63.9, 60.6, 56.6, 56.5, 29.6 |
| 4b | 7.88(d, J=8.2 Hz, 1H), 7.74(t, J=7.7 Hz, 1H), 7.65(d, J=6.8 Hz, 1H), 7.57(t, J=6.8 Hz, 1H), 7.39(s, 1H), 7.36(s, 1H), 6.83(s, 1H), 6.46(s, 1H), 4.53(t, J=6.4 Hz, 2H), 4.09(t, J=6.2 Hz, 2H), 3.91(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.43(s, 3H), 2.27—2.06(m, 2H) | 172.6, 164.2, 160.7, 158.6, 156.2, 154.6, 153.1, 152.1, 148.3, 140.4, 139.6, 138.4, 129.5, 128.2, 126.9, 126.0, 123.7, 108.9, 106.2, 96.3, 93.4, 69.9, 68.9, 63.7, 60.5, 56.4, 29.6, 20.4 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
| 4c | 8.57(s, 1H), 8.07(s, 1H), 7.81(d, J=12.0 Hz, 1H), 7.67(d, J=11.3 Hz, 1H), 7.38(s, 1H), 7.36(s, 1H), 6.81(s, 1H), 6.48(s, 1H), 4.53(t, J=6.5 Hz, 2H), 4.07(t, J=6.2 Hz, 2H), 3.89(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.24—2.00(m, 2H) | 172.6, 164.2, 160.8, 160.7, 158.6, 157.7, 154.4, 153.2, 153.1, 152.1, 141.5, 140.3, 140.2, 139.8, 139.1, 138.9, 134.1, 131.2, 129.1, 128.0, 126.0, 108.9, 106.2, 96.4, 93.5, 68.9, 64.2, 60.6, 60.6, 56.5, 29.5 |
| 4d | 7.69(d, J=10.0 Hz, 1H), 7.43(d, J=9.8 Hz, 1H), 7.40(s, 1H), 7.39(s, 1H), 7.25(t, J=6.6 Hz, 1H), 7.06(t, J=7.1 Hz, 1H), 6.86(s, 1H), 6.50(s, 1H), 4.27(t, J=6.8 Hz, 2H), 4.09(t, J=5.9 Hz, 2H), 3.91(s, 3H), 3.85(s, 3H), 3.83(s, 6H), 3.73(s, 3H), 2.07—2.00(m, 2H) | 172.6, 166.8, 164.3, 160.7, 158.7, 153.2, 152.1, 140.4, 140.2, 139.8, 133.5, 132.1, 129.6, 129.2, 126.3, 126.0, 108.9, 106.1, 96.4, 93.9, 93.5, 73.8, 65.4, 60.6, 56.5, 56.3, 29.5 |
| 4e | 8.54(s, 1H), 7.99(d, J=7.0 Hz, 1H), 7.81(d, J=8.3 Hz, 1H), 7.74(t, J=6.9 Hz, 1H), 7.62(t, J=6.8 Hz, 1H), 7.37(s, 2H), 6.81(s, 1H), 6.47(s, 1H), 4.45(t, J=6.3 Hz, 2H), 4.01(t, J=6.1 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.84(s, 3H), 3.73(s, 3H), 1.95—1.89(m, 2H), 1.88—1.82(m, 2H) | 172.7, 164.2, 160.8, 158.6, 157.5, 153.2, 152.1, 140.4, 140.3, 140.1, 139.9, 138.8, 130.8, 129.1, 127.3, 127.2, 126.1, 108.9, 106.3, 96.4, 93.6, 71.7, 66.3, 60.6, 56.6, 56.5, 26.7, 25.4 |
| 4f | 7.87(d, J=8.1 Hz, 1H), 7.70(t, J=8.9 Hz, 1H), 7.60(d, J=7.0 Hz, 1H), 7.44(s, 1H), 7.41(s, 1H), 7.31(t, J=7.6 Hz, 1H), 6.66(s, 1H), 6.40(s, 1H), 4.52(t, J=6.2 Hz, 2H), 4.13(t, J=6.2 Hz, 2H), 3.91(s, 3H), 3.89(s, 6H), 3.86(s, 3H), 3.79(s, 3H), 2.45(s, 3H), 2.08—2.01(m, 2H), 1.96—1.91(m, 2H) | 173.7, 165.3, 159.9, 157.5, 154.5, 153.1, 153.1, 149.4, 141.4, 139.8, 134.0, 133.9, 130.8, 129.9, 129.3, 127.9, 124.2, 110.3, 107.2, 96.8, 94.0, 72.3, 67.3, 61.1, 56.8, 56.8, 28.1, 26.6, 20.9 |
| 4g | 8.43(s, 1H), 7.98(s, 1H), 7.79(d, J=10.3 Hz, 1H), 7.70(d, J=9.5 Hz, 1H), 7.37(s, 1H), 7.34(s, 1H), 6.82(s, 1H), 6.49(s, 1H), 4.44(t, J=6.4 Hz, 2H), 4.01(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.82(s, 3H), 3.71(s, 3H), 2.05—1.86(m, 2H), 1.85—1.75(m, 2H) | 172.7, 164.2, 160.7, 158.6, 154.4, 153.1, 152.1, 141.6, 139.8, 134.0, 131.7, 131.2, 127.9, 127.7, 117.3, 108.9, 106.2, 106.1, 96.4, 93.6, 71.5, 66.6, 60.6, 56.5, 56.5, 27.4, 26.7 |
| 4h | 7.61(d, J=9.5 Hz, 1H), 7.47(d, J=10.1 Hz, 1H), 7.38(s, 1H), 7.36(s, 1H), 7.30(t, J=7.2 Hz, 1H), 7.21(t, J=6.8 Hz, 1H), 6.81(s, 1H), 6.46(s, 1H), 4.36(t, J=6.4 Hz, 2H), 4.00(t, J=6.9 Hz, 2H), 3.89(s, 3H), 3.83(s, 6H), 3.81(s, 3H), 3.72(s, 3H), 2.01—1.83(m, 2H), 1.79—1.64(m, 2H) | 172.7, 164.2, 160.8, 158.6, 153.9, 153.3, 153.2, 153.1, 152.1, 140.3, 139.9, 126.8, 126.1, 126.0, 124.3, 115.9, 108.9, 106.2, 96.4, 93.6, 71.6, 60.6, 60.5, 56.5, 56.4, 27.5, 26.7 |
| 4i | 8.20(s, 1H), 8.00(d, J=8.1 Hz, 1H), 7.82(d, J=7.3 Hz, 1H), 7.74(t, J=6.9 Hz, 1H), 7.63(t, J=6.5 Hz, 1H), 7.40(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.50(s, 1H), 4.40(t, J=6.5 Hz, 2H), 3.97(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.85(s, 3H), 3.73(s, 3H), 1.86—1.74(m, 2H), 1.72—1.58(m, 2H), 1.57—1.43(m, 2H) | 172.7, 164.2, 160.8, 158.6, 154.6, 153.2, 150.6, 140.3, 140.0, 131.6, 130.8, 130.3, 129.1, 127.3, 126.1, 123.8, 115.2, 109.0, 106.3, 96.4, 93.6, 72.0, 66.6, 60.7, 56.6, 56.5, 29.8, 28.3, 23.3 |
