

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (2): 268.doi: 10.7503/cjcu20190448
收稿日期:2019-08-09
出版日期:2020-02-10
发布日期:2019-11-14
通讯作者:
曹忠
E-mail:zhongcao2004@163.com
基金资助:
PENG Yuyu,WANG Yu,YU Xinyao,ZENG Julan,XIAO Zhongliang,CAO Zhong( )
)
Received:2019-08-09
Online:2020-02-10
Published:2019-11-14
Contact:
Zhong CAO
E-mail:zhongcao2004@163.com
Supported by:摘要:
将单(6-巯基-6-去氧)-β-环糊精(HS-β-CD)通过金硫键自组装在金电极(GE)表面, 构建了一种简单、 快速、 灵敏的超分子识别L-半胱氨酸(L-Cys)的电位型电化学传感器. 通过循环伏安法和交流阻抗法研究了膜表面的电化学行为; 通过扫描电子显微镜(SEM)和X射线光电子能谱(XPS)表征了电极表面的膜组装效果, 其作用机制是固定在金电极表面的HS-β-CD空穴可通过分子间作用力吸附结合带负电的L-Cys, 使电极表面的膜电位发生改变, 导致对L-Cys的超分子选择性识别作用, 从而实现对L-Cys的定量分析. 在优化的实验条件下, 该电极在pH=6.0的磷酸盐缓冲溶液中对L-Cys有良好的电位响应性能, 线性范围为1.0×10 -7~1.0×10 -4 mol/L, 斜率为(-65.29±1.0) mV/pc(25 ℃), 检测下限达到6.0×10 -8 mol/L; 电极响应速度快、 稳定性和重现性好、 抗干扰能力强. 将该电极用于实际猪血清和猪尿液样品中L-Cys含量的测定, 回收率为95.0%~104.7%, 表明该新型电极在生命科学等领域具有良好的应用前景.
TrendMD:
彭与煜,王煜,于鑫垚,曾巨澜,肖忠良,曹忠. 基于单(6-巯基-6-去氧)-β-环糊精修饰金电极对L-半胱氨酸的快速灵敏检测. 高等学校化学学报, 2020, 41(2): 268.
PENG Yuyu,WANG Yu,YU Xinyao,ZENG Julan,XIAO Zhongliang,CAO Zhong. Rapid and Sensitive Detection of L-Cysteine Based on Mono(6-mercapto-6-deoxy)-β-cyclodextrin Modified Gold Electrode †. Chem. J. Chinese Universities, 2020, 41(2): 268.
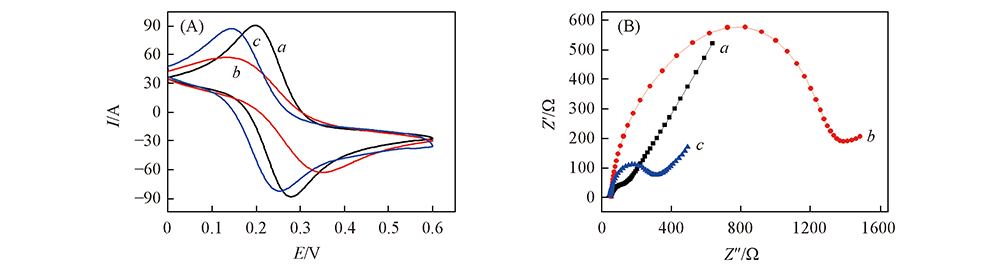
Fig.4 Cyclic voltammetry(A) and impedance(B) curves of bare GE(a), HS-β-CD/GE(b), and L-Cys/HS-β-CD/GE(c) electrodes in the media solution of containing 5.0 mmol/L K3Fe(CN)6/K4Fe(CN)6 and 0.5 mol/L Na2SO4
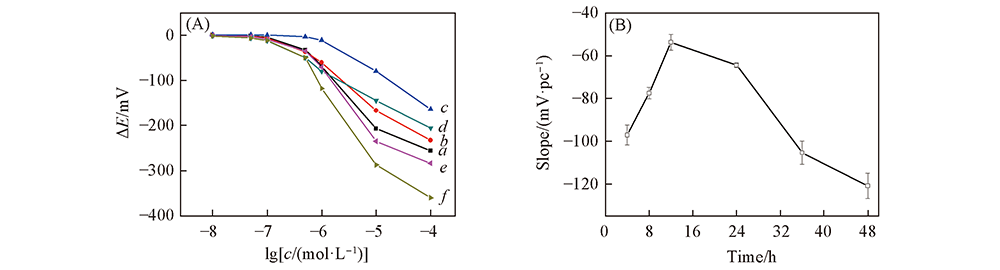
Fig.6 Typical potential response curves to L-Cys at different modification time(A) and effect of electrode sensitivity on modification time(B) (A) Modification time/h: a. 4; b. 8; c. 12; d. 24; d. 36; f. 48.
