

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (11): 2099.doi: 10.7503/cjcu20150741
• Reviews • Previous Articles Next Articles
ZHU Guilian1,2, LIN Tianquan1, HUANG Fuqiang1,2,*( )
)
Received:2015-09-21
Online:2015-11-10
Published:2015-10-23
Contact:
HUANG Fuqiang
E-mail:huangfq@mail.sic.ac.cn
CLC Number:
TrendMD:
ZHU Guilian, LIN Tianquan, HUANG Fuqiang. Black Nano Titania for Efficient Solar Energy Utilization[J]. Chem. J. Chinese Universities, 2015, 36(11): 2099.
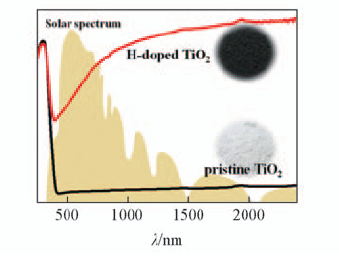
Fig.1 Diffuse reflectance spectra of H-doped black titania(TiO2-xHx) and pristine TiO2(P25, starting material)[26] The backgroud is the total solar spectrum. Copyright from Wiley-VCH.
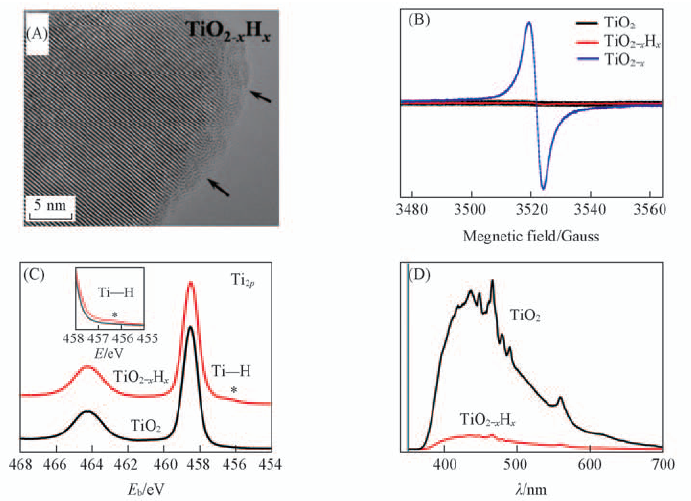
Fig.2 HRTEM micrograph of black TiO2-xHx(A), EPR spectrum(B), Ti2p XPS spectra(C) and photoluminescent spectra(D) of black TiO2-xHx and prisitine TiO2[26] Copyright from Wiley-VCH.
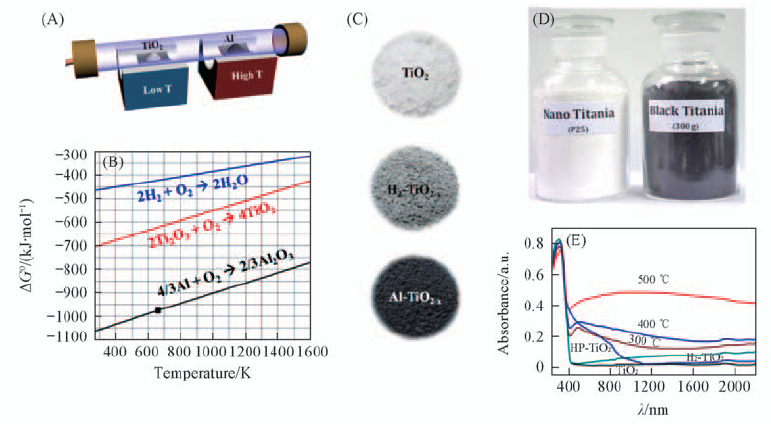
Fig.3 Schematic low-temperature reduction of TiO2 in a two-zone furnace(A), Ellingham diagram of ΔG vs. temperature(B), photographs of pristine TiO2, gray TiO2-x obtained by H2 anneal(H2-TiO2-x), and black TiO2-x obtained by Al reduction(Al-TiO2-x)(C), mass production of black titania(TiO2-x) using our Al-reduction method(D) and absorption spectra of TiO2-x samples reduced at different temperatures, the H2 annealed H2-TiO2-x and high-pressure hydrogenated black titania(HP-TiO2, from ref.[12])(D)[27] Copyright from the Royal Society of Chemistry.