| 4j | 7.89(d, J=7.3 Hz, 1H), 7.77(t, J=7.5 Hz, 1H), 7.63(d, J=8.3 Hz, 1H), 7.54(t, J=7.7 Hz, 1H), 7.39(s, 1H), 7.37(s, 1H), 6.80(s, 1H), 6.46(s, 1H), 4.39(t, J=6.3 Hz, 2H), 3.96(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.88(s, 6H), 3.85(s, 3H), 3.75(s, 3H), 2.43(s, 3H), 1.91—1.75(m, 2H), 1.74—1.64(m, 2H), 1.57—1.46(m, 2H) | 172.6, 164.1, 160.7, 158.6, 156.3, 154.5, 153.1, 151.8, 148.3, 140.5, 139.8, 139.7, 138.4, 130.1, 129.4, 128.2, 126.9, 123.6, 108.9, 106.2, 106.1, 96.3, 93.5, 71.9, 66.6, 60.6, 56.5, 56.5, 29.8, 28.31, 23.3, 20.4 |
| 4k | 8.26(s, 1H), 8.06(s, 1H), 7.80(d, J=9.6 Hz, 1H), 7.70(d, J=9.7 Hz, 1H), 7.38(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.40(t, J=6.4 Hz, 2H), 3.97(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.84(s, 3H), 3.73(s, 3H), 1.94—1.73(m, 2H), 1.72—1.62(m, 2H), 1.59—1.44(m, 2H) | 172.7, 164.2, 160.7, 158.6, 157.8, 153.1, 152.1, 141.6, 140.4, 139.8, 139.0, 138.9, 132.0, 131.2, 129.1, 128.0, 126.1, 108.9, 106.2, 96.4, 93.6, 71.9, 66.9, 65.5, 60.6, 56.5, 29.8, 28.2, 22.6 |
| 4l | 7.70(d, J=10.7 Hz, 1H), 7.51(d, J=9.6 Hz, 1H), 7.39(s, 1H), 7.37(s, 1H), 7.22(t, J=10.4 Hz, 1H), 7.05(t, J=9.2 Hz, 1H), 6.85(s, 1H), 6.50(s, 1H), 4.23(t, J=9.5 Hz, 2H), 3.97(t, J=9.1 Hz, 2H), 3.90(s, 3H), 3.85(s, 6H), 3.82(s, 3H), 3.72(s, 3H), 1.91—1.74(m, 2H), 1.72—1.65(m, 2H), 1.54—1.46(m, 2H) | 172.7, 164.2, 160.8, 158.6, 154.0, 153.2, 153.2, 153.1, 152.1, 140.3, 139.9, 126.8, 126.1, 126.0, 124.3, 115.9, 108.9, 106.2, 96.4, 93.6, 71.6, 60.6, 60.5, 56.5, 56.4, 27.5, 26.7 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
| 4m | 7.66(s, 1H), 7.49(d, J=8.7 Hz, 1H), 7.44(d, J=8.0 Hz, 1H), 7.42(s, 1H), 7.42(s, 1H), 7.30(t, J=10.7 Hz, 1H), 7.21(t, J=11.4 Hz, 1H), 6.67(s, 1H), 6.41(s, 1H), 4.22(t, J=5.4 Hz, 2H), 4.01(t, J=5.1 Hz, 2H), 3.91(s, 3H), 3.90(s, 6H), 3.87(s, 3H), 3.79(s, 3H), 1.81—1.78(m, 2H), 1.72—1.68(m, 2H), 1.52—1.49(m, 2H), 1.46—1.42(m, 2H) | 173.9, 165.3, 162.2, 159.9, 155.1, 154.4, 153.1, 151.4, 141.8, 141.4, 132.5, 132.2, 128.6, 128.1, 127.5, 127.4, 125.1, 107.3, 107.3, 96.8, 94.0, 73.0, 73.0, 61.1, 57.1, 56.7, 27.9, 27.6, 26.9 |
| 4n | 7.74(d, J=9.2 Hz, 1H), 7.63(d, J=8.2 Hz, 1H), 7.49(t, J=6.9 Hz, 1H), 7.41(t, J=8.3 Hz, 1H), 7.32(s, 1H), 7.32(s, 1H), 6.58(s, 1H), 6.32(s, 1H), 4.35(t, J=6.5 Hz, 2H), 3.94(t, J=6.5 Hz, 2H), 3.78(s, 3H), 3.78(s, 6H), 3.77(s, 3H), 3.67(s, 3H), 2.44(s, 3H), 1.97—1.92(m, 2H), 1.92—1.87(m, 2H), 1.49—1.42(m, 2H), 1.41—1.33(m, 2H) | 173.7, 165.3, 162.3, 160.0, 157.6, 154.5, 153.0, 149.4, 141.9, 141.4, 141.1, 139.8, 130.0, 129.3, 127.9, 127.5, 127.4, 110.4, 107.4, 96.8, 94.0, 73.0, 67.5, 61.1, 57.0, 56.8, 27.0, 27.0, 26.9, 20.9 |
| 4o | 8.61(s, 1H), 8.05(s, 1H), 7.83(d, J=8.9 Hz, 1H), 7.74(d, J=7.4 Hz, 1H), 7.38(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.40(t, J=6.6 Hz, 2H), 3.95(t, J=6.7 Hz, 2H), 3.90(s, 3H), 3.86(s, 3H), 3.85(s, 6H), 3.74(s, 3H), 1.80—1.67(m, 2H), 1.66—1.59(m, 2H), 1.49—1.41(m, 2H), 1.40—1.31(m, 2H) | 172.7, 164.2, 160.8, 158.6, 157.8, 153.1, 152.0, 141.7, 140.4, 139.9, 139.0, 138.9, 131.2, 131.1, 129.1, 127.9, 126.1, 108.9, 106.3, 96.4, 93.6, 72.0, 66.8, 60.6, 56.5, 56.5, 30.0, 28.5, 25.7 |
| 4p | 7.84(d, J=7.9 Hz, 1H), 7.66(d, J=7.1 Hz, 1H), 7.60(t, J=6.4 Hz, 1H), 7.39(t, J=7.0 Hz, 1H), 7.40(s, 2H), 7.36(s, 1H), 6.70(s, 1H), 6.44(s, 1H), 4.32(t, J=6.6 Hz, 2H), 4.04(t, J=6.3 Hz, 2H), 3.94(s, 3H), 3.93(s, 3H), 3.92(s, 6H), 3.82(s, 3H), 1.97—1.82(m, 2H), 1.73—1.63(m, 2H), 1.49—1.37(m, 2H), 1.31—1.11(m, 2H) | 173.8, 165.3, 162.2, 159.9, 154.5, 153.0, 151.4, 141.8, 141.4, 134.7, 132.3, 131.5, 130.1, 128.4, 127.5, 124.4, 110.4, 107.3, 96.8, 94.0, 72.9, 67.5, 61.1, 57.1, 56.7, 28.3, 27.6, 26.8 |
Table 2 1H NMR and 13C NMR data of compounds 2c, 2d and 4a—4p
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
