

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (4): 736.doi: 10.7503/cjcu20150681
• Physical Chemistry • Previous Articles Next Articles
WANG Wenliang, GENG Jing, LI Lufei, CHANG Jianmin*( )
)
Received:2015-08-28
Online:2016-04-10
Published:2016-03-16
Contact:
CHANG Jianmin
E-mail:cjianmin168@126.com
CLC Number:
TrendMD:
WANG Wenliang, GENG Jing, LI Lufei, CHANG Jianmin. Catalytic Properties of Fast Pyrolysis Char Loaded with Cu-Zn on Alkali Lignin Pyrolysis for Monophenols†[J]. Chem. J. Chinese Universities, 2016, 37(4): 736.
| Sample | Ultimate analysisa(mass fraction, %) | Proximate analysisc(mass fraction, %) | BET surface aread/ (m2·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | Ob | N | S | A | V | FCb | ||
| Alkali lignin | 62.40 | 6.14 | 29.43 | 0.26 | 1.77 | 6.21 | 66.43 | 27.36 | |
| FPC | 9.04 | 19.39 | 71.57 | 307.56±12.33 | |||||
Table 1 Basic analyses of alkali lignin and FPC
| Sample | Ultimate analysisa(mass fraction, %) | Proximate analysisc(mass fraction, %) | BET surface aread/ (m2·g-1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| C | H | Ob | N | S | A | V | FCb | ||
| Alkali lignin | 62.40 | 6.14 | 29.43 | 0.26 | 1.77 | 6.21 | 66.43 | 27.36 | |
| FPC | 9.04 | 19.39 | 71.57 | 307.56±12.33 | |||||
| Catalyst | D(CuO)/nm | D(ZnO)/nm | Catalyst | D(CuO)/nm | D(ZnO)/nm |
|---|---|---|---|---|---|
| Cu/FPC | 27.9 | Cu0.33Zn0.67/FPC | 18.8 | 38.3 | |
| Cu0.67Zn0.33/FPC | 26.0 | 32.1 | Zn/FPC | 43.0 | |
| Cu0.50Zn0.50/FPC | 25.8 | 33.3 |
Table 2 Average crystal sizes of CuO and ZnO in catalysts
| Catalyst | D(CuO)/nm | D(ZnO)/nm | Catalyst | D(CuO)/nm | D(ZnO)/nm |
|---|---|---|---|---|---|
| Cu/FPC | 27.9 | Cu0.33Zn0.67/FPC | 18.8 | 38.3 | |
