

Chem. J. Chinese Universities ›› 2026, Vol. 47 ›› Issue (4): 20260049.doi: 10.7503/cjcu20260049
• Article • Previous Articles
MA Huan, DONG Shilong, YANG Juncheng( ), ZHU Haitao(
), ZHU Haitao( ), FENG Haitao
), FENG Haitao
Received:2026-01-27
Online:2026-04-10
Published:2026-03-10
Contact:
YANG Juncheng, ZHU Haitao
E-mail:yangjuncheng@bjwlxy.edu.cn;zhuht@bjwlxy.edu.cn
Supported by:CLC Number:
TrendMD:
MA Huan, DONG Shilong, YANG Juncheng, ZHU Haitao, FENG Haitao. Chiral AIEgens Based on Calix[4]arene for Enantioselective Recognition of Acids and Amino Acids[J]. Chem. J. Chinese Universities, 2026, 47(4): 20260049.
| Entry | Analytes | Structure | I1/I2 | State | Solvent |
|---|---|---|---|---|---|
| 1 | Serine | 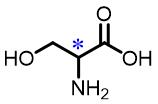 | 42.0(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 2 | Valine | 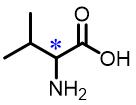 | 13.5(D/L) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 3 | Histidine | 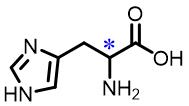 | 12.0(L/D) | Pre/Sol | 4.35×10-4 mol/L in THF/H2O (volume ratio 2∶5) |
| 4 | Arginine | 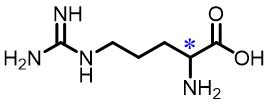 | 4.6(L/D) | Pre/Sol | 3.85×10-4 mol/L in THF/H2O (volume ratio 19∶50) |
| 5 | Proline | 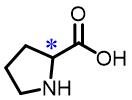 | 2.7(D/L) | Pre/Sol | 4.35×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 6 | N⁃Boc⁃Phenylalanine | 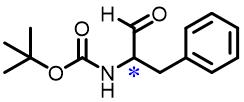 | 19.6(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 7 | N⁃Boc⁃Alanine | 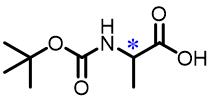 | 32.0(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 8 | N⁃Boc⁃Methionine | 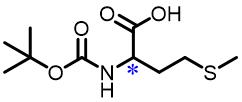 | 5.1(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 9 | Mandelic acid | 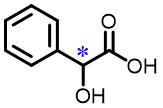 | 28.9(S/R) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 10 | Camphor sulfonic acid | 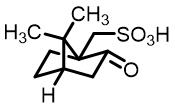 | 20.0(R/S) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 11 | 2⁃Chloromandelic acid | 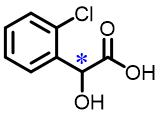 | 3.9(R/S) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 12 | Tartaric acid | 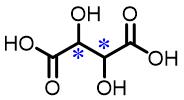 | 2.1(L/D) | Pre/Sol | 4.63×10-4 mol/L in THF/H2O (volume ratio 7∶20) |
Table 1 Enantioselective recognition of chiral acids and amino acids by R/S-9*
| Entry | Analytes | Structure | I1/I2 | State | Solvent |
|---|---|---|---|---|---|
| 1 | Serine |  | 42.0(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 2 | Valine |  | 13.5(D/L) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 3 | Histidine |  | 12.0(L/D) | Pre/Sol | 4.35×10-4 mol/L in THF/H2O (volume ratio 2∶5) |
| 4 | Arginine |  | 4.6(L/D) | Pre/Sol | 3.85×10-4 mol/L in THF/H2O (volume ratio 19∶50) |
