

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (5): 917.doi: 10.7503/cjcu20170660
Previous Articles Next Articles
Received:2017-09-29
Online:2018-03-22
Published:2018-03-22
Supported by:CLC Number:
TrendMD:
| Sample | P1 | P0.5 | P0.25 | P0.1 | P0.05 | P0.01 |
|---|---|---|---|---|---|---|
| m(Pyrene-1)/mg | 3.93 | 1.97 | 0.98 | 0.39 | 0.20 | 0.04 |
| V(TEOS)/μL | 180 | 180 | 180 | 180 | 180 | 180 |
| n(Pyrene-1)∶n(TEOS) | 1∶100 | 0.5∶100 | 0.25∶100 | 0.1∶100 | 0.05∶100 | 0.01∶100 |
Table 1 Preparation conditions of pyrene-doped silica fluorescent nanoparticles*
| Sample | P1 | P0.5 | P0.25 | P0.1 | P0.05 | P0.01 |
|---|---|---|---|---|---|---|
| m(Pyrene-1)/mg | 3.93 | 1.97 | 0.98 | 0.39 | 0.20 | 0.04 |
| V(TEOS)/μL | 180 | 180 | 180 | 180 | 180 | 180 |
| n(Pyrene-1)∶n(TEOS) | 1∶100 | 0.5∶100 | 0.25∶100 | 0.1∶100 | 0.05∶100 | 0.01∶100 |
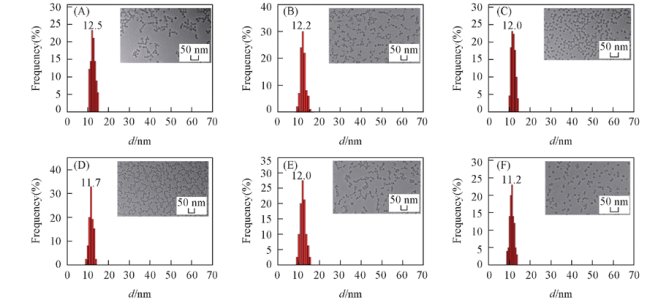
Fig.1 Diameter distribution of pyrene-doped silica fluorescent nanoparticles Note: (A) P1; (B) P0.5; (C) P0.25; (D) P0.1; (E) P0.05; (F) P0.01. Insets: TEM images of corresponding pyrene-doped silica fluorescent nanoparticles.
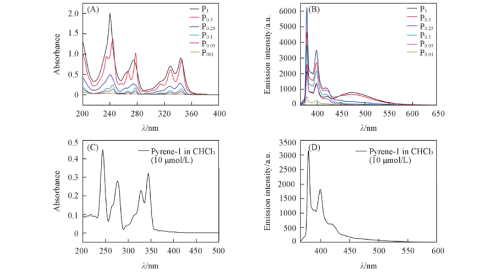
Fig.3 Absorption(A, C) and emission(B, D) spectra of pyrene-doped silica fluorescent nanoparticles(A, B) and pyrene-1 in chloroform(C, D) Note:(A) c=1 μmol/L, λex=345 nm; (B) c=10 μmol/L, λex=345 nm.
| [1] | Yao J., Yang M., Duan Y., Chem. Res., 2014, 114(12), 6130—6178 |
| [2] | Gupta V., Chaudhary N., Srivastava R., Sharma G.D., Bhardwaj R., Chand S., [J]. Am. Chem. Soc., 2011, 133(26), 9960—9963 |
| [3] | Valenti G., Rampazzo E., Bonacchi S., Petrizza L., Marcaccio M., Montalti M., Prodi L., Paolucci F., [J]. Am. Chem. Soc., 2016, 138(49), 15935—15942 |
| [4] | Kesarkar S., Rampazzo E., Valenti G., Marcaccio M., Bossi A., Prodi L., Paolucci F., ChemElectroChem, 2017, 4(7), 1690—1696 |
| [5] | Wang R.F., Peng H. Q., Chen P. Z., Niu L. Y., Gao J. F., Wu L. Z., Tung C. H., Chen Y. Z., Yang Q. Z., Adv. Funct. Mater., 2016, 26(30), 5419—5425 |
| [6] | Lee S.J., Lee J. E., Seo J., Jeong I. Y., Lee S. S., Jung J. H., Adv. Funct. Mater., 2007, 17(17), 3441—3446 |
| [7] | Wolfbeis O.S., Chem. Soc. Res., 2015, 44(14), 4743—4768 |
| [8] | Rosi N.L., Mirkin C. A., Chem. Res., 2005, 105(4), 1547—1562 |
| [9] | Kathiravan A., Renganathan R., Anandan S., Polyhedron, 2009, 28(1), 157—161 |
| [10] | Zhang H.P., Han B., Jia Z. Z., Ding R. B., Xu B., Xu W. Q., Fan Z. M., Chem. [J]. Chinese Universities, 2017, 38(5), 860—865 |
| (张海鹏, 韩冰, 贾致真, 丁荣波, 徐斌, 徐蔚青, 范志民. 高等学校化学学报, 2017, 38(5), 860—865) | |
| [11] | Clarke S.J., Hollmann C. A., Zhang Z., Suffern D., Bradforth S. E., Dimitrijevic N. M., Minarik W. G., Nadeau J. L., Nat. Mater., 2006, 5(5), 409—417 |
| [12] | Chan Y.H., Ye F., Gallina M. E., Zhang X., Jin Y., Wu I. C., Chiu D. T., [J]. Am. Chem. Soc., 2012, 134(17), 7309—7312 |
