

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (6): 20210842.doi: 10.7503/cjcu20210842
宋有为, 安江伟, 王征, 王旭慧, 权燕红, 任军, 赵金仙( )
)
收稿日期:2021-12-20
出版日期:2022-06-10
发布日期:2022-04-04
通讯作者:
赵金仙
E-mail:zhaojinxian@tyut.edu.cn
基金资助:
SONG Youwei, AN Jiangwei, WANG Zheng, WANG Xuhui, QUAN Yanhong, REN Jun, ZHAO Jinxian( )
)
Received:2021-12-20
Online:2022-06-10
Published:2022-04-04
Contact:
ZHAO Jinxian
E-mail:zhaojinxian@tyut.edu.cn
Supported by:摘要:
采用密度泛函理论方法, 构建了Ag, Zn, Pd原子掺杂的Cu(111)和Cu2O(111)活性晶面, 探讨了不同金属掺杂对Cu(111)和Cu2O(111)催化剂的草酸二甲酯(DMO)加氢反应活性和选择性的影响. 研究结果显示, 掺杂Zn可有效阻止乙醇酸甲酯(MG)进一步加氢, 提高MG的选择性, Ag助剂可以有效提高加氢活性; 而Pd助剂的添加使MG的生成能垒增高, 降低了MG的选择性. Ag-Cu(111)表面具有适宜的d带中心, 生成CH3OOCCH2OH的活性最高. 在Ag, Zn, Pd原子掺杂的Cu2O(111)表面, Ag-Cu2O(111)能带带隙小、 价带强度高, 在DMO加氢反应中具有最佳的催化活性. 基于上述结果, 提出铜基催化剂结构调变和性能调控的理论方法, 为高效催化剂的设计提供可靠的理论指导.
中图分类号:
TrendMD:
宋有为, 安江伟, 王征, 王旭慧, 权燕红, 任军, 赵金仙. Ag,Zn,Pd掺杂对铜基催化剂草酸二甲酯选择性加氢反应的影响. 高等学校化学学报, 2022, 43(6): 20210842.
SONG Youwei, AN Jiangwei, WANG Zheng, WANG Xuhui, QUAN Yanhong, REN Jun, ZHAO Jinxian. Effects of Ag,Zn,Pd-doping on Catalytic Performance of Copper Catalyst for Selective Hydrogenation of Dimethyl Oxalate. Chem. J. Chinese Universities, 2022, 43(6): 20210842.
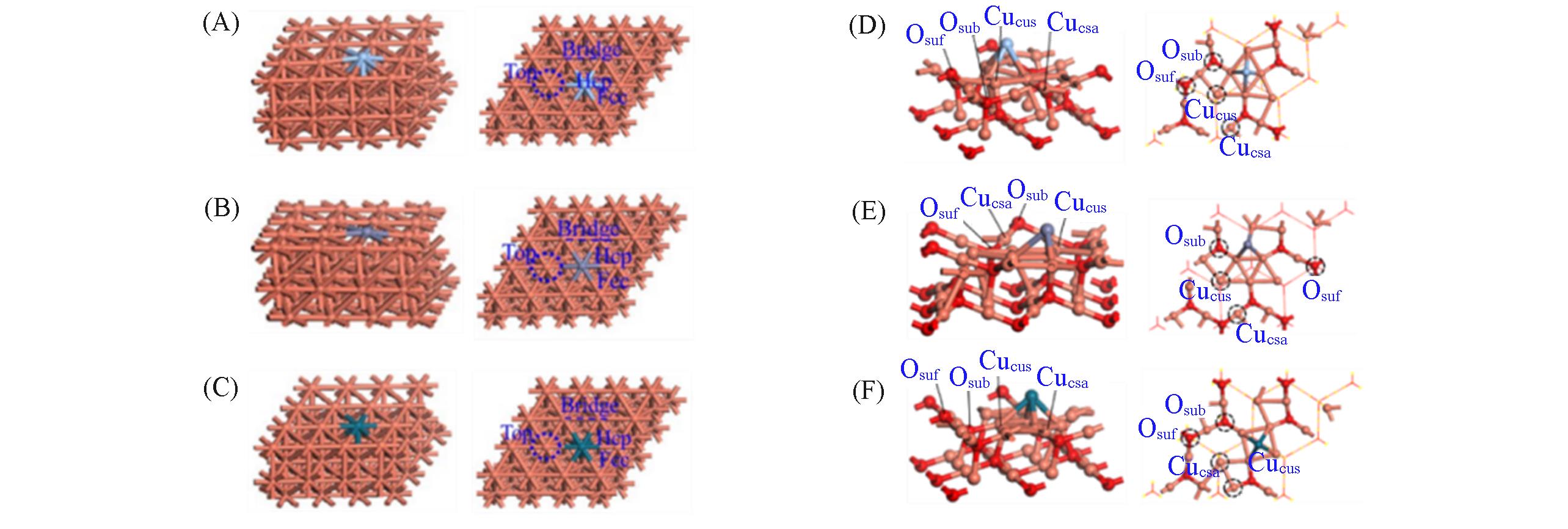
Fig.1 Side views(left) and top views(right) of Ag?Cu(111)(A), Zn?Cu(111)(B), Pd?Cu(111)(C), Ag?Cu2O(111)(D), Zn?Cu2O(111)(E) and Pd?Cu2O(111)(F) models
| Species | Cu(111)[ | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | ||||
|---|---|---|---|---|---|---|---|---|
| Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | |
| H2 | Fcc | 13.3 | Fcc | 40.4 | Fcc | 44.6 | Fcc | 26.3 |
| H | Fcc | 247.6 | Fcc | 236.7 | Fcc | 229.6 | Fcc | 253.3 |
| DMO | — | 46 | — | 55.1 | — | 50.1 | — | 57.8 |
| CH3OOCCO | Top | 152 | Top | 151.1 | Top | 151.4 | Top | 199.6 |
| CH3O | Hcp | 222 | Fcc | 206.2 | Hcp | 221.2 | Fcc | 203.5 |
| CH3OOCCOH | Top(C) Bridge(O) | 48 | Top(C) Top(O) | 301 | Bridge(C) Top(O) | 301 | Top(C) Top(O) | 327.7 |
| CH3OOCCHOH | Top(C) Top(O) | 101.5 | Top | 96.5 | Top | 129 | Top | 146.6 |
| MG | — | 56.1 | — | 50.4 | — | 35.4 | — | 38.2 |
| CH3OH | — | 36.7 | — | 41.5 | — | 43.1 | — | 43.3 |
