

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (11): 2447.doi: 10.7503/cjcu20140195
• Physical Chemistry • Previous Articles Next Articles
FAN Dongli, ZHAI Yan, ZHANG Yan, TU Wei, HUANG Yaodong*( )
)
Received:2014-03-10
Online:2014-11-10
Published:2014-10-09
Contact:
HUANG Yaodong
E-mail:huangyaodong@tju.edu.cn
CLC Number:
TrendMD:
FAN Dongli, ZHAI Yan, ZHANG Yan, TU Wei, HUANG Yaodong. Synthesis and Properties of Photoresponsive Organogels Based on Azobenzene Derivatives[J]. Chem. J. Chinese Universities, 2014, 35(11): 2447.
| Compd. | 1H NMR(300 MHz, CDCl3), δ | m.p./ ℃ | Yield (%) | GC-MS [M+H]+, m/z |
|---|---|---|---|---|
| 1a | 7.90(d, J=9.1 Hz, 2H, HAr), 7.83(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.06(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m,8H,CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)5CH3] | 113—115 | 92 | 409.1 |
| 1b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.84(d, J=8.7 Hz, 2H, H—Ar), 7.62(d, J=8.7 Hz, 2H, H—Ar), 6.98(d, J=9.1 Hz, 2H, H—Ar), 4.02(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)11CH3] | 107—109 | 95 | 493.2 |
| 1c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.99(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)15CH3] | 109—110 | 94 | 549.3 |
| 2a | 7.92(d, J=9.1 Hz, 2H, H—Ar), 7.84(d, J=8.4 Hz, 2H, H—Ar), 7.63(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 3.99(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3], 0.083(s, 9H, CH3) | 100—102 | 81 | 379.1 |
| 2b | 7.91(d, J=9.1Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.95(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3], 0.084(s, 9H, CH3) | 105—106 | 80 | 463.2 |
| 2c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.81(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3], 0.083(s, 9H, CH3) | 86—88 | 78 | 519.3 |
| Compd. | 1H NMR(300 MHz,CDCl3), δ | m.p./ ℃ | Yield (%) | GC-MS [M+H]+, m/z |
| 3a | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 89—91 | 72 | 307.1 |
| 3b | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3] | 90—92 | 75 | 391.2 |
| 3c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 99—101 | 74 | 447.3 |
| 4a | 7.91(d, J=9.0 Hz, 2H, H—Ar), 7.87(d, J=8.7 Hz, 2H, H—Ar), 7.52(d, J=8.2 Hz, 2H, H—Ar), 6.99(d, J=9.0 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H,CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 6H, CH2), 0.9(t, J=6.6 Hz, 3H, —O(CH2)15CH3) | 118—120 | 94 | 313.2 |
| 4b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.55(d, J=8.4 Hz, 2H, H—Ar), 6.99(d, J=8.9 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H, CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 18H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 104—106 | 95 | 397.3 |
| 4c | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.55(d, J=8.4 Hz, 2H, H—Ar), 6.99(d, J=8.9 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H, CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 26H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 118—120 | 95 | 453.4 |
| 5a | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 72—74 | 83 | 331.1 |
| 5b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)11CH3] | 88—89 | 85 | 415.2 |