|---|---|---|
| 2c | 7.37(s, 2H), 6.81(s, 1H), 6.48(s, 1H), 3.94(t, J=5.6 Hz, 2H), 3.90(s, 3H), 3.88(s, 6H), 3.85(s, 3H), 3.77(s, 3H), 3.48(t, J=6.7 Hz, 2H), 1.89—1.72(m, 2H), 1.71—1.62(m, 2H), 1.51—1.40(m, 2H) | |
| 2d | 7.37(s, 2H), 6.84(s, 1H), 6.49(s, 1H), 3.95(t, J=6.0 Hz, 2H), 3.90(s, 3H), 3.87(s, 6H), 3.85(s, 3H), 3.76(s, 3H), 3.58(t, J=6.6 Hz, 2H), 1.67—1.64(m, 2H), 1.58—1.47(m, 2H), 1.43—1.35(m, 2H), 1.29—1.23(m, 2H) | |
| 4a | 8.53(s, 1H), 8.00(d, J=9.2 Hz, 1H), 7.82(d, J=8.6 Hz, 1H), 7.76(t, J=7.6 Hz, 1H), 7.64(t, J=6.8 Hz, 1H), 7.39(s, 1H), 7.38(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.54(t, J=6.6 Hz, 2H), 4.16(t, J=6.1 Hz, 2H), 3.90(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.28—2.07(m, 2H) | 172.6, 164.2, 160.7, 158.6, 157.4, 153.2, 152.2, 140.3, 140.2, 140.1, 139.9, 138.8, 130.9, 129.1, 127.3, 127.2, 126.0, 108.9, 106.2, 96.4, 93.6, 69.0, 63.9, 60.6, 56.6, 56.5, 29.6 |
| 4b | 7.88(d, J=8.2 Hz, 1H), 7.74(t, J=7.7 Hz, 1H), 7.65(d, J=6.8 Hz, 1H), 7.57(t, J=6.8 Hz, 1H), 7.39(s, 1H), 7.36(s, 1H), 6.83(s, 1H), 6.46(s, 1H), 4.53(t, J=6.4 Hz, 2H), 4.09(t, J=6.2 Hz, 2H), 3.91(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.43(s, 3H), 2.27—2.06(m, 2H) | 172.6, 164.2, 160.7, 158.6, 156.2, 154.6, 153.1, 152.1, 148.3, 140.4, 139.6, 138.4, 129.5, 128.2, 126.9, 126.0, 123.7, 108.9, 106.2, 96.3, 93.4, 69.9, 68.9, 63.7, 60.5, 56.4, 29.6, 20.4 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
| 4c | 8.57(s, 1H), 8.07(s, 1H), 7.81(d, J=12.0 Hz, 1H), 7.67(d, J=11.3 Hz, 1H), 7.38(s, 1H), 7.36(s, 1H), 6.81(s, 1H), 6.48(s, 1H), 4.53(t, J=6.5 Hz, 2H), 4.07(t, J=6.2 Hz, 2H), 3.89(s, 3H), 3.85(s, 6H), 3.84(s, 3H), 3.70(s, 3H), 2.24—2.00(m, 2H) | 172.6, 164.2, 160.8, 160.7, 158.6, 157.7, 154.4, 153.2, 153.1, 152.1, 141.5, 140.3, 140.2, 139.8, 139.1, 138.9, 134.1, 131.2, 129.1, 128.0, 126.0, 108.9, 106.2, 96.4, 93.5, 68.9, 64.2, 60.6, 60.6, 56.5, 29.5 |
| 4d | 7.69(d, J=10.0 Hz, 1H), 7.43(d, J=9.8 Hz, 1H), 7.40(s, 1H), 7.39(s, 1H), 7.25(t, J=6.6 Hz, 1H), 7.06(t, J=7.1 Hz, 1H), 6.86(s, 1H), 6.50(s, 1H), 4.27(t, J=6.8 Hz, 2H), 4.09(t, J=5.9 Hz, 2H), 3.91(s, 3H), 3.85(s, 3H), 3.83(s, 6H), 3.73(s, 3H), 2.07—2.00(m, 2H) | 172.6, 166.8, 164.3, 160.7, 158.7, 153.2, 152.1, 140.4, 140.2, 139.8, 133.5, 132.1, 129.6, 129.2, 126.3, 126.0, 108.9, 106.1, 96.4, 93.9, 93.5, 73.8, 65.4, 60.6, 56.5, 56.3, 29.5 |
| 4e | 8.54(s, 1H), 7.99(d, J=7.0 Hz, 1H), 7.81(d, J=8.3 Hz, 1H), 7.74(t, J=6.9 Hz, 1H), 7.62(t, J=6.8 Hz, 1H), 7.37(s, 2H), 6.81(s, 1H), 6.47(s, 1H), 4.45(t, J=6.3 Hz, 2H), 4.01(t, J=6.1 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.84(s, 3H), 3.73(s, 3H), 1.95—1.89(m, 2H), 1.88—1.82(m, 2H) | 172.7, 164.2, 160.8, 158.6, 157.5, 153.2, 152.1, 140.4, 140.3, 140.1, 139.9, 138.8, 130.8, 129.1, 127.3, 127.2, 126.1, 108.9, 106.3, 96.4, 93.6, 71.7, 66.3, 60.6, 56.6, 56.5, 26.7, 25.4 |
| 4f | 7.87(d, J=8.1 Hz, 1H), 7.70(t, J=8.9 Hz, 1H), 7.60(d, J=7.0 Hz, 1H), 7.44(s, 1H), 7.41(s, 1H), 7.31(t, J=7.6 Hz, 1H), 6.66(s, 1H), 6.40(s, 1H), 4.52(t, J=6.2 Hz, 2H), 4.13(t, J=6.2 Hz, 2H), 3.91(s, 3H), 3.89(s, 6H), 3.86(s, 3H), 3.79(s, 3H), 2.45(s, 3H), 2.08—2.01(m, 2H), 1.96—1.91(m, 2H) | 173.7, 165.3, 159.9, 157.5, 154.5, 153.1, 153.1, 149.4, 141.4, 139.8, 134.0, 133.9, 130.8, 129.9, 129.3, 127.9, 124.2, 110.3, 107.2, 96.8, 94.0, 72.3, 67.3, 61.1, 56.8, 56.8, 28.1, 26.6, 20.9 |
| 4g | 8.43(s, 1H), 7.98(s, 1H), 7.79(d, J=10.3 Hz, 1H), 7.70(d, J=9.5 Hz, 1H), 7.37(s, 1H), 7.34(s, 1H), 6.82(s, 1H), 6.49(s, 1H), 4.44(t, J=6.4 Hz, 2H), 4.01(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.82(s, 3H), 3.71(s, 3H), 2.05—1.86(m, 2H), 1.85—1.75(m, 2H) | 172.7, 164.2, 160.7, 158.6, 154.4, 153.1, 152.1, 141.6, 139.8, 134.0, 131.7, 131.2, 127.9, 127.7, 117.3, 108.9, 106.2, 106.1, 96.4, 93.6, 71.5, 66.6, 60.6, 56.5, 56.5, 27.4, 26.7 |