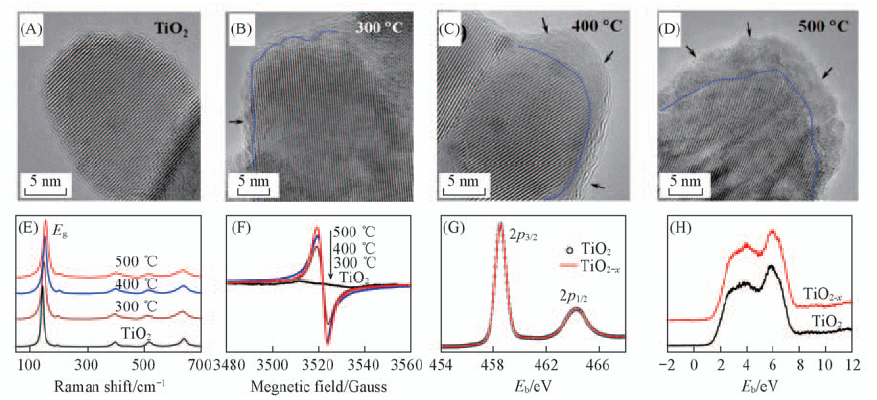
Fig.4 HRTEM images of TiO2 nanocrystals before(A) and after(B—D) Al reduction at different temperatures for 6 h, Raman spectra(E), EPR spectra of black TiO2-x and pristine TiO2 as a reference sample(F), Ti2p XPS spectra(G) and XPS valence band spectra(H) of pristine TiO2 and TiO2-x[27] Copyright from the Royal Society of Chemistry.
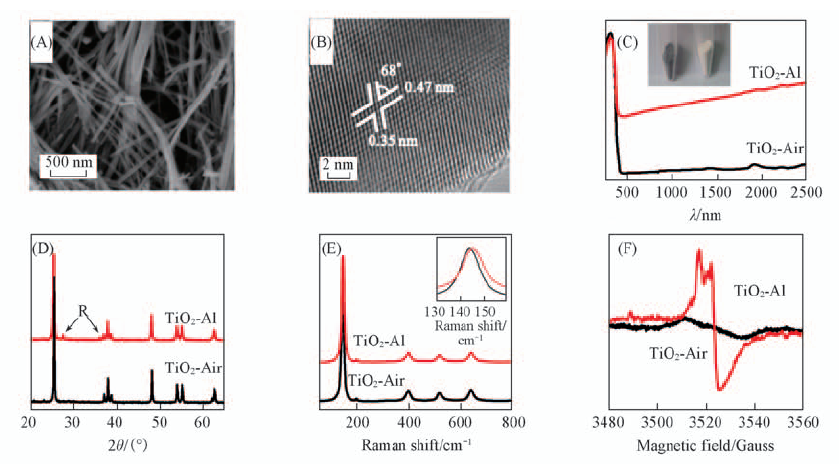
Fig.5 SEM(A), HRTEM images of TiO2-Al(B), UV-Vis absorption spectra(C), XRD patterns(D), Raman spectra(E) and EPR spectra(F) of TiO2-Al and TiO2-air nanowires[37] Copyright from Wiley-VCH.
