

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (11): 2363.doi: 10.7503/cjcu20180347
收稿日期:2018-05-07
出版日期:2018-11-10
发布日期:2018-09-29
作者简介:联系人简介: 李 夏, 女, 博士, 教授, 博士生导师, 主要从事无机配位化学方面的研究. E-mail: 基金资助:Received:2018-05-07
Online:2018-11-10
Published:2018-09-29
Contact:
LI Xia
E-mail:xiali@cnu.edu.cn
Supported by:摘要:
选用2-(3'-羧基苯氧基)苯甲酸(2,3'-H2oba)、 1,3-双(4-吡啶基)-丙烷(DPP)和1,4-二(1,2,4-三氮唑-1-)丁烷(BTB)为配体, 采用水热法得到了过渡金属配合物Cd(2,3'-oba)(DPP)(1)和Zn(2,3'-oba)(BTB)0.5(2). 配合物1为二维网状结构, 中心Cd2+的配位环境为{CdO4N2}, 双齿螯合配位的2,3'-oba和单齿桥联配位的DPP将Cd2+连接成Cd2+-2,3'-oba-Cd2+-DPP两条螺旋链. 配合物1由分子间氢键连接并自组装为三维超分子结构. 配合物2为三维框架结构, 中心Zn2+采取五配位的畸变四方锥构型{ZnO4N}. 2,3'-oba配体以双齿螯合与双齿桥联的配位方式连接相邻Zn2+并形成二维网层, BTB分子桥联Zn2+并连接相邻网层进而形成三维结构. 荧光光谱表明配合物1和2分别在437和359 nm存在最大发射, 均来源于配体的π*-π 跃迁. 广谱型农药甲基磺草酮对配合物2的荧光强度具有明显的猝灭作用, 据此配合物2可作为荧光探针检测甲基磺草酮.
中图分类号:
TrendMD:
李铮, 李睿, 李夏. 2-(3'-羧基苯氧基)苯甲酸和含氮配体构筑的过渡金属配合物的合成、 晶体结构及荧光性质. 高等学校化学学报, 2018, 39(11): 2363.
LI Zheng, LI Rui, LI Xia. Synthesis, Crystal Structure and Fluorescent Properties of Transition Metal Complexes Constructed with 2-(3'-Carboxyphenoxy)benzoic Acid and N-Donor Ligands†. Chem. J. Chinese Universities, 2018, 39(11): 2363.
| Complex | 1 | 2 |
|---|---|---|
| Empirical formula | C27H22CdN2O5 | C18H14N3O5Zn |
| Formula weight | 566.87 | 417.69 |
| Temperature/K | 293(2) | 296(2) |
| Crystal system | Monoclinic | Monoclinic |
| Space group | P21/c | C2/c |
| a/nm | 1.0091(2) | 1.6159(5) |
| b/nm | 1.3008(3) | 1.3546(7) |
| c/nm | 2.0610(6) | 1.6657(8) |
| α/(°) | 90 | 90 |
| β/(°) | 116.83(2) | 113.59(2) |
| γ/(°) | 90 | 90 |
| V/nm3 | 2.4141(10) | 3.3418(31) |
| Z | 4 | 8 |
| Dc/(Mg·m-3) | 1.560 | 1.660 |
| Absorption coefficient/mm-1 | 0.945 | 1.507 |
| F(000) | 1144 | 1704 |
| Crystal size/mm | 0.44×0.31×0.22 | 0.37×0.33×0.29 |
| θ range for data collection/(°) | 3.32—27.63 | 2.89—27.56 |
| Limiting indices | -13≤h≤13, -16≤k≤16, -26≤l≤26 | -20≤h≤20, -17≤k≤17, -21≤l≤21 |
| Reflections collected | 36889 | 25433 |
| Rint | 0.0599 | 0.0849 |
| Data/restraints/parameters | 5569/0/316 | 3853/8/244 |
| Goodness-of-fit on F2 | 1.015 | 1.026 |