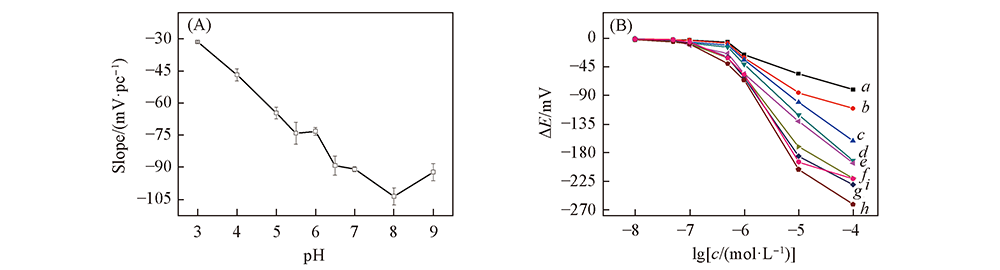
Fig.7 Effect of electrode sensitivity on pH(A) and corresponding potential response curves for L-Cys at different pH values(B) (B) pH value: a. 3; b. 4; c. 5; d. 5.5; e. 6; f. 6.5; g. 7; h. 8 ; i. 9.
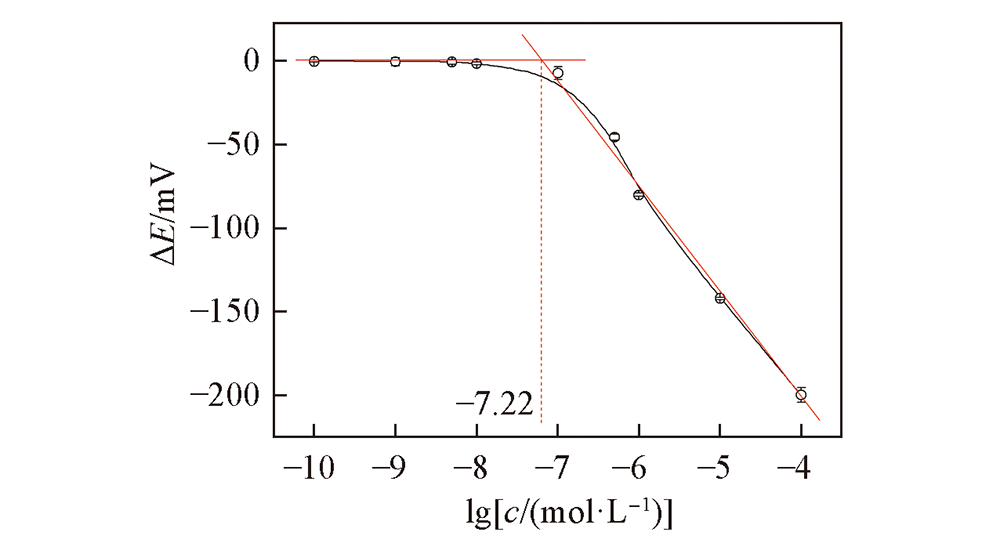
Fig.8 Potential response curve of HS-β-CD/GE to L-Cys in PBS(pH=6.0) with various concentrations The red lines are the fitted curves of potential change vs. logarithmic concentration of L-Cys.
