

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (7): 1475.doi: 10.7503/cjcu20180117
• Physical Chemistry • Previous Articles Next Articles
FANG Sheng, WANG Meiyan*( ), LIU Jingjing, LIU Jingyao
), LIU Jingjing, LIU Jingyao
Received:2018-02-09
Online:2018-07-10
Published:2018-06-14
Contact:
WANG Meiyan
E-mail:mywang858@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
FANG Sheng, WANG Meiyan, LIU Jingjing, LIU Jingyao. Reaction Mechanism of Nickel Complex Catalyzed Isomerization of N-Allylamides†[J]. Chem. J. Chinese Universities, 2018, 39(7): 1475.
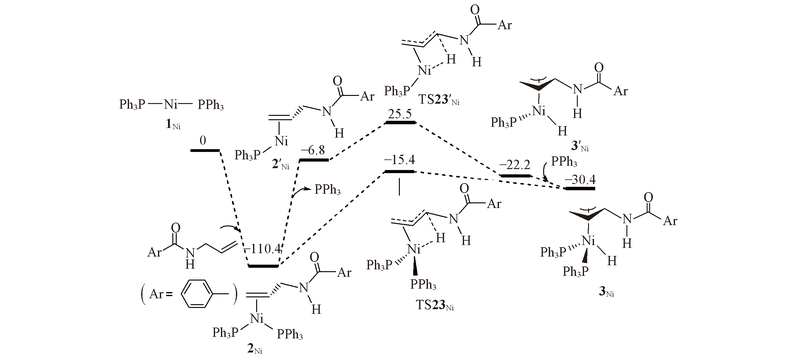
Fig.1 Energy profiles calculated for C—H bond activation by Ni(PPh3)2(1Ni) to form complex 3Ni^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
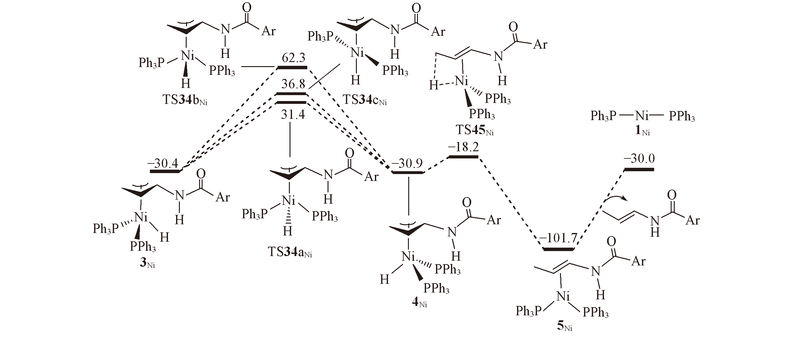
Fig.2 Energy profiles calculated for isomerization of π-allyl complex 3Ni via the π-allyl mechanism to generate E isomer^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
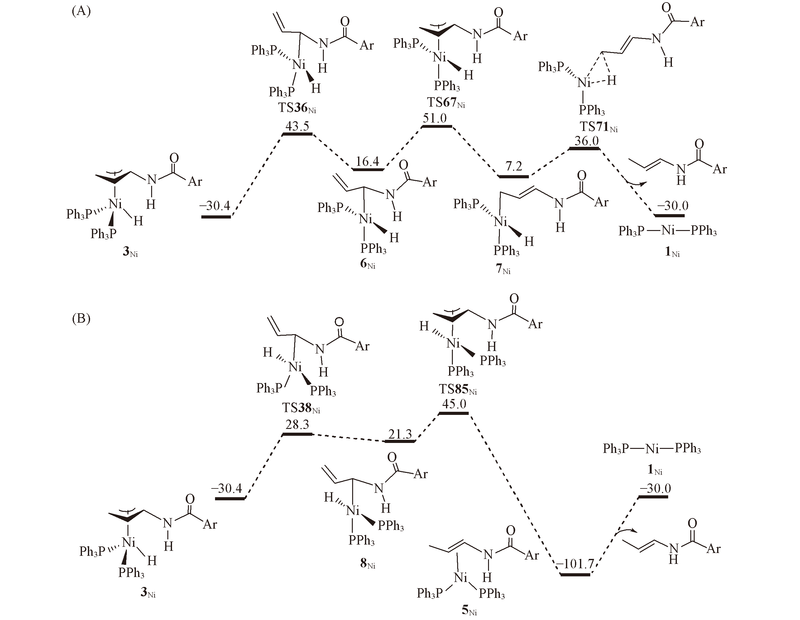