| Final R indices [I>2σ(I)] | R1=0.0471, wR2=0.0996 | R1=0.0494, wR2=0.0886 |
| R indices(all data) | R1=0.1140, wR2=0.1220 | R1=0.1004, wR2=0.1028 |
| CCDC No. | 1437942 | 1817762 |
Table 1 Crystallographic data of complexes 1 and 2
| Complex | 1 | 2 |
|---|---|---|
| Empirical formula | C27H22CdN2O5 | C18H14N3O5Zn |
| Formula weight | 566.87 | 417.69 |
| Temperature/K | 293(2) | 296(2) |
| Crystal system | Monoclinic | Monoclinic |
| Space group | P21/c | C2/c |
| a/nm | 1.0091(2) | 1.6159(5) |
| b/nm | 1.3008(3) | 1.3546(7) |
| c/nm | 2.0610(6) | 1.6657(8) |
| α/(°) | 90 | 90 |
| β/(°) | 116.83(2) | 113.59(2) |
| γ/(°) | 90 | 90 |
| V/nm3 | 2.4141(10) | 3.3418(31) |
| Z | 4 | 8 |
| Dc/(Mg·m-3) | 1.560 | 1.660 |
| Absorption coefficient/mm-1 | 0.945 | 1.507 |
| F(000) | 1144 | 1704 |
| Crystal size/mm | 0.44×0.31×0.22 | 0.37×0.33×0.29 |
| θ range for data collection/(°) | 3.32—27.63 | 2.89—27.56 |
| Limiting indices | -13≤h≤13, -16≤k≤16, -26≤l≤26 | -20≤h≤20, -17≤k≤17, -21≤l≤21 |
| Reflections collected | 36889 | 25433 |
| Rint | 0.0599 | 0.0849 |
| Data/restraints/parameters | 5569/0/316 | 3853/8/244 |
| Goodness-of-fit on F2 | 1.015 | 1.026 |
| Final R indices [I>2σ(I)] | R1=0.0471, wR2=0.0996 | R1=0.0494, wR2=0.0886 |
| R indices(all data) | R1=0.1140, wR2=0.1220 | R1=0.1004, wR2=0.1028 |
| CCDC No. | 1437942 | 1817762 |
| Cd1—O4 | 0.2291(3) | Cd1—O5 | 0.2352(3) | Cd1—O1A | 0.2326(4) | ||
|---|---|---|---|---|---|---|---|
| Cd1—N1 | 0.2312(4) | Cd1—N2 | 0.2296(3) | Cd1—O2A | 0.2373(4) | ||
| O4—Cd1—N2 | 93.79(13) | O4—Cd1—O2A | 93.50(16) | N1—Cd1—O1A | 85.48(14) | ||
| N2—Cd1—N1 | 91.38(13) | N1—Cd1—O2A | 131.47(15) | N2—Cd1—O5 | 134.43(13) | ||
| N2—Cd1—O1A | 116.67(17) | O5—Cd1—O2A | 123.62(17) | O1A—Cd1—O5 | 108.88(17) | ||
| O4—Cd1—O5 | 55.22(11) | O4—Cd1—N1 | 134.98(13) | N2—Cd1—O2A | 86.60(16) | ||
| N1—Cd1—O5 | 90.89(12) | O4—Cd1—O1A | 130.13(17) | O1A—Cd1—O2A | 53.49(14) | ||
Table 2 Selected bond lengths(nm) and angles(°) for complex 1*
| Cd1—O4 | 0.2291(3) | Cd1—O5 | 0.2352(3) | Cd1—O1A | 0.2326(4) | ||
|---|---|---|---|---|---|---|---|
