

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (1): 299.doi: 10.7503/cjcu20200400
所属专题: 分子筛功能材料 2021年,42卷,第1期
收稿日期:2020-06-30
出版日期:2021-01-10
发布日期:2021-01-12
通讯作者:
李激扬
E-mail:lijyang@jlu.edu.cn
基金资助:
WANG Bolun, ZONG Siyu, LI Jiyang( )
)
Received:2020-06-30
Online:2021-01-10
Published:2021-01-12
Contact:
LI Jiyang
E-mail:lijyang@jlu.edu.cn
Supported by:摘要:
除传统的催化、 吸附分离和离子交换外, 主客体组装化学赋予了沸石分子筛材料独特的物理化学性质和广阔的应用前景. 本文聚焦光致发光沸石分子筛复合材料, 综述了这类材料最新的研究进展, 总结了不同发光客体, 如稀土金属、 金属簇、 量子点/碳点等与沸石分子筛形成的复合材料的制备方法与组装策略, 介绍了该类复合材料的光致发光性质和潜在应用, 探究了复合材料中可能存在的量子限域、 分子间相互作用、 能量转移和电子转移等对发光的影响, 并对未来光致发光沸石分子筛复合材料的发展前景进行了展望.
中图分类号:
TrendMD:
王博伦, 宗思宇, 李激扬. 光致发光沸石分子筛复合材料的研究进展. 高等学校化学学报, 2021, 42(1): 299.
WANG Bolun, ZONG Siyu, LI Jiyang. Recent Progress on Photoluminescent Zeolite-based Composite Materials. Chem. J. Chinese Universities, 2021, 42(1): 299.
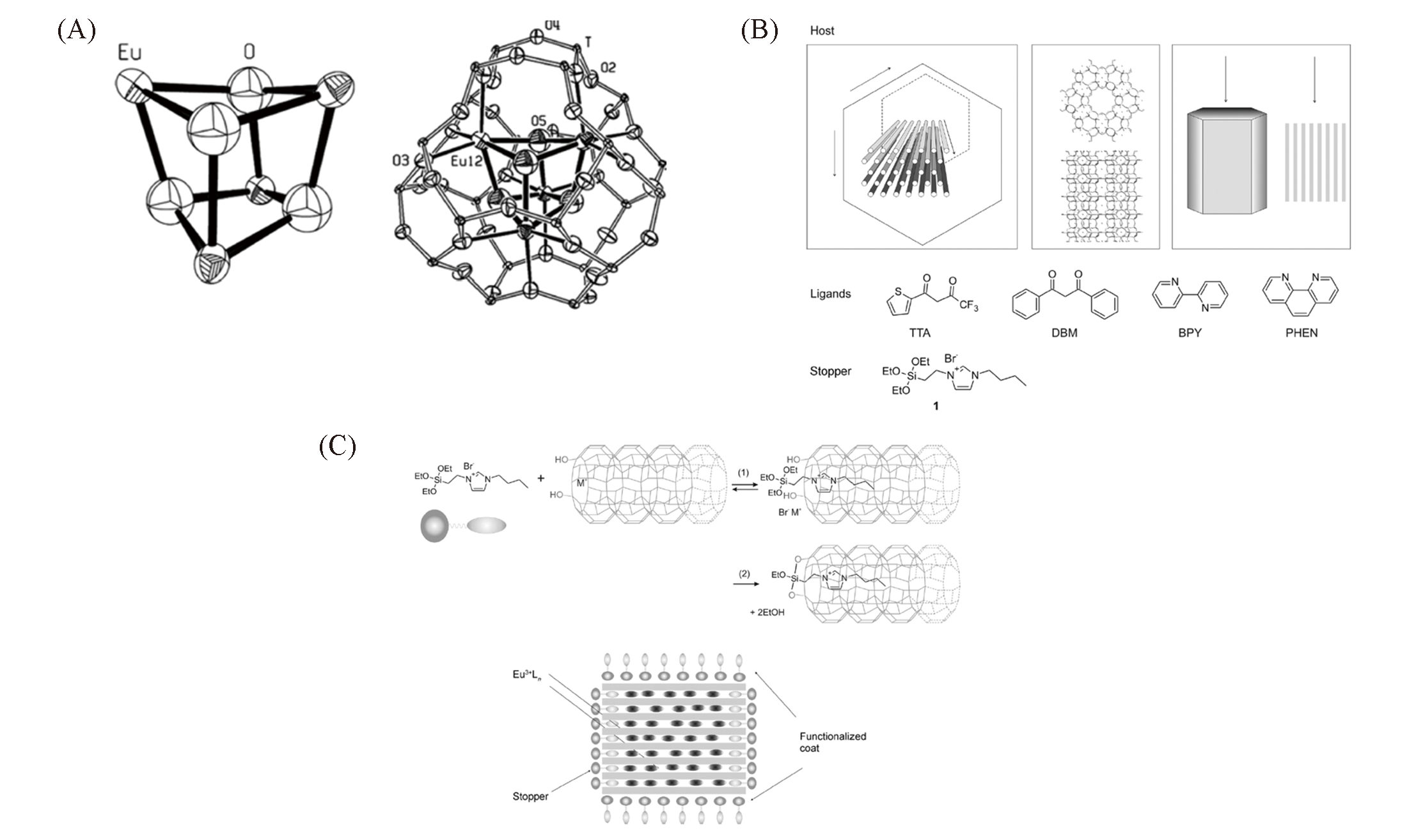
Fig.1 Schematic illustration for different synthetic strategies of rare earth metal?containing zeolites[33,36](A) The structure of Eu4O44+ and it in an SOD cavity. Copyright 2014, American Chemical Society; (B) structures of the host ze-olite L, the ligands and stopper; (C) selective modification of channel entrance of zeolite L(up), and stopper and coat functiona-lized zeolite crystal containing guest Eu3+ complexes(down). Copyright 2014, Wiley-VCH.