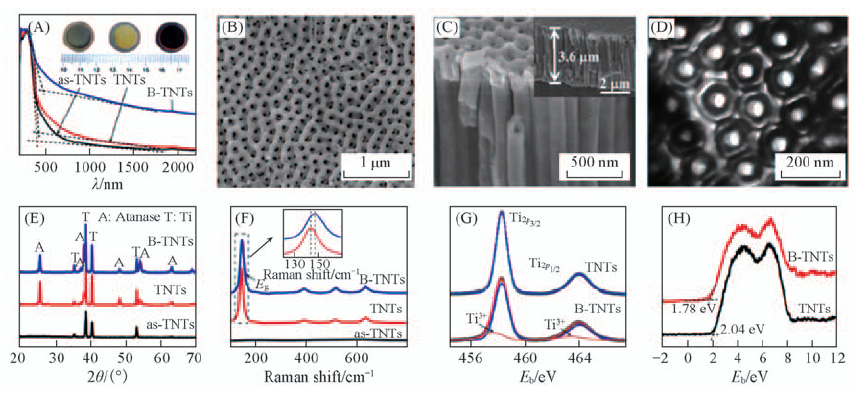
Fig.6 Absorption spectra and photographs(inset) of as-TNTs, TNTs, and B-TNTs(A), typical top view FE-SEM of B-TNTs(B), sideview FE-SEM of B-TNTs(inset is the full side view)(C), typical top view of B-TNTS(D), XRD patterns(E) and Raman spectra of TNTs with different annealing treatment(inset is the magnification of Eg peak)(F), Ti2p XPS(G) and XPS valence band spectra(H) of TNTs and B-TNTs[29]
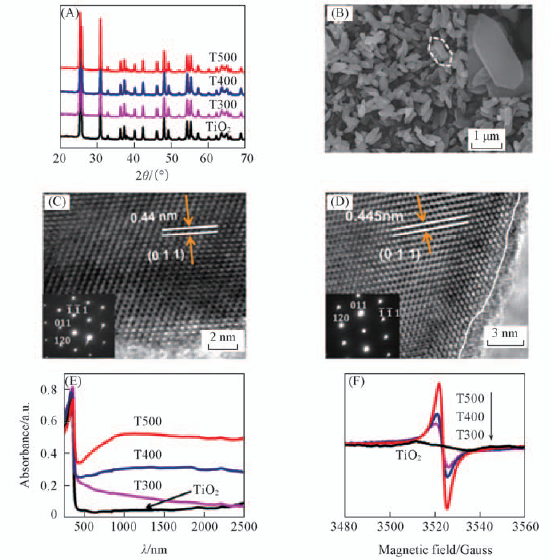
Fig.7 XRD patterns of pristine TiO2 and black TiO2-x after Al reduction at different temperatures for 4 h(A), SEM images of synthesized TiO2(B), HRTEM images of TiO2(C), T500(D), UV-Vis-NIR diffuse reflectance(E) and EPR spectra(F) of the pristine TiO2 and the TiO2-x after Al reduction at different temperatures for 4 h[32] Copyright from the Royal Society of Chemistry.
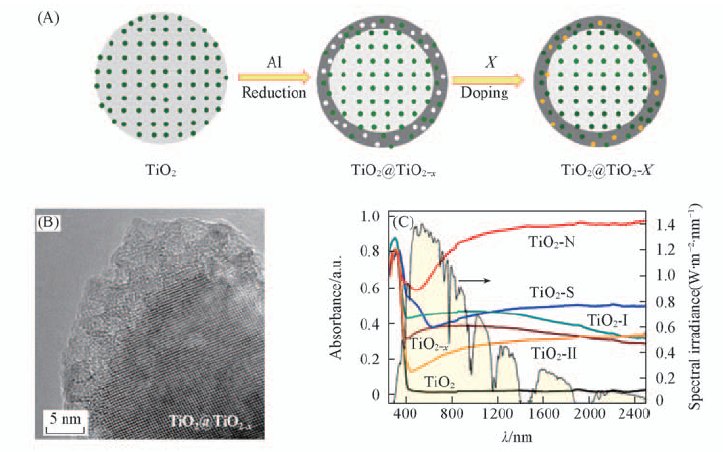
Fig.9 Schematic core/shell structures of TiO2@TiO2-x(denoted as TiO2-x) and TiO2@TiO2-X(denoted as TiO2-X) with Ti4+(oxygen vacancies and X sites in green, white and orange, respectively)(A), HRTEM image of black TiO2-x nanocrystals(B) and diffuse reflectance spectra of TiO2-X(X=H, N, S, I) and solar spectral irradiance(right)(C)[30] Copyright from the Royal Society of Chemistry.