| 4h | 7.61(d, J=9.5 Hz, 1H), 7.47(d, J=10.1 Hz, 1H), 7.38(s, 1H), 7.36(s, 1H), 7.30(t, J=7.2 Hz, 1H), 7.21(t, J=6.8 Hz, 1H), 6.81(s, 1H), 6.46(s, 1H), 4.36(t, J=6.4 Hz, 2H), 4.00(t, J=6.9 Hz, 2H), 3.89(s, 3H), 3.83(s, 6H), 3.81(s, 3H), 3.72(s, 3H), 2.01—1.83(m, 2H), 1.79—1.64(m, 2H) | 172.7, 164.2, 160.8, 158.6, 153.9, 153.3, 153.2, 153.1, 152.1, 140.3, 139.9, 126.8, 126.1, 126.0, 124.3, 115.9, 108.9, 106.2, 96.4, 93.6, 71.6, 60.6, 60.5, 56.5, 56.4, 27.5, 26.7 |
| 4i | 8.20(s, 1H), 8.00(d, J=8.1 Hz, 1H), 7.82(d, J=7.3 Hz, 1H), 7.74(t, J=6.9 Hz, 1H), 7.63(t, J=6.5 Hz, 1H), 7.40(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.50(s, 1H), 4.40(t, J=6.5 Hz, 2H), 3.97(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.85(s, 3H), 3.73(s, 3H), 1.86—1.74(m, 2H), 1.72—1.58(m, 2H), 1.57—1.43(m, 2H) | 172.7, 164.2, 160.8, 158.6, 154.6, 153.2, 150.6, 140.3, 140.0, 131.6, 130.8, 130.3, 129.1, 127.3, 126.1, 123.8, 115.2, 109.0, 106.3, 96.4, 93.6, 72.0, 66.6, 60.7, 56.6, 56.5, 29.8, 28.3, 23.3 |
| 4j | 7.89(d, J=7.3 Hz, 1H), 7.77(t, J=7.5 Hz, 1H), 7.63(d, J=8.3 Hz, 1H), 7.54(t, J=7.7 Hz, 1H), 7.39(s, 1H), 7.37(s, 1H), 6.80(s, 1H), 6.46(s, 1H), 4.39(t, J=6.3 Hz, 2H), 3.96(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.88(s, 6H), 3.85(s, 3H), 3.75(s, 3H), 2.43(s, 3H), 1.91—1.75(m, 2H), 1.74—1.64(m, 2H), 1.57—1.46(m, 2H) | 172.6, 164.1, 160.7, 158.6, 156.3, 154.5, 153.1, 151.8, 148.3, 140.5, 139.8, 139.7, 138.4, 130.1, 129.4, 128.2, 126.9, 123.6, 108.9, 106.2, 106.1, 96.3, 93.5, 71.9, 66.6, 60.6, 56.5, 56.5, 29.8, 28.31, 23.3, 20.4 |
| 4k | 8.26(s, 1H), 8.06(s, 1H), 7.80(d, J=9.6 Hz, 1H), 7.70(d, J=9.7 Hz, 1H), 7.38(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.40(t, J=6.4 Hz, 2H), 3.97(t, J=6.4 Hz, 2H), 3.90(s, 3H), 3.86(s, 6H), 3.84(s, 3H), 3.73(s, 3H), 1.94—1.73(m, 2H), 1.72—1.62(m, 2H), 1.59—1.44(m, 2H) | 172.7, 164.2, 160.7, 158.6, 157.8, 153.1, 152.1, 141.6, 140.4, 139.8, 139.0, 138.9, 132.0, 131.2, 129.1, 128.0, 126.1, 108.9, 106.2, 96.4, 93.6, 71.9, 66.9, 65.5, 60.6, 56.5, 29.8, 28.2, 22.6 |
| 4l | 7.70(d, J=10.7 Hz, 1H), 7.51(d, J=9.6 Hz, 1H), 7.39(s, 1H), 7.37(s, 1H), 7.22(t, J=10.4 Hz, 1H), 7.05(t, J=9.2 Hz, 1H), 6.85(s, 1H), 6.50(s, 1H), 4.23(t, J=9.5 Hz, 2H), 3.97(t, J=9.1 Hz, 2H), 3.90(s, 3H), 3.85(s, 6H), 3.82(s, 3H), 3.72(s, 3H), 1.91—1.74(m, 2H), 1.72—1.65(m, 2H), 1.54—1.46(m, 2H) | 172.7, 164.2, 160.8, 158.6, 154.0, 153.2, 153.2, 153.1, 152.1, 140.3, 139.9, 126.8, 126.1, 126.0, 124.3, 115.9, 108.9, 106.2, 96.4, 93.6, 71.6, 60.6, 60.5, 56.5, 56.4, 27.5, 26.7 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(101 MHz), δ |
| 4m | 7.66(s, 1H), 7.49(d, J=8.7 Hz, 1H), 7.44(d, J=8.0 Hz, 1H), 7.42(s, 1H), 7.42(s, 1H), 7.30(t, J=10.7 Hz, 1H), 7.21(t, J=11.4 Hz, 1H), 6.67(s, 1H), 6.41(s, 1H), 4.22(t, J=5.4 Hz, 2H), 4.01(t, J=5.1 Hz, 2H), 3.91(s, 3H), 3.90(s, 6H), 3.87(s, 3H), 3.79(s, 3H), 1.81—1.78(m, 2H), 1.72—1.68(m, 2H), 1.52—1.49(m, 2H), 1.46—1.42(m, 2H) | 173.9, 165.3, 162.2, 159.9, 155.1, 154.4, 153.1, 151.4, 141.8, 141.4, 132.5, 132.2, 128.6, 128.1, 127.5, 127.4, 125.1, 107.3, 107.3, 96.8, 94.0, 73.0, 73.0, 61.1, 57.1, 56.7, 27.9, 27.6, 26.9 |
| 4n | 7.74(d, J=9.2 Hz, 1H), 7.63(d, J=8.2 Hz, 1H), 7.49(t, J=6.9 Hz, 1H), 7.41(t, J=8.3 Hz, 1H), 7.32(s, 1H), 7.32(s, 1H), 6.58(s, 1H), 6.32(s, 1H), 4.35(t, J=6.5 Hz, 2H), 3.94(t, J=6.5 Hz, 2H), 3.78(s, 3H), 3.78(s, 6H), 3.77(s, 3H), 3.67(s, 3H), 2.44(s, 3H), 1.97—1.92(m, 2H), 1.92—1.87(m, 2H), 1.49—1.42(m, 2H), 1.41—1.33(m, 2H) | 173.7, 165.3, 162.3, 160.0, 157.6, 154.5, 153.0, 149.4, 141.9, 141.4, 141.1, 139.8, 130.0, 129.3, 127.9, 127.5, 127.4, 110.4, 107.4, 96.8, 94.0, 73.0, 67.5, 61.1, 57.0, 56.8, 27.0, 27.0, 26.9, 20.9 |