| 5c | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 93—95 | 84 | 471.2 |
| 6a | 7.82(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s,2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 61—63 | 82 | 331.9 |
| 6b | 7.81(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s, 2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3] | 78—80 | 80 | 422.6 |
| 6c | 7.81(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s, 2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 89—91 | 79 | 472.1 |
Table 1 1H NMR, melting points, yields and GC-MS data for compounds 1—6
| Compd. | 1H NMR(300 MHz, CDCl3), δ | m.p./ ℃ | Yield (%) | GC-MS [M+H]+, m/z |
|---|---|---|---|---|
| 1a | 7.90(d, J=9.1 Hz, 2H, HAr), 7.83(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.06(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m,8H,CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)5CH3] | 113—115 | 92 | 409.1 |
| 1b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.84(d, J=8.7 Hz, 2H, H—Ar), 7.62(d, J=8.7 Hz, 2H, H—Ar), 6.98(d, J=9.1 Hz, 2H, H—Ar), 4.02(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)11CH3] | 107—109 | 95 | 493.2 |
| 1c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.99(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)15CH3] | 109—110 | 94 | 549.3 |
| 2a | 7.92(d, J=9.1 Hz, 2H, H—Ar), 7.84(d, J=8.4 Hz, 2H, H—Ar), 7.63(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 3.99(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3], 0.083(s, 9H, CH3) | 100—102 | 81 | 379.1 |
| 2b | 7.91(d, J=9.1Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.95(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3], 0.084(s, 9H, CH3) | 105—106 | 80 | 463.2 |
| 2c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.81(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3], 0.083(s, 9H, CH3) | 86—88 | 78 | 519.3 |
| Compd. | 1H NMR(300 MHz,CDCl3), δ | m.p./ ℃ | Yield (%) | GC-MS [M+H]+, m/z |
| 3a | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 89—91 | 72 | 307.1 |
| 3b | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3] | 90—92 | 75 | 391.2 |
| 3c | 7.89(d, J=9.1 Hz, 2H, H—Ar), 7.82(d, J=8.4 Hz, 2H, H—Ar), 7.61(d, J=8.7 Hz, 2H, H—Ar), 6.94(d, J=9.1 Hz, 2H, H—Ar), 4.01(t, J=6.6 Hz, 2H, —OCH2), 3.1(s, 1H), 1.32—1.89(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 99—101 | 74 | 447.3 |
| 4a | 7.91(d, J=9.0 Hz, 2H, H—Ar), 7.87(d, J=8.7 Hz, 2H, H—Ar), 7.52(d, J=8.2 Hz, 2H, H—Ar), 6.99(d, J=9.0 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H,CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 6H, CH2), 0.9(t, J=6.6 Hz, 3H, —O(CH2)15CH3) | 118—120 | 94 | 313.2 |
| 4b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.55(d, J=8.4 Hz, 2H, H—Ar), 6.99(d, J=8.9 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H, CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 18H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 104—106 | 95 | 397.3 |
| 4c | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.55(d, J=8.4 Hz, 2H, H—Ar), 6.99(d, J=8.9 Hz, 2H, H—Ar), 4.77(s, 2H, —CH2OH), 4.04(t, J=6.6 Hz, 2H, —OCH2), 1.75—1.86(m, 2H, CH2), 1.68(s, 1H, OH), 1.32—1.51(m, 26H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 118—120 | 95 | 453.4 |
