

Chem. J. Chinese Universities ›› 2026, Vol. 47 ›› Issue (5): 20260013.doi: 10.7503/cjcu20260013
• Review • Previous Articles Next Articles
Received:2026-01-04
Online:2026-05-10
Published:2026-02-10
Contact:
QIN Anjun
E-mail:msqinaj@scut.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Kaiwei, QIN Anjun. Lanthanide Complexes with Aggregation-induced Emission[J]. Chem. J. Chinese Universities, 2026, 47(5): 20260013.
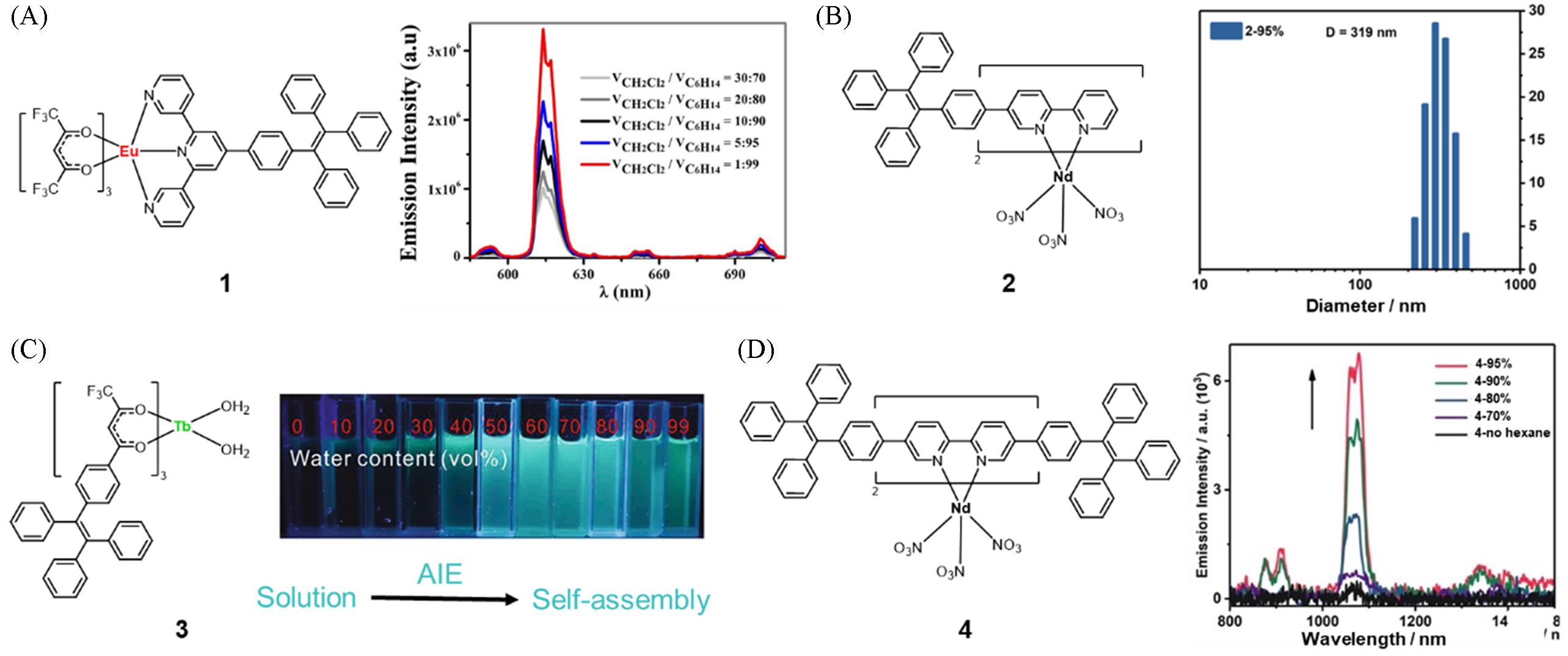
Fig.2 Molecular structure of complex 1 and photoluminescence(PL) spectra of complex 1 in dichloromethane with different volume fractions of n⁃hexane(A)[20], molecular structure of complex 2 and DLS of complex 2 in n⁃hexane/chloroform mixtures with the n⁃hexane content of 95%(B)[22], molecular structure of complex 3 and fluorescent photographs of complex 3(C)[23] and molecular structure of complex 4 and PL spectra of complex 4 in chloroform with different volume fractions of n⁃hexane(D)[22](A) Copyright 2015, Springer Nature; (B, D) Copyright 2021, John Wiley and Sons; (C) Copyright 2021, Elsevier Ltd.
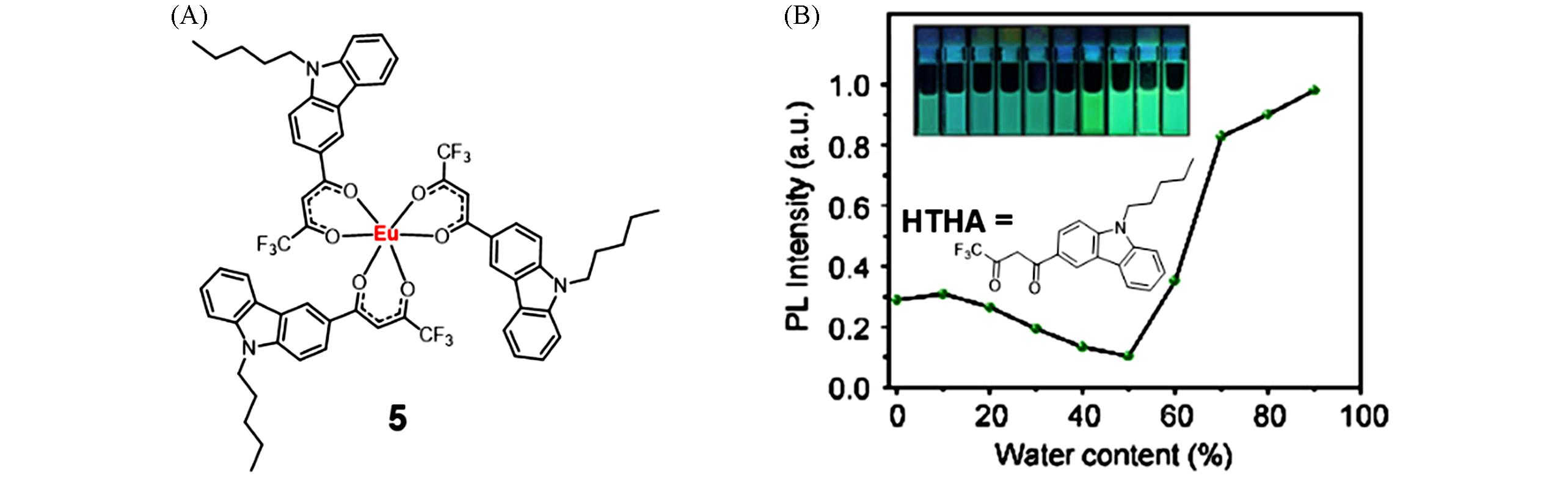
Fig.3 Molecular structure of complex 5(A) and PL intensity profile of ligand HTHA at 470 nm in ethanol with different volume fractions of water and fluorescent photographs of HTHA(B)[25]
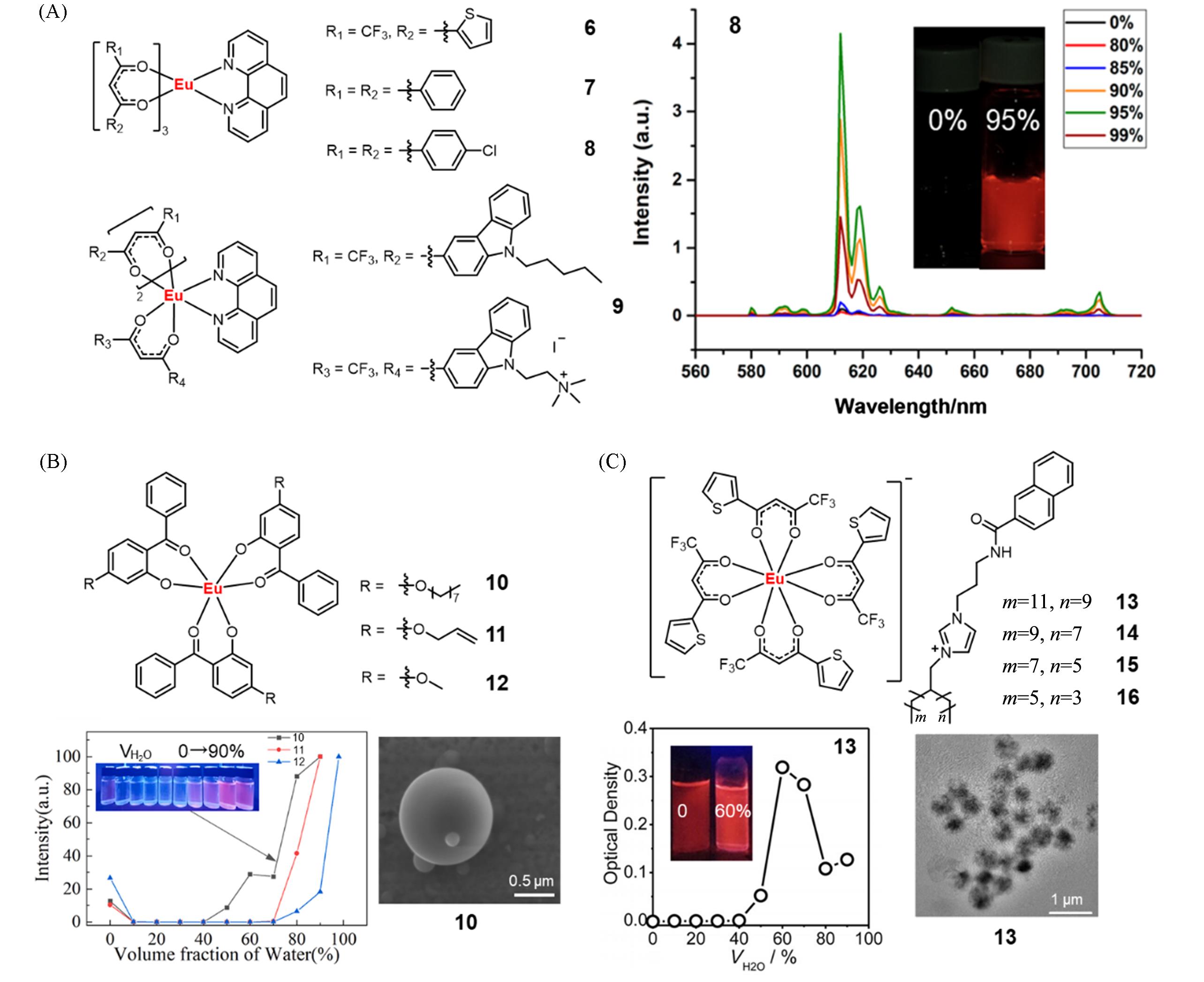
Fig.4 Molecular structure of β⁃diketone complexes 6—9 and PL spectra of complex 8 in THF with different volume fraction of water(A)[29], molecular structure of complexes 10—12 and PL intensity profile of complexes 10—12 at 615 nm in acetone with different volume fractions of water, SEM image of complex 10 with the water volume fraction of 90%(B)[31], molecular structure of complexes 13—16 and optical density of complex 13 at 500 nm in ethanol with different volume fractions of water, TEM image of aggregates formed by complex 13 in ethanol/water mixtures with water volume fraction of 60%(C)[32](A) Copyright 2019, MDPI; (B) Copyright 2025, Elsevier Ltd.; (C) Copyright 2018, John Wiley and Sons.