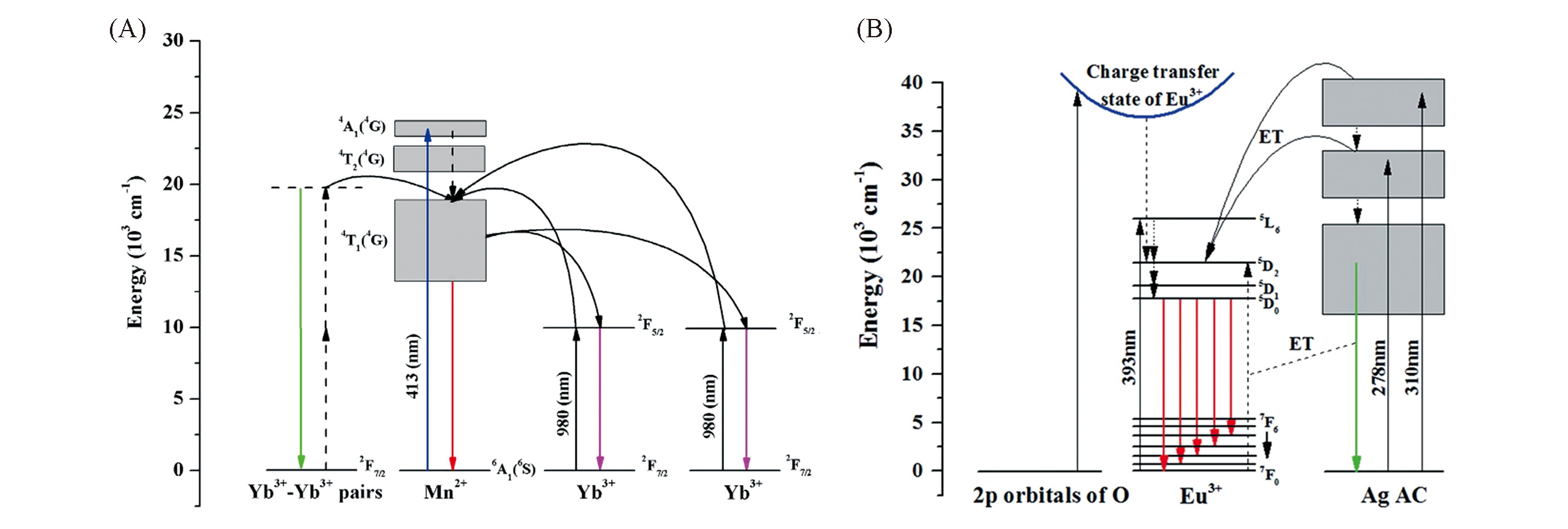
Fig.2 Possible energy level diagram and emission mechanism of rare earth metal?containing zeolites[38,39](A) The possible energy level diagram of Mn2+ and Yb3+ ions, and their ET process. Copyright 2017, Springer Nature;(B) energy levels and ET mechanism from Ag+ active centers to Eu3+ in composite. Copyright 2016, Royal Society of Chemistry.
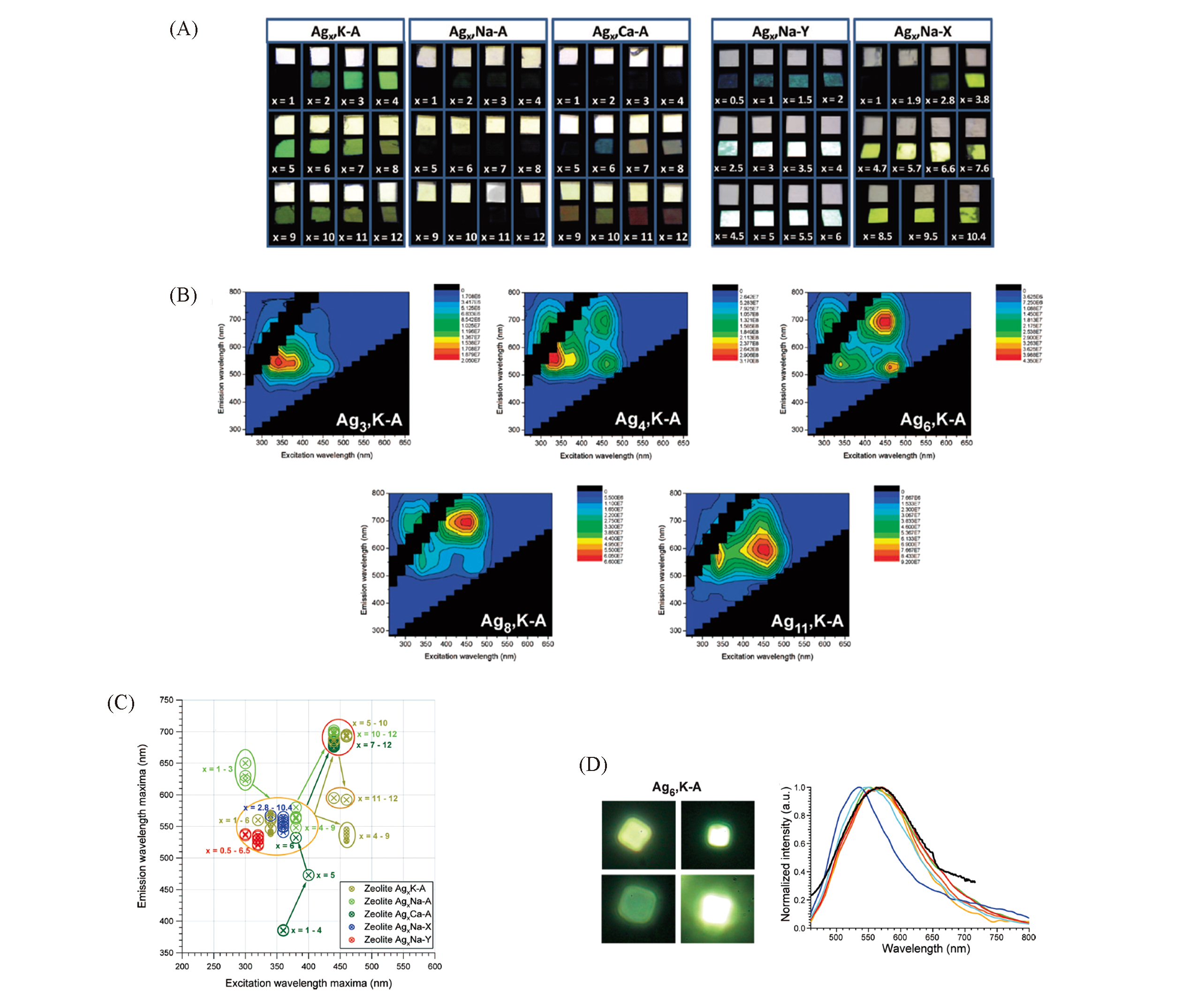
Fig.3 Photoluminescent properties of Ag clusters in zeolites[45](A) Photographs of the heat-treated silver-exchanged zeolites(K-A, Na-A, Ca-A, Na-Y and Na-X zeolites); (B) representative 2D excitation-emission matrixes for Agx, K-A zeolites; (C) position of the most pronounced luminescence bands of the heat-treated silver-exchanged zeolites; (D) single-crystal emission characterization for Ag6,K-A.Copyright 2009, American Chemical Society.