| Cd1—N1 | 0.2312(4) | Cd1—N2 | 0.2296(3) | Cd1—O2A | 0.2373(4) | ||
| O4—Cd1—N2 | 93.79(13) | O4—Cd1—O2A | 93.50(16) | N1—Cd1—O1A | 85.48(14) | ||
| N2—Cd1—N1 | 91.38(13) | N1—Cd1—O2A | 131.47(15) | N2—Cd1—O5 | 134.43(13) | ||
| N2—Cd1—O1A | 116.67(17) | O5—Cd1—O2A | 123.62(17) | O1A—Cd1—O5 | 108.88(17) | ||
| O4—Cd1—O5 | 55.22(11) | O4—Cd1—N1 | 134.98(13) | N2—Cd1—O2A | 86.60(16) | ||
| N1—Cd1—O5 | 90.89(12) | O4—Cd1—O1A | 130.13(17) | O1A—Cd1—O2A | 53.49(14) | ||
| Zn1—O3 | 0.1987(2) | Zn1—N3 | 0.2025(2) | Zn1—O4 | 0.2007(2) | ||
|---|---|---|---|---|---|---|---|
| Zn1—O1 | 0.2002(3) | Zn1—O2 | 0.2303(2) | ||||
| O3—Zn1—O1 | 108.62(10) | O4—Zn1—O2 | 144.29(10) | O4—Zn1—N3 | 93.46(9) | ||
| O1—Zn1—O4 | 95.22(9) | O3—Zn1—O4 | 117.10(9) | O1—Zn1—O2 | 60.02(10) | ||
| O1—Zn1—N3 | 145.90(11) | O3—Zn1—N3 | 96.40(10) | N3—Zn1—O2 | 95.33(10) | ||
| O3—Zn1—O2 | 96.21(9) | ||||||
Table 3 Selected bond lengths(nm) and angles(°) for complex 2
| Zn1—O3 | 0.1987(2) | Zn1—N3 | 0.2025(2) | Zn1—O4 | 0.2007(2) | ||
|---|---|---|---|---|---|---|---|
| Zn1—O1 | 0.2002(3) | Zn1—O2 | 0.2303(2) | ||||
| O3—Zn1—O1 | 108.62(10) | O4—Zn1—O2 | 144.29(10) | O4—Zn1—N3 | 93.46(9) | ||
| O1—Zn1—O4 | 95.22(9) | O3—Zn1—O4 | 117.10(9) | O1—Zn1—O2 | 60.02(10) | ||
| O1—Zn1—N3 | 145.90(11) | O3—Zn1—N3 | 96.40(10) | N3—Zn1—O2 | 95.33(10) | ||
| O3—Zn1—O2 | 96.21(9) | ||||||
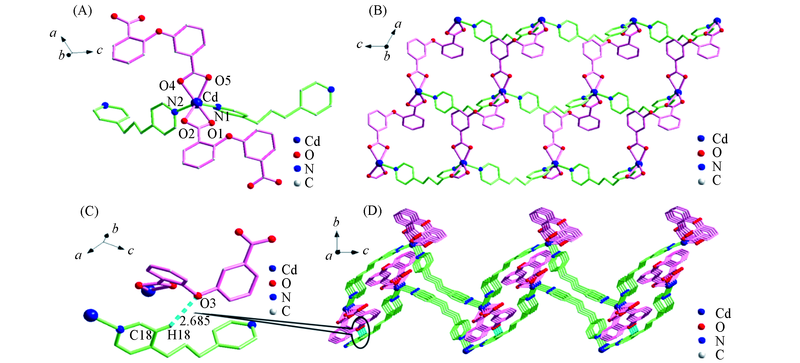
Fig.1 Coordination environment of Cd(Ⅱ)(A), 2D structure(B), hydrogen bond(C) and 3D structure(D) of complex 1All hydrogen atoms are omitted for clarity; symmetry code: A: x-1, y, z.
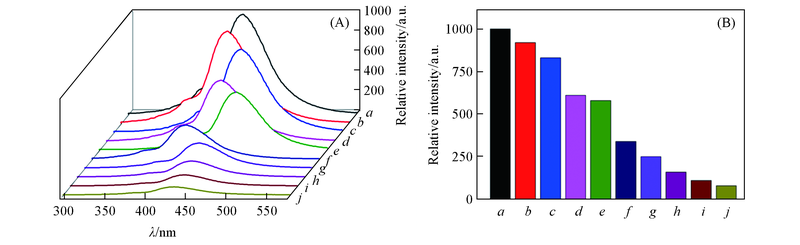
Fig.5 Emission spectra(A) and fluorescence intensity at 437 nm(B) of complex 1 in various solventsSolvent from a to j: methanol; water; ethanol; DMF; ethyl acetate; DMSO; chloroform; acetonitrile;dichloromethane; acetone.
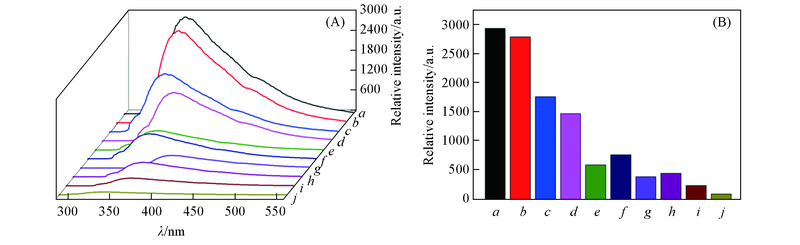
Fig.6 Emission spectra(A) and fluorescence intensity at 359 nm(B) of complex 2 in various solventsSolvent from a to j: methanol; water; ethanol; DMF; ethyl acetate; DMSO; chloroform; acetonitrile; dichloromethane; acetone.