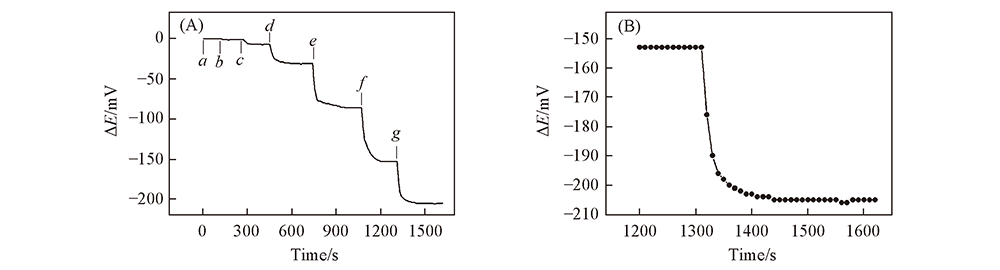
Fig.9 Dynamic curve of HS-β-CD/GE responding to various concentration of L-Cys in PBS(pH=6.0)(A) and enlarged plot of stage G for 1.0×10-4 mol/L of L-Cys(B) c(L-Cys)/( mol·L-1): a. 1.0×10-8; b. 5.0×10-8; c. 1.0×10-7; d. 5.0×10-7; e. 1.0×10-6; f. 1.0×10-5; g. 1.0×10-4.
| No. | Potential/mV | No. | Potential/mV | ||
|---|---|---|---|---|---|
| 1.0×10-5 mol/L | 1.0×10-6 mol/L | 1.0×10-5 mol/L | 1.0×10-6 mol/L | ||
| 1 | -141 | -82 | 6 | -140 | -80 |
| 2 | -142 | -81 | AV | -140.8 | -80.2 |
| 3 | -140 | -79 | SD | ±0.75 | ±1.05 |
| 4 | -141 | -80 | RSD(%) | 0.54 | 1.31 |
| 5 | -141 | -81 | |||
Table 1 Reproducibility of HS-β-CD/GE responding to L-Cys in PBS(pH=6.0)*
| No. | Potential/mV | No. | Potential/mV | ||
|---|---|---|---|---|---|
| 1.0×10-5 mol/L | 1.0×10-6 mol/L | 1.0×10-5 mol/L | 1.0×10-6 mol/L | ||
| 1 | -141 | -82 | 6 | -140 | -80 |
| 2 | -142 | -81 | AV | -140.8 | -80.2 |
| 3 | -140 | -79 | SD | ±0.75 | ±1.05 |
| 4 | -141 | -80 | RSD(%) | 0.54 | 1.31 |
| 5 | -141 | -81 | |||

Fig.10 Effects of interfering substance on HS-β-CD/GE The concentration of L-Cys is 1.0×10-6 mol/L, and the concentrations of other coexisting substances are 1.0×10-4 mol/L. a. L-Cys; b. L-Cys+L-Val; c. L-Cys+L-Trp; d. L-Cys+L-Phe; e. L-Cys+L-Leu; f. L-Cys+L-Thr; g. L-Cys+L-Ala; h. L-Cys+L-Ile; i. L-Cys+L-Gly; j. L-Cys+L-Arg; k. L-Cys+L-Pro; l. L-Cys+L-Cyn; m. L-Cys+L-Met; n. L-Cys+L-His; o. L-Cys+L-Lys; p. L-Cys+Na+; q. L-Cys+K+; r. L-Cys+Ca2+; s. L-Cys+UA; t. L-Cys+GLC.
| Sample | c(L-Cys)a/ (μmol·L-1) | c(L-Cys)b/ (μmol·L-1) | Spiked/ (μmol·L-1) | Found/ (μmol·L-1) | Recovery(%) | RSD(%) |
|---|---|---|---|---|---|---|
| Serum 1 | 0.3213 | 0.3265 | 0.1056 | 0.4371 | 104.7 | 4.2 |
| Serum 2 | 0.4127 | 0.4261 | 0.6065 | 1.0290 | 99.4 | 1.1 |
| Serum 3 | 0.7634 | 0.7528 | 1.0980 | 1.9010 | 104.5 | 1.8 |
| Serum 4 | 0.6516 | 0.6602 | 4.9850 | 5.7010 | 101.1 | 2.5 |
| Serum 5 | 0.1528 | 0.1573 | 9.8870 | 9.8990 | 98.5 | 0.77 |
| Serum 6 | 0.1472 | 0.1396 | 50.12 | 48.58 | 96.6 | 2.9 |
| Urine 1 | —— | —— | 0.1056 | 0.1073 | 101.6 | 5.7 |
| Urine 2 | —— | —— | 0.6065 | 0.6056 | 99.8 | 2.1 |
| Urine 3 | —— | —— | 1.098 | 1.043 | 95.0 | 5.2 |
| Urine 4 | —— | —— | 4.985 | 4.983 | 99.9 | 2.1 |
| Urine 5 | —— | —— | 9.887 | 9.997 | 101.1 | 0.54 |
| Urine 6 | —— | —— | 50.12 | 49.34 | 98.4 | 1.7 |