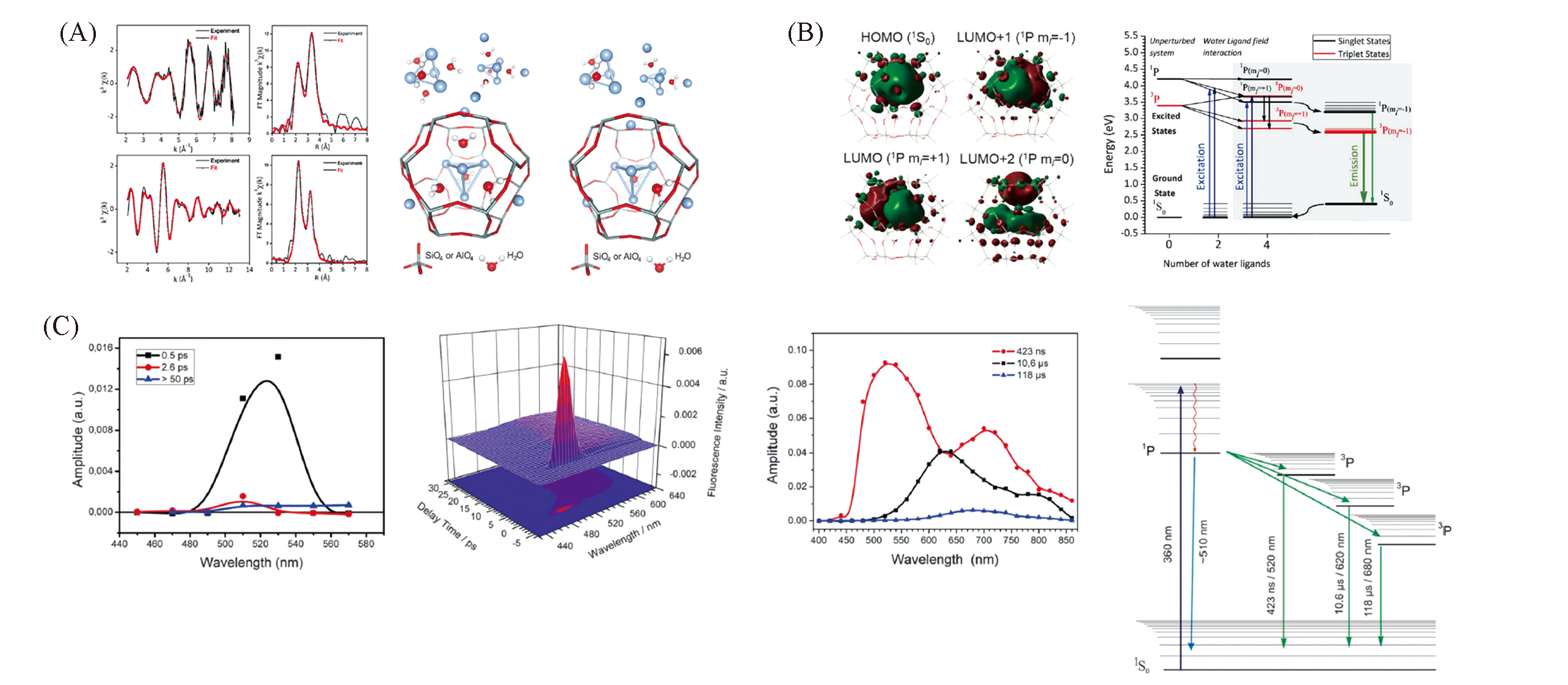
Fig.4 Structure of Ag clusters in LTA zeolite and possible luminescence mechanism[46](A) Ag K-edge X-ray excited optical luminescence(XEOL), transmission-detected X-ray absorption fine structure(EXAFS)and Fourier transforms(FTs) of heat-treated Ag3K9 cluster in LTA zeolite and derived structures; (B) frontier orbitals of [Ag4(H2O)4·(Si24H24O36)]2+ and energy level diagram of Ag4(H2O)22+ and Ag4(H2O)42+ clusters in Ag3K9-LTA. Copyright 2018, AAAS;(C) time-resolved spectra and energy level diagram of Ag3K9-LTA. Copyright 2018, AAAS.
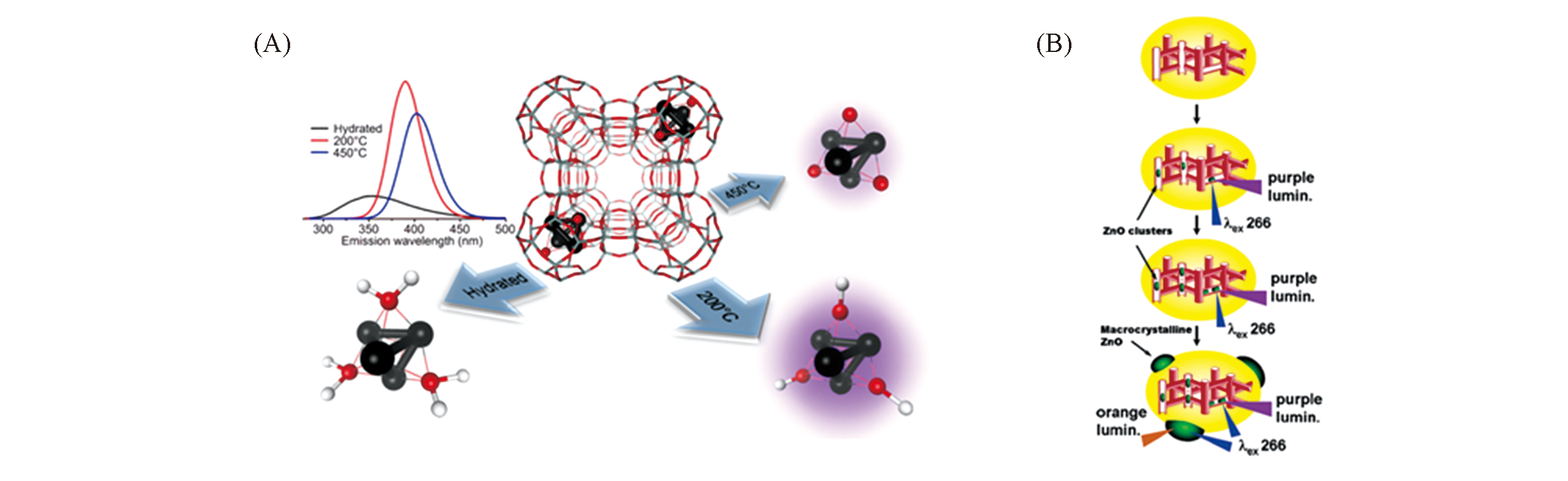
Fig.5 Luminescence and structures of Pb and ZnO clusters in LTA zeolite[49,50](A) The Pb cluster in LTA zeolite under different conditions. Copyright 2018, American Chemical Society;(B) the ZnO clusters in HZSM-5. Copyright 2006, American Chemical Society.