| 5 | Proline |  | 2.7(D/L) | Pre/Sol | 4.35×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 6 | N⁃Boc⁃Phenylalanine |  | 19.6(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 7 | N⁃Boc⁃Alanine |  | 32.0(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 8 | N⁃Boc⁃Methionine |  | 5.1(L/D) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 9 | Mandelic acid |  | 28.9(S/R) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 10 | Camphor sulfonic acid |  | 20.0(R/S) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 11 | 2⁃Chloromandelic acid |  | 3.9(R/S) | Pre/Sol | 4.54×10-4 mol/L in THF/H2O (volume ratio 9∶25) |
| 12 | Tartaric acid |  | 2.1(L/D) | Pre/Sol | 4.63×10-4 mol/L in THF/H2O (volume ratio 7∶20) |
| [1] | Hu M., Feng H. T., Yuan Y. X., Zheng Y. S., Tang B. Z., Coord. Chem. Rev., 2020, 416, 213329 |
| [2] | Calcaterra A., D’Acquarica I., J. Pharm. Biomed. Anal., 2018, 147, 323―340 |
| [3] | Alvarez⁃Rivera G., Bueno M., Ballesteros⁃Vivas D., Cifuentes A., Trend. Anal. Chem., 2020, 123, 115761 |
| [4] | Ito T., Handa H., Congenit. Anom., 2012, 52(1), 1―7 |
| [5] | Saha D., Kharbanda A., Yan W., Lakkaniga N. R., Frett B., Li H. Y., J. Med. Chem., 2019, 63(2), 441―469 |
| [6] | Efimov A. V., Biochemistry(Moscow), 2018, 83, 103―110 |
| [7] | Tan X. P., Wu H. W., Wang D. D., Zhang J., Zhang J. H., Chem. J. Chinese Universities, 2025, 46(2), 20240352 |
| 谭晓萍, 吴宏伟, 王丹丹, 张金, 章俊辉. 高等学校化学学报, 2025, 46(2), 20240352 | |
| [8] | Bai L., Huo S. H., Chen J., Lu X. Q., Chem. J. Chinese Universities, 2019, 40(1), 41―46 |
| 白蕾, 霍淑慧, 陈晶, 卢小泉. 高等学校化学学报, 2019, 40(1), 41―46 | |
| [9] | Xu H., Lin Z., Bai J., Guo Y., Ma S., ACS Catal., 2023, 14(1), 262―270 |
| [10] | Ye F. Y., Hu M., Zheng Y. S., ACS Appl. Mater. Int., 2023, 15(35), 42056―42065 |
| [11] | Qi C. X, Wei K. Y., Li Q. Y., Li Y., Su X. L, Yang J. C., Tian J., Chen P., Feng H. T., Tang B. Z., Aggregate, 2022, 4(3), e299 |
| [12] | Wang X., Xiang S., Qi C. X., Chen M., Su X., Yang J. C., Tian J., Feng H. T., Tang B. Z., ACS Nano, 2022, 16(5), 8223―8232 |
| [13] | Liu J., Su X., Xu Y., Tang W., Gong J., Sensor Actuat. B-Chem., 2025, 429, 137277 |
| [14] | Zhang Y., Wang H. Y., He X. W., Li W. Y., Zhang Y. K., J. Hazard. Mater., 2021, 412, 125249 |
| [15] | Qi C. X., Li Q. Y., Wei K. Y., Zhao W., Zhang W., Chen M. M., Zhang X., Feng H. T., Dyes Pigments, 2025, 239, 112781 |
| [16] | Chen M., Qi C. X., Yin Y. T., Lv P., Xiang S., Tian J., Feng Z. J., Feng H. T., Tang B. Z., Org. Chem. Front., 2022, 9(19), 5160―5167 |
| [17] | Wang J., Meng Q., Yang Y., Zhong S., Zhang R., Fang Y., Gao Y., Cui X., ACS Sens., 2022, 7(9), 2521―2536 |
| [18] | Chen P., Lv P., Guo C. S., Wang R. P., Su X., Feng H. T., Tang B. Z., Chin. Chem. Lett., 2023, 34(6), 108041 |
| [19] | Feng H. T., Yuan Y. X., Xiong J. B., Zheng Y. S., Tang B. Z., Chem. Soc. Rev., 2018, 47(19), 7452―7476 |