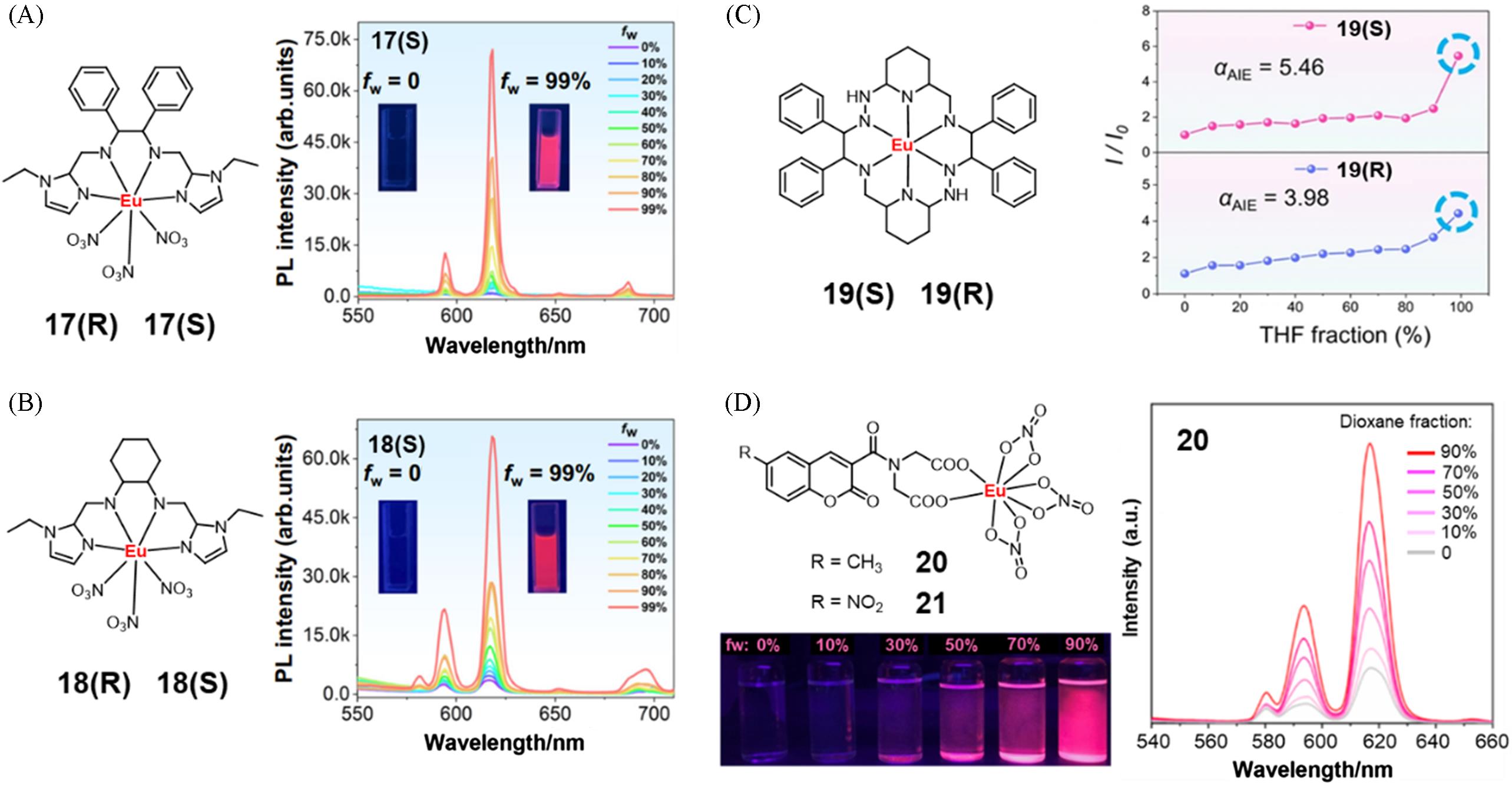
Fig.5 Molecular structure of chiral complexes 17(R) and 17(S) and PL spectra of complex 17(S) in DMSO with different fraction of glycerin(A)[33], molecular structure of chiral complexes 18(R) and 18(S) and PL spectra of complex 18(S) in DMSO with different fraction of glycerin(B)[33], molecular structure of chiral complexes 19(R) and 19(S) and PL intensity profile of complexes 19(R) and 19(S) at 611 nm in DMSO with different fraction of THF(C)[35], and molecular structure of complexes 20 and 21, fluorescent photographs and PL spectra of complex 20 in DMF with different fraction of 1,4⁃dioxane(D)[36](A) Copyright 2024, Springer Nature; (B) Copyright 2024, Springer Nature; (C) Copyright 2025, Springer Nature; (D) Copyright 2020, John Wiley and Sons.