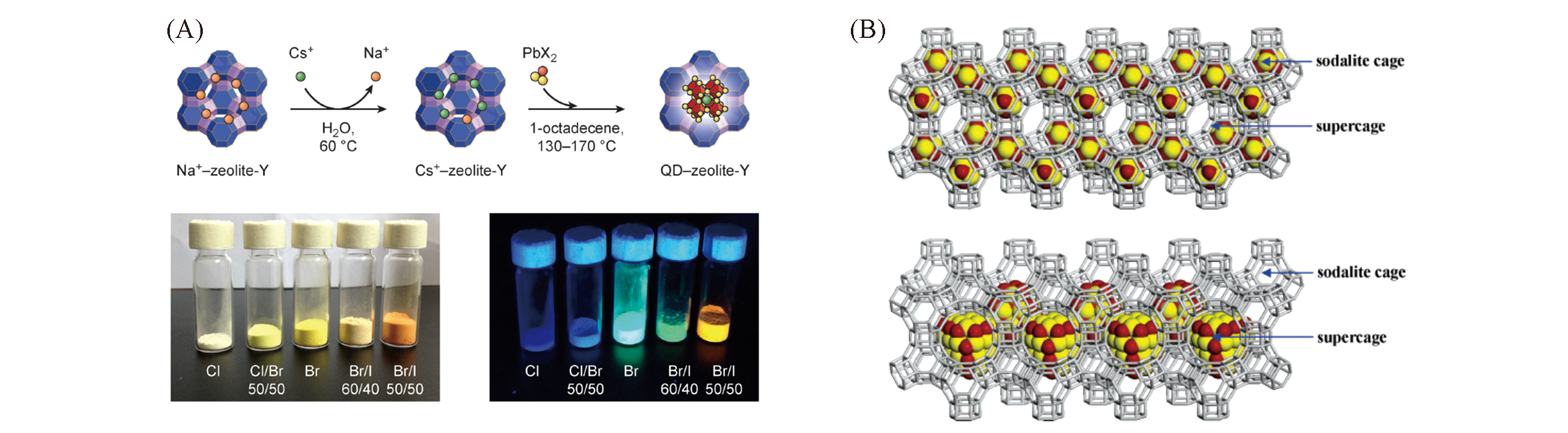
Fig.6 CsPbX3?zeolite?Y and CdS?Y composites[55,58](A) Two-step synthesis of CsPbX3?zeolite-Y composites and their luminescence. Copyright 2017, WILEY-VCH; (B) incorporating a (CdS)4 unit in each SOD cage and a larger CdS QD in each supercagein zeolite Y. Copyright 2007, American Chemical Society.
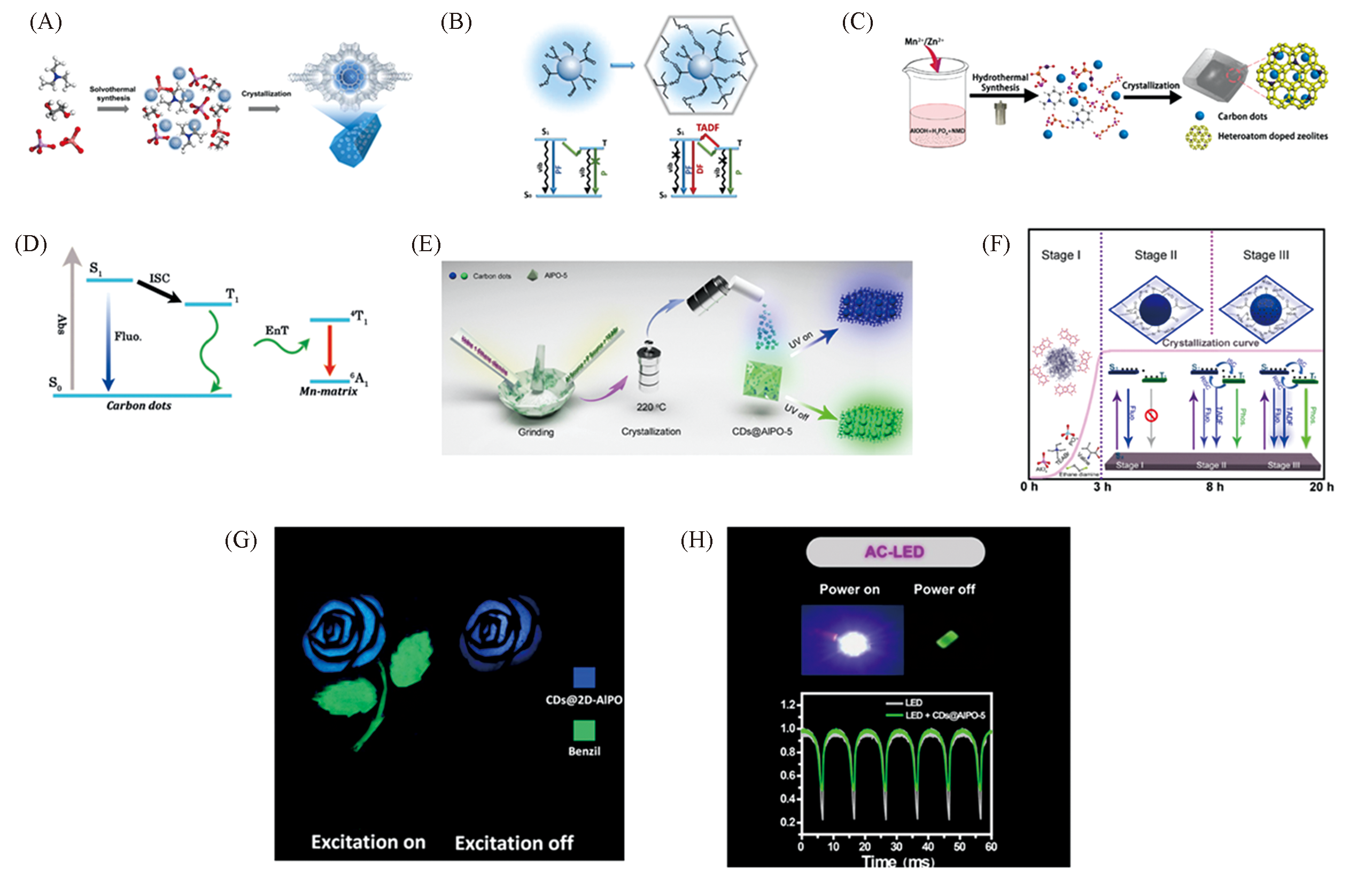
Fig.7 Syntheses of CDs@zeolite composites and their emission mechanism and applications[69,71,72,74](A) CDs-in-zeolite synthetic strategy for synthesis of CDs@AlPO-5 composite; (B) the TADF mechanism of CDs@AlPO-5 compo-site; (C) synthesis of CDs doped heteroatom-containing zeolite composites. Copyright 2019, American Chemical Society; (D) ET process between CDs and octahedral Mn2+ centersin MnAPO-CJ50 matrix. Copyright 2019, Wiley-VCH; (E) solvent-free thermal crystallization synthesis of CDs@AlPO-5 composite; (F) CDs@AlPO-5 