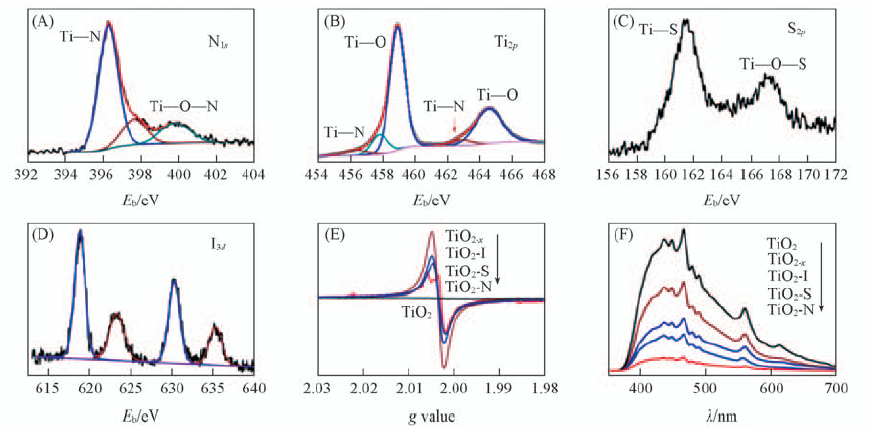
Fig.10 High resolution XPS spectra of N1s and Ti2p of TiO2-N(A, B), S2pXPS spectrum of TiO2-S(C), I3d XPS spectrum acquired from TiO2-I(D), EPR spectra(E) and PL spectra(F) of TiO2-x, TiO2-I, TiO2-N, TiO2-S and pristine TiO2[30] Copyright from the Royal Society of Chemistry.
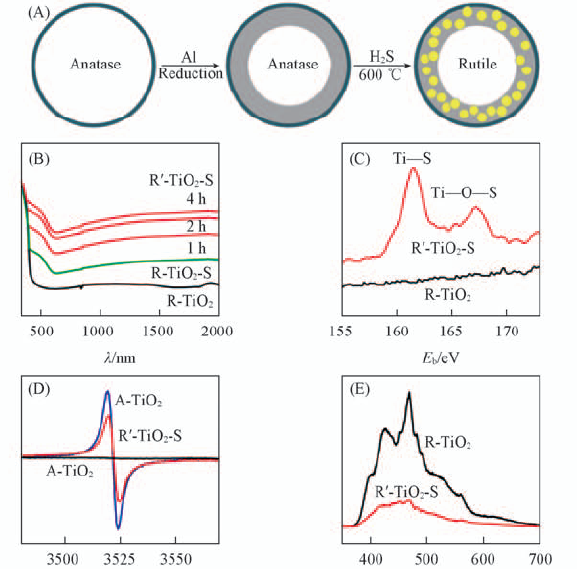
Fig.11 Schematic synthetic route of rutile TiO2 with sulfided surface(R'-TiO2-S)(A), diffuse reflectance spectra of R-TiO2, R-TiO2-S and R'-TiO2-S with different sulfidation time(B), S2p XPS spectra of R'-TiO2-S and R-TiO2(C), EPR spectra of R'-TiO2-S, A-TiO2-x and pristine A-TiO2(D) and photoluminescent spectra of R'-TiO2-S and R-TiO2(E)[28] Copyright from the American Chemical Society.
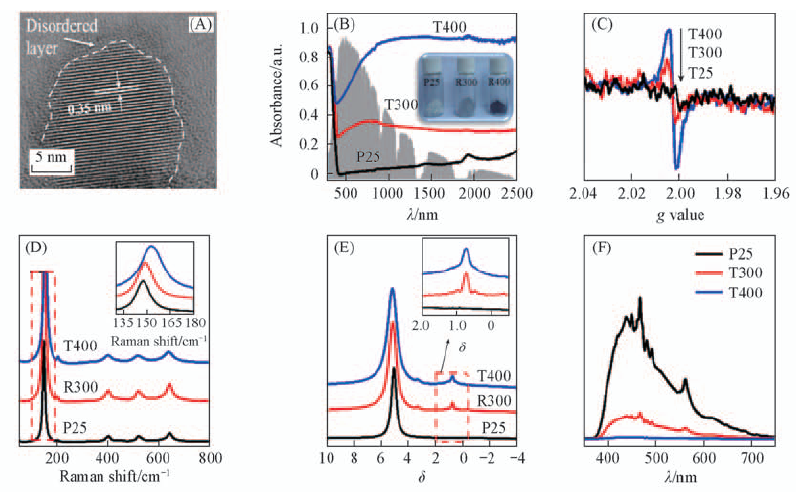
Fig.12 HRTEM image of T400(A), UV-Vis absorption spectra(B), EPR spectra(C), Raman spectra(D), 1H NMR spectra(E) and PL spectra(F) of reference P25 and the reduced samples[31] The insets in (D) and (E) are partial magnification of the marked region with a red border. Copyright from Wiley-VCH.