| Cu0.67Zn0.33/FPC | 26.0 | 32.1 | Zn/FPC | 43.0 | |
| Cu0.50Zn0.50/FPC | 25.8 | 33.3 |
| Sample | Ts/℃ | Tmax/℃ | w(%) | E/(kJ·mol-1) | A/min-1 | R2 |
|---|---|---|---|---|---|---|
| Lignin | 268—463 | 326 | 53.7 | 35.4 | 86.7 | 0.977 |
| 670—779 | 161.3 | 8.5×108 | 0.958 | |||
| Cu | 267—463 | 343 | 48.4 | 29.2 | 19.8 | 0.988 |
| 669—765 | 134.4 | 2.7×107 | 0.964 | |||
| Cu0.67Zn0.33 | 267—463 | 349 | 49.4 | 29.9 | 21.5 | 0.985 |
| 670—765 | 135.0 | 2.3×107 | 0.952 | |||
| Cu0.50Zn0.50 | 268—463 | 345 | 47.5 | 27.9 | 14.5 | 0.984 |
| 669—764 | 111.7 | 8.8×105 | 0.927 | |||
| Cu0.33Zn0.67 | 268—462 | 350 | 48.4 | 29.5 | 19.7 | 0.985 |
| 670—765 | 113.7 | 1.2×106 | 0.940 | |||
| Zn | 268—462 | 344 | 47.3 | 29.4 | 19.2 | 0.983 |
| 670—765 | 104.0 | 3.2×105 | 0.943 |
Table 3 Kinetic parameters of catalytic pyrolysis of alkali lignin*
| Sample | Ts/℃ | Tmax/℃ | w(%) | E/(kJ·mol-1) | A/min-1 | R2 |
|---|---|---|---|---|---|---|
| Lignin | 268—463 | 326 | 53.7 | 35.4 | 86.7 | 0.977 |
| 670—779 | 161.3 | 8.5×108 | 0.958 | |||
| Cu | 267—463 | 343 | 48.4 | 29.2 | 19.8 | 0.988 |
| 669—765 | 134.4 | 2.7×107 | 0.964 | |||
| Cu0.67Zn0.33 | 267—463 | 349 | 49.4 | 29.9 | 21.5 | 0.985 |
| 670—765 | 135.0 | 2.3×107 | 0.952 | |||
| Cu0.50Zn0.50 | 268—463 | 345 | 47.5 | 27.9 | 14.5 | 0.984 |
| 669—764 | 111.7 | 8.8×105 | 0.927 | |||
| Cu0.33Zn0.67 | 268—462 | 350 | 48.4 | 29.5 | 19.7 | 0.985 |
| 670—765 | 113.7 | 1.2×106 | 0.940 | |||
| Zn | 268—462 | 344 | 47.3 | 29.4 | 19.2 | 0.983 |
| 670—765 | 104.0 | 3.2×105 | 0.943 |
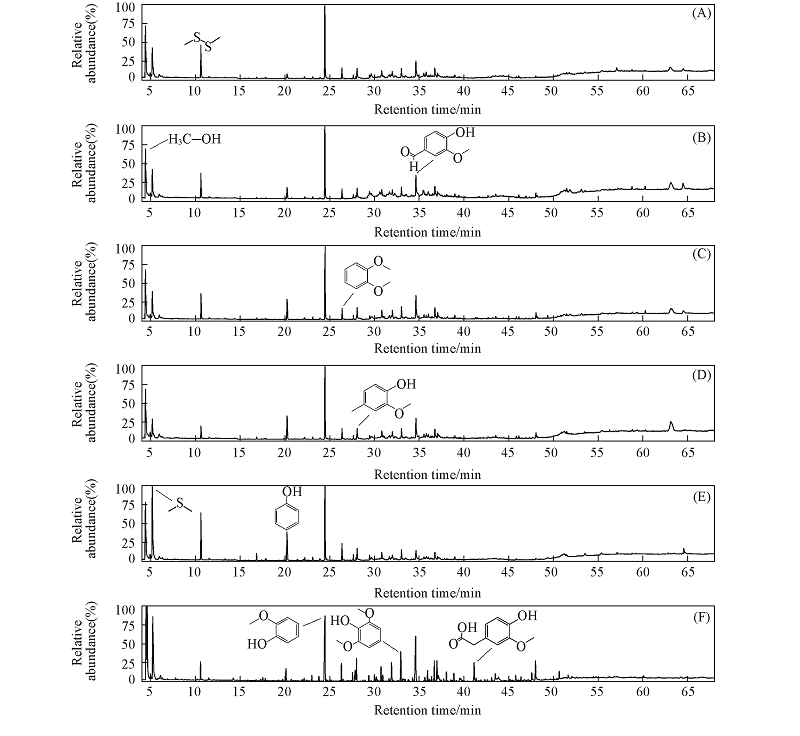
Fig.5 Total ion chromatograms of pyrolysis vapors of alkali lignin with catalysts^(A) Zn/FPC; (B) Cu0.33Zn0.67/FPC; (C) Cu0.50Zn0.50/FPC; (D) Cu0.67Zn0.33/FPC; (E) Cu/FPC; (F) Lignin.