formed with varying crystallization time and the crystallization curve of CDs@AlPO-5; (G) security protection application of CDs@2D-AlPO. Copyright 2017, AAAS; (H) CDs@AlPO-5 for LED. Copyright 2020, CCS Chemistry.
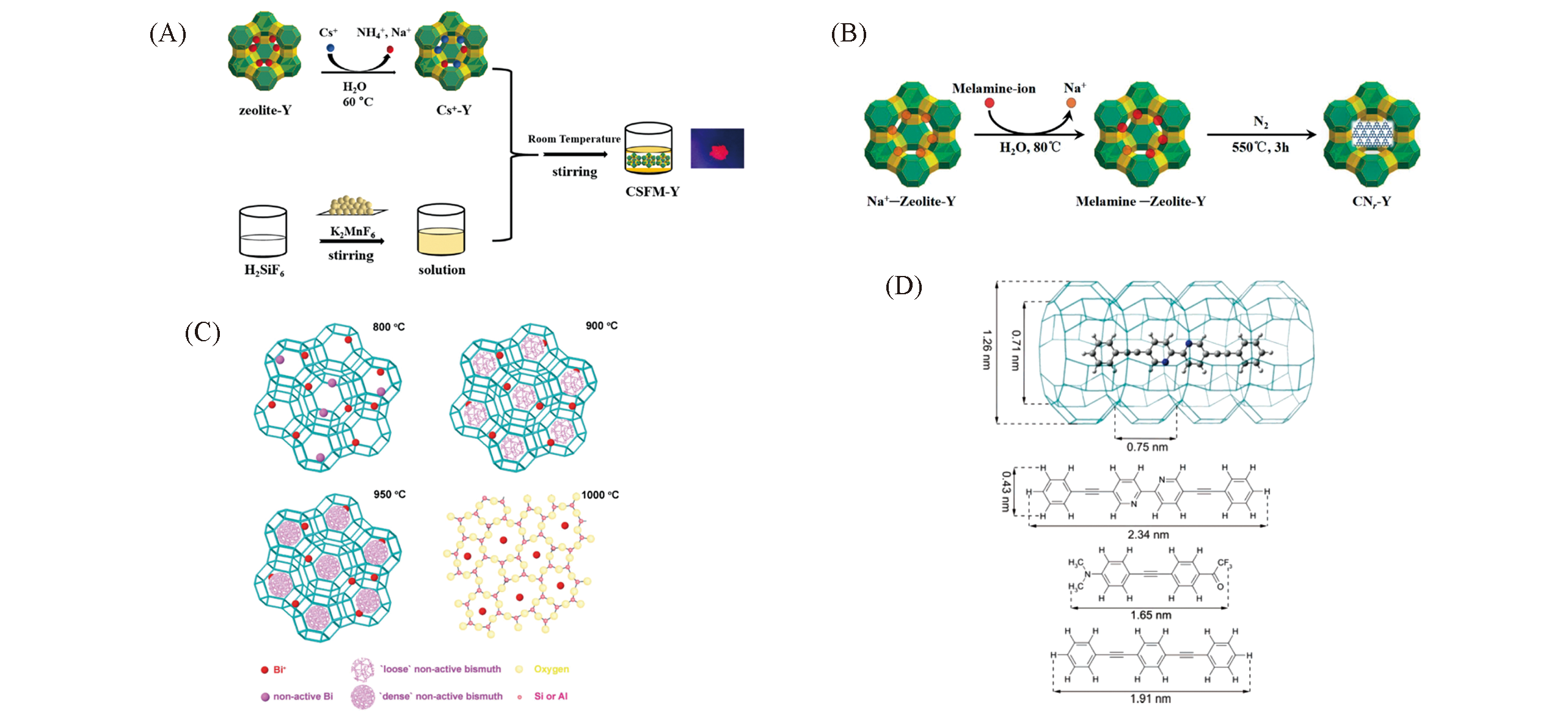
Fig.8 Assemble of other emitters into zeolites[71,78,81,84](A) Encapsulating Cs2SiF6:Mn4+ red phosphors in zeolite Y. Copyright 2019, Royal Society of Chemistry; (B) two-step synthesis of g-C3N4-Y. Copyright 2018, Royal Society of Chemistry; (C) schematic illustration of the structure transformation of Bi+-zeolite Y annealed at different temperatures and 2D representation of the structure of an aluminosilicate network. Copyright 2009, WILEY-VCH; (D) one possible orientation of Bpy2 in a channel of zeolite L, the structure and dimension of different kinds of dyes(Bpy2, Tol2 and BPB). Copyright 2007, American Chemical Society.