| OCCH2OH | Top | 177.3 | Top | 172.4 | Top | 168.7 | Top | 220.6 |
Table 1 Adsorption sites and adsorption energies of all species involved in DMO hydrogenation to MG on Cu(111), Ag-Cu(111), Zn-Cu(111) and Pd-Cu(111) surfaces
| Species | Cu(111)[ | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | ||||
|---|---|---|---|---|---|---|---|---|
| Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | |
| H2 | Fcc | 13.3 | Fcc | 40.4 | Fcc | 44.6 | Fcc | 26.3 |
| H | Fcc | 247.6 | Fcc | 236.7 | Fcc | 229.6 | Fcc | 253.3 |
| DMO | — | 46 | — | 55.1 | — | 50.1 | — | 57.8 |
| CH3OOCCO | Top | 152 | Top | 151.1 | Top | 151.4 | Top | 199.6 |
| CH3O | Hcp | 222 | Fcc | 206.2 | Hcp | 221.2 | Fcc | 203.5 |
| CH3OOCCOH | Top(C) Bridge(O) | 48 | Top(C) Top(O) | 301 | Bridge(C) Top(O) | 301 | Top(C) Top(O) | 327.7 |
| CH3OOCCHOH | Top(C) Top(O) | 101.5 | Top | 96.5 | Top | 129 | Top | 146.6 |
| MG | — | 56.1 | — | 50.4 | — | 35.4 | — | 38.2 |
| CH3OH | — | 36.7 | — | 41.5 | — | 43.1 | — | 43.3 |
| OCCH2OH | Top | 177.3 | Top | 172.4 | Top | 168.7 | Top | 220.6 |
| Elementary step | Reaction |
|---|---|
| R1 | H2+2*?2H* |
| R2 | CH3OOCCOOCH3 + 2*→CH3OOCC*O + CH3O* |
| R3 | CH3OOCC*O + H*?CH3OOCC*OH(CH3OOCC*HO)+ * |
| R4 | CH3OOCC*OH(CH3OOCC*HO) + H*? CH3OOCC*HOH + * |
| R5 | CH3OOCC*HOH + H*? CH3OOCC*H2OH + * |
| R6 | CH3O* + H*? CH3OH* + * |
| R7 | CH3OOCCH2OH+2*?HOCH2C*O+CH3O* |
Table 2 Mechanism of DMO hydrogenation over Cu catalyst#
| Elementary step | Reaction |
|---|---|
| R1 | H2+2*?2H* |
| R2 | CH3OOCCOOCH3 + 2*→CH3OOCC*O + CH3O* |
| R3 | CH3OOCC*O + H*?CH3OOCC*OH(CH3OOCC*HO)+ * |
| R4 | CH3OOCC*OH(CH3OOCC*HO) + H*? CH3OOCC*HOH + * |
| R5 | CH3OOCC*HOH + H*? CH3OOCC*H2OH + * |
| R6 | CH3O* + H*? CH3OH* + * |
| R7 | CH3OOCCH2OH+2*?HOCH2C*O+CH3O* |
Elementary step | Cu(111)[ | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | |
| R1 | — | -13.3 | — | — | -40.4 | — | — | -44.6 | — | — | -26.3 |
| R2 | 220.2 | 86.2 | TS1 | 224.3 | 91.6 | TS5 | 158.5 | 75.6 | TS9 | 327.2 | 39.7 |
| R3 | 60.1 | -39.8 | TS2 | 68.5 | -19.9 | TS6 | 115.1 | -10.3 | TS10 | 134.7 | 2.5 |
| R4 | 23.7 | -38.2 | TS3 | 74.6 | -75 | TS7 | 91.9 | -89.9 | TS11 | 58.2 | -90.7 |
