

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (5): 941.doi: 10.7503/cjcu20170756
Previous Articles Next Articles
XIA Kun, WANG Yi, ZHOU Dan*, HUANG Zhe, WU Zhonghan, XIA Qinghua*
Received:2017-11-22
Online:2018-04-08
Published:2018-04-08
Contact:
ZHOU Dan,XIA Qinghua
Supported by:CLC Number:
TrendMD:
XIA Kun,WANG Yi,ZHOU Dan,HUANG Zhe,WU Zhonghan,XIA Qinghua. Rapid Synthesis of CoSAPO-5 Zeolite and Efficiently Catalytic Epoxidation of α-Pinene with Air†[J]. Chem. J. Chinese Universities, 2018, 39(5): 941.
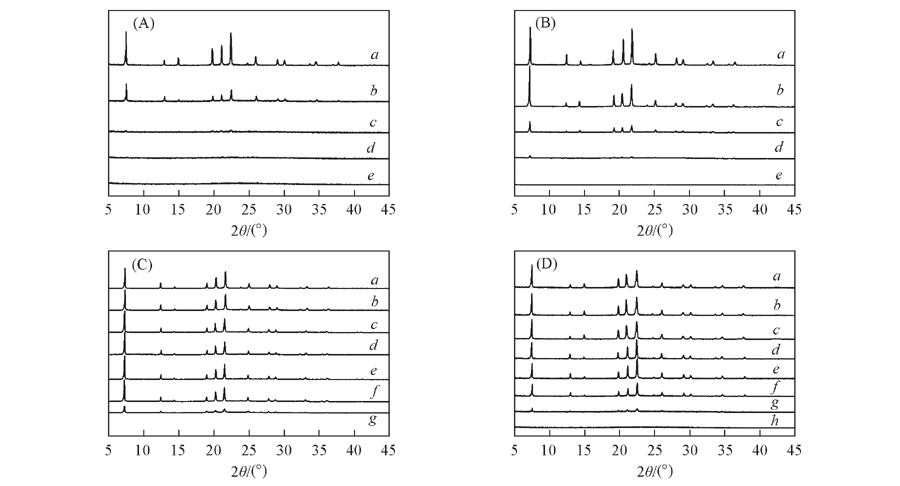
Fig.2 XRD patterns of SAPO-5 samples prepared under static(A) and at rotating rates of 56 r/min(B), 120 r/min(C) and 140 r/min(D), respectively Note:Reaction time/h: a. 24; b. 12; c. 8; d. 6; e. 4; f. 3; g. 2; h. 1.

Fig.3 SEM images of SAPO-5 samples prepared under static and rotating conditions with different magnifications Note:A), (A') SAPO-5-Static-24; (B), (B') SAPO-5-56-24; (C), (C') SAPO-5-56-12; (D), (D') SAPO-5-120-12; (E), (E') SAPO-5-140-24; (F), (F') SAPO-5-140-12; (G), (G') SAPO-5-140-4; (H), (H') SAPO-5-140-3.
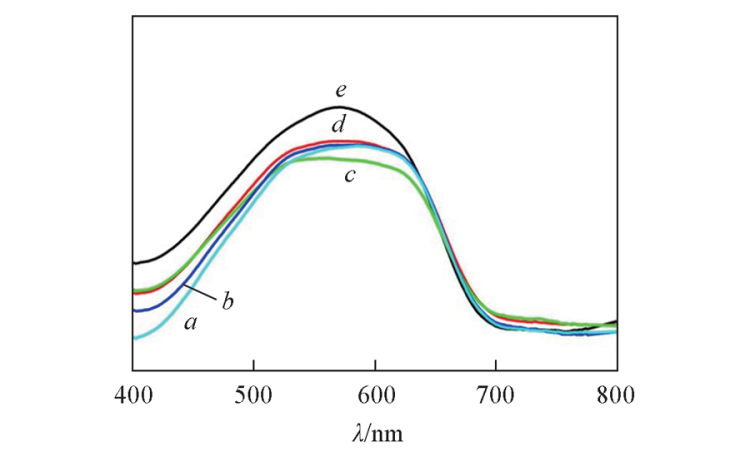
Fig.5 UV-Vis spectra of CoSAPO-5 samples prepared under static and rotating conditions Note:a. CoSAPO-5-Static-24; b. CoSAPO-5-140-3; c. CoSAPO-5-140-4; d. CoSAPO-5-140-12; e. CoSAPO-5-140-24.