| 1 | Wang N., Sun Q. M., Bai R. S., Li X., Guo G. Q., Yu J. H., J. Am. Chem. Soc., 2016, 138, 7484—7487 |
| 2 | Zhu L., Wang L., Zuidema E., Mondal K., Zhang M., Zhang J., Wang C. T., Meng X. J., Yang H. Q., Mesters C., Xiao F. S., Science, 2020, 367, 193—197 |
| 3 | Chai Y. Y., Han X., Li W. Y., Liu S. S., Yao S. K., Wang C., Shi W., daSilva I., Manuel P., Cheng Y. Q., Daemen L. D., RamirezC. A. J., Tang C. C., Jiang L., Yang S. H., Guan N. J., Li L. D., Science, 2020, 368, 1002—1006 |
| 4 | Zhang Q., Mayora A., Terasaki O., Zhang Q., Ma B., Zhao C., Yang G. J., Yu J. H., J. Am. Chem. Soc., 2019, 141, 3772—3776 |
| 5 | Structure Commission of the International Zeolite Association, https://america.iza—structure.org/IZA—SC/ftc_table.php |
| 6 | Li J. Y., Corma A., Yu J. H., Chem. Soc. Rev., 2015, 44, 7112—7127 |
| 7 | Feng G. D., Cheng P., Yan W. F., Boronat M., Su J. H., Wang J. Y., Li Y., Corma A., Xu R. R., Yu J. H., Science, 2016, 351, 1188—1191 |
| 8 | Wang Z. P., Yu J. H., Xu R. R., Chem. Soc. Rev., 2012, 41, 1729—1741 |
| 9 | Li Y., Yu J. H., Chem. Rev., 2014, 114, 7268—7316 |
| 10 | Bai R. S., Song Y., Li Y., Yu J. H., Trends Chem., 2019, 1, 601—611 |
| 11 | Xu R. R., Pang W. Q., Huo Q. S., Molecular Sieve and Porous Material Chemistry, Science Press, Beijing, 2015, 558—594(徐如人, 庞文琴, 霍启升. 分子筛与多孔材料化学, 北京: 科学出版社, 2015, 558—594) |
| 12 | Tzou M. S., Teo B. K., Sachtler W. M. H., J. Catal., 1988, 113, 220—235 |
| 13 | Zhou W. Z., Thomas J. M., Shephard D. S., Johnson B. F. G., Ozkaya D., Maschmeyer T., Bell R. G., Ge Q. F., Science, 1998, 280, 705—708 |
| 14 | Shephard D. S., Maschmeyer T., Johnson B. F. G., Thomas S. J. M., Sankar G., Ozkaya D., Dr. Zhou W. Z., Robert R. D. O., Angew. Chem. Int. Ed., 1997, 36, 2242—2245 |
| 15 | Steele M. R., Macdonald P. M., Ozin G. A., J. Am. Chem. Soc., 1993, 115, 7285—7292 |
| 16 | Ozin G. A., Steele M. R., Holmes A. J., J. Chem. Mater., 1994, 6, 999—1010 |
| 17 | Wickleder M. S., Chem. Rev., 2002, 102, 2011—2087 |
| 18 | Narita K., J. Lumin., 1971, 4, 73—80 |
| 19 | Kasai P. H., J. Chem. Phys., 1965, 43, 3322—3327 |
| 20 | Anderson P. A., Armstrong A. R., Edwards P. P., Angew. Chem. Int. Ed., 1994, 33, 641—643 |
| 21 | Zheng W., Huang P., Tu D. T., Ma E., Zhu H. M., Chen X. Y., Chem. Soc. Rev., 2015, 44, 1379—1415 |
| 22 | Qin Q. S., Zhang P. Z., Sun L. D., Shi S., Chen N. X., Dong H., Zhang X. Y., Li L. M., Yan C. H., Nanoscale, 2017, 9, 4660—4664 |
| 23 | Prodi L ., Rampazzp E., Rastrelli F., Speghini A., Zaccheroni N., Chem. Soc. Rev., 2015, 44, 4922—4952 |
| 24 | Xie J., Gao Z. Y., Zhou E. L., Cheng X. W., Wang Y. B., Xie X. J., Huang L., Huang W., Nanoscale, 2017, 9, 15974—15981 |
| 25 | Wiesholler L. M., Frenzel F., Grauel B., Würth C., Resch-Genger U., Hirsch T., Nanoscale, 2019, 11, 13440—13449 |
| 26 | Yan C. L., Zhao H. G., Perepichka D., Rosel F., Small, 2016, 12, 3888—3907 |
| 27 | Liu B., Chen Y. Y., Li C. H., He F., Hou Z. Y., Huang S. S., Zhu H. M., Chen X. Y., Lin J., Adv. Funct. Mater., 2015, 25, 4717—4729 |
| 28 | Xie X. J., Li Z. J., Zhang Y. W., Guo S. H., Pendharkar A. I., Liu M., Huang L., Huang W., Han G., Small, 2017, 13, 1602843 |
| 29 | Li H. R., Li P., Chem. Commun., 2018, 54, 13884—13893 |
| 30 | Yi X., S J. Y., Jiang X. F., Li Y., Xu Q. H., Zhang Q. Y., Ye S., RSC Adv., 2016, 6, 95925—95935 |
| 31 | Figueiredo B. R., Valente A. A., Lin Z., Silva C. M., Micropor. Mesopor. Mater., 2016, 234, 73—97 |
| 32 | Tian Z. G., Li X., Gao Y. X., Wang Y. H., Peng X. P., J. Lumin., 2019, 211, 62—68 |
| 33 | Kim C. W., Kang H. C., Heo N. H., Seff K., J. Phys. Chem. C, 2015, 119, 24501—24511 |
| 34 | Mass H., Currao A., Calzaferri G., Angew. Chem. Int. Ed., 2002, 41, 2495—2497 |
| 35 | Qin Z. Q., Li H. S., Wang Y. G., Opt. Mater., 2014, 37, 483—487 |