| 5a | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 72—74 | 83 | 331.1 |
| 5b | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H,—O(CH2)11CH3] | 88—89 | 85 | 415.2 |
| 5c | 7.91(d, J=9.1 Hz, 2H, H—Ar), 7.87(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.64(s, 2H, —CH2Cl), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 93—95 | 84 | 471.2 |
| 6a | 7.82(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s,2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 8H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)5CH3] | 61—63 | 82 | 331.9 |
| 6b | 7.81(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s, 2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 20H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)11CH3] | 78—80 | 80 | 422.6 |
| 6c | 7.81(d, J=9.1 Hz, 2H, H—Ar), 7.79(d, J=8.4 Hz, 2H, H—Ar), 7.53(d, J=8.7 Hz, 2H, H—Ar), 6.97(d, J=9.1 Hz, 2H, H—Ar), 4.40(s, 2H, —CH2N3), 4.01(t, J=6.6 Hz, 2H, —OCH2), 1.32—1.87(m, 28H, CH2), 0.9[t, J=6.6 Hz, 3H, —O(CH2)15CH3] | 89—91 | 79 | 472.1 |
| Solvent | Minimum gel concentration/(mg·mL-1) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1a | 1b | 1c | 2a | 2b | 2c | 3a | 3b | 3c | 4a | 4b | 4c | 5a | 5b | 5c | 6a | 6b | 6c | |
| PE | 100 | P | P | 100 | 100 | 55 | 100 | P | P | Ins | Ins | 44 | PG | 20 | P | PG | 67 | P |
| Cyclohexane | PG | P | P | PG | PG | P | PG | P | P | 27 | P | 53 | PG | 40 | 56 | PG | PG | P |
| Hexane | 100 | P | P | PG | 80 | P | 67 | 80 | P | Ins | P | 40 | PG | 23 | P | PG | 50 | P |
| DCM | PG | 40 | 50 | PG | 100 | S | PG | 40 | 100 | 57 | 20 | 10 | PG | 100 | S | PG | PG | S |
| DCE | PG | 33 | 20 | P | P | P | P | P | P | 26 | 20 | 26 | P | P | P | P | P | P |
| Chloroform | PG | 100 | PG | S | PG | S | S | PG | S | 80 | 80 | PG | S | PG | S | S | PG | S |
| CTC | 20 | P | PG | S | PG | PG | PG | P | P | 36 | P | PG | S | PG | P | S | PG | PG |
| Ethyl acetate | PG | 80 | PG | PG | 100 | PG | PG | 80 | P | 80 | 57 | P | S | 50 | PG | S | PG | 55 |
| THF | S | PG | S | S | PG | S | S | PG | P | S | 20 | PG | S | PG | S | S | PG | PG |
| Methanol | PG | 68 | PG | 83 | P | PG | 83 | P | 35 | 33 | 50 | 53 | 56 | 100 | P | PG | 80 | 37 |
| Ethanol | 67 | 71 | 35 | 83 | 48 | 35 | 100 | 40 | PG | 40 | 20 | 53 | 100 | 49 | P | PG | 36 | P |
| Acetonitrile | 67 | 68 | 18 | 43 | 32 | 55 | 77 | 28 | 15 | 57 | 19 | 26 | PG | 24 | 37 | 67 | 22 | 27 |
| Isopropanol | 50 | 100 | 35 | 59 | 100 | PG | 59 | PG | P | 33 | 100 | PG | 100 | 60 | P | 67 | 40 | P |
| Butanol | 56 | 50 | 20 | 83 | 75 | 30 | 56 | 60 | 24 | 40 | 30 | 18 | PG | 40 | 24 | 62 | 40 | 21 |
| Acetone | PG | 100 | 27 | PG | PG | 50 | PG | PG | P | PG | 100 | 29 | S | PG | 56 | S | 80 | 37 |
| Ether | PG | 100 | Ins | PG | PG | Ins | PG | P | Ins | PG | 100 | Ins | PG | 80 | Ins | PG | 80 | Ins |
| Benzene | PG | S | 55 | S | PG | S | S | PG | P | 80 | P | 53 | S | S | P | S | PG | P |
| Toluene | S | PG | P | S | PG | S | S | PG | S | PG | PG | P | S | P | PG | S | PG | S |