| No. | Compound | Relative content(area)(%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Phenols | 7.32 | 4.93 | 4.46 | 3.79 | 1.97 | 1.15 | |||||||
| 1 | Phenol | 1.53 | 4.30 | 3.89 | 3.16 | 1.97 | 1.15 | ||||||
| 2 | o-Cresol | 0.62 | 0.63 | 0.57 | 0.63 | ||||||||
| 3 | p-Cresol | 0.69 | |||||||||||
| 4 | 3-Ethoxyphenol | 1.88 | |||||||||||
| 5 | 4-Methylcatechol | 1.77 | |||||||||||
| 6 | 4-Hydroxybenzaldehyde | 0.58 | |||||||||||
| 7 | 5-tert-Butylpyrogallol | 0.25 | |||||||||||
| Guaiacols | 39.61 | 28.08 | 44.88 | 41.80 | 41.36 | 42.07 | |||||||
| 8 | Guaiacol | 13.74 | 17.82 | 25.26 | 21.98 | 24.77 | 25.84 | ||||||
| 9 | Isocreosol | 0.96 | 1.15 | 1.09 | 1.11 | 1.12 | 1.12 | ||||||
| 10 | Creosol | 2.83 | 3.06 | 3.32 | 3.21 | 3.03 | 3.25 | ||||||
| 11 | 3-Methoxycatechol | 0.55 | |||||||||||
| 12 | 4-Ethylguaiacol | 1.70 | 1.56 | 3.10 | 2.71 | 1.97 | 2.49 | ||||||
| 13 | 4-Acetyl-3-methylphenol | 2.51 | |||||||||||
| 14 | 4-Vinylguaiacol | 1.30 | 1.82 | 2.58 | 1.85 | 1.86 | |||||||
| 15 | 2-Methoxyquinol | 0.45 | |||||||||||
| 16 | Vanillin | 9.29 | 2.74 | 8.67 | 8.66 | 8.62 | 7.51 | ||||||
| 17 | (E)-Isoeugenol | 1.29 | |||||||||||
| 18 | Acetovanillone | 2.77 | 0.45 | 1.62 | 1.55 | ||||||||
| 19 | Guaiacylacetone | 1.04 | |||||||||||
| 20 | Homovanillic acid | 2.48 | |||||||||||
| Syringols | 4.93 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | |||||||
| 21 | Syringol | 4.10 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | ||||||
| 22 | Syringaldehyde | 0.39 | |||||||||||
| 23 | Syringone | 0.44 | |||||||||||
| Alcohols | 10.63 | 17.09 | 19.02 | 16.80 | 18.85 | 21.33 | |||||||
| 24 | Methanol | 8.75 | 17.09 | 18.53 | 16.54 | 18.85 | 21.33 | ||||||
| 25 | Dimethylhexanol | 0.49 | 0.26 | ||||||||||
| 26 | 1-Dodecanol | 0.52 | |||||||||||
| 27 | Pentadecanol | 0.44 | |||||||||||
| 28 | Nonadecanol | 0.92 | |||||||||||
| Ethers | 6.17 | 6.86 | 8.54 | 7.76 | 8.38 | 6.89 | |||||||
| 29 | Methylvanillin | 2.61 | 1.51 | 3.36 | 3.38 | 4.47 | 2.72 | ||||||
| 30 | 4-Vinylveratrole | 0.45 | |||||||||||
| 31 | Anisole | 0.35 | |||||||||||
| 32 | Veratrole | 1.92 | 3.51 | 3.05 | 2.92 | 2.97 | 3.35 | ||||||
| 33 | Homoveratrole | 0.58 | 0.98 | 0.90 | 0.83 | 0.94 | 0.82 | ||||||
| 34 | Triglycol monomethyl ether | 1.23 | 0.63 | ||||||||||
| 35 | 1,2,3-Trimethoxybenzene | 0.25 | 0.32 | ||||||||||
| 36 | 1,2,4-Trimethoxybenzene | 0.36 | 0.19 | ||||||||||
| No. | Compound | Relative content(area)(%) | |||||||||||
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Ketones | 1.77 | 0.59 | 2.54 | 2.29 | 2.28 | 2.30 | |||||||
| 37 | Acetone | 0.80 | 1.78 | 1.31 | 1.43 | 1.52 | |||||||
| 38 | Acetoveratrone | 0.97 | |||||||||||
| 39 | 2,4-Dimethoxyacetophenone | 0.59 | 0.76 | 0.98 | 0.85 | 0.78 | |||||||