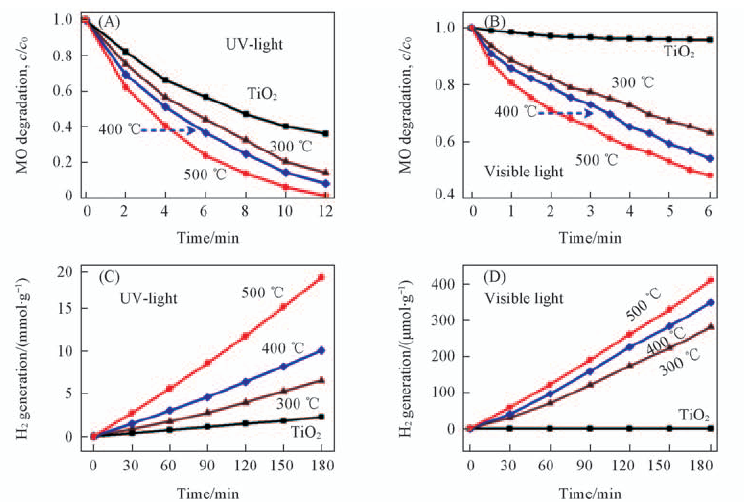
Fig.13 Evaluation of the photocatalytic activities of TiO2-x[27] (A) UV light photo-catalytic degradation of methyl orange; (B) visible light photocatalytic degradation of methyl orange; (C) H2 generation of black TiO2-x under UV light; (D) visible light irradiation. Copyright from the Royal Society of Chemistry.
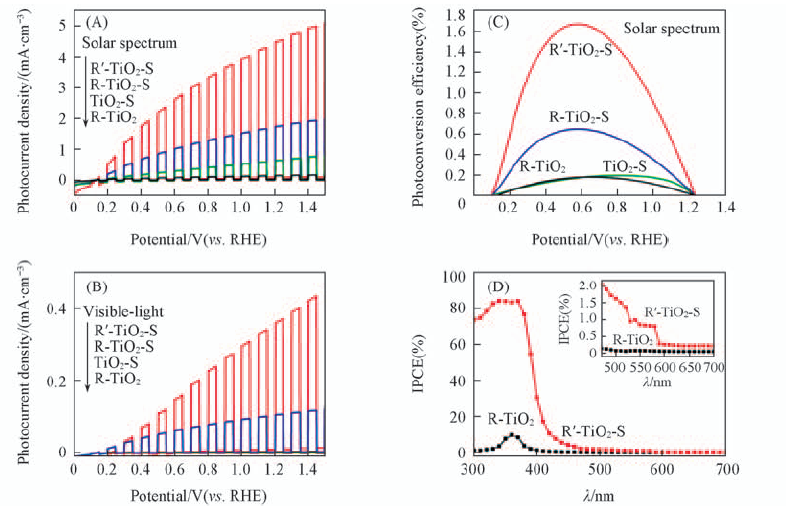
Fig.15 Photoelectrochemical properties of R'-TiO2-S, R-TiO2-S, TiO2-S, and R-TiO2 electrodes (A) Chopped J-V curves under simulated solar light illumination; (B) visible-light illumination via a three-electrode setup(TiO2 working, Pt counter, Ag/AgCl reference electrode, scan rate of 10 mV/s) in 1 mol/L NaOH electrolyte(pH=13.6); (C) photoconversion efficiencies as a function of applied potential; (D) IPCE spectra in the region of 300—700 nm at 0.65 VRHE. Inset of (D) IPCE spectra in the region of 420—700 nm[28]. Copyright from the American Chemical Society.
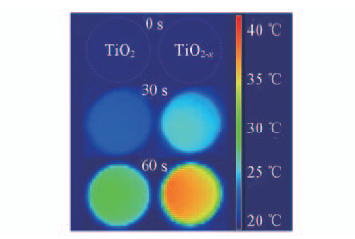
Fig.16 Thermal image map of cool-pressed disks after irradiation under AM 1.5 G Xe lamp solar simulator for different time[27] Copyright from the Royal Society of Chemistry.