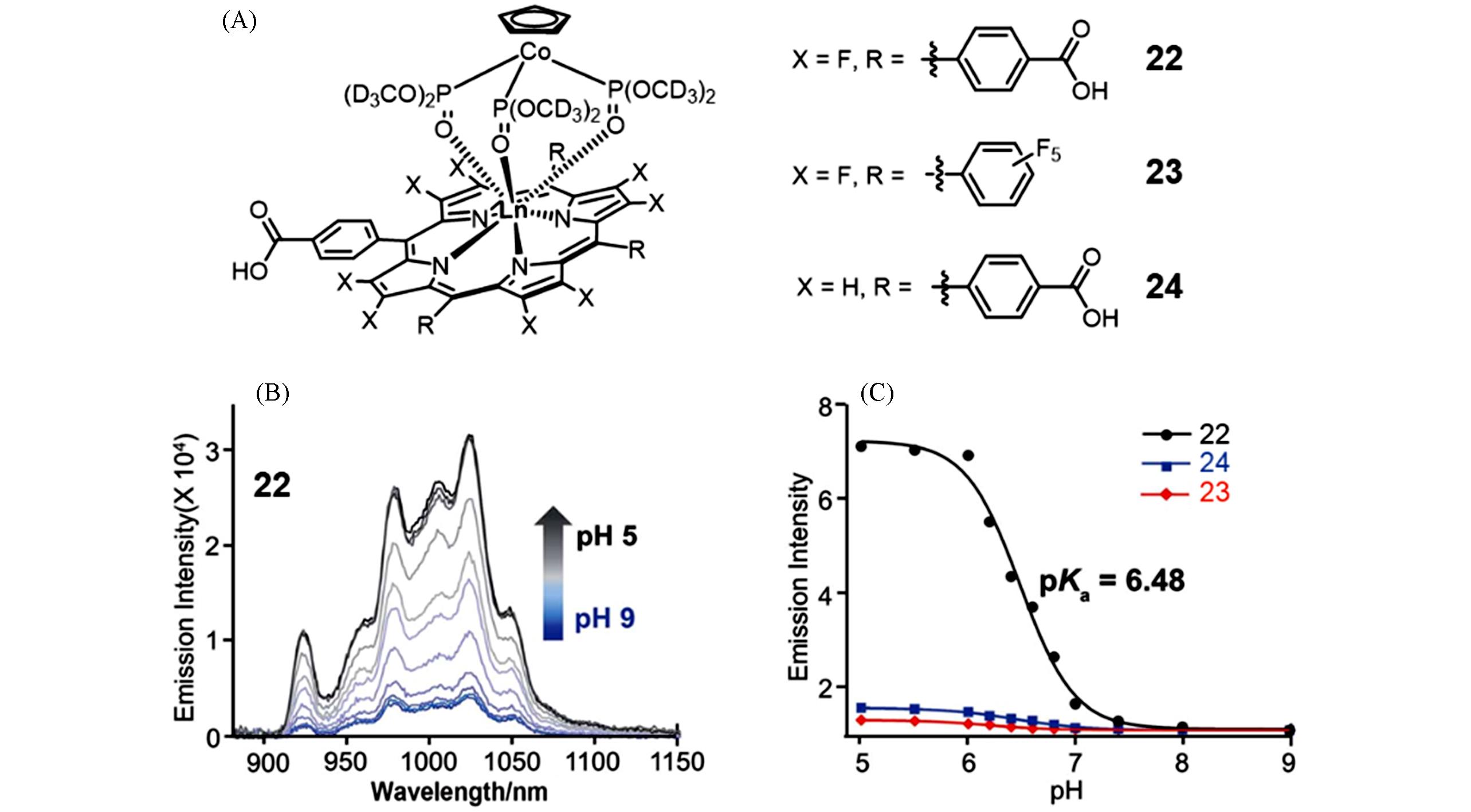
Fig.6 Molecular structure of complexes 22—24(A), fluorescence emission spectra of complex 22 in PBS buffer(pH=5.0—9.0)(B), and the normalized emission intensity ratio of complexes 22—24 according to the value at pH 9.0 versus the pH values in the pH range 5.0—9.0(C)[37]
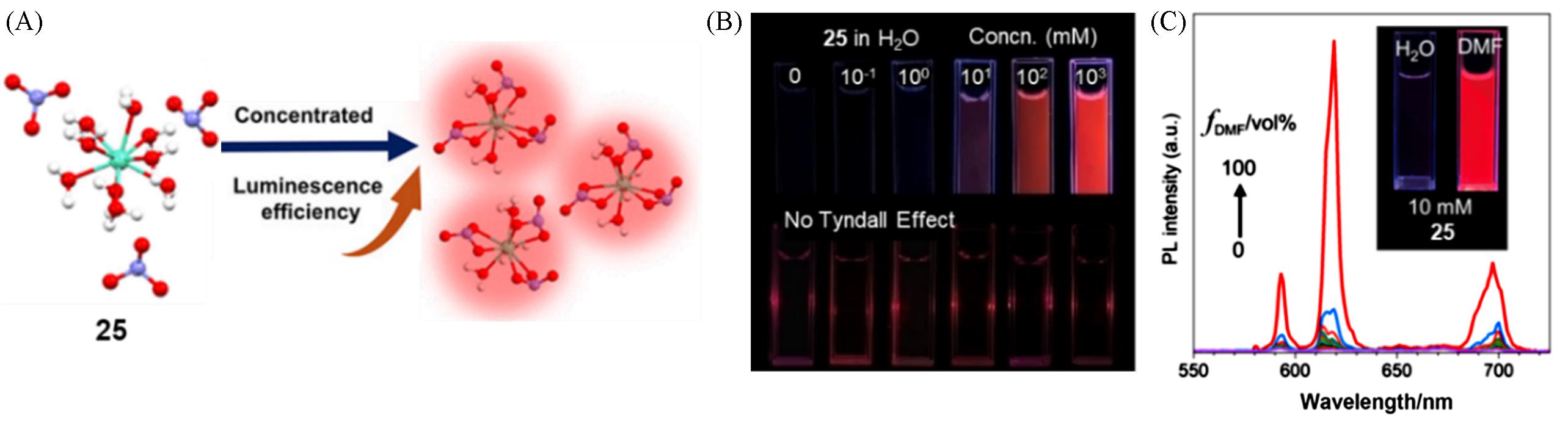
Fig.7 Schematic diagram of the structural changes of complex 25 from low concentration to aggregated state(A), fluorescence photographs of complex 25 solutions and laser for Tyndall effect test(B) and fluorescence emission spectra of complex 25 in water with different fraction of DMF and fluorescence photograph complex 25 in water and DMF(C)[39]
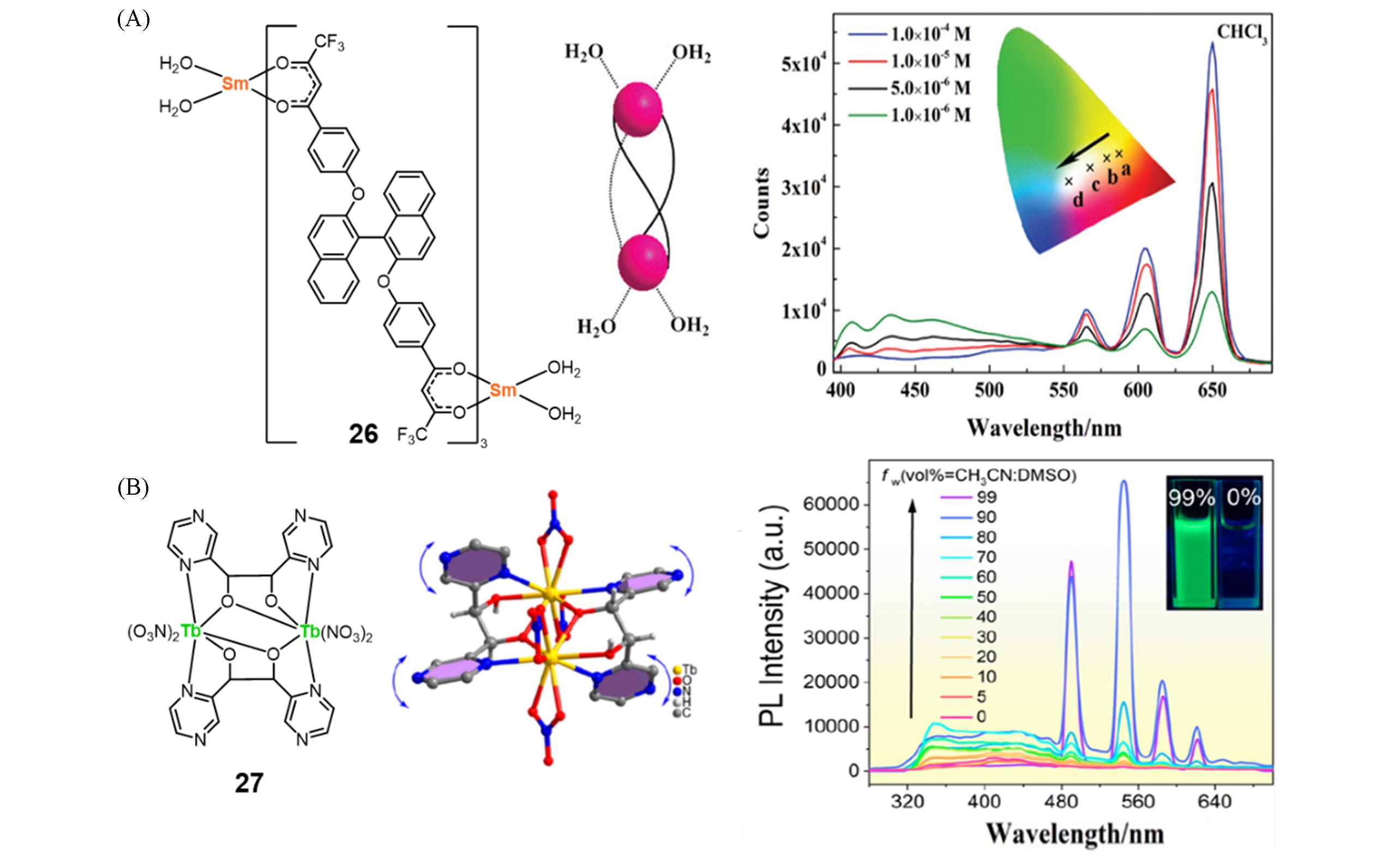
Fig.8 Molecular structure and schematic diagram of complex 26, PL spectra of complex 26 in THF at different concentrations(A)[40] and molecular structure and structural diagram of complex 27, PL spectra of complex 27 in DMSO with different fraction of CH3CN(B)[41](A) Copyright 2014, the Royal Society of Chemistry; (B) copyright 2023, American Chemical Society.