| Esters | 0.36 | 1.90 | 1.50 | 3.60 | 1.96 | 1.24 | |||||||
| 40 | Allyl acetate | 1.48 | |||||||||||
| 41 | Butyrolactone | 0.36 | |||||||||||
| 42 | Aceteugenol | 0.42 | 0.78 | 0.98 | 0.78 | ||||||||
| 43 | Diisobutyl phthalate | 0.72 | 0.57 | 0.71 | 0.46 | ||||||||
| 44 | Dibutyl phthalate | 0.39 | |||||||||||
| 45 | Isopropyl Palmitate | 0.57 | |||||||||||
| 46 | Mono(2-ethylhexyl) phthalate | 1.09 | 1.25 | ||||||||||
| Hydrocarbons | 0.25 | 1.64 | 2.15 | 2.79 | 4.33 | 1.37 | |||||||
| 47 | o-Cymene | 0.25 | 0.42 | 0.28 | |||||||||
| 48 | 2,4-Dimethyl styrene | 0.29 | |||||||||||
| 49 | 1-Ethylidene-1H-indene | 0.42 | 0.68 | ||||||||||
| 50 | α-Cedrene | 0.51 | 2.15 | 1.83 | 4.33 | 1.37 | |||||||
| Carboxylic acids | 5.96 | 1.28 | 2.54 | 3.36 | 1.37 | ||||||||
| 51 | Acetic acid | 0.40 | 1.28 | 0.72 | 0.87 | ||||||||
| 52 | Myristic acid | 0.75 | 0.50 | ||||||||||
| 53 | Pentadecylic acid | 0.66 | |||||||||||
| 54 | Petroselic acid | 1.07 | |||||||||||
| 55 | Palmitic acid | 2.57 | |||||||||||
| 56 | Tridecylic acid | 1.82 | 1.99 | 1.37 | |||||||||
| 57 | Oleic Acid | 0.51 | |||||||||||
| Sulfur compounds | 23.00 | 35.24 | 10.72 | 14.01 | 16.36 | 20.17 | |||||||
| 58 | Methanethiol | 8.28 | |||||||||||
| 59 | Dimethyl sulfide | 12.76 | 24.31 | 6.79 | 8.68 | 10.13 | 11.68 | ||||||
| 60 | 2,3-Dithiabutane | 1.96 | 9.57 | 3.25 | 5.33 | 6.23 | 8.49 | ||||||
| 61 | 2,4-Dithiapentane | 1.36 | 0.68 | ||||||||||
Table 4 Main components of pyrolysis vapors of alkali lignin with catalysts
| No. | Compound | Relative content(area)(%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Phenols | 7.32 | 4.93 | 4.46 | 3.79 | 1.97 | 1.15 | |||||||
| 1 | Phenol | 1.53 | 4.30 | 3.89 | 3.16 | 1.97 | 1.15 | ||||||
| 2 | o-Cresol | 0.62 | 0.63 | 0.57 | 0.63 | ||||||||
| 3 | p-Cresol | 0.69 | |||||||||||
| 4 | 3-Ethoxyphenol | 1.88 | |||||||||||
| 5 | 4-Methylcatechol | 1.77 | |||||||||||
| 6 | 4-Hydroxybenzaldehyde | 0.58 | |||||||||||
| 7 | 5-tert-Butylpyrogallol | 0.25 | |||||||||||
| Guaiacols | 39.61 | 28.08 | 44.88 | 41.80 | 41.36 | 42.07 | |||||||
| 8 | Guaiacol | 13.74 | 17.82 | 25.26 | 21.98 | 24.77 | 25.84 | ||||||
| 9 | Isocreosol | 0.96 | 1.15 | 1.09 | 1.11 | 1.12 | 1.12 | ||||||
| 10 | Creosol | 2.83 | 3.06 | 3.32 | 3.21 | 3.03 | 3.25 | ||||||
| 11 | 3-Methoxycatechol | 0.55 | |||||||||||
| 12 | 4-Ethylguaiacol | 1.70 | 1.56 | 3.10 | 2.71 | 1.97 | 2.49 | ||||||
| 13 | 4-Acetyl-3-methylphenol | 2.51 | |||||||||||
| 14 | 4-Vinylguaiacol | 1.30 | 1.82 | 2.58 | 1.85 | 1.86 | |||||||
| 15 | 2-Methoxyquinol | 0.45 | |||||||||||
| 16 | Vanillin | 9.29 | 2.74 | 8.67 | 8.66 | 8.62 | 7.51 | ||||||
| 17 | (E)-Isoeugenol | 1.29 | |||||||||||
| 18 | Acetovanillone | 2.77 | 0.45 | 1.62 | 1.55 | ||||||||
| 19 | Guaiacylacetone | 1.04 | |||||||||||