| Dioxane | PG | S | 55 | S | PG | PG | S | PG | P | 50 | S | PG | S | 80 | PG | S | PG | 55 |
| Triethylamine | PG | PG | PG | PG | PG | P | PG | PG | P | S | P | PG | PG | PG | 30 | PG | PG | P |
| Pyridine | S | PG | 37 | S | PG | S | S | PG | P | S | S | PG | S | S | 37 | S | 80 | S |
Table 2 Gelation properties of compounds 1—6*
| Solvent | Minimum gel concentration/(mg·mL-1) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1a | 1b | 1c | 2a | 2b | 2c | 3a | 3b | 3c | 4a | 4b | 4c | 5a | 5b | 5c | 6a | 6b | 6c | |
| PE | 100 | P | P | 100 | 100 | 55 | 100 | P | P | Ins | Ins | 44 | PG | 20 | P | PG | 67 | P |
| Cyclohexane | PG | P | P | PG | PG | P | PG | P | P | 27 | P | 53 | PG | 40 | 56 | PG | PG | P |
| Hexane | 100 | P | P | PG | 80 | P | 67 | 80 | P | Ins | P | 40 | PG | 23 | P | PG | 50 | P |
| DCM | PG | 40 | 50 | PG | 100 | S | PG | 40 | 100 | 57 | 20 | 10 | PG | 100 | S | PG | PG | S |
| DCE | PG | 33 | 20 | P | P | P | P | P | P | 26 | 20 | 26 | P | P | P | P | P | P |
| Chloroform | PG | 100 | PG | S | PG | S | S | PG | S | 80 | 80 | PG | S | PG | S | S | PG | S |
| CTC | 20 | P | PG | S | PG | PG | PG | P | P | 36 | P | PG | S | PG | P | S | PG | PG |
| Ethyl acetate | PG | 80 | PG | PG | 100 | PG | PG | 80 | P | 80 | 57 | P | S | 50 | PG | S | PG | 55 |
| THF | S | PG | S | S | PG | S | S | PG | P | S | 20 | PG | S | PG | S | S | PG | PG |
| Methanol | PG | 68 | PG | 83 | P | PG | 83 | P | 35 | 33 | 50 | 53 | 56 | 100 | P | PG | 80 | 37 |
| Ethanol | 67 | 71 | 35 | 83 | 48 | 35 | 100 | 40 | PG | 40 | 20 | 53 | 100 | 49 | P | PG | 36 | P |
| Acetonitrile | 67 | 68 | 18 | 43 | 32 | 55 | 77 | 28 | 15 | 57 | 19 | 26 | PG | 24 | 37 | 67 | 22 | 27 |
| Isopropanol | 50 | 100 | 35 | 59 | 100 | PG | 59 | PG | P | 33 | 100 | PG | 100 | 60 | P | 67 | 40 | P |
| Butanol | 56 | 50 | 20 | 83 | 75 | 30 | 56 | 60 | 24 | 40 | 30 | 18 | PG | 40 | 24 | 62 | 40 | 21 |
| Acetone | PG | 100 | 27 | PG | PG | 50 | PG | PG | P | PG | 100 | 29 | S | PG | 56 | S | 80 | 37 |
| Ether | PG | 100 | Ins | PG | PG | Ins | PG | P | Ins | PG | 100 | Ins | PG | 80 | Ins | PG | 80 | Ins |
| Benzene | PG | S | 55 | S | PG | S | S | PG | P | 80 | P | 53 | S | S | P | S | PG | P |
| Toluene | S | PG | P | S | PG | S | S | PG | S | PG | PG | P | S | P | PG | S | PG | S |
| Dioxane | PG | S | 55 | S | PG | PG | S | PG | P | 50 | S | PG | S | 80 | PG | S | PG | 55 |
| Triethylamine | PG | PG | PG | PG | PG | P | PG | PG | P | S | P | PG | PG | PG | 30 | PG | PG | P |
| Pyridine | S | PG | 37 | S | PG | S | S | PG | P | S | S | PG | S | S | 37 | S | 80 | S |
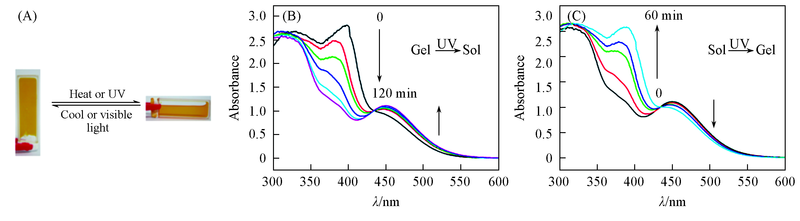
Fig.2 Pictures(A) and UV-Vis spectra(B, C) of the gel-sol transitions of gel 4c (A) Heated/cooled or irradiated by UV/visible light; (B) UV irradiation(0.1 cm path length); (C) visible irradiation.

Fig.3 Teas plot of solubility parameters(A) and pictures of gelation behaviors of compound 4b(100 mg/mL) at mixed solvents(B1—B3) (A) Compound 4b and the single/mixed solvents. EAC: ethanol; CYH: cyclohexane; DCM: dichloromethane. (B1—B2) Gelation behavior and DCM:CYH(volume ratios): (B1) gel, 9:1—7:3; (B2) precipitation, 2:8, 1:9; (B3) partial gel, 6:4—3:7.