| 4o | 8.61(s, 1H), 8.05(s, 1H), 7.83(d, J=8.9 Hz, 1H), 7.74(d, J=7.4 Hz, 1H), 7.38(s, 1H), 7.37(s, 1H), 6.83(s, 1H), 6.49(s, 1H), 4.40(t, J=6.6 Hz, 2H), 3.95(t, J=6.7 Hz, 2H), 3.90(s, 3H), 3.86(s, 3H), 3.85(s, 6H), 3.74(s, 3H), 1.80—1.67(m, 2H), 1.66—1.59(m, 2H), 1.49—1.41(m, 2H), 1.40—1.31(m, 2H) | 172.7, 164.2, 160.8, 158.6, 157.8, 153.1, 152.0, 141.7, 140.4, 139.9, 139.0, 138.9, 131.2, 131.1, 129.1, 127.9, 126.1, 108.9, 106.3, 96.4, 93.6, 72.0, 66.8, 60.6, 56.5, 56.5, 30.0, 28.5, 25.7 |
| 4p | 7.84(d, J=7.9 Hz, 1H), 7.66(d, J=7.1 Hz, 1H), 7.60(t, J=6.4 Hz, 1H), 7.39(t, J=7.0 Hz, 1H), 7.40(s, 2H), 7.36(s, 1H), 6.70(s, 1H), 6.44(s, 1H), 4.32(t, J=6.6 Hz, 2H), 4.04(t, J=6.3 Hz, 2H), 3.94(s, 3H), 3.93(s, 3H), 3.92(s, 6H), 3.82(s, 3H), 1.97—1.82(m, 2H), 1.73—1.63(m, 2H), 1.49—1.37(m, 2H), 1.31—1.11(m, 2H) | 173.8, 165.3, 162.2, 159.9, 154.5, 153.0, 151.4, 141.8, 141.4, 134.7, 132.3, 131.5, 130.1, 128.4, 127.5, 124.4, 110.4, 107.3, 96.8, 94.0, 72.9, 67.5, 61.1, 57.1, 56.7, 28.3, 27.6, 26.8 |
| Compd. | n | R | Inhibition ratea (%) | |||
|---|---|---|---|---|---|---|
| X. citri | X. oryzae | |||||
| 100 μg/mL | 50 μg/mL | 100 μg/mL | 50 μg/mL | |||
| 4a | 3 | H | 73.69±5.97 | 67.82±2.64 | 40.78±3.60 | 33.63±5.14 |
| 4b | 3 | 3-CH3 | 83.52±2.19 | 68.13±1.20 | 49.33±2.85 | 44.71±4.65 |
| 4c | 3 | 6-Cl | 73.17±7.31 | 63.18±6.12 | 50.23±4.81 | 43.66±8.00 |
| 4d | 3 | 3-OH | 77.50±2.97 | 67.71±7.99 | 52.78±5.55 | 43.77±5.57 |
| 4e | 4 | H | 75.23±9.52 | 62.10±2.25 | 55.17±5.65 | 42.52±5.98 |
| 4f | 4 | 3-CH3 | 74.61±2.36 | 65.50±5.74 | 61.30±4.24 | 53.27±8.16 |
| 4g | 4 | 6-Cl | 74.20±9.83 | 61.79±7.18 | 47.19±5.92 | 37.91±6.71 |
| 4h | 4 | 3-OH | 77.96±1.47 | 68.07±6.74 | 53.22±6.21 | 42.31±2.88 |
| 4i | 5 | H | 80.38±6.90 | 69.88±4.33 | 47.57±5.99 | 37.48±2.00 |
| 4j | 5 | 3-CH3 | 76.67±2.63 | 66.63±6.14 | 50.23±5.71 | 44.80±1.41 |
| 4k | 5 | 6-Cl | 81.00±8.11 | 69.41±4.79 | 36.18±0.32 | 31.99±6.24 |
| 4l | 5 | 3-OH | 73.07±4.23 | 67.35±7.10 | 49.61±3.08 | 38.20±8.51 |
| 4m | 6 | H | 76.00±7.20 | 65.55±4.94 | 54.74±8.50 | 46.65±7.34 |
| 4n | 6 | 3-CH3 | 77.45±1.75 | 67.35±6.36 | 47.95±7.65 | 39.92±9.61 |
| 4o | 6 | 6-Cl | 76.21±5.41 | 68.13±7.63 | 29.61±2.46 | 18.79±4.44 |
| 4p | 6 | 3-OH | 77.45±3.28 | 66.37±3.42 | 33.72±1.93 | 21.23±6.28 |
| Myricetin | 49.38±4.06 | 40.37±0.41 | 41.38±5.94 | 32.32±2.85 | ||
| Bismerthiazolb | 59.47±7.65 | 49.49±5.74 | 47.03±3.06 | 39.59±6.08 | ||
| Thiodiazole-copperb | 56.90±1.47 | 47.94±8.47 | 43.23±3.01 | 38.18±6.32 | ||
Table 3 Antibacterial activities of target compounds 4a—4p
| Compd. | n | R | Inhibition ratea (%) | |||
|---|---|---|---|---|---|---|
| X. citri | X. oryzae | |||||
| 100 μg/mL | 50 μg/mL | 100 μg/mL | 50 μg/mL | |||
| 4a | 3 | H | 73.69±5.97 | 67.82±2.64 | 40.78±3.60 | 33.63±5.14 |
| 4b | 3 | 3-CH3 | 83.52±2.19 | 68.13±1.20 | 49.33±2.85 | 44.71±4.65 |
| 4c | 3 | 6-Cl | 73.17±7.31 | 63.18±6.12 | 50.23±4.81 | 43.66±8.00 |
| 4d | 3 | 3-OH | 77.50±2.97 | 67.71±7.99 | 52.78±5.55 | 43.77±5.57 |
| 4e | 4 | H | 75.23±9.52 | 62.10±2.25 | 55.17±5.65 | 42.52±5.98 |
| 4f | 4 | 3-CH3 | 74.61±2.36 | 65.50±5.74 | 61.30±4.24 | 53.27±8.16 |
| 4g | 4 | 6-Cl | 74.20±9.83 | 61.79±7.18 | 47.19±5.92 | 37.91±6.71 |
| 4h | 4 | 3-OH | 77.96±1.47 | 68.07±6.74 | 53.22±6.21 | 42.31±2.88 |
| 4i | 5 | H | 80.38±6.90 | 69.88±4.33 | 47.57±5.99 | 37.48±2.00 |
| 4j | 5 | 3-CH3 | 76.67±2.63 | 66.63±6.14 | 50.23±5.71 | 44.80±1.41 |
| 4k | 5 | 6-Cl | 81.00±8.11 | 69.41±4.79 | 36.18±0.32 | 31.99±6.24 |
| 4l | 5 | 3-OH | 73.07±4.23 | 67.35±7.10 | 49.61±3.08 | 38.20±8.51 |
| 4m | 6 | H | 76.00±7.20 | 65.55±4.94 | 54.74±8.50 | 46.65±7.34 |
| 4n | 6 | 3-CH3 | 77.45±1.75 | 67.35±6.36 | 47.95±7.65 | 39.92±9.61 |
| 4o | 6 | 6-Cl | 76.21±5.41 | 68.13±7.63 | 29.61±2.46 | 18.79±4.44 |
| 4p | 6 | 3-OH | 77.45±3.28 | 66.37±3.42 | 33.72±1.93 | 21.23±6.28 |
| Myricetin | 49.38±4.06 | 40.37±0.41 | 41.38±5.94 | 32.32±2.85 | ||
| Bismerthiazolb | 59.47±7.65 | 49.49±5.74 | 47.03±3.06 | 39.59±6.08 | ||
| Thiodiazole-copperb | 56.90±1.47 | 47.94±8.47 | 43.23±3.01 | 38.18±6.32 | ||