| 20 | Homovanillic acid | 2.48 | |||||||||||
| Syringols | 4.93 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | |||||||
| 21 | Syringol | 4.10 | 2.39 | 3.65 | 3.80 | 3.14 | 3.48 | ||||||
| 22 | Syringaldehyde | 0.39 | |||||||||||
| 23 | Syringone | 0.44 | |||||||||||
| Alcohols | 10.63 | 17.09 | 19.02 | 16.80 | 18.85 | 21.33 | |||||||
| 24 | Methanol | 8.75 | 17.09 | 18.53 | 16.54 | 18.85 | 21.33 | ||||||
| 25 | Dimethylhexanol | 0.49 | 0.26 | ||||||||||
| 26 | 1-Dodecanol | 0.52 | |||||||||||
| 27 | Pentadecanol | 0.44 | |||||||||||
| 28 | Nonadecanol | 0.92 | |||||||||||
| Ethers | 6.17 | 6.86 | 8.54 | 7.76 | 8.38 | 6.89 | |||||||
| 29 | Methylvanillin | 2.61 | 1.51 | 3.36 | 3.38 | 4.47 | 2.72 | ||||||
| 30 | 4-Vinylveratrole | 0.45 | |||||||||||
| 31 | Anisole | 0.35 | |||||||||||
| 32 | Veratrole | 1.92 | 3.51 | 3.05 | 2.92 | 2.97 | 3.35 | ||||||
| 33 | Homoveratrole | 0.58 | 0.98 | 0.90 | 0.83 | 0.94 | 0.82 | ||||||
| 34 | Triglycol monomethyl ether | 1.23 | 0.63 | ||||||||||
| 35 | 1,2,3-Trimethoxybenzene | 0.25 | 0.32 | ||||||||||
| 36 | 1,2,4-Trimethoxybenzene | 0.36 | 0.19 | ||||||||||
| No. | Compound | Relative content(area)(%) | |||||||||||
| Lignin | Cu | Cu0.67Zn0.33 | Cu0.5Zn0.5 | Cu0.33Zn0.67 | Zn | ||||||||
| Ketones | 1.77 | 0.59 | 2.54 | 2.29 | 2.28 | 2.30 | |||||||
| 37 | Acetone | 0.80 | 1.78 | 1.31 | 1.43 | 1.52 | |||||||
| 38 | Acetoveratrone | 0.97 | |||||||||||
| 39 | 2,4-Dimethoxyacetophenone | 0.59 | 0.76 | 0.98 | 0.85 | 0.78 | |||||||
| Esters | 0.36 | 1.90 | 1.50 | 3.60 | 1.96 | 1.24 | |||||||
| 40 | Allyl acetate | 1.48 | |||||||||||
| 41 | Butyrolactone | 0.36 | |||||||||||
| 42 | Aceteugenol | 0.42 | 0.78 | 0.98 | 0.78 | ||||||||
| 43 | Diisobutyl phthalate | 0.72 | 0.57 | 0.71 | 0.46 | ||||||||
| 44 | Dibutyl phthalate | 0.39 | |||||||||||
| 45 | Isopropyl Palmitate | 0.57 | |||||||||||
| 46 | Mono(2-ethylhexyl) phthalate | 1.09 | 1.25 | ||||||||||
| Hydrocarbons | 0.25 | 1.64 | 2.15 | 2.79 | 4.33 | 1.37 | |||||||
| 47 | o-Cymene | 0.25 | 0.42 | 0.28 | |||||||||
| 48 | 2,4-Dimethyl styrene | 0.29 | |||||||||||
| 49 | 1-Ethylidene-1H-indene | 0.42 | 0.68 | ||||||||||
| 50 | α-Cedrene | 0.51 | 2.15 | 1.83 | 4.33 | 1.37 | |||||||
| Carboxylic acids | 5.96 | 1.28 | 2.54 | 3.36 | 1.37 | ||||||||
| 51 | Acetic acid | 0.40 | 1.28 | 0.72 | 0.87 | ||||||||
| 52 | Myristic acid | 0.75 | 0.50 | ||||||||||
| 53 | Pentadecylic acid | 0.66 | |||||||||||
| 54 | Petroselic acid | 1.07 | |||||||||||
| 55 | Palmitic acid | 2.57 | |||||||||||
| 56 | Tridecylic acid | 1.82 | 1.99 | 1.37 | |||||||||
| 57 | Oleic Acid | 0.51 | |||||||||||
| Sulfur compounds | 23.00 | 35.24 | 10.72 | 14.01 | 16.36 | 20.17 | |||||||
| 58 | Methanethiol | 8.28 | |||||||||||
| 59 | Dimethyl sulfide | 12.76 | 24.31 | 6.79 | 8.68 | 10.13 | 11.68 | ||||||