Table 2 Recovery of the proposed HS-β-CD/GE for determination of L-Cys in pig serum and urine samples
| Sample | c(L-Cys)a/ (μmol·L-1) | c(L-Cys)b/ (μmol·L-1) | Spiked/ (μmol·L-1) | Found/ (μmol·L-1) | Recovery(%) | RSD(%) |
|---|---|---|---|---|---|---|
| Serum 1 | 0.3213 | 0.3265 | 0.1056 | 0.4371 | 104.7 | 4.2 |
| Serum 2 | 0.4127 | 0.4261 | 0.6065 | 1.0290 | 99.4 | 1.1 |
| Serum 3 | 0.7634 | 0.7528 | 1.0980 | 1.9010 | 104.5 | 1.8 |
| Serum 4 | 0.6516 | 0.6602 | 4.9850 | 5.7010 | 101.1 | 2.5 |
| Serum 5 | 0.1528 | 0.1573 | 9.8870 | 9.8990 | 98.5 | 0.77 |
| Serum 6 | 0.1472 | 0.1396 | 50.12 | 48.58 | 96.6 | 2.9 |
| Urine 1 | —— | —— | 0.1056 | 0.1073 | 101.6 | 5.7 |
| Urine 2 | —— | —— | 0.6065 | 0.6056 | 99.8 | 2.1 |
| Urine 3 | —— | —— | 1.098 | 1.043 | 95.0 | 5.2 |
| Urine 4 | —— | —— | 4.985 | 4.983 | 99.9 | 2.1 |
| Urine 5 | —— | —— | 9.887 | 9.997 | 101.1 | 0.54 |
| Urine 6 | —— | —— | 50.12 | 49.34 | 98.4 | 1.7 |
| [1] |
Paul B. D., Sbodio J. I., Snyder S. H., Trends Pharmacol. Sci., 2018,39(5), 513— 524
doi: 10.1016/j.tips.2018.02.007 URL |
| [2] |
Gazit V., Ben-Abraham R., Coleman R., Weizman A., Katz Y ., Amino Acids, 2004,26(2), 163— 168
doi: 10.1007/s00726-003-0045-5 URL |
| [3] |
Harris J. R., Trends Biochem. Sci., 2003,28(1), 32— 40
doi: 10.1016/S0968-0004(02)00003-8 URL |
| [4] |
Ferreira L. G., Andricopulo A. D., Pharmacol. Therap., 2017,180, 49— 61
doi: 10.1016/j.pharmthera.2017.06.004 URL |
| [5] |
Alcock L. J., Perkins M. V., Chalker J. M., Chem. Soc. Rev., 2018,47, 231— 268
doi: 10.1039/C7CS00607A URL |
| [6] |
Pišlar A., Jewett A., Kos J., Semin. Cancer Biol., 2018,53, 168— 177
doi: 10.1016/j.semcancer.2018.07.010 URL |
| [7] |
Maurais A. J., Weerapana E., Curr. Opin. Chem. Biol., 2019,50, 29— 36
doi: 10.1016/j.cbpa.2019.02.010 URL |
| [8] |
Borase H. P., Patil C. D., Salunkhe R. B., Suryawanshi R. K., Kim B. S., Bapat V. A., Patil S. V., Appl. Biochem. Biotech., 2015,175(7), 3479— 3493
doi: 10.1007/s12010-015-1519-0 URL |
| [9] | Yang Z., Zhu Y., Nie G., Li M., Wang C., Lu X ., Dalton Transactions, 2017, 1— 9 |
| [10] |
Bulatov A. V., Petrova A. V., Vishnikin A. B., Moskvin L. N., Microchem. J., 2013,110(2), 369— 373
doi: 10.1016/j.microc.2013.04.020 URL |
| [11] |
Cebi N., Dogan C. E., Develioglu A., Yayla M. E., Sagdic O., Food Chem., 2017,228, 116-124
doi: 10.1016/j.foodchem.2017.01.132 URL |
| [12] |