| Sample | Mass fraction of Co(%) | Sample | Mass fraction of Co(%) |
|---|---|---|---|
| CoSAPO-5-Static-24 | 0.421 | CoSAPO-5-140-12 | 0.711 |
| CoSAPO-5-140-3 | 0.545 | CoSAPO-5-140-24 | 0.738 |
| CoSAPO-5-140-4 | 0.696 |
Table 1 ICP characterization results of CoSAPO-5 samples
| Sample | Mass fraction of Co(%) | Sample | Mass fraction of Co(%) |
|---|---|---|---|
| CoSAPO-5-Static-24 | 0.421 | CoSAPO-5-140-12 | 0.711 |
| CoSAPO-5-140-3 | 0.545 | CoSAPO-5-140-24 | 0.738 |
| CoSAPO-5-140-4 | 0.696 |
| Sample | Relative crystallinity(%) | Sample | Relative crystallinity(%) |
|---|---|---|---|
| SAPO-5-Static-24 | 76.7% | SAPO-5-140-12 | 96.3% |
| SAPO-5-140-3 | 81.1% | SAPO-5-140-24 | 100% |
| SAPO-5-140-4 | 87.5% |
Table 2 Relative crystallinity of SAPO-5 samples prepared under rotation and static conditions
| Sample | Relative crystallinity(%) | Sample | Relative crystallinity(%) |
|---|---|---|---|
| SAPO-5-Static-24 | 76.7% | SAPO-5-140-12 | 96.3% |
| SAPO-5-140-3 | 81.1% | SAPO-5-140-24 | 100% |
| SAPO-5-140-4 | 87.5% |
| Catalyst | Conversion(%) | Selectivity(%) | Yield(%) |
|---|---|---|---|
| CoSAPO-5-Static-8 | 15.3 | 92.3 | 14.6 |
| CoSAPO-5-Static-12 | 19.2 | 92.6 | 17.7 |
| CoSAPO-5-Static-24 | 90.2 | 92.4 | 83.3 |
| CoSAPO-5-56-8 | 22.6 | 92.9 | 21.0 |
| CoSAPO-5-56-12 | 91.5 | 92.2 | 84.4 |
| CoSAPO-5-56-24 | 92.1 | 92.5 | 85.1 |
| CoSAPO-5-120-3 | 93.7 | 92.3 | 86.5 |
| CoSAPO-5-120-4 | 93.9 | 92.4 | 86.8 |
| CoSAPO-5-120-8 | 94.3 | 92.2 | 86.9 |
| CoSAPO-5-120-12 | 93.8 | 92.5 | 86.8 |
| CoSAPO-5-140-3 | 96.5 | 92.2 | 89.0 |
| CoSAPO-5-140-4 | 96.3 | 92.3 | 88.9 |
| CoSAPO-5-140-8 | 95.9 | 92.1 | 88.3 |
| CoSAPO-5-140-12 | 96.0 | 92.2 | 88.5 |
Table 3 Epoxidation of α-pinene with air over various CoSAPO-5 catalysts*