| [13] | Sun Q., Wang Y.A., Li L. S., Wang D., Zhu T., Xu J., Yang C., Li Y., Nat. Photon., 2007, 1(12), 717—722 |
| [14] | Kramer I.J., Sargent E. H., Chem. Rev., 2014, 114(1), 863—882 |
| [15] | Huang Y., Lan Y., Yi Q., Huang H., Wang Y., Lu J., Chem. Res. Chinese Universities, 2016, 32(1), 16—19 |
| [16] | Mandal S., Jana N.R., J. Phys. Chem. C, 2017, 121(42), 23727—23735 |
| [17] | Feng L., Zhu C., Yuan H., Liu L., Lv F., Wang S., Chem. Soc. Rev., 2013, 42(16), 6620—6633 |
| [18] | Peng H.Q., Sun C. L., Niu L. Y., Chen Y. Z., Wu L. Z., Tung C. H., Yang Q. Z., Adv. Funct. Mater., 2016, 26(30), 5483—5489 |
| [19] | Huang H., He W., Ma Y., Chem. Res. Chinese Universities, 2016, 32(4), 695—701 |
| [20] | Zhou J., Yang Y., Zhang C.Y., Chem. Rev., 2015, 115(21), 11669—11717 |
| [21] | Tang Y., Han S., Liu H., Chen X., Huang L., Li X., Zhang J., Biomaterials, 2013, 34(34), 8741—8755 |
| [22] | Wu C., Chiu D.T., Angew. Chem. Int. Ed., 2013, 52(11), 3086—3109 |
| [23] | Liu H.Y., Wu P. J., Kuo S. Y., Chen C. P., Chang E. H., Wu C. Y., Chan Y. H., [J]. Am. Chem. Soc., 2015, 137(32), 10420—10429 |
| [24] | Singh K., Sareen D., Kaur P., Miyake H., Tsukube H., Chem. Eur. J., 2013, 19(22), 6914—6936 |
| [25] | Bonacchi S., Genovese D., Juris R., Montalti M., Prodi L., Rampazzo E., Zaccheroni N., Angew. Chem. Int.Ed., 2011, 50(18), 4056—4066 |
| [26] | Song J.T., Yang X. Q., Zhang X. S., Yan D. M., Wang Z. Y., Zhao Y. D., ACS Appl. Mater. Interfaces, 2015, 7(31), 17287—17297 |
| [27] | Kim E., Kim H.J., Bae D. R., Lee S. J., Cho E. J., Seo M. R., Kim J. S., Jung J. H., New [J]. Chem., 2008, 32(6), 1003—1007 |
| [28] | Garcia-Acosta B., Comes M., Bricks J.L., Kudinova M. A., Kurdyukov V. V., Tolmachev A. I., Descalzo A. B., Marcos M. D., Marti-nez-Manez R., Moreno A., Sancenon F., Soto J., Villaescusa L. A., Rurack K., Barat J. M., Escriche I., Amoros P., Chem. Commun., 2006, (21), 2239—2241 |
| [29] | Genovese D., Rampazzo E., Bonacchi S., Montalti M., Zaccheroni N., Prodi L., Nanoscale, 2014, 6(6), 3022—3036 |
| [30] | Genovese D., Montalti M., Prodi L., Rampazzo E., Zaccheroni N., Tosic O., Chem. Commun., 2011, 47(39), 10975—10977 |
| [31] | Genovese D., Bonacchi S., Juris R., Montalti M., Prodi L., Rampazzo E., Zaccheroni N., Angew. Chem. Int.Ed., 2013, 52(23), 5965—5968 |
| [32] | Scott B.J., Wirnsberger G., Stucky G. D., Chem. Mater., 2001, 13(10), 3140—3150 |
| [33] | Huang W., Wu D.Y., Wu G. H., Wang Z. Q., Dalton Trans., 2012, 41(9), 2620—2625 |
| [34] | Lee S.J., Bae D. R., Han W. S., Lee S. S., Jung J. H., Eur. [J]. Inorg. Chem., 2008, 2008(10), 1559—1564 |
| [35] | Herz E., Ow H., Bonner D., Burns A., Wiesner U., [J]. Mater. Chem., 2009, 19(35), 6341—6347 |
| [36] | Ow H., Larson D.R., Srivastava M., Baird B. A., Webb W. W., Wiesner U., Nano Lett., 2005, 5(1), 113—117 |
| [37] | Burns A., Ow H., Wiesner U., Chem. Soc. Res., 2006, 35(11), 1028—1042 |