Xiao Q. Y., Gao H. L., Yuan Q. P., Lu C., Lin J. M., J. Chromatogr. A, 2013,1274, 145— 150
doi: 10.1016/j.chroma.2012.12.016 URL |
| [13] |
Tsikas D., Evans C. E., Denton T. T., Mitschke A., Gutzki F. M., Pinto J. T., Khomenko T., Szabo S., Arthur J. L., Anal. Biochem., 2012,430(1), 4— 15
doi: 10.1016/j.ab.2012.07.022 URL |
| [14] |
Forgacsova A., Galba J., Mojzisova J., Mikus P., Piestansky J., Kovac A ., J. Pharmac. Biomed. Anal., 2019,164, 442— 451
doi: 10.1016/j.jpba.2018.10.053 URL |
| [15] |
Zhang Y., Yao W., Liang D., Sun M., Wang S., Huang D ., Sens. Actuators B Chem., 2018,259, 768— 774
doi: 10.1016/j.snb.2017.12.059 URL |
| [16] |
Gao Y. Q., Tao L., Wang X. T., Li L., Sens. Actuators B Chem., 2017,238, 1293— 1301
doi: 10.1016/j.snb.2016.09.106 URL |
| [17] | Li X. G., Guo B. Q., Qin T. C., Hao J., Yu P., Mao L. Q., Chinese J. Anal. Chem., 2016,44(9), 1465— 1470 |
| ( 李晓钢, 郭彬乾, 秦泰春, 郝洁, 于萍, 毛兰群 . 分析化学, 2016,44(9), 1465— 1470) | |
| [18] |
Fan Y., Liu J. T., Wang Y., Luo J. P., Xu H. R., Xu S. W., Cai X. X., Biosens. Bioelectron., 2017,95, 60— 66
doi: 10.1016/j.bios.2017.04.003 URL |
| [19] |
Sheng Y. Y., You Y., Cao Z., Liu L., Wu H. C., Analyst, 2018,143, 2411— 2415
doi: 10.1039/C8AN00580J URL |
| [20] |
You Y., Zhou K., Guo B. Y., Liu Q. S., Cao Z., Liu L., Wu H. C., ACS Sens., 2019,4, 774— 779
doi: 10.1021/acssensors.9b00408 URL |
| [21] | Cao Z., Li W. F., Liu C., Peng Y. Y., Huang Y., Xiao Z. L., Chinese J. Anal. Chem., 2019,47(2), 229— 236 |
| ( 曹忠, 李文锋, 彭与煜, 刘陈, 黄颖, 肖忠良 . 分析化学, 2019,47(2), 229— 236) | |
| [22] | Xiao S. F., Wang Z. H., Luo G. A., Wang Y. M., Chem. J. Chinese Universities, 2004,25(10), 1833— 1835 |
| ( 肖素芳, 王宗花, 罗国安, 王义明 . 高等学校化学学报, 2004,25(10), 1833— 1835) | |
| [23] | Wu S., Lan X., Huang F., Luo Z., Ju H., Meng C., Duan C., Biosens. Bioelectron ., 2012,32(1), 293— 296 |
| [24] | Yang J., Zhang Y. Y., Liu C., Li J. X., Xiao Z. L., Li D., Zhang L., Cao Z., Chem. J. Chinese Universities, 2018,39(11), 2386— 2394 |
| ( 杨佳, 张煜杨, 刘陈, 李佳欣, 肖忠良, 李丹, 张玲, 曹忠 . 高等学校化学学报, 2018,39(11), 2386— 2394) | |
| [25] | Li Y. Q., Zhu Q., Xiao Z. L., Lü C. Z., Feng Z. M., Yin Y. L., Cao Z., Chem. J. Chinese Universities, 2018,39(4), 636— 644 |
| ( 李雨晴, 朱钦, 肖忠良, 吕超志, 冯泽猛, 印遇龙, 曹忠 . 高等学校化学学报, 2018,39(4), 636— 644) | |
| [26] | Wang Y., Li D., Kang J., Guan S., Wu D., Int. J. Electrochem. Sci., 2019,14, 5448— 5461 |
| [27] |