| Catalyst | Conversion(%) | Selectivity(%) | Yield(%) |
|---|---|---|---|
| CoSAPO-5-Static-8 | 15.3 | 92.3 | 14.6 |
| CoSAPO-5-Static-12 | 19.2 | 92.6 | 17.7 |
| CoSAPO-5-Static-24 | 90.2 | 92.4 | 83.3 |
| CoSAPO-5-56-8 | 22.6 | 92.9 | 21.0 |
| CoSAPO-5-56-12 | 91.5 | 92.2 | 84.4 |
| CoSAPO-5-56-24 | 92.1 | 92.5 | 85.1 |
| CoSAPO-5-120-3 | 93.7 | 92.3 | 86.5 |
| CoSAPO-5-120-4 | 93.9 | 92.4 | 86.8 |
| CoSAPO-5-120-8 | 94.3 | 92.2 | 86.9 |
| CoSAPO-5-120-12 | 93.8 | 92.5 | 86.8 |
| CoSAPO-5-140-3 | 96.5 | 92.2 | 89.0 |
| CoSAPO-5-140-4 | 96.3 | 92.3 | 88.9 |
| CoSAPO-5-140-8 | 95.9 | 92.1 | 88.3 |
| CoSAPO-5-140-12 | 96.0 | 92.2 | 88.5 |
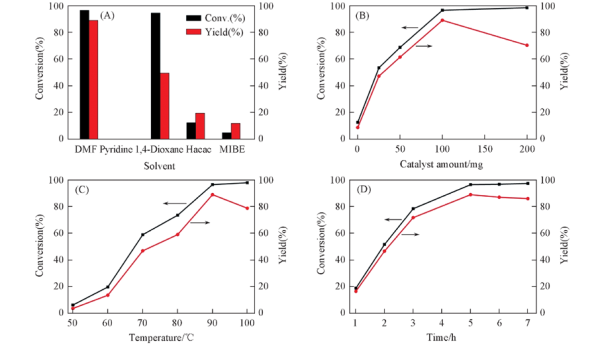
Fig.6 Effects of solvent(A), catalyst amount(B), reaction temperature(C) and time(D) on the epoxidation of α-pinene over CoSAPO-5-140-3 Note:Reaction conditions:α-pinene: 3 mmol; solvent: 10.0 g; flow rate of dry air: 40 mL/min; catalyst: 0—200 mg; CHP: 0.3 mmol; reaction temperature: 50—100 ℃; reaction time: 1—7 h.
| Alkene | Epoxide | Conversion(%) | Selectivity(%) | Yield(%) |
|---|---|---|---|---|
| 98.6 | 83.7 | 82.5 | ||
| 84.4 | 85.1 | 71.8 | ||
| 61.8 | 100 | 61.8 | ||
| 42.5b | 62.5b | 26.6 | ||
| 4.1 | 100 | 4.1 |
Table 4 Epoxidation of different substrates over Co-SAPO-5-140-3 catalystsa
| Alkene | Epoxide | Conversion(%) | Selectivity(%) | Yield(%) |
|---|---|---|---|---|
| 98.6 | 83.7 | 82.5 | ||
| 84.4 | 85.1 | 71.8 | ||
| 61.8 | 100 | 61.8 | ||
| 42.5b | 62.5b | 26.6 | ||
| 4.1 | 100 | 4.1 |
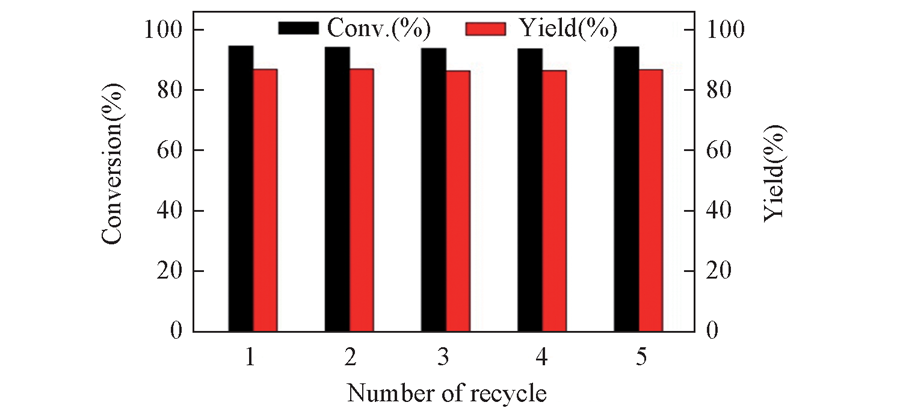
Fig.7 Recycling tests of CoSAPO-5-140-3 catalyst Note:Reaction conditions:α-pinene: 3 mmol; DMF: 10.0 g; catalyst: 100 mg; CHP: 0.3 mmol; time: 5 h; temperature: 90 ℃; flow rate of air: 40 mL/min.