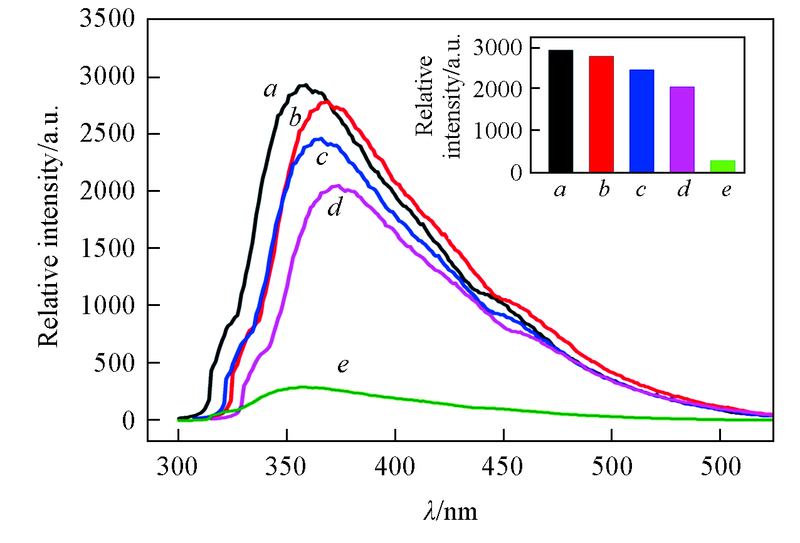
Fig.8 Emission spectra and peak intensity(inset) of complex 2 dispersed in methanol containing hymexazol(b), carbendazim(c), tebuconazole(d) and mesotrione(e)a. Blank.

Fig.9 Emission spectra and peak intensity(inset) of complex 2 dispersed in methanol solvent containing mesotrione with various concentrationsc(Mesotrione)/(mol·L-1) for a—j: 0, 2.5×10-7, 5×10-7, 10-6, 2.5×10-6, 5×10-6, 10-5, 2.5×10-5, 5×10-5, 1×10-4.
| [1] | Zhang X., Wang W., Hu Z., Wang G., Uvdal K., Coordination Chemistry Reviews, 2015, 284, 206—235 |
| [2] | Wu M. X., Gao J., Wang F., Yang J., Song N., Jin X., Mi P., Tian J., Luo J., Liang F., Yang Y. W., Small, 2018, 14(17), e1704440 |
| [3] | Vinogradov A. V., Zaake-Hertling H., Hey-Hawkins E., Agafonov A. V., Seisenbaeva G. A., Kessler V. G., Vinogradov V. V., Chemical Communications, 2014, 50, 10210—10213 |
| [4] | He Y., Zhou W., Qian G., Chen B., Chemical Society Reviews, 2014, 43, 5657—5678 |
| [5] | Wang S., Shan L., Fan Y., Jia J., Xu J., Wang L., Journal of Solid State Chemistry, 2017, 245, 132—137 |
| [6] | Chen Y. Z., Wang Z. U., Wang H., Lu J., Yu S. H., Jiang H. L., Journal of the American Chemical Society, 2017, 139, 2035—2044 |
| [7] | Wang H.J., Zhu G. S., Zhang K. Y., Sun F. X., Qiu S. L., Chem. [J]. Chinese Universities, 2009, 30(1), 11—13 |
| (王海君, 朱广山, 张可勇, 孙福兴, 裘式纶. 高等学校化学学报, 2009, 30(1), 11—13) | |
| [8] | Hu Z., Deibert B. J., Li J., Chemical Society Reviews, 2014, 43, 5815—5840 |
| [9] | Zhao S. N., Song X. Z., Song S. Y., Zhang H. J., Coordination Chemistry Reviews, 2017, 337, 80—96 |
| [10] | Zhang Z., Zhao Y., Gong Q., Li Z., Li J., Chemical Communications, 2013, 49, 653—661 |
| [11] | Ahn D. Y., Lee D. Y., Shin C. Y., Bui H. T., Shrestha N. K., Giebeler L., Noh Y. Y., Han S. H., ACS Applied Materials & Interfaces, 2017, 9, 12930—12935 |
| [12] | Coronado E., MinguezEspallargas G., Chemical Society Reviews, 2013, 42, 1525—1539 |
| [13] | Heine J., Muller-Buschbaum K., Chemical Society Reviews, 2013, 42, 9232—9242 |
| [14] | Yi F. Y., Gu M., Wang S. C., Zheng J. Q., Pan L., Han L., Inorganic Chemistry, 2018, 57, 2654—2662 |
| [15] | Schoedel A., Li M., Li D., O’Keeffe M., Yaghi O. M., Chemical Reviews, 2016, 116, 12466—12535 |