| [20] | Qu H., Wang Y., Li Z., Wang X., Fang H., Tian Z., Cao X., J. Am. Chem. Soc., 2017, 139(50), 18142―18145 |
| [21] | Zhang X., Yu Q., Chen S., Dai Z., New J. Chem., 2018, 42(6), 4045―4051 |
| [22] | Li Y., Yu K., Xu Z., Zeng J., Wei J., Jiang H., Zhu Y., Gu S., Ma X., Nat. Commun., 2026, 17(1), 96 |
| [23] | Sun M. X., Ni C. Z., Zhang F. Q., Zhu Y. Y., Zeng J., Gu S. X., Chin. Chem. Lett., 2023, 34(12), 108345 |
| [24] | Zhao L., Cheng L., Yang Y., Wang P., Tian P., Yang T., Nian H., Cao L., Angew. Chem. Int. Ed., 2024, 63(25), e202405150 |
| [25] | Wang Y., Liao Q., Feng Y., Meng Q., J. Mol. Struct., 2024, 1304, 137695 |
| [26] | Duan H., Yang T., Li Q., Cao F., Wang P., Cao L., Chin. Chem. Lett., 2024, 35(1), 108878 |
| [27] | Chen L., Chen Y., Zhang Y., Liu Y., Angew. Chem. Inter. Ed., 2021, 60(14), 7654―7658 |
| [28] | Che Y., Li K., Xie C., Sun F., Yuan Y., Zhao X., Zhao C. X., Chang G., Adv. Funct. Mater., 2025, 35(36), 2506753 |
| [29] | Xiang S., Lv P., Guo C., Qi C. X., Yang J. C., Tian J., Yang D. S., Feng H. T., Tang B. Z., Chem. Commun., 2021, 57(98), 13321―13324 |
| [30] | Yang F. M., Luo J., Chen Y. C., Chung W. S., Synthesis, 2023, 55(17), 2786―2796 |
| [31] | Xue Y., Zeng Y., Shi Y., Tian Y., Sun C., Zhang N., Zheng X., Chen P., Sci. China Chem., 2025, 68(7), 3100―3107 |
| [32] | Liang B., Cheng Y., Liu X., Jia L., Wei X., Zheng Q., Wang P., Xia D., Yan X., Sci. China Chem., 2024, 67(10), 3373―3381 |
| [33] | Su T., Yang M., Wang S., Song Y. Y., Gao Z., Zhao C., Anal. Chem., 2025, 97(39), 21660―21667 |
| [34] | Li D., Yu X., Chen S., Fan C., Chi W., Dong Q., Zeng C., Talanta, 2025, 293, 128023 |
| [35] | Zhang Y. X., Zhang F. Q., Peng A. P., Jiang T., Meng Y. X., Li Y., Gu S. X., Zhu Y. Y., Chin. Chem. Lett., 2026, 37(1), 111500 |
| [36] | Zhang L., Zhang K., Wu M., Ding J., Feng M., Li X., Yang S., Ma H., Anal. Chem., 2025, 97(6), 3344―3351 |
| [37] | Yang J. C., Dong S. L., Ding Y. C., Qi C., Feng H. T., Chem. Commun., 2025, 61(53), 9654―9657 |
| [1] | YANG Zhan, DENG Huangjun, CHI Zhenguo. Research Progress on Hydrogen-bonded Organic Frameworks with Aggregation-induced Emission [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260012. |
| [2] | LI Yin, TANG Ruilin, QU Chao, CHENG Lianghui, HU Yuxi, WU Yuxiang, WANG Zhiming. Ionization Strategy for the Preparation of a Water-soluble Maleic Anhydride-based Photosensitive Probe and Its Application in High-efficiency Antibacterial Therapy [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250413. |
| [3] | REN Aocheng, LI Qingyun, JI Xiaofan. Fluorescent Supramolecular Polymer Networks [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250390. |
| [4] | SUN Yan, ZHU Dongxia. Construction of Near-infrared Triggered Organic Photosensitive Materials and Their Applications in Disease Treatment [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260002. |