| 36 | Li P., Wang Y. G., Li H. R., Calzaferri G., Angew. Chem. Int. Ed., 2014, 53, 2904—2909 |
| 37 | Bai Z. H., Fujii M., Imakita K., Hayashi S., Micropor. Mesopor. Mater., 2013, 173, 43—46 |
| 38 | Ye S., Sun J., Yi X., Wang Y., Zhang Q., Sci. Rep.,2017, 7, 46219—46228 |
| 39 | Yi X., Sun J., Jiang X. F., Li Y., Xu Q. H., Zhang Q., Ye S., RSC Adv., 2016, 6, 95925—95935 |
| 40 | Mech A., Monguzzi A., Cucinotta F., Meinardi F., Mezyk J., Cola L. D., Tubino R., Phys. Chem. Chem. Phys., 2011, 13, 5605—5609 |
| 41 | Royon A., Bourhis K., Bellec M., Papon G., Bousquet B., Deshayes Y., Cardinal T., Canioni L., Adv. Mater., 2010, 22, 5282—5286 |
| 42 | Díez I., Ras R. H., Nanoscale, 2011, 3, 1963—1970 |
| 43 | Cuong N. T., Nguyen H. M. T., Pham⁃Ho M. P., Nguyen M. T., Phys. Chem. Chem. Phys., 2016, 18, 18128—18136 |
| 44 | Leiggener C., Brühwilera D., Calzaferri G., J. Mater. Chem., 2003, 13, 1969—1977 |
| 45 | Cremer G. D., Gonzalez E. C., Roeffaers M. B. J., Moens B., Ollevier J., Auweraer M. V., Schoonheyd R., Jacobs P., Schryver F. C. D., Hofkens Johan., Vos D. E. De., Sels B. F., Vosch Tom., J. Am. Chem. Soc.,2009, 131, 3049—3056 |
| 46 | Grandjean D., Coutiño⁃Gonzalez E., Cuong N. T., Fron E., Baekelant W., Aghakhani S., Schlexer P., D’Acapito F., Banerjee D., Roeffaers M. B. J., Nguyen M. T., Hofkens J., Lievens P., Science, 2018, 361, 686—690 |
| 47 | Fron E., Aghakhani S., Baekelant W., Grandjean D., CoutinoGonzalez E., Auweraer M. V., Roeffaers M. B. J., Lievens P., Hofkens J., J. Phys. Chem. C, 2019, 123, 10630—10638 |
| 48 | Baekelant W., Aghakhani S., CoutinoGonzalez E., Kennes K., D’Acapito F., Grandjean D., Auweraer M. V., Lievens P., Roeffaers M. B. J., Hofkens J., Steele J., J. Phys. Chem. Lett., 2018, 9, 5344—5350 |
| 49 | Baekelant W., Aghakhani S., CoutinoGonzalez E., Grandjean D., Kennes K., Jonckheere D., Fron E., D’Acapito F., Longo A., Lievens P., Roeffaers M. B. J., Hofkens J., J. Phys. Chem. C, 2018, 122, 13953—13961 |
| 50 | Shi J. Y., Chen J., Feng Z. C., Chen T., Wang X. L., Ying P. L., Li C., J. Phys. Chem. B, 2006, 110, 25612—25618 |
| 51 | Chen W., Sammynaiken R., Huang Y. N., J. Appl. Phys., 2000, 88, 5188—5193 |
| 52 | Chen W., Zhang X. H., Huang Y. N., Appl. Phys. Lett., 2000, 76, 2328—2330 |
| 53 | Xu Y. H., Wang X. X., Zhang W. L., Lv F., Guo S. J., Chem. Soc. Rev., 2018, 47, 586—625 |
| 54 | Stucky G. D., MacDougall J. E., Science, 1990, 247, 669—678 |
| 55 | Sun J. Y., Rabouw F. T., Yang X. F., Huang X. Y., Jing X. P., Ye S., Zhang Q. Y., Adv. Funct. Mater., 2017, 27, 1704371 |
| 56 | Ye S., Sun J. Y., Han Y . H., Zhou Y. Y., Zhang Q. Y., ACS Appl. Mater. Interfaces, 2018, 10, 24656—24664 |
| 57 | Sadjadi M. S., Pourahmad A., Sohrabnezhad Sh., Zare K., Mater. Lett., 2007, 61, 2923—2926 |
| 58 | Jeong N. C., Kim H. S., Yoon K. B., J. Phys. Chem. C, 2007, 111, 10298—10312 |
| 59 | Nath S. S., Chakdar D., Gope G., Kakati J., Kalita B., Talukdar A., Avasthi D. K., J. Appl. Phys., 2009, 105, 094305 |
| 60 | Xu X. Y., Ray R., Gu Y. L., Ploehn H. J., Gearheart L., Raker K., Scrivens W. A., J. Am. Chem. Soc., 2004, 126, 12736—12737 |
| 61 | Zhu S. J., Zhang J. H., Wang L., Song Y. B., Zhang G. Y., Wang H. Y., Yang B., Chem. Commun., 2012,48, 10889—10891 |
| 62 | Xiao L., Sun H., Nanoscale Horiz., 2018, 3, 565—597 |
| 63 | Liu R. L., Wu D. Q., Liu S. H., Koynov K., Knoll W., Li Q., Angew. Chem. Int. Ed., 2009, 48, 4598—4601 |
| 64 | Wang F., Xie Z., Zhang H., Liu C. Y., Zhang Y. G., Adv. Funct. Mater., 2011, 21, 1027—1031 |
| 65 | Xiu Y., Gao Q., Li G. D., Wang K. X., Chen J. S., Inorg. Chem., 2010, 49, 5859—5867 |
| 66 | Baldovi H. G., Valencia S., Alvaro M., Asiri A. M., Garcia H., Nanoscale, 2015, 7, 1744—1752 |
| 67 | Wang Y. Y., Li Y., Yan Y., Xu J., Guan B. Y., Wang Q., Li J. Y., Yu J. H., Chem. Commun., 2013, 49, 9006—9008 |