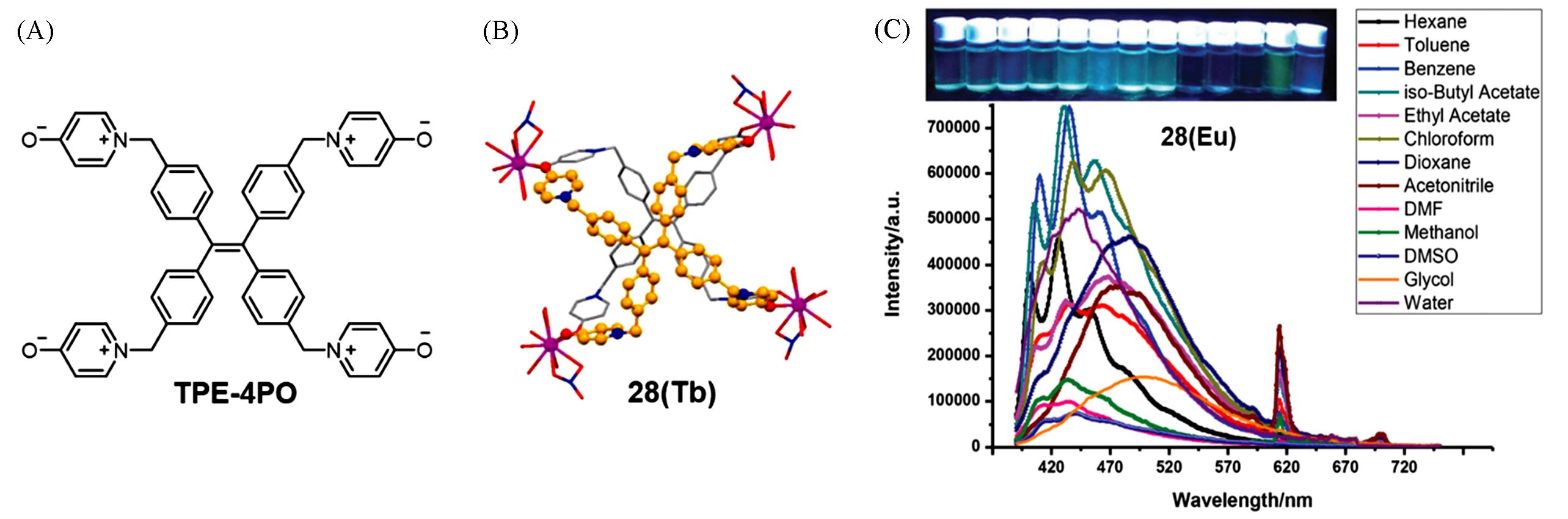
Fig.9 Molecular structure of ligand TPE⁃4PO(A), the coordination environment of Tb3+ ions and TPE⁃4PO ligands in the crystal of complex 28(Tb)(B) and fluorescent photographs and PL spectra of complex 28(Eu) in various solvents(C)[45]
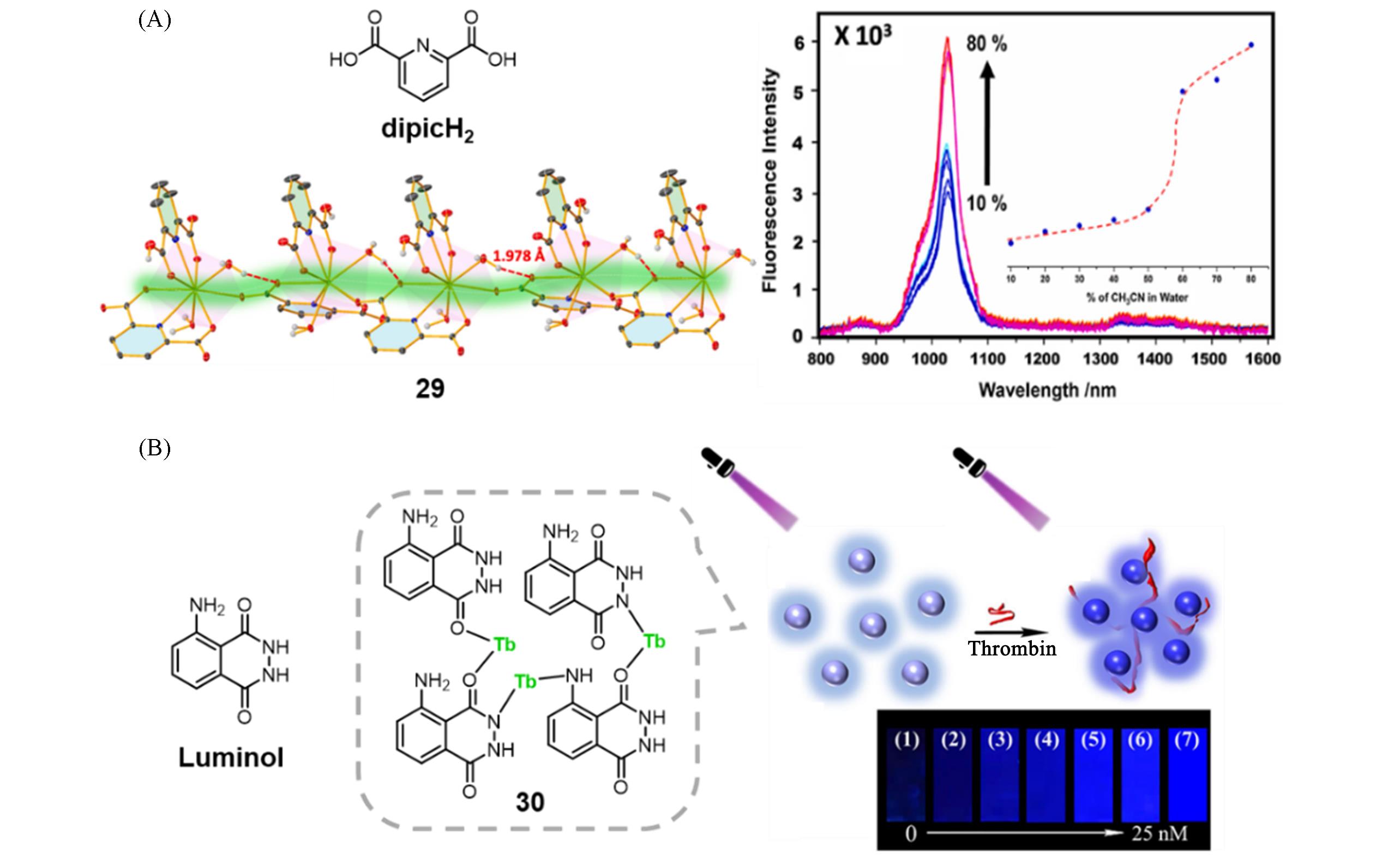
Fig.10 Molecular structure of ligand dipicH2 and crystal structure of polymer 29, PL spectra of polymer 29 in CH3CN with different fraction of water(A)[46] and molecular structure of ligand luminol and polymer 30, fluorescent photographs of the polymer 30 dispersed in Tris buffer in the presence of various concentrations of thrombin(B)[49](A) Copyright 2025, Elsevier Ltd.; (B) Copyright 2019, Springer Nature.
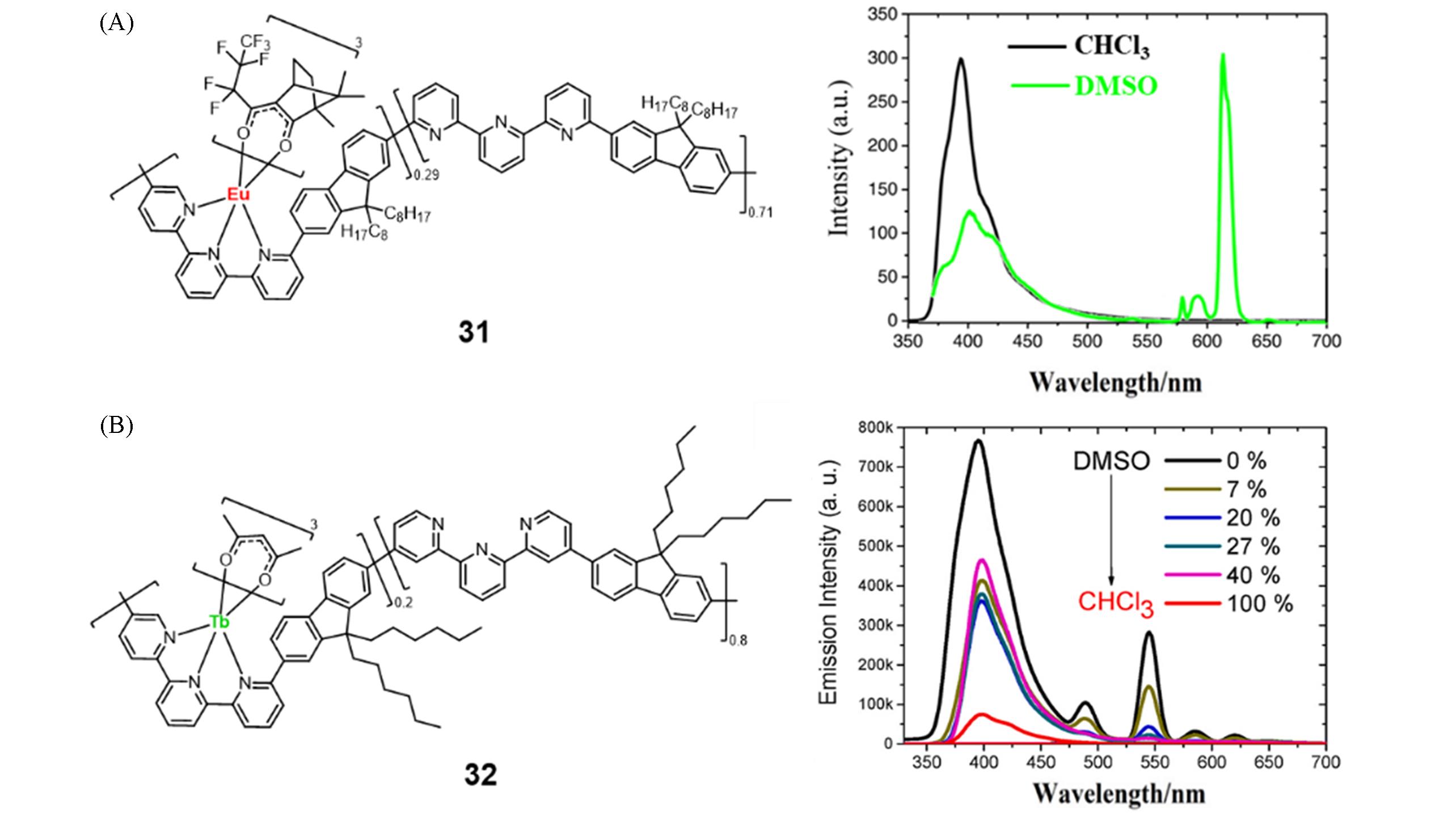
Fig.11 Molecular structure, PL spectra in chloroform and DMSO of metallopolymer 31(A)[50] and molecular structure of metallopolymer 32 and PL spectra of metallopolymer 32 in DMSO/chloroform mixture with different chloroform fractions(B)[51](A) Copyright 2020, Elsevier Ltd.; (B) copyright 2021, Elsevier Ltd.