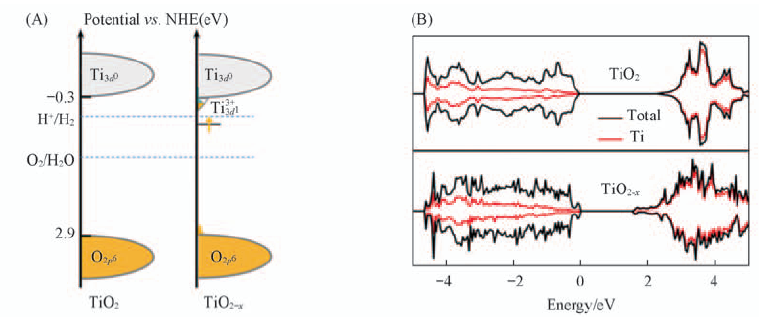
Fig.17 Schematic electronic structures for TiO2 and TiO2-x(A) and total DOS and partial DOS(Ti) for TiO2 and TiO2-x(B)[27] Copyright from the Royal Society of Chemistry.
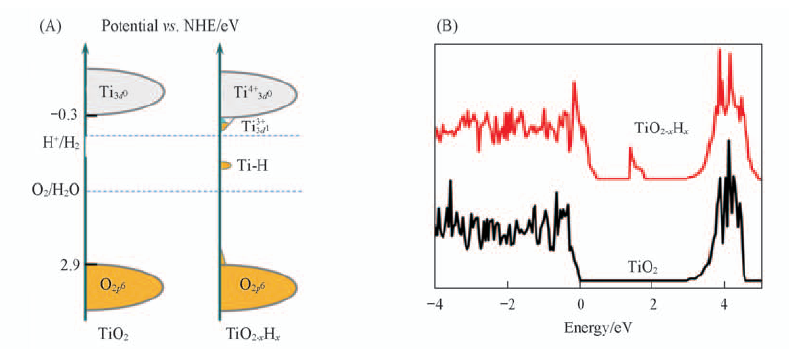
Fig.18 Schematic electronic structures for hydrogenated titania(TiO2-xHx) and pristine TiO2(A) and calculated DOS of pristine TiO2 and black TiO2-xHx(B)[26] Copyright from Wiley-VCH.
| [1] | Kubacka A., Fernandez-Garcia M., Colon G., Chem. Rev., 2012, 112, 1555—1614 |
| [2] | Kudo A., Miseki Y., Chem. Soc. Rev., 2009, 38, 253—278 |
| [3] | Chen X., Mao S. S., Chem. Rev., 2007, 107, 2891—2959 |
| [4] | Lü X., Mou X., Wu J., Zhang D., Zhang L., Huang F., Xu F., Huang S., Adv. Funct. Mater., 2010, 20, 509—515 |
| [5] | Lu X., Huang F., Mou X., Wang Y., Xu F., Adv. Mater., 2010, 22, 3719—3722 |
| [6] | Gratzel M., Nature, 2001, 414, 338—344 |
| [7] | Batzill M., Morales E., Diebold U., Phys. Rev. Lett., 2006, 96, 02613 |
| [8] | Subramanian V., Wolf E. E., Kamat P. V., Langmuir, 2003, 19, 469—474 |
| [9] | Schneider J., Matsuoka M., Takeuchi M., Zhang J., Horiuchi Y., Anpo M., Bahnemann D. W., Chem. Rev., 2014, 114, 9919—9986 |
| [10] | Zhou H., Qu Y., Zeid T., Duan X., Energy Environ. Sci., 2012, 5, 6732—6743 |
| [11] | Yi Z., Ye J., Kikugawa N., Kako T., Ouyang S., Stuart-Williams H., Yang H., Cao J., Luo W., Li Z., Liu Y., Withers R. L., Nat. Mater., 2010, 9, 559—564 |
| [12] | Chen X., Liu L., Yu P. Y., Mao S. S., Science, 2011, 331, 746—750 |
| [13] | Hoang S., Berglund S. P., Hahn N. T., Bard A. J., Mullins C. B., J. Am. Chem. Soc., 2012, 134, 3659—3662 |
| [14] | Jiang X., Zhang Y., Jiang J., Rong Y., Wang Y., Wu Y., Pan C., J. Phys. Chem. C, 2012, 116, 22619—22624 |
| [15] | Liu L., Yu P. Y., Chen X., Mao S. S., Shen D. Z., Phys. Rev. Lett., 2013, 111, 0065505 |
| [16] | Yang Y., Ling Y. C., Wang G. M., Li Y., Eur. J. Inorg. Chem., 2014, 4, 760—766 |
| [17] | Zhu W. D., Wang C. W., Chen J. B., Li D. S., Zhou F., Zhang H. L., Nanotechnology, 2012, 23, 455204—455209 |
| [18] | Xia T., Zhang C., Oyler N. A., Chen X. B., Adv. Mater., 2013, 25, 6905—6910 |
| [19] | Chen X., Liu L., Huang F., Chem. Soc. Rev., 2015, 46, 1861—1885 |
| [20] | Zheng Z., Huang B., Lu J., Wang Z., Qin X., Zhang X., Dai Y., Whangbo M. H., Chem. Commun., 2012, 48, 5733—5735 |
| [21] | Lu J., Dai Y., Jin H., Huang B., Phys. Chem. Chem. Phys., 2011, 13, 18063—18068 |
| [22] | Wang G., Wang H., Ling Y., Tang Y., Yang X., Fitzmorris R. C., Wang C., Zhang J. Z., Li Y., Nano Lett., 2011, 11, 3026—3033 |
| [23] | Wei W., Yaru N., Chunhua L., Zhongzi X., RSC Adv., 2012, 2, 8286—8288 |
| [24] | Leshuk T., Parviz R., Everett P., Krishnakumar H., Varin R. A., Gu F., ACS Appl. Mater. Interfaces, 2013, 5, 1892—1895 |
| [25] | Zhang S., Zhang S., Peng B., Wang H., Yu H., Wang H., Peng F., Electrochem. Commun., 2014, 40, 24—27 |