| R5 | 91.5 | -73.3 | TS4 | 47.3 | -107 | TS8 | 104.2 | -58.3 | TS12 | 171.5 | -35.1 |
| R6 | 111.3 | -20.3 | TS13 | 141.7 | -39.8 | TS14 | 138.9 | -43.1 | TS15 | 141.3 | -36.1 |
| R7 | 192.6 | 88.4 | TS16 | 321.4 | 101 | TS17 | 305.9 | 68.9 | TS18 | 75.6 | 40 |
Table 3 Activation energies(Ea) and reaction energies(ΔH) for elementary reactions(R1—R5) of DMO hydrogenation to MG and side reactions(R6, R7) on Cu(111) and M-Cu(111) surfaces
Elementary step | Cu(111)[ | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | |
| R1 | — | -13.3 | — | — | -40.4 | — | — | -44.6 | — | — | -26.3 |
| R2 | 220.2 | 86.2 | TS1 | 224.3 | 91.6 | TS5 | 158.5 | 75.6 | TS9 | 327.2 | 39.7 |
| R3 | 60.1 | -39.8 | TS2 | 68.5 | -19.9 | TS6 | 115.1 | -10.3 | TS10 | 134.7 | 2.5 |
| R4 | 23.7 | -38.2 | TS3 | 74.6 | -75 | TS7 | 91.9 | -89.9 | TS11 | 58.2 | -90.7 |
| R5 | 91.5 | -73.3 | TS4 | 47.3 | -107 | TS8 | 104.2 | -58.3 | TS12 | 171.5 | -35.1 |
| R6 | 111.3 | -20.3 | TS13 | 141.7 | -39.8 | TS14 | 138.9 | -43.1 | TS15 | 141.3 | -36.1 |
| R7 | 192.6 | 88.4 | TS16 | 321.4 | 101 | TS17 | 305.9 | 68.9 | TS18 | 75.6 | 40 |
| Species | Cu2O(111)[ | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | ||||
|---|---|---|---|---|---|---|---|---|
| Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | |
| H2 | Cucus | 31.7 | Cucus | 29.1 | Cucus | 77.6 | Cucus | 32.3 |
| H | Osuf | 231.1 | Cucus | 276.1 | Cucus | 265.0 | Bridge (Cucus?Cucsa) | 255.1 |
| DMO | Cucus | 101.3 | Cucus | 98.4 | Cucus | 98.6 | Cucus | 100.0 |
| CH3OOCCO | Cucus | 200.7 | Cucus | 226.2 | Cucus | 242.6 | Cucus | 246.6 |
| CH3O | Cucus | 231.1 | Cucus | 277.1 | Cucus | 242.4 | Cucus | 250.0 |
| CH3OOCCHO | Cucus | 88.3 | Cucus | 126.2 | Cucus | 117.6 | Cucus | 151.6 |
| CH3OOCCHOH | Cucus | 140.4 | Cucus | 198.5 | Cucus | 175.8 | Cucus | 181.8 |
| MG | Cucus | 107.1 | Cucus | 111.0 | Cucus | 121.3 | Cucus | 129.4 |
| CH3OH | Cucus | 97.2 | Cucus | 86.3 | Cucus | 110.1 | Cucus | 98.1 |
| OCCH2OH | Cucus | 323.1 | Cucus | 267.0 | Cucus | 250.9 | Cucus | 232.3 |
Table 4 Adsorption sites and adsorption energies(Eads) of all species involved in DMO hydrogenation to MG on Cu2O(111) and M?Cu2O(111) surfaces