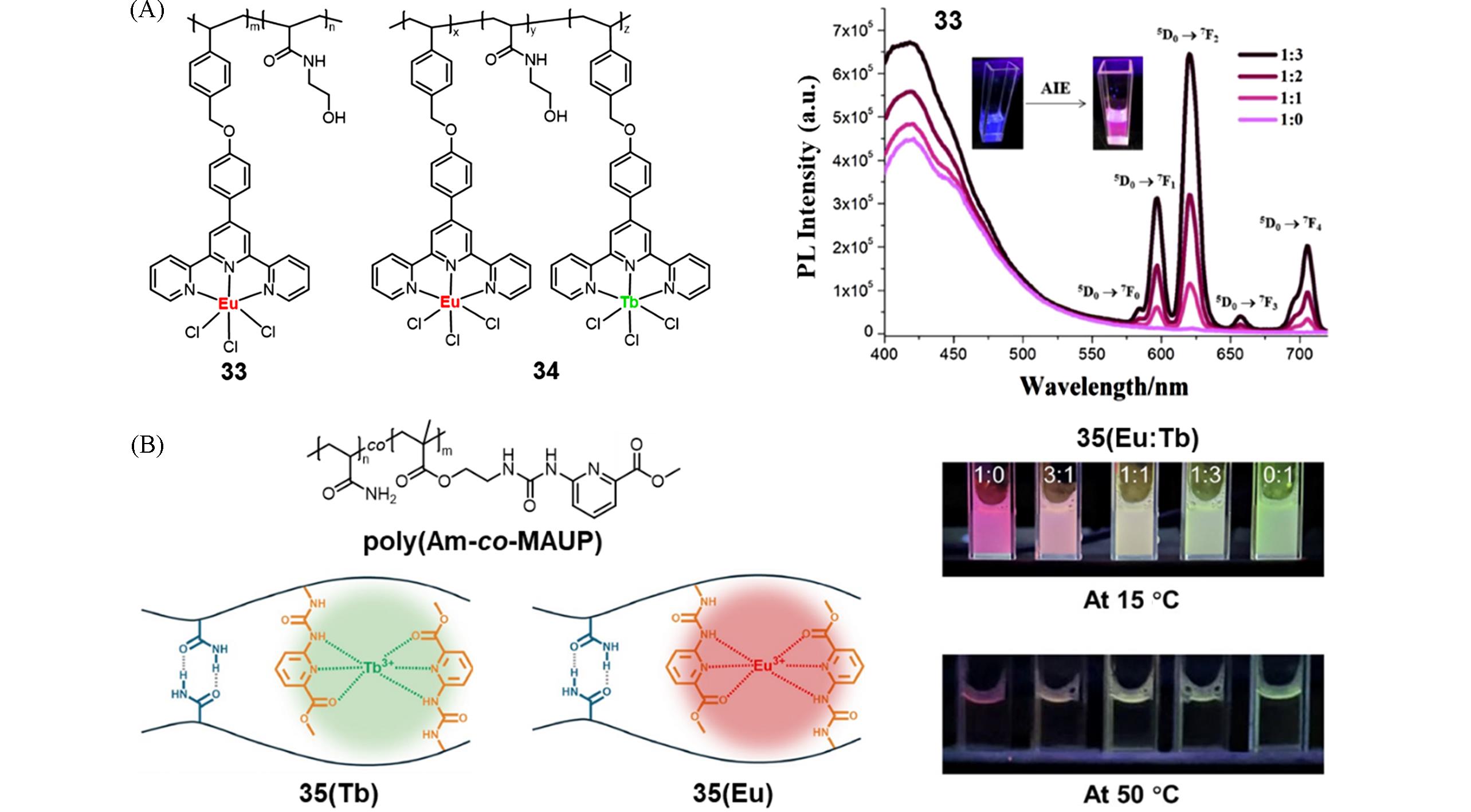
Fig.12 Molecular structure of polymers 33 and 34 and PL spectra in DMF with different fraction of 1,4⁃dioxane(A)[56] and molecular structure of poly(Am⁃co⁃MAUP) and schematic diagram of 35(Tb) and 35(Eu), fluorescent photographs of 35(Ln) consisting of different ratios of Eu3+ to Tb3+ at 15 ℃ and 50 ℃(B)[58](A) Copyright 2018, Elsevier Ltd.; (B) copyright 2025, Science China Press.
| System type | Structural flexibility | Representative applications |
|---|---|---|
| Mononuclear | High | Chemical sensing, bioimaging, and anti⁃counterfeiting |
| Dinuclear | Moderate | Solid⁃state lighting and stimulus⁃responses |
| Polynuclear | Low to moderate | Solid⁃state lighting, chemical sensing, and biosensing |
Table 1 Comparative summary of AIE lanthanide complexes with different number of Ln centers
| System type | Structural flexibility | Representative applications |
|---|---|---|
| Mononuclear | High | Chemical sensing, bioimaging, and anti⁃counterfeiting |
| Dinuclear | Moderate | Solid⁃state lighting and stimulus⁃responses |
| Polynuclear | Low to moderate | Solid⁃state lighting, chemical sensing, and biosensing |
| Ln3+ ion | Emission range(Luminescent color) | Characteristic emission peak/nm | Luminescence lifetime |
|---|---|---|---|
| Tb3+ | Visible(Green) | 545 | ms scale(0.5—2.0 ms) |
| Eu3+ | Visible(Red) | 613 | ms scale(0.2—1.0 ms) |
| Sm3+ | Visible(Orange) | 645 | μs scale(1—50 μs) |
| Yb3+ | NIR⁃II | 980 | μs scale(1—200 μs) |
| Nd3+ | NIR⁃II | 1064 | μs scale(<10 μs) |
Table 2 Comparative properties of Ln3+ ions in AIE lanthanide complexes
| Ln3+ ion | Emission range(Luminescent color) | Characteristic emission peak/nm | Luminescence lifetime |
|---|---|---|---|
| Tb3+ | Visible(Green) | 545 | ms scale(0.5—2.0 ms) |
| Eu3+ | Visible(Red) | 613 | ms scale(0.2—1.0 ms) |
| Sm3+ | Visible(Orange) | 645 | μs scale(1—50 μs) |
| Yb3+ | NIR⁃II | 980 | μs scale(1—200 μs) |
| Nd3+ | NIR⁃II | 1064 | μs scale(<10 μs) |
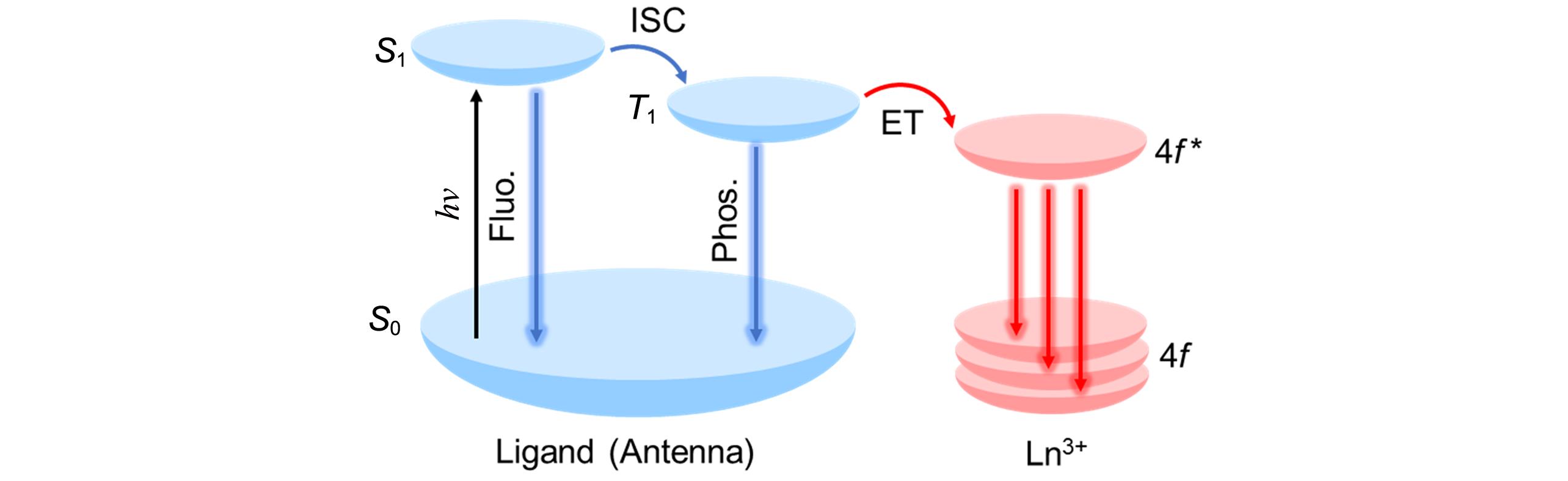
Fig.14 Energy level diagram illustrating the antenna effect and sequential energy transfer processes for lanthanide complexes with characteristic f⁃f electronic transitions