| [38] | Larson D.R., Ow H., Vishwasrao H. D., Heikal A. A., Wiesner U., Webb W. W., Chem. Mater., 2008, 20(8), 2677—2684 |
| [39] | Bagwe R.P., Yang C., Hilliard L. R., Tan W., Langmuir, 2004, 20(19), 8336—8342 |
| [40] | Zanarini S., Rampazzo E., Ciana L.D., Marcaccio M., Marzocchi E., Montalti M., Paolucci F., Prodi L., [J]. Am. Chem. Soc., 2009, 131(6), 2260—2267 |
| [41] | Stöber W., Fink A., Bohn E., [J]. Colloid Interface Sci., 1968, 26(1), 62—69 |
| [42] | van Blaaderen A., Imhof A., Hage W., Vrij A., Langmuir, 1992, 8(6), 1514—1517 |
| [43] | Verhaegh N.A. M., van Blaaderen A., Langmuir, 1994, 10(5), 1427—1438 |
| [44] | Rampazzo E., Bonacchi S., Montalti M., Prodi L., Zaccheroni N., [J]. Am. Chem. Soc., 2007, 129(46), 14251—14256 |
| [45] | Yong K.T., Roy I., Swihart M. T., Prasad P. N., [J]. Mater. Chem., 2009, 19(27), 4655—4672 |
| [46] | Zanarini S., Rampazzo E., Bonacchi S., Juris R., Marcaccio M., Montalti M., Paolucci F., Prodi L., [J]. Am. Chem. Soc., 2009, 131(40), 14208—14209 |
| [47] | Rampazzo E., Bonacchi S., Juris R., Montalti M., Genovese D., Zaccheroni N., Prodi L., Rambaldi D.C., Zattoni A., Reschiglian P., J. Phys. Chem. B, 2010, 114(45), 14605—14613 |
| [48] | Figueira-Duarte T.M., Müllen K., Chem. Res., 2011, 111(11), 7260—7314 |
| [49] | Fujiwara Y., Amao Y., Sens. Actuators B: Chem., 2003, 89(1), 58—61 |
| [50] | Ikeda H., Nakamura M., Ise N., Oguma N., Nakamura A., Ikeda T., Toda F., Ueno A., [J]. Am. Chem. Soc., 1996, 118(45), 10980—10988 |
| [51] | Monahan C., Chem. Commun., 1998, (3), 431—432 |
| [52] | Fernández-González M.Á., Rivero D., García-Iriepa C., Sampedro D., Frutos L. M., [J]. Chem. Theo. Comput., 2017, 13(2), 727—736 |
| [53] | Anthony J.E., Angew. Chem. Int. Ed., 2008, 47(3), 452—483 |
| [54] | Shirota Y., Kageyama H., Chem. Res., 2007, 107(4), 953—1010 |
| [55] | Wu J., Pisula W., Müllen K., Chem. Res., 2007, 107(3), 718—747 |
| [56] | Zhang R., Zhao Y., Li G., Yang D., Ni Z., RSC Adv., 2016, 6(11), 9037—9048 |
| [57] | Laia C.A. T., Costa S. M. B., Chem. Phys. Lett., 1998, 285(5—6), 385—390 |
| [58] | Hamai S., Hirayama F., [J]. Phy. Chem., 1983, 87(1), 83—89 |
| [59] | Peng H.Q., Chen Y. Z., Zhao Y., Yang Q. Z., Wu L. Z., Tung C. H., Zhang L. P., Tong Q. X., Angew. Chem. Int. Ed., 2012, 51(9), 2088—2092 |
| [60] | Miller R.A., Presley A. D., Francis M. B., [J]. Am. Chem. Soc., 2007, 129(11), 3104—3109 |
| [61] | Förster T., Kasper K.,Elektrochem., 1955, 59(10), 976—980 |
| [1] | CHANG Tonghang, CHENG Zhen. Research Progress of Organic Small Molecule Theranostic Probes Integrating Fluorescence Imaging and Chemotherapy [J]. Chem. J. Chinese Universities, 0, (): 20220430. |
| [2] | LIN Zhongqiao, CHEN Peipei, WANG Lei. Application of Mineralocorticoid Receptor Antagonists with Different Chemical Structures in Cardiovascular Diseases [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220059. |