| Species | Cu2O(111)[ | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | ||||
|---|---|---|---|---|---|---|---|---|
| Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | Site | Eads/(kJ?mol-1) | |
| H2 | Cucus | 31.7 | Cucus | 29.1 | Cucus | 77.6 | Cucus | 32.3 |
| H | Osuf | 231.1 | Cucus | 276.1 | Cucus | 265.0 | Bridge (Cucus?Cucsa) | 255.1 |
| DMO | Cucus | 101.3 | Cucus | 98.4 | Cucus | 98.6 | Cucus | 100.0 |
| CH3OOCCO | Cucus | 200.7 | Cucus | 226.2 | Cucus | 242.6 | Cucus | 246.6 |
| CH3O | Cucus | 231.1 | Cucus | 277.1 | Cucus | 242.4 | Cucus | 250.0 |
| CH3OOCCHO | Cucus | 88.3 | Cucus | 126.2 | Cucus | 117.6 | Cucus | 151.6 |
| CH3OOCCHOH | Cucus | 140.4 | Cucus | 198.5 | Cucus | 175.8 | Cucus | 181.8 |
| MG | Cucus | 107.1 | Cucus | 111.0 | Cucus | 121.3 | Cucus | 129.4 |
| CH3OH | Cucus | 97.2 | Cucus | 86.3 | Cucus | 110.1 | Cucus | 98.1 |
| OCCH2OH | Cucus | 323.1 | Cucus | 267.0 | Cucus | 250.9 | Cucus | 232.3 |
Elementary step | Cu2O(111)[ | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | |
| R1 | 299 | -15.1 | TS37 | 66.3 | -9.1 | — | — | -77.6 | TS38 | 57.6 | -9.8 |
| R2 | 140.5 | 139.8 | TS19 | 178.9 | 121.7 | TS23 | 187.4 | 105.6 | TS27 | 203.5 | 137.6 |
| R3 | 99.2 | 5.1 | TS20 | 65.1 | -61.7 | TS24 | 83.7 | -71.6 | TS28 | 106.4 | -116.3 |
| R4 | 68.2 | -16.5 | TS21 | 66.9 | -72.1 | TS25 | 52.6 | -93.8 | TS29 | 87.9 | -15 |
| R5 | 244 | -190.7 | TS22 | 32.5 | -77.8 | TS26 | 82.7 | -40.7 | TS30 | 52.7 | -91.9 |
| R6 | 6.7 | -53.1 | TS31 | 73.6 | -76.7 | TS32 | 83.8 | -88.4 | TS33 | 31.5 | -73.3 |
| R7 | 229 | 147.3 | TS34 | 316 | -12.1 | TS35 | 306.6 | 11.1 | TS36 | 138.7 | 74.6 |
Table 5 Activation energies(Ea) and reaction energies(ΔH) for elementary reactions(R1—R5) of DMO hydrogenation to MG and side reactions(R6, R7) on Cu2O(111) and M-Cu2O(111) surfaces
Elementary step | Cu2O(111)[ | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | TS | Ea/ (kJ?mol-1) | ΔH/ (kJ?mol-1) | |
| R1 | 299 | -15.1 | TS37 | 66.3 | -9.1 | — | — | -77.6 | TS38 | 57.6 | -9.8 |
| R2 | 140.5 | 139.8 | TS19 | 178.9 | 121.7 | TS23 | 187.4 | 105.6 | TS27 | 203.5 | 137.6 |
| R3 | 99.2 | 5.1 | TS20 | 65.1 | -61.7 | TS24 | 83.7 | -71.6 | TS28 | 106.4 | -116.3 |