| 68 | Mu Y., Shi H., Wang Y., Ding H., Li J., J. Mater. Chem. C, 2017, 5, 10894—10899 |
| 69 | Liu J. C., Wang N., Yu Y., Yan Y., Zhang H. Y., Li J. Y., Yu J. H., Sci. Adv., 2017, 3, e1603171 |
| 70 | Liu J. C., Zhang H. Y., Wang N., Yu Y., Cui Y. Z., Li J. Y., Yu J. H., ACS Mater. Lett., 2019, 1, 58—63 |
| 71 | Wang B. L., Mu Y., Zhang H. Y., Shi H. Z., Chen G. R., Yu Y., Yang Z. Q., Li J. Y., Yu J. H., ACS Cent. Sci., 2019, 5, 349—356 |
| 72 | Wang B. L., Yu Y., Zhang H. Y., Xuan Y. Z., Chen G. R., Ma W. Y., Li J. Y., Yu J. H., Angew. Chem. Int. Ed., 2019, 58, 18443—18448 |
| 73 | Zhang H. Y., Liu J. C., Wang B. L., Liu K. K., Chen G. R., Yu X. W., Li J. Y., Yu J. H., Mater. Chem. Front., 2020, 4, 1404—1410 |
| 74 | Zhang H. Y., Liu K. K., Liu J. C., Wang B. L., Li C. Y., Song W., Li J. Y., Liu H., Yu J. H., CCS Chem., 2020, 2, 118—127 |
| 75 | Xu X. T., Chen J. Q., Shi W. T., Sun D. L., Chu S. W., Sun L., Zhai J. P., Pei J. H., Wang L., Ruan S. C., Tang Z. K., J. Alloys Compd., 2019, 782, 837—844 |
| 76 | Xu X. T., Chen J. Q., Shi W. T., Sun D. L ., Chu S. W., Sun L., Zhang W. F., Chen Y. P., Zhai J. P., Ruan S. C., Tang Z. K ., Pho⁃tonics Res., 2019, 7, 1182—1187 |
| 77 | Meng S. Q., Zhou Y. Y., Wan W., Ye S., Zhang Q. Y., J. Mater. Chem. C, 2019, 7, 1345—1352 |
| 78 | Wan W., Sun J. Y., Ye S., Zhang Q. Y., RSC Adv., 2018, 8, 25057—25064 |
| 79 | Sun H. T., Sakka Y. S., Shirahata N., Matsushita Y., Deguchi K., Shimizu T., J. Phys. Chem. C, 2013, 117, 6399—6408 |
| 80 | Sun H. T., Matsushita Y., Sakka Y., Shirahata N., Tanaka M., Katsuya Y., Gao H., Kobayashi K., J. Am. Chem. Soc., 2012, 134, 2918—2921 |
| 81 | Sun H. T., Hosokawa A., Miwa Y., Shimaoka F., Fujii M., Mizuhata M., Hayashi S., Deki S., Adv. Mater., 2009, 21, 3694—3698 |
| 82 | Yoon S. Y., Kim H. J., Heo N. H., Seff K., Micropor. Mesopor. Mater., 2019, 288, 109552 |
| 83 | Park G. B., Kim J. Y., Kim H. J., Heo N. H., Seff K., J. Porous Mater., 2019, 26, 1079—1089 |
| 84 | Bussemer B., Munsel D., Wünscher H., Mohr G. J., Grummt U., J. Phys. Chem. B, 2007, 111, 8—15 |
| 85 | Pham T. C. T., Kim H. S., Yoon K. B., Angew. Chem. Int. Ed., 2013, 52, 5539—5543 |
| [1] | 王瑞娜, 孙瑞粉, 钟添华, 池毓务. 大尺寸石墨烯量子点组装体的制备及电化学发光性能[J]. 高等学校化学学报, 2022, 43(8): 20220161. |
| [2] | 周永辉, 李尧, 吴雨轩, 田晶, 徐龙权, 费旭. 一种新型光致发光自愈合水凝胶的合成[J]. 高等学校化学学报, 2022, 43(2): 20210606. |
| [3] | 伍泽鑫, 朱渊杰, 王泓中, 王均安, 贺英. 甲基修饰的咔唑/二苯砜基AIE-TADF蓝光材料及其OLED器件[J]. 高等学校化学学报, 2022, 43(11): 20220371. |
| [4] | 矫龙, 代学民, 牟建新, 杜志军, 王汉夫, 董志鑫, 邱雪鹏. 柔性OLED用高耐热聚酰亚胺薄膜的制备与性能[J]. 高等学校化学学报, 2022, 43(11): 20220390. |
| [5] | 李伦, 张静妍, 罗静, 刘仁, 朱乙. UV/Vis-LED激发的香豆素吡啶鎓盐光引发剂的合成及性能[J]. 高等学校化学学报, 2022, 43(10): 20220178. |
| [6] | 韩宗甦, 于晓泳, 闵辉, 师唯, 程鹏. 一个基于偶氮六酸的稀土金属-有机框架[J]. 高等学校化学学报, 2022, 43(1): 20210342. |
| [7] | 周永慧, 黄如军, 严健洋, 李亚军, 邱欢欢, 杨进轩, 郑佑轩. 两种基于氮杂环结构铱(Ⅲ)配合物的合成及有机电致发光性能[J]. 高等学校化学学报, 2022, 43(1): 20210415. |
| [8] | 杜顺福, 王文经, EL-SAYED El-Sayed M., 苏孔钊, 袁大强, 洪茂椿. 一种具有化学发光性能的锆基金属有机四面体[J]. 高等学校化学学报, 2022, 43(1): 20210628. |
| [9] | 袁博, 祁超超, 张相挺, 栾国颜, 邹海峰. 基于能量传递可调光色荧光粉Ca2LaTaO6∶ Dy3+, Sm3+的发光性质及其发光二极管器件应用[J]. 高等学校化学学报, 2021, 42(9): 2717. |
| [10] | 魏敏敏, 袁泽, 闾敏, 马辉, 谢小吉, 黄岭. 稀土掺杂上转换纳米颗粒-金属有机骨架复合材料的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2313. |
| [11] | 王坤华, 姚纪松, 杨俊楠, 宋永慧, 刘雨莹, 姚宏斌. 金属卤化物钙钛矿纳米晶高效发光二极管的制备与器件性能优化[J]. 高等学校化学学报, 2021, 42(5): 1464. |
| [12] | 孟妍, 王秀凤, 张莉, 刘鸣华. 不同取代位置的萘衍生两亲分子在气/液界面组装膜中的螺旋结构与圆偏振发光[J]. 高等学校化学学报, 2021, 42(4): 1253. |
| [13] | 孟利利, 陈琳琳, 张小亮, 解令海, 刘欢. 可控液体输运制备取向聚合物薄膜:面向性能增强的发光二极管[J]. 高等学校化学学报, 2021, 42(4): 1260. |
| [14] | 李健, 于明明, 孙源, 冯文华, 冯兆池, 吴剑峰. 水溶液pH对甲烷低温氧化制备甲醇的影响[J]. 高等学校化学学报, 2021, 42(3): 776. |
| [15] | 张慧东, 谷盼盼, 张芳, 都明旭, 叶开其, 刘宇. 窄光谱磷光配合物的设计及电致发光性能[J]. 高等学校化学学报, 2021, 42(12): 3571. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||