| Compd. | n | R | Toxic regression equation | r | EC50/(μg·mL-1) |
|---|---|---|---|---|---|
| 4a | 3 | H | y=0.8555x+3.9292 | 0.9368 | 17.85±2.27 |
| 4b | 3 | 3-CH3 | y=0.9079x+4.0182 | 0.9033 | 12.06±2.84 |
| 4c | 3 | 6-Cl | y=0.7629x+4.0511 | 0.9859 | 17.53±2.94 |
| 4d | 3 | 3-OH | y=0.8701x+3.9962 | 0.9876 | 14.24±2.13 |
| 4e | 4 | H | y=0.7769x+4.0440 | 0.9713 | 17.00±3.11 |
| 4f | 4 | 3-CH3 | y=0.7701x+4.1052 | 0.9911 | 14.52±1.80 |
| 4g | 4 | 6-Cl | y=0.9324x+3.7319 | 0.9869 | 22.91±4.26 |
| 4h | 4 | 3-OH | y=0.9139x+3.9345 | 0.9988 | 14.65±3.15 |
| 4i | 5 | H | y=0.8168x+4.0913 | 0.8317 | 12.38±1.74 |
| 4j | 5 | 3-CH3 | y=0.7878x+4.1041 | 0.9667 | 13.72±1.31 |
| 4k | 5 | 6-Cl | y=0.9325x+3.9408 | 0.9719 | 13.67±2.35 |
| 4l | 5 | 3-OH | y=0.6705x+4.2733 | 0.9944 | 12.13±1.91 |
| 4m | 6 | H | y=0.7829x+4.0987 | 0.9867 | 14.17±1.37 |
| 4n | 6 | 3-CH3 | y=0.8137x+4.0627 | 0.9771 | 14.19±1.66 |
| 4o | 6 | 6-Cl | y=0.6998x+4.2666 | 0.9710 | 11.17±2.20 |
| 4p | 6 | 3-OH | y=0.7367x+4.2104 | 0.9845 | 11.80±0.72 |
| Myricetin | y=0.6520x+3.6763 | 0.9838 | 107.20±1.63 | ||
| Bismerthiazola | y=0.8357x+3.5466 | 0.9876 | 54.85±1.37 | ||
| Thiediazole-coppera | y=0.8874x+3.4149 | 0.9984 | 61.13±3.37 |
Table 4 EC50 values of some compounds against X. citri
| Compd. | n | R | Toxic regression equation | r | EC50/(μg·mL-1) |
|---|---|---|---|---|---|
| 4a | 3 | H | y=0.8555x+3.9292 | 0.9368 | 17.85±2.27 |
| 4b | 3 | 3-CH3 | y=0.9079x+4.0182 | 0.9033 | 12.06±2.84 |
| 4c | 3 | 6-Cl | y=0.7629x+4.0511 | 0.9859 | 17.53±2.94 |
| 4d | 3 | 3-OH | y=0.8701x+3.9962 | 0.9876 | 14.24±2.13 |
| 4e | 4 | H | y=0.7769x+4.0440 | 0.9713 | 17.00±3.11 |
| 4f | 4 | 3-CH3 | y=0.7701x+4.1052 | 0.9911 | 14.52±1.80 |
| 4g | 4 | 6-Cl | y=0.9324x+3.7319 | 0.9869 | 22.91±4.26 |
| 4h | 4 | 3-OH | y=0.9139x+3.9345 | 0.9988 | 14.65±3.15 |
| 4i | 5 | H | y=0.8168x+4.0913 | 0.8317 | 12.38±1.74 |
| 4j | 5 | 3-CH3 | y=0.7878x+4.1041 | 0.9667 | 13.72±1.31 |
| 4k | 5 | 6-Cl | y=0.9325x+3.9408 | 0.9719 | 13.67±2.35 |
| 4l | 5 | 3-OH | y=0.6705x+4.2733 | 0.9944 | 12.13±1.91 |
| 4m | 6 | H | y=0.7829x+4.0987 | 0.9867 | 14.17±1.37 |
| 4n | 6 | 3-CH3 | y=0.8137x+4.0627 | 0.9771 | 14.19±1.66 |
| 4o | 6 | 6-Cl | y=0.6998x+4.2666 | 0.9710 | 11.17±2.20 |
| 4p | 6 | 3-OH | y=0.7367x+4.2104 | 0.9845 | 11.80±0.72 |
| Myricetin | y=0.6520x+3.6763 | 0.9838 | 107.20±1.63 | ||
| Bismerthiazola | y=0.8357x+3.5466 | 0.9876 | 54.85±1.37 | ||
| Thiediazole-coppera | y=0.8874x+3.4149 | 0.9984 | 61.13±3.37 |
| Compd. | n | R | Toxic regression equation | r | EC50/(μg·mL-1) |
|---|---|---|---|---|---|
| 4c | 3 | 6-Cl | y=0.7670x+3.5327 | 0.9862 | 81.85±3.03 |
| 4d | 3 | 3-OH | y=0.6330x+3.7945 | 0.9972 | 80.25±2.31 |
| 4e | 4 | H | y=0.8403x+3.4619 | 0.9880 | 67.67±1.81 |
| 4f | 4 | 3-CH3 | y=0.7279x+3.8807 | 0.9811 | 34.49±2.14 |
| 4g | 4 | 6-Cl | y=0.7356x+3.4632 | 0.9966 | 122.79±0.94 |
| 4h | 4 | 3-OH | y=0.8820x+3.3230 | 0.9929 | 79.68±2.18 |
| 4i | 5 | H | y=0.9043x+3.1931 | 0.9840 | 99.57±2.57 |
| 4j | 5 | 3-CH3 | y=0.6945x+3.6513 | 0.9897 | 87.49±2.42 |
| 4m | 6 | H | y=0.9009x+3.3445 | 0.9956 | 68.80±2.52 |
| 4n | 6 | 3-CH3 | y=0.7117x+3.5476 | 0.9975 | 109.84±1.09 |
| Myricetin | y=0.6512x+3.4715 | 0.9862 | 222.44±2.37 | ||
| Bismerthiazol* | y=0.5525x+3.8806 | 0.9947 | 148.20±2.44 | ||
| Thiediazole-copper* | y=0.5876x+3.6813 | 0.9950 | 175.47±2.09 |
Table 5 EC50 values of some compounds against X. oryzae
| Compd. | n | R | Toxic regression equation | r | EC50/(μg·mL-1) |
|---|---|---|---|---|---|
| 4c | 3 | 6-Cl | y=0.7670x+3.5327 | 0.9862 | 81.85±3.03 |
| 4d | 3 | 3-OH | y=0.6330x+3.7945 | 0.9972 | 80.25±2.31 |
| 4e | 4 | H | y=0.8403x+3.4619 | 0.9880 | 67.67±1.81 |
| 4f | 4 | 3-CH3 | y=0.7279x+3.8807 | 0.9811 | 34.49±2.14 |
| 4g | 4 | 6-Cl | y=0.7356x+3.4632 | 0.9966 | 122.79±0.94 |