| Mechanism | Dominant motion/vibration targeted | Key trigger for AIE |
|---|---|---|
Restriction of intramolecular rotation(RIR) | Intramolecular rotation(flexible moieties) | Aggregation⁃induced steric hindrance/intermolecular interactions |
Restriction of intramolecular vibration(RIV) | Intramolecular vibration(ligand backbones) | Aggregation⁃induced conformational rigidity |
Electronic⁃vibrational decoupling(EVD) | Electronic⁃vibrational coupling | Concentration increase/solvent⁃induced ligand exchange |
Ln3+⁃coordination⁃induced aggregation | Cluster dispersion⁃aggregation transition | Ln3+⁃ligand coordination |
Table 3 Summary of core AIE mechanisms in lanthanide complexes
| Mechanism | Dominant motion/vibration targeted | Key trigger for AIE |
|---|---|---|
Restriction of intramolecular rotation(RIR) | Intramolecular rotation(flexible moieties) | Aggregation⁃induced steric hindrance/intermolecular interactions |
Restriction of intramolecular vibration(RIV) | Intramolecular vibration(ligand backbones) | Aggregation⁃induced conformational rigidity |
Electronic⁃vibrational decoupling(EVD) | Electronic⁃vibrational coupling | Concentration increase/solvent⁃induced ligand exchange |
Ln3+⁃coordination⁃induced aggregation | Cluster dispersion⁃aggregation transition | Ln3+⁃ligand coordination |
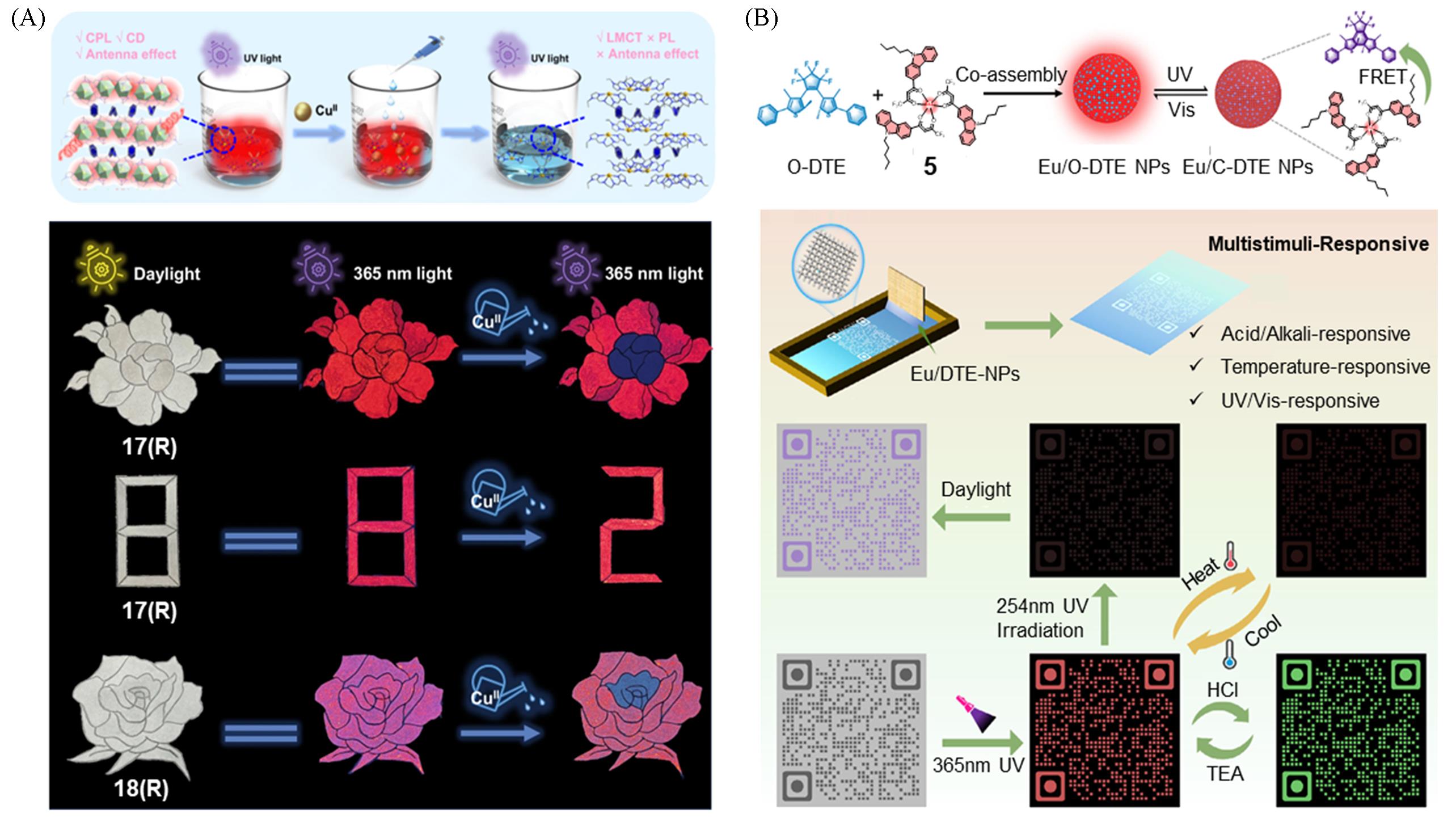
Fig.15 Schematic diagram of the response mechanism of the complexes 17(R, S) and 18(R, S) to copper ions and its real applications(A)[33] and schematic illustration of 5 NPs assemblies and Eu/DTE⁃NPs co⁃assemblies and application of Eu/DTE⁃NPs in multidimensional dynamic information encryption(B)[25](A) Copyright 2024, Springer Nature; (B) copyright 2025, John Wiley and Sons.
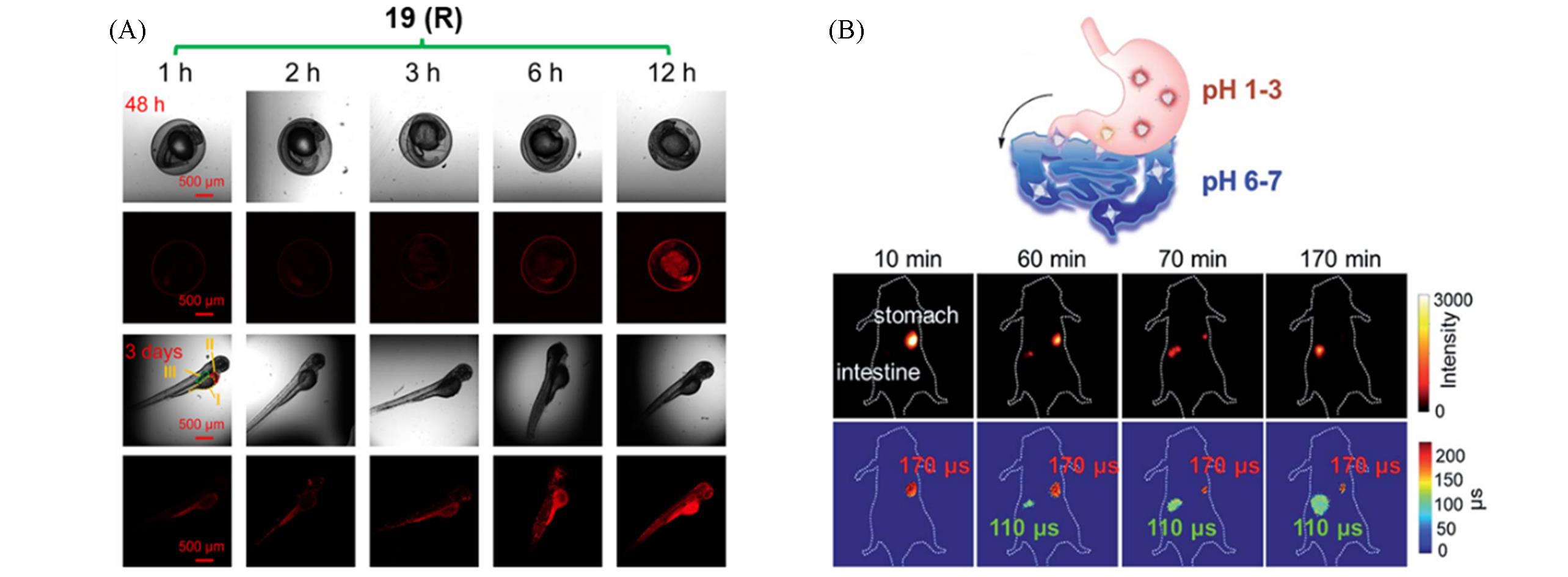
Fig.16 CLSM images and fluorescence intensity diagrams of zebrafish embryos(48 h after fertilization) and larvae(day 3) after incubation with complex 19(R)(A)[35] and schematic illustration of the metabolic process of complex 22 from the stomach to the intestine, and NIR fluorescence intensity imaging of complex 22(B)[37](A) Copyright 2025, Springer Nature; (B) copyright 2019, the Royal Society of Chemistry.