| 60 | 2,3-Dithiabutane | 1.96 | 9.57 | 3.25 | 5.33 | 6.23 | 8.49 | ||||||
| 61 | 2,4-Dithiapentane | 1.36 | 0.68 | ||||||||||
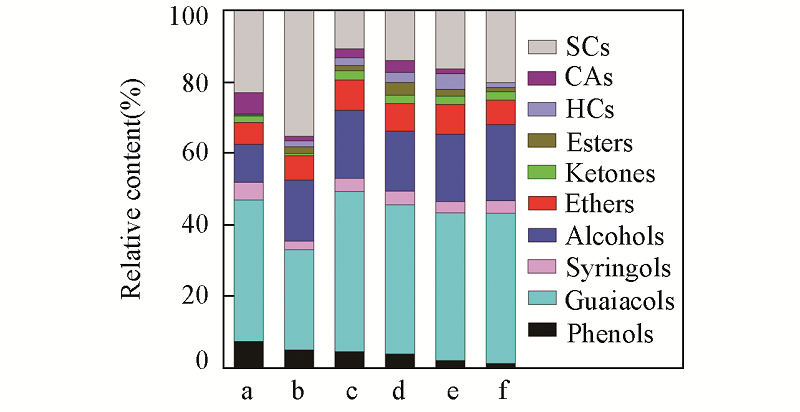
Fig.6 Component distribution of pyrolysis vapors of alkali lignin with catalysts^a. Lignin; b. Cu; c. Cu0.67Zn0.33; d. Cu0.50Zn0.50; e. Cu0.33Zn0.67; f. Zn.
| [1] | Zakzeski J., Bruijnincx P. C. A., Jongerius A. L., Weckhuysen B. M., Chem. Rev., 2010, 110(6), 3552—3599 |
| [2] | Jiang T.D., Lignin, Chemical Industry Press, Beijing, 2009, 19—45 |
| (蒋挺大. 木质素, 北京:化学工业出版社, 2009, 19—45) | |
| [3] | Ouyang X. P., Tan Y. D., Qiu X. Q., J. Fuel Chem. Technol., 2014, 42(6), 677—682 |
| (欧阳新平, 谭友丹, 邱学青. 燃料化学学报, 2014, 42(6), 677—682) | |
| [4] | Yang Z. X., Kumar A., Huhnke R. L., Renew. Sust. Energ. Rev., 2015, 50, 859—870 |
| [5] | Jiang X. X., Ellis N., Zhong Z. P., Chinese J. Chem. Eng., 2010, 18(6), 1018—1022 |
| [6] | Fan D. B., Chang J. M., Gou J. S., Xia B. H., Ren X. Y., J. Adhesion, 2010, 86(8), 834—843 |
| [7] | Kim J. S., Bioresource Technol., 2015, 178, 90—98 |
| [8] | Amen-Chen C., Pakdel H., Roy C., Bioresource Technol., 2001, 79, 277—299 |
| [9] | Jung K. A., Woo S. H., Lim S. R., Park J. M., Chem. Eng. J., 2015, 259, 107—116 |
| [10] | Yan L., Pang H., Huang Y. B., Fu Y., Acta Chim. Sinica, 2014, 72(9), 1005—1011 |
| (严龙, 庞欢, 黄耀兵, 傅尧. 化学学报, 2014, 72(9), 1005—1011) | |
| [11] | Wang W. L., Ren X. Y., Li L. F., Chang J. M., Cai L. P., Geng J., Fuel Process. Technol., 2015, 134, 345—351 |
| [12] | Wang W. L., Ren X. Y., Chang J. M., Cai L. P., Shi S. Q., Fuel Process. Technol., 2015, 138, 605—611 |
| [13] | Peng C. N., Zhang G. Y., Yue J. R., Xu G. W., Fuel Process. Technol., 2014, 124, 212—221 |
| [14] | Singh S. K., Ekhe J. D., Catal. Sci. Technol., 2015, 5(4), 2117—2124 |
| [15] | Shen D. K., Zhao J., Xiao R., Gu S., J. Anal. Appl. Pyrol., 2015, 111, 47—54 |
| [16] | Deepa A. K., Dhepe P. L., ACS Catal., 2015, 5(1), 365—379 |
| [17] | Adhikari S., Srinivasan V., Fasina O., Energy Fuels, 2014, 28(7), 4532—4538 |
| [18] | Bu Q., Lei H. W., Wang L., Wei Y., Zhu L., Zhang X. S., Liu Y. P., Yadavalli G., Tang J. M., Bioresource Technol., 2014, 162, 142—147 |
| [19] | Li Z. X., Na W., Wang H., Gao W. G., Chem. J. Chinese Universities, 2014, 35(12), 2616—2623 |
| (李志雄, 纳薇, 王华, 高文桂. 高等学校化学学报, 2014, 35(12), 2616—2623) | |
| [20] | Xi J. Y., Lü G. X., Wang Z. F., Acta Phys-Chim. Sin., 2001, 17(7), 655—658 |
| (席靖宇, 吕功煊, 王志飞. 物理化学学报, 2001, 17(7), 655—658) | |
| [21] | Wang D., Yang G. H., Ma Q. X., Yoneyama Y., Tan Y. S., Han Y. Z., Tsubaki N., Fuel, 2013, 109, 54—60 |
| [22] | Wang W. L., Yu Y. X., Chang J. M., Bai T. T., J. Fuel Chem. Technol., 2013, 41(11), 1310—1315 |
| (王文亮, 虞宇翔, 常建民, 白甜甜. 燃料化学学报, 2013, 41(11), 1310—1315) | |