Fig.3 Energy profiles calculated for isomerization of π-allyl complex 3Ni via the σ-allyl mechanism to generate E isomer^ The H atom bonded to Ni is directed outwards(A) and inwards(B). The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
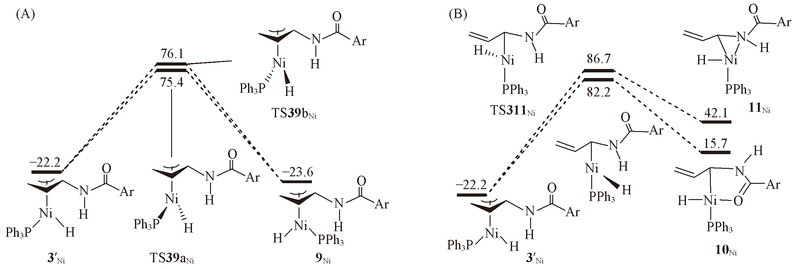
Fig.4 Energy profiles calculated for isomerization of π-allyl complex 3'Ni via the π-allyl(A) and σ-allyl(B) mechanism^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
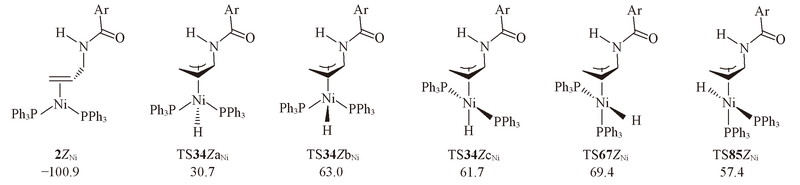
Fig.5 Structures and energies of the rate-determining intermediate 2ZNi and five lowest transition states on the energy profiles to generate Z isomer^The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
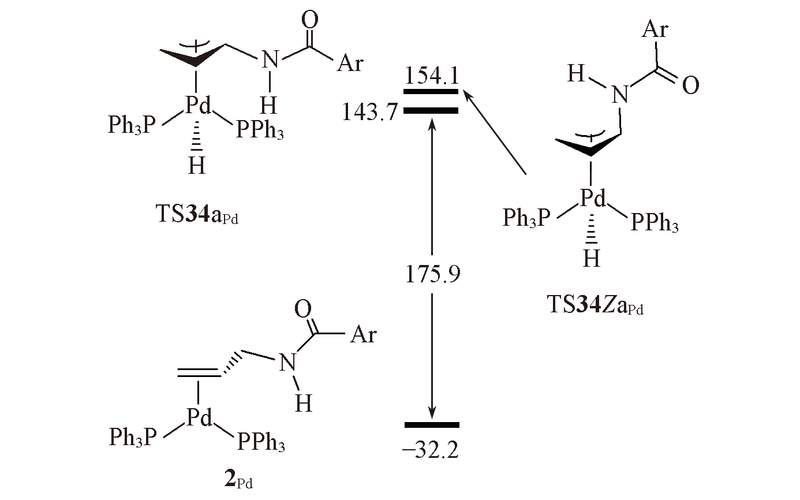
Fig.6 Structures and energies of the rate-determining intermediate 2Pd and transition states TS34aPd and TS34ZaPd on the energy profiles to generate E and Z isomer^The calculated free energies relative to 1Pd+N-allyl-4-methylbenzamide are given in kJ/mol.