| [16] | Fan K., Bao S. S., Nie W. X., Liao C. H., Zheng L. M., Inorganic Chemistry, 2018, 57, 1079—1089 |
| [17] | Yi F. Y., Chen D. X., Wu M. K., Han L., Hai L. J., Chem. Plus. Chem, 2016, 81, 675—690 |
| [18] | Karmakar A., Desai A. V., Ghosh S. K., Coordination Chemistry Reviews, 2016, 307, 313—341 |
| [19] | Guo L., Wang M., Cao D., Small, 2018, 14(17), e1703822 |
| [20] | Devkota J., Kim K. J., Ohodnicki P., Culp J. T., Greve D., Lekse J. W., Nanoscale, 2018, 10(17), 8075—8087 |
| [21] | Xu X., Guo Y., Wang X., Li W., Qi P., Wang Z., Wang X., Gunasekaran S., Wang Q., Sensors and Actuators B: Chemical, 2018, 260, 339—345 |
| [22] | Wen L., Xu X., Lv K., Huang Y., Zheng X., Zhou L., Sun R., Li D., ACS Applied Materials & Interfaces, 2015, 7, 4449—4455 |
| [23] | Wang C., Tian L., Zhu W., Wang S., Wang P., Liang Y., Zhang W., Zhao H., Li G., ACS Applied Materials & Interfaces, 2017, 9, 20076—20085 |
| [24] | Wang K., Tian X., JinY., Sun J., Zhang Q., Crystal Growth & Design, 2017, 17, 1836—1842 |
| [25] | Xue L. P., Chang X. H., Li S. H., Ma L. F., Wang L. Y., Dalton Transactions, 2014, 43, 7219—7226 |
| [26] | Wang H. H., Yang H. Y., Shu C. H., Chen Z. Y., Hou L., Wang Y. Y., Crystal Growth & Design, 2016, 16, 5394—5402 |
| [27] | Hu X. L., Liu F. H., Qin C., Shao K. Z., Su Z. M., Dalton Transactions, 2015, 44, 7822—7827 |
| [28] | Wang Q.S., Zheng X. L., Qu X. L., Li R., Li X., Chem. [J]. Chinese Universities, 2017, 38(7), 1125—1133 |
| (王秋爽, 郑晓丽, 屈相龙, 李睿, 李夏. 高等学校化学学报, 2017, 38(7), 1125—1133) | |
| [29] | Xu X., Guo Y., Wang X., Li W., Qi P., Wang Z., Wang X., Gunasekaran S., Wang Q., Sensors and Actuators B: Chemical, 2018, 260, 339—345 |
| [30] | Yang Q., Wang J., Chen X., Yang W., Pei H., Hu N., Li Z., Suo Y., Li T., Wang J., Journal of Materials Chemistry A, 2018, 6, 2184—2192 |
| [31] | Sun P., Gao Y. L., Xu C., Lian Y. F., RSC Advances, 2018, 8, 10277—10283 |
| [32] | Liao Y., Berthion J. M., Colet I., Merlo M., Nougadere A., Hu R., Journal of Chromatography A, 2018, 1549, 31—38 |
| [33] | Jin D., Gong A., Zhou H., RSC Advances, 2017, 7, 17489—17496 |
| [34] | Zhao Y., Xu X., Qiu L., Kang X., Wen L., Zhang B., ACS Applied Materials & Interfaces, 2017, 9, 15164—15175 |
| [35] | Fan L., Li D., Wei P., Tang P., Li M., Yuan D., Liu G., Zhang X., Dou J., Journal of Coordination Chemistry, 2011, 64, 3031—3040 |