| [1] | Xia Q.H., Ge H. Q., Ye C. P., Liu Z. M., Su K. X., Chem. Rev., 2005, 105, 1603—1662 |
| [2] | Chu X., Zhou D., Li D., Xia K., Gan N., Lu X.H., Nie R. F., Xia Q. H., Micropor. Mesopor. Mater., 2016, 230, 166—176 |
| [3] | Kubi<inline-formula><mml:math xmlns:mml="http://www.w3.org/1998/Math/MathML" id="Mml1-0251-0790-39-5-941"><mml:mtable frame="none" columnlines="none" rowlines="none"><mml:mtr><mml:mtd><mml:maligngroup/><mml:mrow><mml:mover><mml:mrow><mml:mi mathvariant="normal">c</mml:mi></mml:mrow><mml:mrow><mml:mi mathvariant="normal">˙</mml:mi></mml:mrow></mml:mover></mml:mrow></mml:mtd></mml:mtr></mml:mtable></mml:math></inline-formula>ka D., Kubi<inline-formula><mml:math xmlns:mml="http://www.w3.org/1998/Math/MathML" id="Mml2-0251-0790-39-5-941"><mml:mtable frame="none" columnlines="none" rowlines="none"><mml:mtr><mml:mtd><mml:maligngroup/><mml:mrow><mml:mover><mml:mrow><mml:mi mathvariant="normal">c</mml:mi></mml:mrow><mml:mrow><mml:mi mathvariant="normal">˙</mml:mi></mml:mrow></mml:mover></mml:mrow></mml:mtd></mml:mtr></mml:mtable></mml:math></inline-formula>kov�� I., <inline-formula><mml:math xmlns:mml="http://www.w3.org/1998/Math/MathML" id="Mml3-0251-0790-39-5-941"><mml:mtable frame="none" columnlines="none" rowlines="none"><mml:mtr><mml:mtd><mml:maligngroup/><mml:mrow><mml:mover><mml:mrow><mml:mi mathvariant="normal">C</mml:mi></mml:mrow><mml:mrow><mml:mi mathvariant="normal">˙</mml:mi></mml:mrow></mml:mover></mml:mrow></mml:mtd></mml:mtr></mml:mtable></mml:math></inline-formula>ejka [J]., Catal. Rev., 2013, 55, 1—78 |
| [4] | Monteiro J., Veloso C., Top. Catal., 2004, 27, 80—169 |
| [5] | Milchert E., Goc W., Meissner E., Myszkowski J., [J]. Chem. Techmol. Biotechmol., 1994, 61(3), 251—254 |
| [6] | Solati Z., Hashemi M., Ebrahimi L., Catal. Lett., 2011, 141, 163—167 |
| [7] | Li K., Zhou D., Deng J.J., Lu X. H., Xia Q. H., J. Mol. Catal. A: Chem., 2014, 387, 31—37 |
| [8] | Zhou D., Zhang T.J., Xia Q. H., Zhao Y. R., Lv K. X., Lu X. H., Nie R. F., Chem. Sci., 2016, 7, 4966—4972 |
| [9] | Wang L., Sun J., Meng X.J., Zhang W. P., Zhang J., Pan S. X., Shen Z., Xiao F. S., Chem. Commun., 2014, 50, 2012—2014 |
| [10] | Qi B., Lu X.H., Zhou D., Xia Q. H., Tang Z. R., Fang S. Y., Pang T., Dong Y. L., J. Mol. Catal. A: Chem., 2010, 322, 73—79 |
| [11] | Shan Z.C., Wang H., Meng X. J., Liu S. Y., Wang L., Wang C. Y., Li F., Lewis J. P.,Xiao F. S., Chem. Commun., 2011, 47, 1048—1050 |