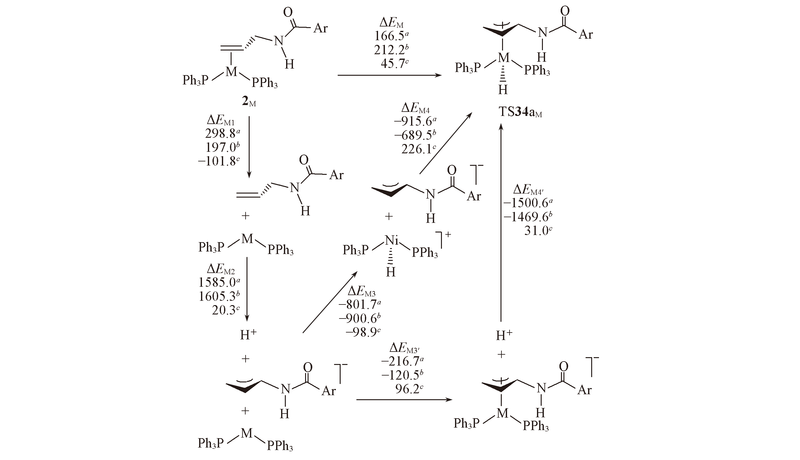
Fig.7 Energy decomposition analyses of the energy barriers(kJ/mol) from rate-determining intermediates 2M to transition states TS34aM^ a. The energy of Ni; b. the energy of Pd; c. the energy difference of Ni and Pd.
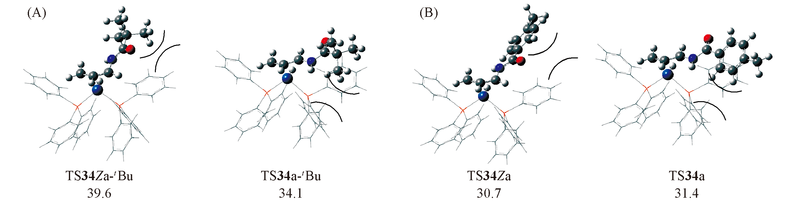
Fig.8 Structures and energies(kJ/mol) of two rate-determining transition states TS34a-tBu and TS34Za-tBu(A) for reactant with R=tBu (relative to 1Ni+N-allyl-pivaloylamide), as well as TS34a and TS34Za(B) for reactant with R=p-MeC6H4 (relative to 1Ni+N-allyl-4-methylbenzamide)
| [1] | Van Santen R.A.,van Leeuwen P. W. N. M.,Moulijn J A.,Averill B. A., Catalysis: An Integrated Approach, Elsevier,Amsterdam, 1999 |
| [2] | Mirza-Aghayan M., Boukherroub R., Bolourtchian M., Hoseini M., Tabar-Hydar K., J. Organomet. Chem., 2003, 678, 1—4 |
| [3] | Donohoe T. J., O'Riordan T. J. C., Rosa C. P., Angew. Chem. Int. Ed., 2009, 48, 1014—1017 |
| [4] | Krompiec S., Krompiec M., Penczek H., Ignasiak R., Coord. Chem. Rev., 2008, 252, 1819—1841 |
| [5] | Escoubet S., Gastaldi S., Bertrand M.,Eur. J. Org. Chem., 2005, 3855—3873 |
| [6] | Kramer S., Mielby J., Buss K., Kasama T., Kegnæs S., Chem. Cat. Chem., 2017, 9, 2930—2934 |
| [7] | Wu Q., Wang L., Jin R., Kang C., Bian Z., Du Z., Ma X., Guo H., Gao L.,Eur. J. Org. Chem., 2016, 5415—5422 |
| [8] | Kocen A. L., Brookhart M., Daugulis O., Chem. Commun., 2017, 53, 10010—10013 |
| [9] | Zhuo L. G., Yao Z. K., Yu Z. X., Org. Lett., 2013, 15, 4634—4637 |
| [10] | Stille J.K., Becker Y., J. Org. Chem., 1980, 45, 2139—2145 |
| [11] | Sergeyev S. A., Hesse M., Helvetica Chimica Acta, 2003, 86, 750—755 |
| [12] | Yamada H., Sodeoka M., Shibasaki M., J. Org. Chem., 1991, 56, 4569—4574 |
| [13] | Zacuto M. J., Xu F., J. Org. Chem., 2007, 72, 6298—6300 |
| [14] | Krompiec S., Pigulla M., Kuz'nik N., Krompiec M., Marciniec B., Chadyniak D., Kasperczyk J., J. Mol. Cat. A: Chem., 2005, 225, 91—101 |
| [15] | Krompiec S., Pigulla M., Bieg T., Szczepankiewicz W., Kuz'nik N., Krompiec M., Kubicki M., J. Mol. Cat. A: Chem., 2002, 189, 169—185 |
| [16] | Krompiec S., Pigulla M., Szczepankiewicz W., Bieg T., Kuznik N., Leszczynska-Sejda K., Kubicki M., Borowiak T., Tetrahedron Lett., 2001, 42, 7095—7098 |
| [17] | Neugnot B., Cintrat J. C., Rousseau B., Tetrahedron, 2004, 60, 3575—3579 |
| [18] | Nakanishi S., Otsuji Y., Itoh K., Hayashi N., Bull. Chem. Soc. Jpn., 1990, 63, 3595—3600 |
| [19] | Couture A., Deniau E., Grandclaudon P., Lebrun S., Tetrahedron Lett., 1996, 37, 7749—7752 |