| [26] | Wang Z., Yang C., Lin T., Yin H., Chen P., Wan D., Xu F., Huang F., Lin J., Xie X., Jiang M., Adv. Funct. Mater., 2013, 23, 5444—5450 |
| [27] | Wang Z., Yang C., Lin T., Yin H., Chen P., Wan D., Xu F., Huang F., Lin J., Xie X., Jiang M., Energy Environ. Sci., 2013, 6, 3007—3014 |
| [28] | Yang C., Wang Z., Lin T., Yin H., Lu X., Wan D., Xu T., Zheng C., Lin J., Huang F., Xie X., Jiang M., J. Am. Chem. Soc., 2013, 135, 17831—17838 |
| [29] | Cui H., Zhao W., Yang C., Yin H., Lin T., Shan Y., Xie Y., Gu H., Huang F., J. Mater. Chem. A, 2014, 2, 8612—8616 |
| [30] | Lin T. Q., Yang C. Y., Wang Z., Yin H., Lu X. J., Huang F. Q., Lin J. H., Xie X. M., Jiang M. H., Energy Environ. Sci., 2014, 7, 967—972 |
| [31] | Zhu G., Yin H., Yang C., Cui H., Wang Z., Xu J., Lin T., Huang F., ChemCatChem, 2015, 7, 2614—2619 |
| [32] | Zhu G., Lin T., Lü X., Zhao W., Yang C., Wang Z., Yin H., Liu Z., Huang F., Lin J., J. Mater. Chem. A, 2013, 1, 9650—9653 |
| [33] | Yu J., Dai G., Huang B., J. Phys. Chem. C, 2009, 113, 16394—16401 |
| [34] | Yu J., Tao H., Cheng B., ChemPhysChem, 2010, 11, 1617—1618 |
| [35] | Prokes S. M., Gole J. L., Chen X., Burda C., Carlos W. E., Adv. Funct. Mater., 2005, 15, 161—167 |
| [36] | Lazarus M. S., Sham T. K., Chem. Phys. Lett., 1982, 92, 670—674 |
| [37] | Yin H., Lin T., Yang C., Wang Z., Zhu G., Xu T., Xie X., Huang F., Jiang M., Chemistry Eur. J., 2013, 19, 13313—13316 |
| [38] | Iskandar F., Nandiyanto A. B. D., Yun K. M., Hogan C. J., Okuyama K., Biswas P., Adv. Mater., 2007, 19, 1408—1412 |
| [39] | Kandiel T.A., Feldhoff A., Robben L., Dillert R., Bahnemann D. W., Chem. Mater., 2010, 22, 2050—2060 |
| [40] | Zhao W., Zhao W., Zhu G., Lin T., Xu F., Huang F., CrystEngComm, 2015, 17, 7528—7534 |
| [41] | Henderson M. A., Epling W. S., Peden C. H. F., Perkins C. L., J. Phys. Chem. B, 2003, 107, 534—545 |
| [42] | Finazzi E., Di Valentin C., Pacchioni G., Selloni A., J. Chem. Phys., 2008, 129, 154113 |
| [43] | Tojo S., Tachikawa T., Fujitsuka M., Majima T., J. Phys. Chem. C, 2008, 112, 14948—14954 |
| [44] | Morgan W. E., van Wazer J. R., Stec W. J., J. Am. Chem. Soc., 1973, 95, 751—755 |
| [45] | Wang J., Tafen D. N., Lewis J. P., Hong Z., Manivannan A., Zhi M., Li M., Wu N., J. Am. Chem. Soc., 2009, 131, 12290—12297 |
| [1] | TENG Zhenyuan, ZHANG Qitao, SU Chenliang. Charge Separation and Surface Reaction Mechanisms for Polymeric Single-atom Photocatalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220325. |
| [2] | QIN Yongji, LUO Jun. Applications of Single-atom Catalysts in CO2 Conversion [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220300. |
| [3] | LIN Zhi, PENG Zhiming, HE Weiqing, SHEN Shaohua. Single-atom and Cluster Photocatalysis: Competition and Cooperation [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220312. |