| [23] | Chang J. M., Wang W. L., Ren X. Y., Li L. F., Si H., Yu Y. X., Preparation and Application of the Catalysts Used for Biomass Fast Pyrolysis Process, CP 103691429B, 2015-06-10 |
| (常建民, 王文亮, 任学勇, 李露霏, 司慧, 虞宇翔. 用于生物质快速热裂解液化的催化剂及其制备方法和应用, CP 103691429B, 2015-06-10) | |
| [24] | Wang Y. E., Cao S., Wu W. H., Wu M., Tang Y. W., Lu T. H., Chem. J. Chinese Universities, 2014, 35(11), 2455—2459 |
| (王彦恩, 曹爽, 武伟红, 吴敏, 唐亚文, 陆天虹. 高等学校化学学报, 2014, 35(11), 2455—2459) | |
| [25] | Lu X. C., Jiang J. C., Sun K., Xie X.P., B. Korean Chem. Soc., 2014, 35(1), 103—110 |
| [26] | Wang J., Zhang M. X., Chen M. Q., Min F. F., Zhang S. P., Ren Z. W., Yan Y. J., Thermochim. Acta, 2006, 444(1), 110—114 |
| [27] | Collard F. X., Blin J., Bensakhria A., Valette J., J. Anal. Appl. Pyrol., 2012, 95, 213—226 |
| [28] | Ye J. W., Jin C. D., Song P. A., Wu Q., Chem. Ind. For. Prod., 2010, 30)6), 40—44 |
| (叶结旺, 金春德, 宋平安, 吴强. 林产化学与工业, 2010, 30(6), 40—44) | |
| [29] | Wang X. Y., Wan X. J., Wu F. Y., Appl. Chem. Ind., 2010, 39(3), 377—379 |
| (王新运, 万新军, 吴凤义. 应用化工, 2010, 39(3), 377—379) | |
| [30] | Coats A. W., Redfern J. P., Nature, 1964, 201, 68—69 |
| [31] | Liu J. Y., Wu S. B., Guo Y. L., Chem. Ind. For. Prod., 2008, 28(4), 65—70 |
| (刘江燕, 武书彬, 郭伊丽. 林产化学与工业, 2008, 28(4), 65—70) | |
| [32] | Li Z.Z., Chemistry of Plant Fiber Resources, China Light Industry Press, Beijing, 2012, 203—204 |
| (李忠正. 植物纤维化学, 北京: 中国轻工业出版社, 2012, 203—204) | |
| [33] | Cheng H., Yu J., Yao M. Q., Xu G. W., CIESC Journal, 2013, 64(5), 1757—1765 |
| (程辉, 余剑, 姚梅琴, 许光文. 化工学报, 2013, 64(5), 1757—1765) | |
| [34] | Geng J., Wang W. L., Ren X. Y., Chang J. M., Chin. Sciencepaper, 2015, 10(12), 1450—1454 |
| (耿晶, 王文亮, 任学勇, 常建民. 中国科技论文, 2015, 10(12), 1450—1454) |
| [1] | ZHAO Mengyang, HUANG Ziyang. Preparation and in vitro Bioactivity of HA/CuO/SrCO3 Gradiently Composite Coating [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210644. |
| [2] | DONG Yanhong, LU Xinhuan, YANG Lu, SUN Fanqi, DUAN Jingui, GUO Haotian, ZHANG Qinjun, ZHOU Dan, XIA Qinghua. Preparation of Bifunctional Metal-organic Framework Materials and Application in Catalytic Olefins Epoxidation [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220458. |
| [3] | CHEN Xiangyun, ZHU Benqiang, YUAN Bing, YU Fengli, XIE Congxia, YU Shitao. Hydrogenation of α-Pinene Catalyzed by Ru Nanoparticles Stabilized by Magnetic Alkali Lignin Amine [J]. Chem. J. Chinese Universities, 2020, 41(8): 1826. |
| [4] | ZHAO Peng,ZHANG Jinteng,LIN Yanhong. Excellent Ultraviolet Photocatalytic Efficiency of Mg 2+ Doped ZnO and Analysis on Its Synergetic Effect † [J]. Chem. J. Chinese Universities, 2020, 41(3): 538. |
| [5] |
HAN Hongjing,GE Qin,CHEN Yanguang,WANG Haiying,ZHAO Hongzhi,WANG Yizhen,ZHANG Yanan,DENG Jitong,SONG Hua,ZHANG Mei.