| R4 | 68.2 | -16.5 | TS21 | 66.9 | -72.1 | TS25 | 52.6 | -93.8 | TS29 | 87.9 | -15 |
| R5 | 244 | -190.7 | TS22 | 32.5 | -77.8 | TS26 | 82.7 | -40.7 | TS30 | 52.7 | -91.9 |
| R6 | 6.7 | -53.1 | TS31 | 73.6 | -76.7 | TS32 | 83.8 | -88.4 | TS33 | 31.5 | -73.3 |
| R7 | 229 | 147.3 | TS34 | 316 | -12.1 | TS35 | 306.6 | 11.1 | TS36 | 138.7 | 74.6 |
| Species | Charge transfer, Q/|e| | |||
|---|---|---|---|---|
| Cu(111) | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | |
| DMO | 0.0194 | -0.0942 | 0.0038 | 0.1498 |
| MG | -0.0630 | -0.0851 | -0.0702 | -0.0283 |
Table 6 Charge transfer(Q) between DMO, MG and Cu(111), Ag?Cu(111) and Zn?Cu(111) catalysts
| Species | Charge transfer, Q/|e| | |||
|---|---|---|---|---|
| Cu(111) | Ag?Cu(111) | Zn?Cu(111) | Pd?Cu(111) | |
| DMO | 0.0194 | -0.0942 | 0.0038 | 0.1498 |
| MG | -0.0630 | -0.0851 | -0.0702 | -0.0283 |
| Species | Charge transfer, Q/|e| | |||
|---|---|---|---|---|
| Cu2O(111) | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | |
| DMO | 0.0934 | 0.0917 | 0.0913 | 0.1507 |
| MG | 0.0129 | 0.0048 | -0.0047 | -0.0002 |
Table 7 Charge transfer(Q) between DMO, MG and Cu2O(111), Ag-Cu2O(111) and Zn?Cu2O(111) catalysts
| Species | Charge transfer, Q/|e| | |||
|---|---|---|---|---|
| Cu2O(111) | Ag?Cu2O(111) | Zn?Cu2O(111) | Pd?Cu2O(111) | |
| DMO | 0.0934 | 0.0917 | 0.0913 | 0.1507 |
| MG | 0.0129 | 0.0048 | -0.0047 | -0.0002 |
| 1 | Yang X., Liu B. Y., Shanghai Chemical Industry, 2014, 39(9), 27—30 |
| 杨西, 刘宝勇. 上海化工, 2014, 39(9), 27—30 | |
| 2 | Zhao Y. J., Zhao S., Wang B., Lü J., Ma X. B., Chem. Ind. & Eng. Pro., 2013, 32(4), 721—731 |
| 赵玉军, 赵硕, 王博, 吕静, 马新宾. 化工进展, 2013, 32(4), 721—731 | |
| 3 | Wang B. W., Xu Q., Song H., Xu G. H., Nat. Gas Chem., 2007, 78—80 |
| 4 | Li J. H., Duan X. P., Lin H.Q.,Ye L. M., Yuan Y. Z., Petrochem. Technol., 2014, 43(9), 985—994 |
| 李建辉, 段新平, 林海强, 叶林敏, 袁友珠. 石油化工, 2014, 43(9), 985—994 | |
| 5 | Tian K. S., Wang B. W., Xu G.H., J. Nat. Gas Chem., 2006, 31(6), 60—63 |
| 田克胜, 王保伟, 许根慧. 天然气化工, 2006, 31(6), 60—63 | |
| 6 | Ye R. P., Lin L., Chen C., Yang J. X., Li F., Zhang X., Li D. J., Qin Y. Y., Zhou Z. F., Yao Y. G., ACS Catal., 2018, 8(4), 3382—3394 |