| [12] | Tang B., Lu X.H., Zhou D., Lei J., Niu Z. H., Fan J., Xia Q. H., Catal. Commun., 2012, 21, 68—71 |
| [13] | Wei X.L., Lu X. H., Zhang T. J., Chu X., Zhou D., Nie R.F., Xia Q. H., Micropor. Mesopor. Mater., 2015, 214, 80—87 |
| [14] | Zhou D., Tang B., Lu X.H., Wei X. L., Li K., Xia Q. H., Catal. Commun., 2014, 45, 124—128 |
| [15] | Wang Z., Yu J.H., Xu R. R., Chem. Soc. Rev., 2012, 41, 1729—1741 |
| [16] | Chang X.W., Yan W. F., Shi W., Xu R. R., Chem. [J]. Chinese Universities, 2018, 39(1), 12—18 |
| (常晓文, 闫文付, 师唯, 徐如人. 高等学校化学学报, 2018, 39(1), 12—18) | |
| [17] | Liu R., Zha F., Yang A.M., Chang Y., Chem. [J]. Chinese Universities, 2016, 37(5), 964—971 |
| (刘蓉, 查飞, 杨爱梅, 常玥. 高等学校化学学报, 2016, 37(5), 964—971) | |
| [18] | Liu N., Wang J.Q., Chen B. H., Li Y. X., Zhang R. D., Chem. [J]. Chinese Universities, 2016, 37(10), 1817—1825 |
| (刘宁, 王继琼, 陈标华, 李英霞, 张润铎. 高等学校化学学报, 2016, 37(10), 1817—1825) | |
| [19] | Li Y., Yu J.H., Chem. Rev., 2014, 114, 7268—7316 |
| [20] | Xu R.R., Pang W. Q., Yu J. H., Huo Q. S., Chen J. S., Chemistry of Zeolites and Related Porous Materials, John Wiley & Sons,(Asia) Pte Ltd., 2007 |
| [21] | Zhou D., Chen L., Yu J.H., Li Y., Yan W. F., Deng F., Xu R. R., Inorg. Chem., 2005, 44, 4391—4397 |
| [22] | Zhou D., Lu X.H., Xu J., Yu A. A., Li J. Y., Deng F., Xia Q. H., Chem. Mater., 2012, 24, 4160—4165 |
| [23] | Xi D.Y., Sun Q. M., Chen X. X., Wang N., Yu J. H., Chem. Commun., 2015, 51, 11987—11989 |
| [24] | Li M., Zeng C.F., Zhang L. X., CrystEngComm, 2012, 14, 3787—3792 |
| [25] | Shen W.L., Li X., Wei Y. X., Tian P., Deng F., Han X. W., Bao X. H., Micropor. Mesopor. Mater., 2012, 158, 19—25 |
| [1] | YAO Yiting, LYU Jiamin, YU Shen, LIU Zhan, LI Yu, LI Xiaoyun, SU Baolian, CHEN Lihua. Preparation of Hierarchical Microporous-mesoporous Fe2O3/ZSM-5 Hollow Molecular Sieve Catalytic Materials and Their Catalytic Properties for Benzylation [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220090. |
| [2] | LI Zhiguang, QI Guodong, XU Jun, DENG Feng. Role of Catalyst Acidity in Glucose Conversion over Sn-Al-β Zeolite as Studied by Solid-state NMR [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220138. |
| [3] | CHEN Weiqin, LYU Jiamin, YU Shen, LIU Zhan, LI Xiaoyun, CHEN Lihua, SU Baolian. Preparation of Organic Hybrid Mesoporous Beta Zeolite for Alkylation of Mesitylene with Benzyl Alcohol [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220086. |
| [4] | LI Jiafu, ZHANG Kai, WANG Ning, SUN Qiming. Research Progress of Zeolite-encaged Single-atom Metal Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220032. |