Zhu Q., Liu C., Zhou L., Wu L., Bian K., Zeng J., Wang J., Feng Z., Yin Y., Cao Z., Biosens. Bioelectron., 2019,140, 111356
doi: 10.1016/j.bios.2019.111356 URL |
| [28] |
Selvarajan S., Alluri N. R., Chandrasekhar A., Kim S. J., Biosens. Bioelectron., 2017,91, 203— 210
doi: 10.1016/j.bios.2016.12.006 URL |
| [29] |
Amini M. K., Khorasani J. H., Khaloo S. S., Tangestaninejad S., Anal. Biochem., 2003,320(1), 32— 38
doi: 10.1016/S0003-2697(03)00355-5 URL |
| [30] |
Bai Y., Xu J., Chen H., Biosens. Bioelectron., 2009,24(10), 2985— 2990
doi: 10.1016/j.bios.2009.03.008 URL |
| [31] |
Bakker E., Anal. Chem., 2016,88(1), 395— 413
doi: 10.1021/acs.analchem.5b04034 URL |
| [32] |
Ma Y. H., Yuan R., Chai Y. Q., Wu X., Zhou W., Liu X. L., Deng F., Anal. Lett., 2009,42(15), 2411— 2429
doi: 10.1080/00032710903061147 URL |
| [33] |
Cao Z., Gong F. C., Li H. P., Xiao Z. L., Long S., Zhang L., Peng S. J., Anal. Chim. Acta, 2007,581(1), 19— 26
doi: 10.1016/j.aca.2006.08.003 URL |
| [34] | Cao T. T., Cao Z., He J. L., Liang H. Q., Xiao Z. L., Chem. J. Chinese Universities, 2014,35(7), 1388— 1395 |
| ( 曹婷婷, 曹忠, 何婧琳, 梁海琴, 肖忠良 . 高等学校化学学报, 2014,35(7), 1388— 1395) | |
| [35] |
Li X. Q., Liang H. Q., Cao Z., Xiao Q., Xiao Z. L., Song L. B., Chen D., Wang F. L., Mater. Sci. Eng. C, 2017,72, 26— 33
doi: 10.1016/j.msec.2016.11.034 URL |
| [36] |
Shahrokhian S., Anal. Chem., 2001,73(24), 5972— 5978
doi: 10.1021/ac010541m URL |
| [37] |
Cuartero M., Pérez S., García M. S., Talanta, 2018,180, 316— 322
doi: 10.1016/j.talanta.2017.12.029 URL |
| [38] |
Wang Z. Z., Tang W. X., Yu J., Zhang F., He P. G., J. Electroanal. Chem., 2019,835, 137— 142
doi: 10.1016/j.jelechem.2019.01.031 URL |
| [39] | Chen Y., Liu Y., Chem. Soc. Rev., 2010,41(22), 495— 505 |
| [40] |
Bellia F., La M. D., Pedone C., Rizzarelli E., Saviano M., Vecchio G., Chem. Soc. Rev., 2009,38(9), 2756— 2781
doi: 10.1039/b718436k URL |
| [41] |
Morin-Crini N., Winterton P., Fourmentin S., Wilson L. D., Fenyvesi É., Crini G., Prog. Polym. Sci., 2018,78, 1— 23
doi: 10.1016/j.progpolymsci.2017.07.004 URL |
| [42] |
Feng L., Tong C., He Y., Liu B., Wang C., Sha J., Lü C ., J. Lumin., 2014,146, 502— 507
doi: 10.1016/j.jlumin.2013.10.039 URL |
| [43] |
Ma M., Zhe T., Song W., Guo P., Wang J., Wang J ., Sens. Actuators B: Chem., 2017,253, 818— 829
doi: 10.1016/j.snb.2017.07.003 URL |
| [44] |