| 7 | Wang D. H., Zhang C. C., Zhu M. Y., Yu F., Dai B., Chem. Select, 2017, 2(17), 4823—4829 |
| 8 | An J. W., Wang X. H., Zhao J. X., Jiang S. H., Quan Y. H., Pei Y. L., Wu M. M., Ren J., Mol. Catal., 2019, 482, 110667 |
| 9 | Zhao Y. J., Kong L. X., Xu Y. X., Huang H. J., Yao Y. Q., Zhang J. W., Wang S. P., Ind. Eng. Chem. Res., 2020, 59, 12381—12388 |
| 10 | Yin A., Wen C., Guo X., Dai W., Fan K., J. Catal., 2011, 280(1), 77—88 |
| 11 | Zheng X., Lin H., Zheng J., Duan X., Yuan Y., ACS Catal., 2013, 3(12), 2738—2749 |
| 12 | Zhang C. C., Wang D. H., Zhu M. Y., Yu F., Dai B., Chem. Sel., 2016, 1(11), 2857—2863 |
| 13 | Wang Q., Qiu L., Ding D., Chen Y. Z., Shi C. W., Cui P., Wang Y., Catal. Commun., 2018, 108, 68—72 |
| 14 | Delley B., Zu C., J. Phys. Chem., 1996, 100(15), 6107—6110 |
| 15 | Perdew J. P., Burke K., Ernzerhof M., Phys. Rev. Lett., 1996, 77(18), 3865—3868 |
| 16 | Ernzerhof M., Scuseria G. E., J. Chem. Phys., 1999, 110, 5029—5036 |
| 17 | Dolg M., Wedig U., Stoll H., J. Chem. Phys., 1986, 86, 866—872 |
| 18 | Wang Y. Z., Wisesa P., Comput. Mater. Sci., 2020, 187, 110100 |
| 19 | Halgren T. A., Lipscomb W. N., Chem. Phys. Lett., 1977, 49(12), 225—232 |
| 20 | Hirshfeld F. L., Theor. Chim. Acta, 1977, 44, 129—138 |
| 21 | Zhao Y. T., Cui C. N., Han J. Y., Wang H., Zhu X. L., Ge Q. F., J. Am. Chem. Soc., 2016, 138(32), 10191—10198 |
| 22 | Yue H., Ma X., Gong J., Acc. Chem. Res., 2014, 47(5), 1483—1492 |
| 23 | Li S. M., Wang Y., Zhang J., Wang S. P., Xu Y., Zhao Y. J., Ma X. B., Ind. Eng. Chem. Res., 2015, 54(4), 1243—1250 |
| 24 | Hui S. G., Zhang B., Zhang S. H., Li W., J. Nat. Gas Chem., 2012, 21(6), 753—758 |
| 25 | Zhang R. G., Zhang J., Jiang Z., Wang B. J., Fan M. H., Chem. Eng. J., 2018, 351, 732—746 |
| 26 | Yang B., Burch R., Hardacre C., Headdock G., Hu P., ACS Catal., 2012, 2(6), 1027—1032 |
| 27 | Zhang R. G., Zhang J., Zhao B., He L., Wang A. J., Wang B. J., J. Phys. Chem. C, 2017, 121(50), 27936—27949 |
| 28 | Larmier K., Liao W. C., Tada S., Lam E., Verel R., Bansode A., Urakawa A., Comas⁃Vives A., Copéret C., Angew. Chem. Int. Ed., 2017, 56(9), 2318—2323 |
| 29 | Li J., Croiset E., Ricardez⁃Sandova L., J. Mol. Catal. A: Chem., 2012, 365, 103—114 |