| [20] | Naito T., Yuumoto Y., Kiguchi T., Ninomiya I., J. Chem. Soc., Perkin Trans., 1996, 1, 281—288 |
| [21] | Wang L., Liu C., Bai R., Pan Y., Lei A., Chem. Commun., 2013, 49, 7923—7925 |
| [22] | Kozuch S., Shaik S., J. Am. Chem. Soc., 2017, 11, 4075—4086 |
| [23] | Zhang X., Tutkowski B., Oliver A., Helquist P., Wiest O., ACS Catal., 2018, 8, 1740—1747 |
| [24] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Keith T., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas O., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision E. 01, Gaussian Inc., Wallingford CT, 2013 |
| [25] | Becke A. D., Phys. Rev.A, 1988, 38, 3098—3100 |
| [26] | Lee C., Yang W., Parr R. G., Phys. Rev. B, 1988, 37, 785—789 |
| [27] | Becke A. D., J. Chem.Phys., 1993, 98, 5648—5652 |
| [28] | Hay P. J., Wadt W. R., J. Chem. Phys., 1985, 82, 270—283 |
| [29] | Hay P. J., Wadt W. R., J. Chem. Phys., 1985, 82, 299—310 |
| [30] | Ehlers A. W., Böhme M., Dapprich S., Gobbi A., Höllwarth A., Jonas V., Köhler K. F., Stegmann R., Veldkamp A., Frenking G., Chem. Phys. Lett., 1993, 208, 111—114 |
| [31] | Fukui K., Acc. Chem.Res., 1981, 14, 363—368 |
| [32] | Zhao Y., Truhlar D. G., J. Chem. Phys., 2006, 125, 194101 |
| [33] | Dolg M., Wedig U., Stoll H., Preuss H., J. Chem.Phys., 1987, 86, 866—872 |
| [34] | Andrae D., Haeussermann U., Dolg M., Stoll H., Preuss H., Theor. Chem. Acc., 1990, 77, 123—141 |
| [35] | Marenich A. V., Cramer C. J., Truhlar D. G., J. Phys. Chem. B, 2009, 113, 6378—6396 |
| [36] | Kozuch S., Shaik S., Acc. Chem. Res., 2011, 44, 101—110 |
| [37] | Xie H., Kuang J., Wang L., Li Y., Huang L., Fan T., Lei Q., Fang W., Organometallics, 2017, 36, 3371—3381 |
| [1] | ZHANG Mi, TIAN Yafeng, GAO Keli, HOU Hua, WANG Baoshan. Molecular Dynamics Simulation of the Physicochemical Properties of Trifluoromethanesulfonyl Fluoride Dielectrics [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220424. |
| [2] | LIU Yang, LI Wangchang, ZHANG Zhuxia, WANG Fang, YANG Wenjing, GUO Zhen, CUI Peng. Theoretical Exploration of Noncovalent Interactions Between Sc3C2@C80 and [12]Cycloparaphenylene Nanoring [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220457. |
| [3] | WANG Sijia, HOU Lu, LI Chenglong, LI Wencui, LU Anhui. Recent Advances in Synthesis and Applications of Hollow Nano-carbons [J]. Chem. J. Chinese Universities, 0, (): 20220637. |
| [4] | WU Qingying, ZHU Zhenyu, WU Jianming, XU Xin. A Dataset Representativeness Metric and A Slicing Sampling Strategy for the Kennard-Stone Algorithm [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220397. |