| [13] | Furet E., Costuas K., Rabiller P., Maury O., J. Am. Chem. Soc., 2008, 130(7), 2180—2183 |
| [14] | Thibon A., Pierre V. C., Anal. Bioanal. Chem., 2009, 394(1), 107—120 |
| [15] | Wang T., Wang S. F., Liu Z. Y., He Z. Y., Yu P., Zhao M. Y., Zhang H. X., Lu L. F., Wang Z. X., Wang Z. Y., Zhang W. A., Fan Y., Sun C. X., Zhao D. Y., Liu W. M., Bünzli J. C. G., Zhang F., Nat. Mater., 2021, 20(11), 1571—1578 |
| [16] | Xu J., Luo R., Luo Z. C., Xu J., Mu Z., Bian H. Y., Chan S., Y., Tan. B. Y. H., Chi D. Z., An Z. F., Xing G. C., Qin X., Gong C. Y., Wu Y. M., Liu X. G., Nat. Photon., 2025, 19(1), 71—78 |
| [17] | Andres J., Hersch R. D., Moser J. E., Chauvin A. S., Adv. Funct. Mater., 2014, 24(32), 5029—5036 |
| [18] | Yang Y. L., Hu X. M., Yang Z., Huang W., Adv. Funct. Mater., 2025, 35(2), 2412970 |
| [19] | Fernández⁃Fariña S., Kotova O., Donohoea S. R., Gunnlaugsson T., Chem. Soc. Rev., 2025, 54(23), 11226—11265 |
| [20] | Zhang Y., Jiao P. C., Xu H. B., Tang M. J., Yang X. P., Huang S. M., Deng J. G., Sci. Rep., 2015, 5(1), 9335 |
| [21] | Wu J. J., Zhao C., Zhu Z. H., Li X. L., Ashebr T. G., Tang J. K., Chem. Asian J., 2022, 17(23), e202200913 |
| [22] | Zhuo H., Guan D. B., He J. C., Xu H. B., Zeng M. H., Chem. Eur. J., 2021, 27(65), 16204—16211 |
| [23] | Su P. R., Liang L. J., Wang T., Zhou P. P., Cao J., Liu W. S., Tang Y., Chem. Eng. J., 2021, 413, 127408 |
| [24] | Wang S. Y., Wang L., Fang F., Ma X., Guo Y. Y., Wang R. D., Zhang S. S., Zhang Z., Du L., Zhao Q. H., Inorg. Chem. Front., 2023, 10(18), 5258—5269 |
| [25] | Liang L. J., Yang X., Yan X. Y., Kou Y., Zhang Y. H., Su P. R., Tang Y., Adv. Mater., 2026, 38(2), e14252 |
| [26] | de Sá G. F., Malta O. L., de Mello⁃Donegá C., Simas A. M., Longo R. L., Santa⁃Cruz P. A., da Silva E. F. Jr., Coord. Chem. Rev., 2000, 196(1), 165—195 |
| [27] | Kai T., Kishimoto M., Akita M., Yoshizawa M., Chem. Commun., 2018, 54(8), 956—959 |
| [28] | Xu M. J., Wang J. H., Liao Z. X., Liu J. Q., Yi H., Hao H. X., Li F., Xu S. Y., Liu F. F., Peng Y. L., Wang S. L., Cao X. D., Zhang P. F., Luminescence, 2025, 40(8), e70289 |
| [29] | Wong H. Y., Chan W. T. K., Law G. L., Molecules, 2019, 24(4), 662 |
| [30] | Su P. R., Wang T., Zhou P. P., Yang X. X., Feng X. X., Zhang M. N., Liang L. J., Tang Y., Yan C. H., Natl. Sci. Rev., 2022, 9(1), nwab016 |
| [31] | Chen F. F., Wang J. M., Xu W., Ren Z. Z., Peng G., Huang T., Zhao F., J. Alloy. Compd., 2025, 1010, 177421 |
| [32] | Zhang G. P., Zhu H. X., Chen M. J., Pietraszkiewicz M., Pietraszkiewicz O., Li H. G., Hao J. C., Chem. Eur. J., 2018, 24(59), 15912—15920 |
| [33] | Li Y. L. Wang H. L., Zhu Z. H., Wang Y. F., Liang F. P., Zou H. H., Nat. Commun., 2024, 15(1), 2896 |
| [34] | Wang H. L., Yu B., Liang F. P., Zou H. H., Chin. Chem. Lett., 2025, 111346 |
| [35] | Qin W. W., Yu B., Zhang G. H., Tang M. J., Zou H. H., Liang F. P., Zhu Z. H., Rare Met., 2025, 44(12), 10390—10403 |
| [36] | Yang S. S., Guo Z. P., Hu Z. Q., Guo D. C., Luminescence, 2021, 36(2), 306—315 |
| [37] | Ning Y. Y., Cheng S. M., Wang J. X., Liu Y. W., Feng W., Li F. Y., Zhang J. L., Chem. Sci., 2019, 10(15), 4227—4235 |
| [38] | Bodedla G. B., Zhu X., Wong W. Y., Aggregate, 2023, 4(3), e330 |
| [39] | Wang Y. F., Wang J. J., Zhang S. W., Tang N., Ou X. W., Jiang J. H., Ma F. L., Alam P., Qiu Z. J., Wang W. J., Zhao Z., Lam J. W. Y., Tang B. Z., ACS Nano, 2025, 19(27), 25042—25051 |
| [40] | Leng J. Q., Li H. F., Chen P., Sun W. B., Gao T., Yan P. F., Dalton Trans., 2014, 43(32), 12228—12235 |
| [41] | Ai J. F., Li Y. L., Wang H. L., Liang F. P., Zhu Z. H., Zou H. H., Inorg. Chem., 2023, 62(48), 19552—19564 |
| [42] | Nakai T., Shima K., Shoji S., Fushimi K., Hasegawa Y., Kitagawa Y., Front. Chem., 2023, 11, 1154012 |
| [43] | Pan J. K., Chen Z., Wang H., Li X. P., Yu X. J., Sci. China Mater., 2025, 68(10), 3485—3510 |
| [44] | Yang D. Q., Li H. M., Li H. R., Coord. Chem. Rev., 2024, 514, 215875 |
| [45] | Zhu Y. X., Wei Z. W., Pan M., Wang H. P., Zhang J. Y., Su C. Y., Dalton Trans., 2016, 45(3), 943—950 |
| [46] | Raghuvanshi K., Verma A., Sunkari S. S., J. Lumin., 2025, 283, 121284 |
| [47] | Tong Y. J., Yu L. D., Wu L. L., Cao S. P., Zhang L., Xia X. H., Qiu J. D., Chem. Commun., 2018, 54(54), 7487—7490 |
| [48] | Tong Y. J., Song A. M., Yu L. D., Liang R. P., Qiu J. D., Microchim. Acta, 2019, 187(1), 53 |
| [49] | Qi W. J., Zhao M. Y., Fu Y. L., He H. K., Tian X., Wu D., Zhang Y., Hu P. P., Dyes Pigm., 2020, 172, 107797 |
| [50] | Campos E. C. G., Turchetti D. A., Santana A. J., Domingues R. A., Duarte L., Atvars T. D. Z., Akcelrud L., Synth. Met., 2021, 273, 116686 |
| [51] | Campos E. C. G., Turchetti D. A., Zanlorenzi C., Domingues R. A, Duarte L. G. T. A., Atvars T. D. Z., Akcelrud L., Polymer, 2021, 229, 123990 |
| [52] | Campos E. C. G., Turchetti D. A., Domingues R. A., Akcelrud L. C., Synth. Met., 2025, 311, 117802 |
| [53] | Zhang S. X., Yin W. D., Yang Z. M., Shah I., Yang Y., Li Z., Zhang S. J., Zhang B., Lei Z. Q., Ma H. C., Anal. Chem., 2020, 92(1), 7808—7815 |
| [54] | Huang Y. J., Feng W. X., Zhou Z. P., Zheng H. Z., Zhao Y., Yan H. X., Lv X. Q., J. Mater. Chem. C, 2022, 10(19), 7586—7593 |
| [55] | Feng W. X., Huang Y. J., Zhao Y., Tian W., Yan H. X., ACS Appl. Mater. Interfaces, 2023, 15(13), 17211—17221 |
| [56] | Zhang Z., Chang H., Kang Y. F., Li X. P., Jiang H. E., Xue B. L., Wang Y. Y., Lv X. Q., Zhu X. J., Sensor. Actuat. B⁃Chem., 2019, 282, 999—1007 |
| [57] | Zhang Z., Chen Y. X., Chang H., Wang Y. Y., Li X. P., Zhu X. J., J. Mater. Chem. C, 2020, 8(6), 2205—2210 |
| [58] | Fan X. Z., Zhou Z. H., Luo M. Q., Banquy X., Zhang J. W., Zhu X. X., Zhang C. Z., Sci. China Chem., 2025, 68(11), 5949—5959 |
| [59] | You J. G., Lu C. Y., Kumar A. S. K., Tseng W. L., Nanoscale, 2018, 10(37), 17691—17698 |
| [60] | Ma F. H., Deng L., Wang T. T., Zhang A. M., Yang M. H., Li X. Q., Chen X., Microchim. Acta, 2023, 190(8), 291 |
| [61] | Ma J., Cong X., Ou K. D., Liao Y. G., Yang Y. J., Wang H., Sensor. Actuat. B⁃Chem., 2023, 390, 133904 |
| [62] | Huang X. M., Chen H. Y., Huang R., Shi Y. D., Ye R. H., Qiu B., Microchim. Acta, 2024, 191(1), 54 |
| [63] | Mu J., Zhang H. F., Huang Z. Z., Jia Q., Spectrochim. Acta A, 2023, 291, 122388 |
| [64] | Pan T. T., Zhou T., Tu Y. F., Yan J. L., Talanta, 2021, 227, 122197 |
| [65] | Miao W. J., Wang L., Liu Q., Guo S., Zhao L. Z., Peng J. J., Chem. Asian J., 2021, 16(3), 247—251 |
| [66] | Madni A., Zhang Y. C., Zhang L. Y., Qiang Y. N., Qi S. D., Zhai H. L., Food Compos. Anal., 2025, 148, 108429— |