Seidi F., Shamsabadi A. A., Amini M., Shabaniand M., Crespy D., Polym. Chem., 2019,10, 3674— 3711
doi: 10.1039/C9PY00495E URL |
| [45] |
Pawar S., Shende P., Trotta F., Int. J. Pharm., 2019,565, 333— 350
doi: 10.1016/j.ijpharm.2019.05.015 URL |
| [46] |
Staden S. V., Holo L., Sens. Actuators B: Chem., 2004,98(1), 97— 100
doi: 10.1016/j.snb.2003.09.029 URL |
| [47] |
Funasaki N ., J. Inclusion Phenom. Macrocyclic Chem., 2004,50(1/2), 43— 50
doi: 10.1007/s10847-004-8837-5 URL |
| [48] |
Cao Z., Zhang L., Guo C., Gong F. C., Long S., Tan S. Z., Xia C. B., Xu F., Sun L. X., Mater. Sci. Eng. C, 2009,29(3), 1051— 1056
doi: 10.1016/j.msec.2008.09.010 URL |
| [49] | Zhao C. F., Zhou X. Y., Zhang W. M., Zhao W. K., Instrumental Analysis, Higher Education Press, Beijing, 1990,284 |
| ( 赵藻藩, 周性尧, 张悟铭, 赵文宽. 仪器分析, 北京: 高等教育出版社., 1990,284) | |
| [50] |
Liang R., Ding J., Gao S., Qin W., Angew. Chem. Int. Ed., 2017,56, 6833— 6837
doi: 10.1002/anie.201701892 URL |
| [1] | 王博东, 潘美辰, 卓颖. 二氧化硅纳米颗粒表面原位还原银纳米簇电化学发光传感界面的构建与分子识别[J]. 高等学校化学学报, 2021, 42(11): 3519. |
| [2] | 付可飞, 连惠婷, 魏晓峰, 孙向英, 刘斌. 环糊精基阻抗型传感器的制备及对L-半胱氨酸的识别[J]. 高等学校化学学报, 2020, 41(4): 706. |
| [3] | 李逊, 薛玉瑞, 宋宇, 张文科. 硫醚-金配位相互作用的单分子力谱研究[J]. 高等学校化学学报, 2018, 39(12): 2774. |
| [4] | 杨佳, 张煜杨, 刘陈, 李佳欣, 肖忠良, 李丹, 张玲, 曹忠. 基于聚二硫二丙烷磺酸膜修饰FET延长栅极的L-胱氨酸传感器[J]. 高等学校化学学报, 2018, 39(11): 2386. |
| [5] | 周心慧, 王海水. 利用L-半胱氨酸自组装膜和混合溶剂协同作用手性拆分DL-谷氨酸[J]. 高等学校化学学报, 2017, 38(6): 1076. |
| [6] | 李映图, 李理波, 周健. 分子动力学模拟多巴在自组装膜上的黏附性[J]. 高等学校化学学报, 2017, 38(5): 798. |
| [7] | 张鲁格, 薛泽旭, 张翀, 延辉. 分子动力学模拟3(5)-(9-蒽基)吡唑分子在自组装膜上的选择性沉积[J]. 高等学校化学学报, 2016, 37(3): 505. |
| [8] | 曹婷婷, 曹忠, 何婧琳, 梁海琴, 肖忠良. 一种简单快速检测汞离子的1,4-二硫苏糖醇膜修饰金平板电极[J]. 高等学校化学学报, 2014, 35(7): 1388. |
| [9] | 周婷, 曹忠, 戴云林, 曹婷婷, 何婧琳, 徐雷涛, 龙姝. 基于氢键作用的杯芳烃超分子识别乙醇的传感机理及分析应用[J]. 高等学校化学学报, 2013, 34(6): 1339. |
| [10] | 李聪聪, 孙向英, 杨传孝. 基于石墨烯和量子点自组装膜的能量转移构筑的高灵敏度DNA界面荧光传感[J]. 高等学校化学学报, 2012, 33(07): 1367. |
| [11] | 杨胜园, 谭慧, 刘艳梅, 吴朝阳, 沈国励, 俞汝勤. 非标记液晶型免疫传感器检测赭曲霉素A[J]. 高等学校化学学报, 2011, 32(5): 1065. |
| [12] | 沈广宇, 沈国励. 基于网状混合自组装膜的压电免疫传感器及其应用[J]. 高等学校化学学报, 2010, 31(11): 2196. |
| [13] | 汪志芳, 孙向英, 刘辉, 刘斌. 新型“内标”式双重荧光自组装膜的制备和DNA的界面传感[J]. 高等学校化学学报, 2008, 29(9): 1747. |
| [14] | 王宜冰,夏即雅,高丽华,王科志 . 三种双偶极半菁衍生物与H3PMo12O40自组装多层膜的研究[J]. 高等学校化学学报, 2007, 28(6): 1175. |
| [15] | 程昱川, 王蕴馨, 宋薇, 于苗, 徐蔚青, 赵冰 . 表面增强拉曼光谱研究基于轴向配位键结合的金属卟啉自组装膜[J]. 高等学校化学学报, 2007, 28(1): 121. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||