| [5] | WANG Yuanyue, AN Suosuo, ZHENG Xuming, ZHAO Yanying. Spectroscopic and Theoretical Studies on 5-Mercapto-1,3,4-thiadiazole-2-thione Microsolvation Clusters [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220354. |
| [6] | ZHANG Lingyu, ZHANG Jilong, QU Zexing. Dynamics Study of Intramolecular Vibrational Energy Redistribution in RDX Molecule [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220393. |
| [7] | SHEN Qi, CHEN Haiyao, GAO Denghui, ZHAO Xi, NA Risong, LIU Jia, HUANG Xuri. A Study on the Interaction Mechanism of the Natural Product Falcarindiol with Human GABAA Receptor [J]. Chem. J. Chinese Universities, 0, (): 0. |
| [8] | CHEN Shaochen, CHENG Min, WANG Shihui, WU Jinkui, LUO Lei, XUE Xiaoyu, JI Xu, ZHANG Changchun, ZHOU Li. Transfer Learning Modeling for Predicting the Methane and Hydrogen Delivery Capacity of Metal-Organic Frameworks [J]. Chem. J. Chinese Universities, 0, (): 20220459. |
| [9] | PENG Xinzhe, GE Jiaoyang, WANG Fangli, YU Guojing, ZHOU Dong, RAN Xueqin, YANG Lei, XIE Linghai. A Theoretical Study on Tension and Reorganization Energy of Benzothiophene Grid [J]. Chem. J. Chinese Universities, 0, (): 20220313. |
| [10] | GUO Cheng, ZHANG Wei, TANG Yun. Ordered Mesoporous Materials: History, Progress and Perspective [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220167. |
| [11] | TANG Qiaowei, CAI Xiaoqing, LI Jiang, ZHU Ying, WANG Lihua, TIAN Yang, FAN Chunhai, HU Jun. Synchrotron-based X-ray Microscopy for Brain Imaging [J]. Chem. J. Chinese Universities, 0, (): 20220379. |
| [12] | YANG Dan, LIU Xu, DAI Yihu, ZHU Yan, YANG Yanhui. Research Progress in Electrocatalytic CO2 Reduction Reaction over Gold Clusters [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220198. |
| [13] | DAI Wei, HOU Hua, WANG Baoshan. Theoretical Investigations on the Electronic Structures and Reactivity of Heptafluoro-iso-butyronitrile Anion [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220044. |
| [14] | SHI Naike, ZHANG Ya, SANSON Andrea, WANG Lei, CHEN Jun. Uniaxial Negative Thermal Expansion and Mechanism in Zn(NCN) [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220124. |
| [15] | REN Nana, XUE Jie, WANG Zhifan, YAO Xiaoxia, WANG Fan. Effects of Thermodynamic Data on Combustion Characters of 1,3-Butadiene [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220151. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||