| [67] | Saraci F., Quezada⁃Novoa V., Donnarumma P. R., Howarth A. J., Chem. Soc. Rev., 2020, 49(22), 7949—7977 |
| [68] | Xu H., Cheng P., Aggregate, 2024, 5(3), e518 |
| [69] | Yin H. Q., Wang X. Y., Yin X. B., J. Am. Chem. Soc., 2019, 141(38), 15166—15173 |
| [70] | Yang L., Dou Y., Qin L., Chen L. L., Xu M. Z., Kong C., Zhang D. P., Zhou Z., Wang S. N., Inorg. Chem., 2020, 59(22), 16644—16653 |
| [71] | Li G. Y., Tong C. L., Anal. Chim. Acta, 2020, 1133, 11—19 |
| [72] | Li Z. J., Jiang F. L., Yu M. X., Li S. C., Chen L., Hong M. C., Nat. Commun., 2022, 13(1), 2142 |
| [73] | Xu S. J., Xiong G., Zhang X. Y., Huang K., Qin D. B., Zhao B., Cryst. Growth Des., 2023, 23(6), 4214—4221 |
| [74] | Zhai X. Y., Kou Y., Liang L. J., Liang P. Y., Su P. R., Tang Y., Inorg. Chem., 2023, 62(45), 18533—18542 |
| [75] | Yan Z. W., Ling Y., Zeng Q., Sun Z., Li N. B., Luo H. Q., Sensor. Actuat. B⁃Chem., 2024, 417, 136169 |
| [76] | Li Y. L., Wang H. L., Xiao Z. X., Ai J. F., Liang F. P., Zhu Z. H., Zou H. H., ACS Appl. Mater. Interfaces, 2024, 16(45), 62301—62313 |
| [77] | Wang H. L., Li Y. H., Zou H. H., Liang F. P., Zhu Z. H., Adv. Mater., 2025, 37(29), 2502742 |
| [78] | Li Z. J., Zhang J. P., Wu Z. Y., Lei H., Zhao Y. F., Qi W. Q., Gao X., Jiang F. L., Liu Y. S., Chen L., Hong M. C., Adv. Sci., 2025, 12(4), e10147 |
| [79] | Li X. Q., Zhao J. W., Wang Y. L., Liu W., Zhao S. S., Chen X., Tian T. Y., Zhang H., Sun Q., Zhao Z., Inorg. Chem., 2025, 64(34), 17313—17321 |
| [80] | Zhang J., He B. Z., Hu Y. P., Alam P., Zhang H. K., Lam J. W. Y., Tang B. Z., Adv. Mater., 2021, 33(32), 2008071 |
| [1] | Luo J., Xie Z., Lam J. W. Y., Cheng L., Chen H., Qiu C., Kwok H. S., Zhan X., Liu Y., Zhu D., Tang B. Z., Chem. Commun., 2001, (18), 1740—1741 |
| [2] | Han P. B., Xia E. H., Qin A. J., Tang B. Z., Coord. Chem. Rev., 2022, 473, 214843 |
| [3] | Yang S. Y., Zhang L., Kong F. C., Chen Y. Y., Li W. J., Wang F., Liu C., He X., Xiao X. D., Wang J., Sun J. W., Chow P. C. Y., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem, 2025, 11(5), 102534 |
| [4] | Yan D., Wang D., Tang B. Z., Nat. Rev. Bioeng., 2025, 3(11), 976—991 |
| [5] | Chen W. Z., Guan H. T., Lu Y. F., Zeng G. H., Gu D., Guo K. Y., Jiang C., Liu H. X., Aggregate, 2025, 6(4), e70008 |
| [6] | Mei J., Hong Y. N., Lam J. W. Y., Qin A. J., Tang Y. H., Tang B. Z., Adv. Mater., 2014, 26(31), 5429—5479 |
| [7] | Mei J., Leung N. L. C., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Rev., 2015, 115(21), 11718—11940 |
| [8] | Yang S. Y., Chen Y. Y., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Soc. Rev., 2024, 53(11), 5366—5393 |
| [9] | Pei Y., Sun Y., Huang M. J., Zhang Z. J., Yan D. Y., Cui J., Zhu D. X., Zeng Z. B., Wang D., Tang B. Z., Biosensors, 2022, 12(12), 1104 |
| [10] | Mauro M., Cebrián C., Isr. J. Chem., 2018, 58(8), 901—914 |
| [11] | Sathish V., Ramdass A., Thanasekaran P., Lu K. L., Rajagopal S., J. Photoch. Photobio. C, 2015, 23, 25—44 |
| [12] | Wang F., Liu X. G., Chem. Soc. Rev., 2009, 38(4), 976—989 |
| [1] | WU Rui, LI Zheng, LI Qi, SHI Jiajun, ZHAO Yan, FENG Weixu, YAN Hongxia. Recent Progress on Unconventional Hyperbranched Luminescent Polymers Containing Si, P and B [J]. Chem. J. Chinese Universities, 2026, 47(5): 20250411. |
| [2] | LI Weijian, XU Xiaoqin, WANG Wei, YANG Haibo. Recent Advances in AIE-active Dendrimers [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260003. |
| [3] | YIN Shiqi, ZHENG Zhigang, HE Xintong, WANG Shimin, GU Xinggui, WANG Erjing. From Protogenesis to Functionalization: Luminescence, Modification and Application of AIE⁃active Natural Products [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260042. |
| [4] | JIAO Haili, ZHENG Xiaoyan. Progress on Theoretical Study of Organic Luminescence Enhancement Induced by Confined Environments [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260060. |
| [5] | ZENG Xianping, QIN Yi, WANG Dong. Aggregation-induced Emission Probes for Imaging-guided Surgery [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260066. |
| [6] | ZHAI Zhe, LIU Leijing, TIAN Wenjing. Molecular Design of Aggregation-induced Emission Probes and Their Applications in Organelle Imaging [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260078. |
| [7] | ZHU Gaohua, SHU Ju, GENG Jiangtao, MA Fulong, XIONG Linghong, HE Xuewen. In situ Activating NIR-II AIE Probe for Highly Sensitive Hydrogen Peroxide Imaging in Tumor [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260058. |
| [8] | PAN Jingke, QI Xin, ZHANG Lulu, WANG Beibei, LU Chao. Synthesis and Application of Aggregation-induced Emission Supramolecular Polymers Constructed by Host-guest Interaction [J]. Chem. J. Chinese Universities, 2026, 47(5): 20260130. |
| [9] | CHENG Jianshuo, YE Wenyan, ZHOU Lulu, LIU Mouwei, LI Zhongyu, TANG Ziran, YU Wanting, ZHU Liangliang. Photoexcitation-induced Biomacromolecular Self-assembly [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250398. |
| [10] | GAO Xin, QING Jia, HU Yichen, SHANGGUAN Zhichun, LIANG Tongling, ZHOU Yongsheng, ZHANG Guanxin, ZHANG Deqing. Novel AIE Fluorescent Probes for Ultrahigh Sensitivity and High Photostability in Lipid Droplets Imaging [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250410. |
| [11] | ZHANG Yangdaiyi, SHAO Yan, JIANG Shimei. Multi-responsive Hydrogel Featuring Synergistic Regulation of AIE and Mechanical Behaviors via Dynamic Hydrogen Bonding Network [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250381. |
| [12] | MA Huan, DONG Shilong, YANG Juncheng, ZHU Haitao, FENG Haitao. Chiral AIEgens Based on Calix[4]arene for Enantioselective Recognition of Acids and Amino Acids [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260049. |
| [13] | LI Yuting, LUO Liang. Cutting-edge Advances in Raman Imaging Technology and Its Interdisciplinary Research with Aggregate Science [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260008. |
| [14] | FANG Jinyu, HUANG Hanwei, SONG Hang, WU Qian, ZHAO Zheng, TANG Ben Zhong. Intelligent Response, Precision Activation: Novel Theranostic Strategies of Enzyme-responsive Aggregation-induced Emission Materials in Biomedicine [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260009. |
| [15] | SUN Yan, ZHU Dongxia. Construction of Near-infrared Triggered Organic Photosensitive Materials and Their Applications in Disease Treatment [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260002. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