| Solvent volume ratio | 9:1 | 8:2 | 7:3 | 6:4 | 5:5 | 4:6 | 3:7 | 2:8 | 1:9 |
|---|---|---|---|---|---|---|---|---|---|
| EAC:CYH | 25 | 30 | 50 | 100 | 50 | 30 | 50 | 30 | 50 |
| DCM:CYH | 50 | 50 | 100 | PG | PG | PG | PG | P | P |
Table 3 Gelation test of compound 4b in the mixed solvents(mg/mL)*
| Solvent volume ratio | 9:1 | 8:2 | 7:3 | 6:4 | 5:5 | 4:6 | 3:7 | 2:8 | 1:9 |
|---|---|---|---|---|---|---|---|---|---|
| EAC:CYH | 25 | 30 | 50 | 100 | 50 | 30 | 50 | 30 | 50 |
| DCM:CYH | 50 | 50 | 100 | PG | PG | PG | PG | P | P |
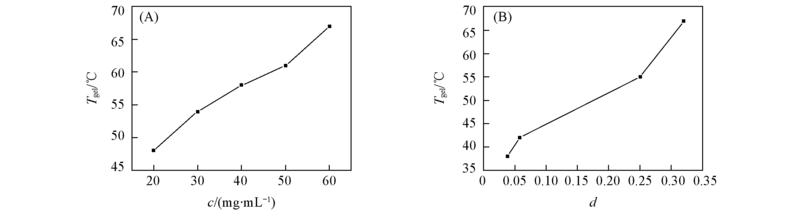
Fig.4 Plots of Tgel against the influence factors of the thermal stability of the gel (A) Concentration of compound 4b in ethanol; (B) distance between compound 4b and solvents on the Teas plot(dichloromethane, THF, butano and ethanol).
| [1] | Terech P., Weiss R. G., Chem. Rev., 1997, 97(8), 3133—3159 |
| [2] | Raeburn J., Zamith Cardoso A., Adams D. J., Chem. Soc. Rev., 2013, 42(12), 5143—5156 |
| [3] | Gawlitza K., Wu C. Z., Georgieva R., Wang D. Y., Ansorge-Schumacher M. B., von Klitzing R., Phys. Chem. Chem. Phys., 2012, 14(27), 9594—9600 |
| [4] | Jiang H. L., Zhu Y. H., Chen C., Shen J. H., Bao H., Peng L. M., Yang X. L., Li C. Z., New J. Chem., 2012, 36(4), 1051—1056 |
| [5] | Sugiyasu K., Fujita N., Shinkai S., Angew. Chem. Int. Ed., 2004, 43(10), 1229—1233 |
| [6] | Zhu S. J., Liu F. Q., Wang J. C., Su F., Li S. M., Chem. J. Chinese Universities,2014, 35(4), 863—868 |
| (朱寿进, 刘法谦, 王璟朝, 宿烽, 李速明. 高等学校化学学报,2014, 35(4), 863—868) | |
| [7] | Kubo W., Murakoshi K., Kitamura T., Yoshida S., Haruki M., Hanabusa K., Shirai H., Wada Y., Yanagida S., J. Phys. Chem., 2001, 105(51), 12809—12815 |
| [8] | Liu Z. X., Feng Y., Yan Z. C., He Y. M., Liu C. Y., Fan Q. H., Chem. Mater., 2012, 24(19), 3751—3757 |
| [9] | Wu Y. P., Wu S., Zou G., Zhang Q. J., Soft Matter,2011, 7(19), 9177—9183 |
| [10] | Deindorfer P., Davis R., Zentel R., Soft Matter,2007, 3(10), 1308—1311 |
| [11] | Edwards W., Lagadec C. A., Smith D. K., Soft Matter,2011, 7(1), 110—117 |
| [12] | Löfman M. L., Koivukorpi J., Noponen V., Salo H., Sieüanen E., Colloid Interface Sci., 2011, 360(2), 633—644 |
| [13] | Zhu G. Y., Dordick J. S., Chem. Mater., 2006, 18(25), 5988—5995 |
| [14] | Bielejewski M., Lapinski A., Luboradzki R., Tritt-Goc J., Langmuir,2009, 25(14), 8274—8279 |
| [15] | Tong C. Q., Fan K. Q., Niu L. B., Li J. J., Guan X. D., Tao N. M., Shen H. H., Song J., Soft Matter,2014, 10(5), 767—772 |
| [16] | Matthieu R., Laurent B., Chem. Commun., 2011, 47(29), 8271—8273 |
| [17] | Xu H., Zeng X. W., Bioorg. Med. Chem. Lett., 2010, 20(14), 4193—4195 |
| [18] | Huang Y. D., Dong X. L., Zhang L. L., Chai W., Chang J. Y., J. Mol. Struct., 2013, 1031(1), 43—48 |
| [19] | Austin W. B., Bilow N., Kelleghan W. J., Lau K. S. Y., J. Org. Chem., 1981, 46(11), 2280—2286 |
| [20] | Jiao T. F., Wang Y. J., Gao F. Q., Zhou J. X., Gao F. M., Prog. Nat. Sci. Mater. Int., 2012, 22(1), 64—70 |
| [21] | Wu S., Zhang Q. J., Bubeck C., Macromolecules,2010, 43(14), 6142—6151 |
| [22] | Barton A. F. M., Chem. Rev., 1975, 75(6), 731—753 |
| [23] | Stefanis E., Panayiotou C., Int. J. Thermophys,2008, 29(2), 568—585 |