| [5] | MENG Xianglong, YANG Ge, GUO Hailing, LIU Chenguang, CHAI Yongming, WANG Chunzheng, GUO Yongmei. Synthesis of Nano-zeolite and Its Adsorption Performance for Hydrogen Sulfide [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210687. |
| [6] | WEI Lina, PENG Li, ZHU Feng, GU Pengfei, GU Xuehong. Preparation of Au-CeZr/FAU Catalytic Membranes for Preferential Oxidation of CO in H2-rich Stream [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220175. |
| [7] | LI Yichuan, ZHU Guofu, WANG Yu, CHAI Yongming, LIU Chenguang, HE Shengbao. Effects of Substrate Surface Properties and Precursor Chemical Environment on In⁃situ Oriented Construction of Titanium Silicalite Zeolite Membranes [J]. Chem. J. Chinese Universities, 2021, 42(9): 2934. |
| [8] | LUO Qiangqiang, JIN Shaoqing, SUN Hongmin, YANG Weimin. Post-synthesis of Ti-MWW Zeolite via Titanium Incorporation in Liquid Acid Solution [J]. Chem. J. Chinese Universities, 2021, 42(9): 2742. |
| [9] | ZHANG Xu, QUE Jiaqian, HOU Yuexin, LYU Jiamin, LIU Zhan, LEI Kunhao, YU Shen, LI Xiaoyun, CHEN Lihua, SU Baolian. Hierarchical Mesoporous-microporous TS-1 Single Crystal Catalysts for Epoxidation of Allyl Chloride [J]. Chem. J. Chinese Universities, 2021, 42(8): 2529. |
| [10] | WANG Lei, SUN Tantan, YAN Nana, MA Chao, LIU Xiaona, TIAN Peng, GUO Peng, LIU Zhongmin. Exploring Organic Structure-directing Agents Used for SAPO-34 to Synthesize SSZ-13 [J]. Chem. J. Chinese Universities, 2021, 42(6): 1716. |
| [11] | LI Jian, YU Mingming, SUN Yuan, FENG Wenhua, FENG Zhaochi, WU Jianfeng. Effect of Aqueous Solution pH on the Oxidation of Methane to Methanol at Low Temperature [J]. Chem. J. Chinese Universities, 2021, 42(3): 776. |
| [12] | WANG Juan, WANG Linying, ZHU Dali, CUI Wenhao, WANG Yifeng, TIAN Peng, LIU Zhongmin. Progress in Direct Synthesis of High Silica Zeolite Y [J]. Chem. J. Chinese Universities, 2021, 42(1): 1. |
| [13] | WEN Jiali, ZHANG Junhao, JIANG Jiuxing. Extra-large Pore Zeolites: a Ten-year Updated Review [J]. Chem. J. Chinese Universities, 2021, 42(1): 101. |
| [14] | WANG Jianyu, ZHANG Qiang, YAN Wenfu, YU Jihong. Roles of Hydroxyl Radicals in Zeolite Synthesis [J]. Chem. J. Chinese Universities, 2021, 42(1): 11. |
| [15] | LIU Yi, LIU Yi. Research Progress on Zeolite Layer Preparation via Oriented Seeded Growth [J]. Chem. J. Chinese Universities, 2021, 42(1): 117. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||