| 4h | 4 | 3-OH | y=0.8820x+3.3230 | 0.9929 | 79.68±2.18 |
| 4i | 5 | H | y=0.9043x+3.1931 | 0.9840 | 99.57±2.57 |
| 4j | 5 | 3-CH3 | y=0.6945x+3.6513 | 0.9897 | 87.49±2.42 |
| 4m | 6 | H | y=0.9009x+3.3445 | 0.9956 | 68.80±2.52 |
| 4n | 6 | 3-CH3 | y=0.7117x+3.5476 | 0.9975 | 109.84±1.09 |
| Myricetin | y=0.6512x+3.4715 | 0.9862 | 222.44±2.37 | ||
| Bismerthiazol* | y=0.5525x+3.8806 | 0.9947 | 148.20±2.44 | ||
| Thiediazole-copper* | y=0.5876x+3.6813 | 0.9950 | 175.47±2.09 |
| Compd. | n | R | Inhibition rate(%) | ||
|---|---|---|---|---|---|
| Curative | Protection | Inactivation | |||
| 4a | 3 | H | 39.2±4.5 | 40.1±3.9 | 63.2±6.6 |
| 4b | 3 | 3-CH3 | 38.6±4.7 | 40.2±4.9 | 60.0±5.5 |
| 4c | 3 | 6-Cl | 43.8±1.0 | 47.3±4.6 | 69.2±8.4 |
| 4d | 3 | 3-OH | 35.4±7.5 | 46.3±2.5 | 44.5±5.7 |
| 4e | 4 | H | 41.5±5.0 | 50.5±5.6 | 75.1±5.1 |
| 4f | 4 | 3-CH3 | 50.3±3.3 | 52.6±5.2 | 67.5±8.1 |
| 4g | 4 | 6-Cl | 51.6±6.6 | 59.8±5.1 | 77.7±6.4 |
| 4h | 4 | 3-OH | 33.7±3.5 | 43.3±4.4 | 65.8±4.2 |
| 4i | 5 | H | 47.9±0.7 | 49.1±8.5 | 66.4±7.3 |
| 4j | 5 | 3-CH3 | 44.4±5.1 | 40.7±4.5 | 62.8±3.6 |
| 4k | 5 | 6-Cl | 49.0±7.2 | 53.0±5.6 | 73.6±2.5 |
| 4l | 5 | 3-OH | 40.5±5.7 | 44.0±2.6 | 54.0±4.1 |
| 4m | 6 | H | 51.4±2.6 | 62.3±4.9 | 78.3±1.5 |
| 4n | 6 | 3-CH3 | 48.7±6.6 | 47.3±9.2 | 51.6±2.7 |
| 4o | 6 | 6-Cl | 50.1±3.2 | 48.1±4.8 | 54.7±0.4 |
| 4p | 6 | 3-OH | 46.2±6.0 | 54.3±4.1 | 74.9±3.6 |
| Myricetin | 31.6±7.2 | 42.1±6.4 | 50.9±6.3 | ||
| Ribavirin | 39.9±3.7 | 51.8±6.0 | 73.3±2.9 | ||
| Ningnanmycin | 52.7±1.3 | 65.7±1.9 | 90.4±3.1 | ||
Table 6 Antiviral activities of the test compounds against TMV in vivo at 500 mg/L
| Compd. | n | R | Inhibition rate(%) | ||
|---|---|---|---|---|---|
| Curative | Protection | Inactivation | |||
| 4a | 3 | H | 39.2±4.5 | 40.1±3.9 | 63.2±6.6 |
| 4b | 3 | 3-CH3 | 38.6±4.7 | 40.2±4.9 | 60.0±5.5 |
| 4c | 3 | 6-Cl | 43.8±1.0 | 47.3±4.6 | 69.2±8.4 |
| 4d | 3 | 3-OH | 35.4±7.5 | 46.3±2.5 | 44.5±5.7 |
| 4e | 4 | H | 41.5±5.0 | 50.5±5.6 | 75.1±5.1 |
| 4f | 4 | 3-CH3 | 50.3±3.3 | 52.6±5.2 | 67.5±8.1 |
| 4g | 4 | 6-Cl | 51.6±6.6 | 59.8±5.1 | 77.7±6.4 |
| 4h | 4 | 3-OH | 33.7±3.5 | 43.3±4.4 | 65.8±4.2 |
| 4i | 5 | H | 47.9±0.7 | 49.1±8.5 | 66.4±7.3 |
| 4j | 5 | 3-CH3 | 44.4±5.1 | 40.7±4.5 | 62.8±3.6 |
| 4k | 5 | 6-Cl | 49.0±7.2 | 53.0±5.6 | 73.6±2.5 |
| 4l | 5 | 3-OH | 40.5±5.7 | 44.0±2.6 | 54.0±4.1 |
| 4m | 6 | H | 51.4±2.6 | 62.3±4.9 | 78.3±1.5 |
| 4n | 6 | 3-CH3 | 48.7±6.6 | 47.3±9.2 | 51.6±2.7 |
| 4o | 6 | 6-Cl | 50.1±3.2 | 48.1±4.8 | 54.7±0.4 |
| 4p | 6 | 3-OH | 46.2±6.0 | 54.3±4.1 | 74.9±3.6 |
| Myricetin | 31.6±7.2 | 42.1±6.4 | 50.9±6.3 | ||
| Ribavirin | 39.9±3.7 | 51.8±6.0 | 73.3±2.9 | ||
| Ningnanmycin | 52.7±1.3 | 65.7±1.9 | 90.4±3.1 | ||
| [1] | Li B. C., Lin X. R., Zhang Y. M., Zhang D. W., Xiao Y., Lin F., Chem. Res. Chinese Universities,2017, 33(1), 70—73 |
| [2] | Xu B. X., Ding X. D., Wu Y. C., Cui L., Qian P., Wang D., Zhao Y. F., Chem. Res. Chinese Universities,2018, 34(1), 51—56 |
| [3] | Huang M. G., Ruan X. H., Zhang J. P., Li Q., Wang Y. H., Chen L. J., Zhang C., Li P., Xue W., Chin. J. Org. Chem.,2017, 37(8), 2145—2152 |
| (黄民国, 阮祥辉, 张菊平, 李琴, 王一会, 陈丽娟, 张橙, 李普, 薛伟. 有机化学,2017, 37(8), 2145—2152) | |
| [4] | Zhou J. R., Mukheriee P., Gugger E. T., Tanaka T., Blackbum G. L., Clinton S. K., Cancer Res.,1998, 58(22), 5231—5238 |
| [5] | Zhang X. J., Huang Q. L., Ji Y. B., Tianjin Pharmacy,2008, 20(5), 57—60 |
| (张秀娟, 黄清玲, 季宇彬. 天津药学,2008, 20(5), 57—60) | |
| [6] | Grenier D., Chen H., Ben L. A., Fournier L. J., Pierre M. M., Plos One,2015, 10(6), e0131758 |
| [7] | Yu M. S., Lee J., Lee J. M., Kim Y., Chin Y. W., Jee J. G., Keum Y. S., Jeong Y. J., Bioorg. Med. Chem. Lett.,2012, 22(12), 4049—4054 |
| [8] | Ruan X. H., Zhao H. J., Zhang C., Chen L. J., Li P., Wang Y. H., He M., Xue W., Chem. J. Chinese Universities,2018, 39(6), 1197—1204 |