| [1] | LIU Suyu, DING Fei, LI Qian, FAN Chunhai, FENG Jing. Azobenzene-integrated DNA Nanomachine [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220122. |
| [2] | GAO Jian, FENG Yiyu, FANG Wenyu, WANG Hui, GE Jing, FENG Wei. Alkane Grafted Phase Change Azobenzene Materials Based on Low Temperature Heat Release [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220146. |
| [3] | HUANG Yi, LYU Lingling, PAN Xiaopeng, SUN Guangdong, LI Yongqiang, YAO Juming, SHAO Jianzhong. Three-dimensional Printing of Photocrosslinked Self-supporting Silk Fibroin Hydrogels [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210841. |
| [4] | WANG Gaobo, MA Jing. Binding Selectivity Between Diazobenzene and Different Nucleophilic Reagents: Covalent and Noncovalent Interactions [J]. Chem. J. Chinese Universities, 2021, 42(7): 2238. |
| [5] | JIANG Lei, YAN Shengdi, LIN Yu, WU Guozhang. Studies on Viscosity and Chemical Structure Changes in Polycarbonate Melts under Nitrogen Protection [J]. Chem. J. Chinese Universities, 2021, 42(3): 884. |
| [6] | DENG Yaqian, WU Zhitan, LV Wei, TAO Ying, YANG Quanhong. Gelation of Two⁃dimensional Materials for Energy Storage Applications [J]. Chem. J. Chinese Universities, 2021, 42(2): 380. |
| [7] | LI Shanshan, ZHAO Wenjuan, LI Hui, FANG Qianrong. A Photoresponsive Azobenzene-functionalized Covalent Organic Framework † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1384. |
| [8] | ZHAO Ruiyang,YU Chunyan,HAN Jishu,FU Yunlei,LI Ming,HU Dehua,LIU Fusheng. Preparation of Photo-responsive Film by Electrochemical Deposition Method and the Application in Optical Information Storage† [J]. Chem. J. Chinese Universities, 2019, 40(2): 358. |
| [9] | ZHANG Chanchan, ZHANG Fanghui, DING Lei, NI Zhenjie, JIANG Lang, DONG Huanli, ZHANG Xiaotao, LI Rongjin, HU Wenping. Organic Phototransistor Based on Surface Plasmon Resonance Effect† [J]. Chem. J. Chinese Universities, 2018, 39(1): 102. |
| [10] | LI Zhiming, DING Qiang, GU Xiaojun, XIN Hong, BAI Binglian, LI Min. Organogel and Photo-responsive Behaviour of Hydrazide Derivatives Containing Azobenzene Groups† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1695. |
| [11] | YAN Chao, XIAO Yulong, DAI Heng, CHENG Xiaohong. Synthesis and Properties of Symmetric Azobenzene Derivative† [J]. Chem. J. Chinese Universities, 2016, 37(3): 475. |
| [12] | AI Hui, LI Wen, ZHANG Bin, LI Bao, WU Lixin. Light Modulated Host-guest Recognition of Organic Modified Polyoxometalate Complex† [J]. Chem. J. Chinese Universities, 2015, 36(11): 2297. |
| [13] | ZHANG Qingzhong, ZHANG Junmei, LIU Yuping, REN Xiao, HUANG Wei. Synthesis and Characterization of Electro-optic Materials Based on Intermolecular Lateral Hydrogen Bonding† [J]. Chem. J. Chinese Universities, 2015, 36(10): 2053. |
| [14] | JIANG Hongbo, GONG Chengbin, WANG Qiang, TANG Qian, MA Xuebing. Synthesis and Characterization of Novel Photo- and pH-Responsive Poly[8-1] [J]. Chem. J. Chinese Universities, 2014, 35(9): 2043. |
| [15] | HONG Xiaozhong, GU Fang, LI Jiangtao, WANG Haijun. Statistical Properties on the Aggregation of Patchy Particles† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1579. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||