| [36] | Carlucci L., Ciani G., Proserpio D. M., Rizzato S., Cryst. Eng. Comm., 2002, 4, 121—129 |
| [37] | Li X., Gu Y. K., Deng X. B., Zhao K, Jin L. P., Cryst. Eng. Comm., 2011, 13, 6665—6673 |
| [38] | Sheldrick G. M., SHELXL-97, Program for Crystal Structure Solution, University of Göttingen, Göttingen, 1997 |
| [39] | Sheldrick G. M., SHELXS-97, Program for Crystal Structure Refinement, University of Göttingen, Göttingen, 1997 |
| [40] | Guo J., Ma J. F., Liu B., Kan W. Q., Yang J., Crystal Growth & Design, 2011, 11, 3609—3621 |
| [41] | Zhang L. P., Ma J. F., Yang J., Pang Y. Y., Ma J. C., Inorganic Chemistry, 2010, 49, 1535—1550 |
| [42] | Pramanik S., Zheng C., Zhang X., Emge T. J., Li J., Journal of the American Chemical Society, 2011, 133, 4153—4155 |
| [1] | 施耐克, 张娅, SANSON Andrea, 王蕾, 陈骏. Zn(NCN)单轴的负热膨胀性及机理研究[J]. 高等学校化学学报, 2022, 43(6): 20220124. |
| [2] | 李巧, 赵洋, 王恩举. 基于芳叉丙二腈的高活性迈克尔系统的吸湿反应及荧光性质[J]. 高等学校化学学报, 2022, 43(3): 20210690. |
| [3] | 岳胜利, 武光宝, 李星, 李康, 黄高胜, 唐翌, 周惠琼. 准二维钙钛矿太阳能电池的研究进展[J]. 高等学校化学学报, 2021, 42(6): 1648. |
| [4] | 田霞,杨福群,袁伟,赵雷,姚雷,甄小丽,韩建荣,刘守信. 含噁二唑大环冠醚的合成、 结构及金属离子识别性能[J]. 高等学校化学学报, 2020, 41(3): 490. |
| [5] | 刘东枚,苏雅静,李姗姗,许奇炜,李夏. 4-(4-羧基苯氧基)间苯二甲酸构筑的过渡金属配位聚合物: 合成、 晶体结构、 荧光传感与光催化[J]. 高等学校化学学报, 2020, 41(2): 253. |
| [6] | 秦刘磊,刘洋,关小琴,郑晓媛,张子钰,刘尊奇. 无机-有机杂化化合物[(H2DABCO)CuCl4]·H2O的合成及开关型介电性质[J]. 高等学校化学学报, 2020, 41(1): 70. |
| [7] | 李冰, 王学敏, 白凤英, 刘淑清. 稀土氮杂环配合物的合成、 结构及抑菌活性[J]. 高等学校化学学报, 2019, 40(4): 632. |
| [8] | 王冬梅, 刘子华, 李光华, 刘云凌, 李春霞. 铟基双金属配位聚合物的合成、 结构及荧光性质[J]. 高等学校化学学报, 2018, 39(9): 1886. |
| [9] | 田欢, 张梦龙, 王莉莎, 童碧海, 赵卓. 4,13-二硫杂苯并-18-冠-6的合成及对Ag+的选择性萃取[J]. 高等学校化学学报, 2018, 39(6): 1191. |
| [10] | 赵国政, 范荣荣, 颜熹琳, 唐维, 唐明峰, 贾建峰, 武海顺. 高压下晶体5-硝胺基-3,4-二硝基吡唑肼结构转变的周期性密度泛函理论研究[J]. 高等学校化学学报, 2018, 39(2): 292. |
| [11] | 陈奇丹, 唐俊杰, 方千荣. 高稳定性的含氟多级孔共价有机框架材料[J]. 高等学校化学学报, 2018, 39(11): 2357. |
| [12] | 贠吉星, 胡志莉, 李禹蒙, 金晶, 陈冲, 鄢欣, 刘永华, 丁榆, 迟玉贤, 牛淑云. 由芳香羧酸构筑的系列Ni(Ⅱ)配合物的合成、 结构及光电性能[J]. 高等学校化学学报, 2018, 39(10): 2161. |
| [13] | 王秋爽, 郑晓丽, 屈相龙, 李睿, 李夏. 1,3-二(4-吡啶基)-丙烷与邻苯二甲酸构筑的过渡金属配合物的合成、结构和荧光性质[J]. 高等学校化学学报, 2017, 38(7): 1125. |
| [14] | 辛丙靖, 李鹏, 罗力莎, 夏添, 李光华. 一维链状卤化亚铜化合物的溶剂热原位合成、 结构及性质[J]. 高等学校化学学报, 2017, 38(4): 530. |
| [15] | 赵邦屯, 马书修, 陶晶晶, 朱卫民. 含吡啶基四硫富瓦烯衍生物的合成、 结构和电化学性质[J]. 高等学校化学学报, 2017, 38(2): 193. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