| [4] | ZHAO Yingzhe, ZHANG Jianling. Applications of Metal-organic Framework-based Material in Carbon Dioxide Photocatalytic Conversion [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220223. |
| [5] | XIA Wu, REN Yingyi, LIU Jing, WANG Feng. Chitosan Encapsulated CdSe QDs Assemblies for Visible Light-induced CO2 Reduction in an Aqueous Solution [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220192. |
| [6] | QIU Liqi, YAO Xiangyang, HE Liangnian. Visible-light-driven Selective Reduction of Carbon Dioxide Catalyzed by Earth-abundant Metalloporphyrin Complexes [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220064. |
| [7] | WANG Guangqi, BI Yiyang, WANG Jiabo, SHI Hongfei, LIU Qun, ZHANG Yu. Heterostructure Construction of Noble-metal-free Ternary Composite Ni(PO3)2-Ni2P/CdS NPs and Its Visible Light Efficient Catalytic Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220050. |
| [8] | TAO Yu, OU Honghui, LEI Yongpeng, XIONG Yu. Research Progress of Single-atom Catalysts in Photocatalytic Reduction of Carbon Dioxide [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220143. |
| [9] | FENG Li, SHAO Lanxing, LI Sijun, QUAN Wenxuan, ZHUANG Jinliang. Synthesis of Ultrathin Sm-MOF Nanosheets and Their Visible-light Induced Photodegradation of Mustard Simulant [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210867. |
| [10] | MENG Xiangyu, ZHAN Qi, WU Yanan, MA Xiaoshuang, JIANG Jingyi, SUN Yueming, DAI Yunqian. Photothermal Enhanced Photocatalytic Hydrogenation Performance of Au/RGO/Na2Ti3O7 [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210655. |
| [11] | GUO Biao, ZHAO Chencan, LIU Xinxin, YU Zhou, ZHOU Lijing, YUAN Hongming, ZHAO Zhen. Effects of Surface Hydrothermal Carbon Layer on the Photocatalytic Activity of Magnetic NiFe2O4 Octahedron [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220472. |
| [12] | LI Chenchen, NA Yong. g-C3N4/CdS/Ni Composite as a Bifunctional Photocatalyst for H2 Generation and 5-Hydroxymethylfurfural Oxidation [J]. Chem. J. Chinese Universities, 2021, 42(9): 2896. |
| [13] | LI Yishan, GUO Liang, PENG Sifan, ZHANG Qingmao, ZHANG Yuhao, XU Shiqi. Cobalt Substitutions in Lanthanum Manganate Photocatalyst: First-principles and Visible-light Photocatalytic Ability Investigation [J]. Chem. J. Chinese Universities, 2021, 42(6): 1881. |
| [14] | WANG Peng, YANG Min, TANG Sengpei, CHEN Feitai, LI Youji. Preparation of Cellular C3N4/CoSe2/GA Composite Photocatalyst and Its CO2 Reduction Activity [J]. Chem. J. Chinese Universities, 2021, 42(6): 1924. |
| [15] | YANG Sixian, ZHONG Wenyu, LI Chaoxian, SU Qiuyao, XU Bingjia, HE Guping, SUN Fengqiang. Photochemical Fabrication and Performance of Polyaniline Nanowire/SnO2 Composite Photocatalyst [J]. Chem. J. Chinese Universities, 2021, 42(6): 1942. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||