Production of Phenolic Compounds from Bagasse Lignin via Catalytic Pyrolysis of Ca1-xPrxFe |
| [6] | WANG Yongpeng,XU Zibo,LIU Mengzhu,ZHANG Haibo,JIANG Zhenhua. Non-enzymatic Glucose Sensor Based on the Electrospun Porous Foamy Copper Oxides Micro-nanofibers† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1310. |
| [7] | HUANG Rui,YAO Zhilong,SUN Peiyong,ZHANG Shenghong. Effect of Structure and Properties of CuO-WO3-ZrO2 on Hydrogenation Catalytic of Benzaldehyde† [J]. Chem. J. Chinese Universities, 2019, 40(5): 1005. |
| [8] | Long TIAN,Yan LONG,Shuyan SONG,Cheng WANG. Synthesis of Flower-like Structured Mn/CuO-CeO2 and the Catalytic Performance for CO Oxide Reaction † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2549. |
| [9] |
YANG Xiurong,ZHANG Chi,GAO Hongxu,ZHAO Fengqi,NIU Shiyao,GUO Zhaoqi,MA Haixia.
Density Functional Theory Study of NO, NO2 Adsorbed on ZnO(10 |
| [10] | WANG Fuxiang,CHEN Ziyu,YANG Weiting,LIU Lijuan,REN Guojian,LIU Yanfeng,PAN Qinhe. Preparation and Adsorption Performance for U(Ⅵ) of ZnO@ZIF-8 Core@Shell Microspheres† [J]. Chem. J. Chinese Universities, 2019, 40(1): 24. |
| [11] | HUANG Yuting,YING Zuping,ZHENG Jixing,ZHUANG Sigeng,LIU Lu,FENG Wei. Hierarchical Porous ZnO Nanomaterial Synthesized with Corn Straw as Biological Templates and Its Photocatalytic Performance† [J]. Chem. J. Chinese Universities, 2018, 39(9): 2031. |
| [12] | HAN Zhiying, LI Youji, LIN Xiao, WANG Ziyu, LI Ziqin, WANG Hao. Preparation and Photoelectrocatalytic Performance of Fe2O3/ZnO Composite Electrode Loading on Conductive Glass† [J]. Chem. J. Chinese Universities, 2018, 39(4): 771. |
| [13] | ZHOU Lie, WU Qingyun, XU Ying, WANG Chenguang, MA Longlong, LI Wenzhi, CHEN Peili. Depolymerization of the Alkali Lignin for Aromatic Compounds over Ni/SiO2-Al2O3 Solid Acid Catalysts† [J]. Chem. J. Chinese Universities, 2018, 39(4): 735. |
| [14] | XUE Jiao, WANG Runwei, ZHANG Zongtao, QIU Shilun. Preparation and Photocatalytic Performance of Novel Zn-doped C/Nb2O5 Nanoparticles Catalyst† [J]. Chem. J. Chinese Universities, 2018, 39(2): 319. |
| [15] | ZHANG Jin, SHI Tiancai, LUO Liwen, LIU Jia, LIU Rong, LIU Le, LIANG Ming, MA Yangmin. One-pot Synthesis of β-Carboline Derivatives Catalyzed by CuO Nanoparticles† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2411. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||