| [5] | LI Yuting, LUO Liang. Cutting-edge Advances in Raman Imaging Technology and Its Interdisciplinary Research with Aggregate Science [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260008. |
| [6] | FANG Jinyu, HUANG Hanwei, SONG Hang, WU Qian, ZHAO Zheng, TANG Ben Zhong. Intelligent Response, Precision Activation: Novel Theranostic Strategies of Enzyme-responsive Aggregation-induced Emission Materials in Biomedicine [J]. Chem. J. Chinese Universities, 2026, 47(4): 20260009. |
| [7] | CHENG Jianshuo, YE Wenyan, ZHOU Lulu, LIU Mouwei, LI Zhongyu, TANG Ziran, YU Wanting, ZHU Liangliang. Photoexcitation-induced Biomacromolecular Self-assembly [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250398. |
| [8] | GAO Xin, QING Jia, HU Yichen, SHANGGUAN Zhichun, LIANG Tongling, ZHOU Yongsheng, ZHANG Guanxin, ZHANG Deqing. Novel AIE Fluorescent Probes for Ultrahigh Sensitivity and High Photostability in Lipid Droplets Imaging [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250410. |
| [9] | WU Zeyi, SI Wenni, QI Chunxuan, LI Shuo, FENG Haitao. Construction and Enantiorecognition Property of Red Emission Chiral Probes Based on Triphenylamine [J]. Chem. J. Chinese Universities, 2026, 47(4): 208. |
| [10] | ZHANG Yangdaiyi, SHAO Yan, JIANG Shimei. Multi-responsive Hydrogel Featuring Synergistic Regulation of AIE and Mechanical Behaviors via Dynamic Hydrogen Bonding Network [J]. Chem. J. Chinese Universities, 2026, 47(4): 20250381. |
| [11] | TAN Xiaoping, WU Hongwei, WANG Dandan, ZHANG Jin, ZHANG Junhui. Chiral Gold Nanoprobes for Colorimetric Detection of the Drug Enantiomers [J]. Chem. J. Chinese Universities, 2025, 46(2): 20240352. |
| [12] | LIU Kejun, WEI Ruiqi, ZHOU Ruiyuan, SHEN Ripei, HAN Jie. Synthesis, Photochromism and Fluorescence of Naphthopyran-tetraphenylethylene Hybrid [J]. Chem. J. Chinese Universities, 2024, 45(9): 20240201. |
| [13] | DONG Yiran, LI Fengjiao, MIAO Jinling, NIE Yong, LI Tianrui, XU Chunyue, LIU Wei, LIU Guangning, JIANG Xuchuan. Synthesis, Photoluminescence and Stimulus-responsiveness Properties of Bridged Fluorinated Tetraphenylethene Compounds [J]. Chem. J. Chinese Universities, 2024, 45(7): 20240051. |
| [14] | WU Zexin, ZHU Yuanjie, WANG Hongzhong, WANG Junan, HE Ying. Methyl-modified Carbazole/Diphenyl Sulfone-based AIE-TADF Blue Emitter and Its OLEDs [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220371. |
| [15] | LIU Wei, YAO Wei, ZHOU Mingming, YOU Qi, NIE Yong, JIANG Xuchuan. Synthesis, Aggregation-induced Emission and Piezofluorochromic Properties of 9,10-Bis(N-phenylindole-3-vinyl)anthracene [J]. Chem. J. Chinese Universities, 2021, 42(8): 2668. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||