| 30 | Huo C. F., Li Y. W., Wang J. G., Jiao H. J., J. Am. Chem. Soc., 2009, 131(41), 14713—14721 |
| 31 | Zhang R., Wang B., Ling L., Liu H., Huang W., Appl. Surf. Sci., 2010, 257(4), 1175—1180 |
| 32 | Kumar A., De Leeuw N. H., Biochem. Pharmacol., 2016, 15, 96—106 |
| 33 | Zhang R. G., Zhao B., He L., Wang A. J., Wang B. J., PhysChemChemPhys, 2018, 20(25), 17487—17496 |
| 34 | Zuo Z. J., Ramírez P. J., Senanayake S. D., Liu P., Rodriguez J. A., J. Am. Chem. Soc., 2016, 138(42), 13810—13813 |
| 35 | Liu Z. M., Ma L., Junaid A. S. M., J. Phys. Chem. C, 2010, 114(10), 4445—4450 |
| [1] | 刘恒烁,遇治权,孙志超,王瑶,刘颖雅,王安杰. COF固载铜盐催化苯硼酸与咪唑的Chan-Lam偶联反应[J]. 高等学校化学学报, 2020, 41(5): 1091. |
| [2] | 何小可, 李小云, 王朝, 胡念, 邓兆, 陈丽华, 苏宝连. 金属有机骨架高温自还原制备高性能等级孔碳负载Co基催化剂[J]. 高等学校化学学报, 2020, 41(4): 639. |
| [3] | 曹勇勇, 蒋军辉, 倪哲明, 夏盛杰, 钱梦丹, 薛继龙. Au19Pt团簇性质及对肉桂醛选择性加氢机理研究[J]. 高等学校化学学报, 2016, 37(7): 1342. |
| [4] | 蒋军辉, 夏盛杰, 倪哲明, 张连阳. 巴豆醛在Au(111)面上的吸附及选择性加氢机理研究[J]. 高等学校化学学报, 2016, 37(4): 693. |
| [5] | 方超, 陈亚君, 毛卉, 赵俊, 蒋云福, 赵仕林, 马骏. 基于杨梅单宁制备Pt/SiO2-C催化剂及其对肉桂醛选择性催化加氢性能[J]. 高等学校化学学报, 2015, 36(1): 124. |
| [6] | 江欣, 秦晓宇, 宫孟娣, 李秀玲, 李光植, 杨立滨, 赵冰. 金属镍掺杂改进纳米TiO2的表面增强拉曼散射性能[J]. 高等学校化学学报, 2014, 35(3): 488. |
| [7] | 王艳, 张小超, 赵丽军, 赵晓霞, 史宝萍, 樊彩梅. 采用第一性原理研究非金属掺杂BiOCl的电子结构和光吸收性质[J]. 高等学校化学学报, 2014, 35(12): 2624. |
| [8] | 王成杰, 唐春梅, 张轶杰, 高凤志. 不同金属外掺杂富勒烯C20M(M=Li, Ti, Fe)的储氢性能[J]. 高等学校化学学报, 2014, 35(10): 2131. |
| [9] | 刘淼, 冷粟, 陈嵩岳, 张亮, 张美秀, 刘宛宜, 沈力, 张振斌, 刘南, 焦昕倩, 陈力可, 全福民. 改性Ti/SnO2-Sb电极降解硝基苯废水[J]. 高等学校化学学报, 2013, 34(8): 1899. |
| [10] | 孙翠枝 周仁贤. 乙醇/水溶剂体系中Pt\|Co/ZrO2催化剂上肉桂醛的选择性加氢性能[J]. 高等学校化学学报, 2011, 32(7): 1551. |
| [11] | 陈媛梅, 李卿, 黄元河. H2分子在碱金属掺杂碳纳米管上的吸附特性[J]. 高等学校化学学报, 2010, 31(6): 1235. |
| [12] | 念保义,徐刚,吴坚平,杨立荣 . 离子液体介质中用Cu/ZrO2-SiO2催化香茅醛加氢合成薄荷醇[J]. 高等学校化学学报, 2007, 28(9): 1677. |
| [13] | 熊伟, 马红霞, 陈华, 黄艳轶, 黄裕林, 李贤均, 黎星术, 戚建英, 陈新滋. 奎宁修饰的纳米铑簇合物催化丙酮酸乙酯对映选择性加氢反应研究[J]. 高等学校化学学报, 2002, 23(9): 1758. |
| [14] | 李卫华, 乔学斌, 高恩勤, 杨迈之, 郝彦忠, 蔡生民. 3d过渡金属掺杂TiO2纳米晶膜电极的光电化学研究[J]. 高等学校化学学报, 2000, 21(10): 1534. |
| [15] | 梁贤振, 赵维君, 杨瑞华, 冯鸣华, 王忠文, 马新宾, 徐根慧. CO和CH3ONO在Al2O3负载钯铜合金催化剂上合成草酸二甲酯的原位红外光谱研究[J]. 高等学校化学学报, 1997, 18(5): 777. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||