| [3] | WU Fengtian, LI Jun, SUN Yijia, ZENG Rong, REN Han, LIU Xiuping, ZHANG Caihong, YAN Fangming, WU Ling, CUI Chunna. Fibroin Assisted Copper-promoted C-N Coupling Reaction [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210707. |
| [4] | PU Weiwen, SHI Yongsen, LIU Jianfeng, KE Dehong, WU Xiaolan, XU Sheng. Synthesis of (E)-α-Hydroxyethyl-α,β-unsaturated Aldehydes with Tetrahydrofuran as Carbonyl Source [J]. Chem. J. Chinese Universities, 2021, 42(12): 3641. |
| [5] | PENG Haiyue, WANG Ting, LI Guorui, HUANG Jing. Synthesis of Melanin and Its Function Regulation by Small Molecules [J]. Chem. J. Chinese Universities, 2021, 42(11): 3357. |
| [6] | ZUO Huailong, LEI Simin, ZHANG Rui, LI Yuxin, CHEN Wei. Design, Synthesis and Antifungal Activity of Novel Isoquinoline Derivatives [J]. Chem. J. Chinese Universities, 2021, 42(9): 2766. |
| [7] | LIU Wei, YAO Wei, ZHOU Mingming, YOU Qi, NIE Yong, JIANG Xuchuan. Synthesis, Aggregation-induced Emission and Piezofluorochromic Properties of 9,10-Bis(N-phenylindole-3-vinyl)anthracene [J]. Chem. J. Chinese Universities, 2021, 42(8): 2668. |
| [8] | HUANG Chibao, KANG Shuai, PAN Qi, LYU Guoling. Carbazole-derived Dicyanostilbene Two-photon Fluorescence Probe for Lipid Raft [J]. Chem. J. Chinese Universities, 2021, 42(8): 2443. |
| [9] | CAO Cheng,CAO Tian,YAN Penji,WANG Qingyun,YUE Guoren,JI Xiangdong. High Selectivity of Enol-keto Tautomers Imine Derivative for the Recognition of Co2+† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1544. |
| [10] | XIE Ziyi, LIU Dan, ZHANG Yihan, LIU Qingqing, DONG Huanli, HU Wenping. Recent Advances on High Mobility Emissive Anthracene-derived Organic Semiconductors † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1179. |
| [11] | HUA Tao, LI Shengnan, LI Fengxiang, WANG Haonan. Treatment of Naphthalene by Microbial Electrochemical System and the Analysis of Microbial Communities † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1964. |
| [12] | QIAN Wenhao,HUANG Wei,CONG Yufeng,LI Fusheng. Selective Oxidation of Benzyl Alcohols Catalyzed by UiO-67-Sal-CuCl2 in Air† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1178. |
| [13] | HUANG Rui,YAO Zhilong,SUN Peiyong,ZHANG Shenghong. Effect of Structure and Properties of CuO-WO3-ZrO2 on Hydrogenation Catalytic of Benzaldehyde† [J]. Chem. J. Chinese Universities, 2019, 40(5): 1005. |
| [14] | LI Pu,CHEN Ying,XIA Rongjiao,GUO Tao,ZHANG Min,JIANG Shichun,ANG Xu,HE Ming,XUE Wei. Synthesis and Biological Activities of Myricetin Derivatives Containing Quinoxaline† [J]. Chem. J. Chinese Universities, 2019, 40(5): 909. |
| [15] | LÜ Mingjun,LI Wen,YANG Xinying,FANG Hao. Synthesis and Antitumor Activity of N9 Position Aromatic Substituted Purine-8-one Derivatives† [J]. Chem. J. Chinese Universities, 2019, 40(2): 254. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||