| (阮祥辉, 赵洪菊, 张橙, 陈丽娟, 李普, 王一会, 贺鸣, 薛伟. 高等学校化学学报,2018, 39(6), 1197—1204) | |
| [9] | Liu I. M., Tzeng T. F., Liou S. S., Lan T. W., Planta Med.,2007, 73(10), 1054—1060 |
| [10] | Guo J. J., Meng Y. H., Zhao Y., Hu Y. Y., Rena D. Y., Yang X. B., Food Funct.,2015, 6(5), 1620—1634 |
| [11] | Shimmyo Y., Kihara T., Akaike A., Niidome T., Sugimoto H. J., Neurosci. Res.,2008, 86(6), 1836—1845 |
| [12] | Xiao W., Ruan X. H., Li Q., Zhang J. P., Zhong X. M., Xie Y., Wang X. B., Huang M. G., Xue W., Chem. J. Chinese Universities,2017, 38(1), 35—40 |
| (肖维, 阮祥辉, 李琴, 张菊平, 钟新敏, 谢艳, 王晓斌, 黄民国, 薛伟. 高等学校化学学报,2017, 38(1), 35—40) | |
| [13] | Su X. W., D’Souza D. H., Food Environ. Virol.,2013, 5(2), 97—102 |
| [14] | Suguna M., Kumar N. S., Subbaiah M. V., Krishnaiah A. J., Chem. Pharm. Res.,2010, 2(1), 7—20 |
| [15] | Burguete A., Pontiki E., Hadjipavlou-Litina D., Villar R., Vicente E., Solano B., Ancizu S., Pérez-Silanes S., Aldana I., Monge A., Bioorg. Med. Chem. Lett.,2007, 17(23), 6439—6443 |
| [16] | Ganley B., Chowdhury G., Bhansali J., Daniels J. S., Gates K. S., Bioorg. Med. Chem.,2001, 9(9), 2395—2401 |
| [17] | Rodrigues F. A., Bomfim I. S., Cavalcanti B. C., Pessoa C. Ó., Wardell J. L., Wardell S. M., Pinheiro A. C., Kaiser C. R., Nogueira T. C., Low J. N., Gomes L. R., de Souza M. V., Bioorg. Med. Chem. Lett.,2014, 24(3), 934—939 |
| [18] | Khan S. A., Saleem K., Khan Z., Eur. J. Med. Chem.,2007, 42(1), 103—108 |
| [19] | Li Q., Synthesis and Biological Activity of 1,4-Pentadien-3-one(oxime) Ether Compounds Containing Quinazoline(Quinoxaline) Groups, Guizhou University, Guiyang, 2018 |
| (李琴. 含喹唑(喔)啉基团的1,4-戊二烯-3-酮(肟)醚类化合物的合成及生物活性研究, 贵阳: 贵州大学, 2018) | |
| [20] | Xue W., Song B. A., Zhao H. J., Qi X. B., Huang Y. J., Liu X. H., Eur. J. Med. Chem.,2015, 97, 155—163 |
| [21] | Li P., Yin J., Xu W. M., Wu J., He M., Hu D. Y., Yang S., Song B. A., Chem. Biol. Drug Des.,2013, 82(5), 546—556 |
| [22] | Xu W. M., Han F. F., He M., Hu D. Y., He J., Yang S., Song B. A., J. Agric. Food Chem.,2012, 60(4), 1036—1041 |
| [23] | Chen Y., Wang Z. B., Zhang X., Xia L. J., Gong H. Y., Zhao H. J., Xue W., Chin. J. Org. Chem.,2014, 34(8), 1662—1668 |
| (陈玉, 王忠波, 张贤, 夏丽娟, 龚华玉, 赵洪菊, 薛伟. 有机化学,2014, 34(8), 1662—1668) |
| [1] | 左怀龙, 雷思敏, 张锐, 李玉新, 陈伟. 新型异喹啉衍生物的设计合成及抑菌活性[J]. 高等学校化学学报, 2021, 42(9): 2766. |
| [2] | 胡皓程, 李文利, 张嘉宁, 刘宇博. 黑木耳寡糖的提取、 结构表征及生物活性[J]. 高等学校化学学报, 2021, 42(8): 2465. |
| [3] | 潘一骁, 李艳稳, 韩佳宏, 赵浩强, 冯宇, 丁相元, 徐立进, 范青华, 时茜. 环化及亚胺/酰胺部分氢化一锅法串联反应合成1,2,3,4⁃四氢喹喔啉[J]. 高等学校化学学报, 2020, 41(10): 2239. |
| [4] | 李冰, 王学敏, 白凤英, 刘淑清. 稀土氮杂环配合物的合成、 结构及抑菌活性[J]. 高等学校化学学报, 2019, 40(4): 632. |
| [5] | 常俊朋, 赵佳瑞, 陈思佳, 孟凯, 石微妮, 李瑞芳. 抗菌肽SAMP1及其类似肽的构效关系[J]. 高等学校化学学报, 2019, 40(4): 705. |
| [6] | 万金林, 巫受群, 甘宜远, 孟娇, 王贞超, 欧阳贵平. 含1,3,4-噻二唑结构的查尔酮缩氨基脲类化合物的合成及抗细菌活性[J]. 高等学校化学学报, 2018, 39(8): 1683. |
| [7] | 阮祥辉, 赵洪菊, 张橙, 陈丽娟, 李普, 王一会, 贺鸣, 薛伟. 含哌嗪酰胺类杨梅素衍生物的合成及生物活性[J]. 高等学校化学学报, 2018, 39(6): 1197. |
| [8] | 谭英, 肖梦武, 叶姣, 胡艾希, 曾子清, 欧晓明. (Z)-3,3-二甲基-1-(1H-1,2,4-三唑-1-基)-2-丁酮肟(5-芳基-1,3,4-噁二唑-2-基)甲基醚的合成和抑菌活性[J]. 高等学校化学学报, 2017, 38(8): 1375. |
| [9] | 肖维, 阮祥辉, 李琴, 张菊平, 钟新敏, 谢艳, 王晓斌, 黄民国, 薛伟. 酰胺类杨梅素衍生物的合成及抑菌活性[J]. 高等学校化学学报, 2017, 38(1): 35. |
| [10] | 邹海民, 周琛, 孙成均, 李永新, 杨晓松, 文君, 曾红燕. 市售保健品中7种醒酒护肝功效成分的毛细管电泳测定[J]. 高等学校化学学报, 2016, 37(7): 1276. |
| [11] | 贾长青, 杨冬燕, 车传亮, 马永强, 芮昌辉, 闫晓静, 覃兆海. 1H-1,2,4-三唑-5-氨基甲酸酯类化合物的合成、 结构表征及杀虫、 抑菌活性[J]. 高等学校化学学报, 2016, 37(5): 892. |
| [12] | 陈伟, 魏巍, 刘明, 华学文, 李玉新, 李永红, 张晓, 李正名. 新型含二甲氧基甲基嘧啶基磺酰脲衍生物的合成及生物活性[J]. 高等学校化学学报, 2015, 36(7): 1291. |
| [13] | 陈伟, 魏巍, 李玉新, 万莹莹, 刘巧霞, 李永红, 于淑晶, 李正名. 2-甲基-6-硝基苯磺酰脲衍生物的合成及生物活性[J]. 高等学校化学学报, 2015, 36(5): 907. |
| [14] | 陈伟, 魏巍, 周莎, 李永红, 张晓, 童军, 李玉新, 李正名. 新型含苯基取代嘧啶基磺酰脲衍生物的设计、 合成及生物活性[J]. 高等学校化学学报, 2015, 36(4): 672. |
| [15] | 王娇, 田克情, 薛子桥, 武云云, 杨田, 赵惠敏, 张萍. 4-取代苯基-1,5-苯并硫氮杂䓬-2-甲酸的合成及抑菌活性[J]. 高等学校化学学报, 2015, 36(3): 505. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||