

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (11): 2015.doi: 10.7503/cjcu20170159
• Physical Chemistry • Previous Articles Next Articles
Received:2017-03-20
Online:2017-11-10
Published:2017-09-30
Contact:
HU Jiawen
E-mail:jwhu@hnu.edu.cn
Supported by:CLC Number:
TrendMD:
SHEN Xiaohua, HU Jiawen. Synthesis of Polystyrene/silver Nanoparticels Composite Spheres and Their Catalytical Application†[J]. Chem. J. Chinese Universities, 2017, 38(11): 2015.
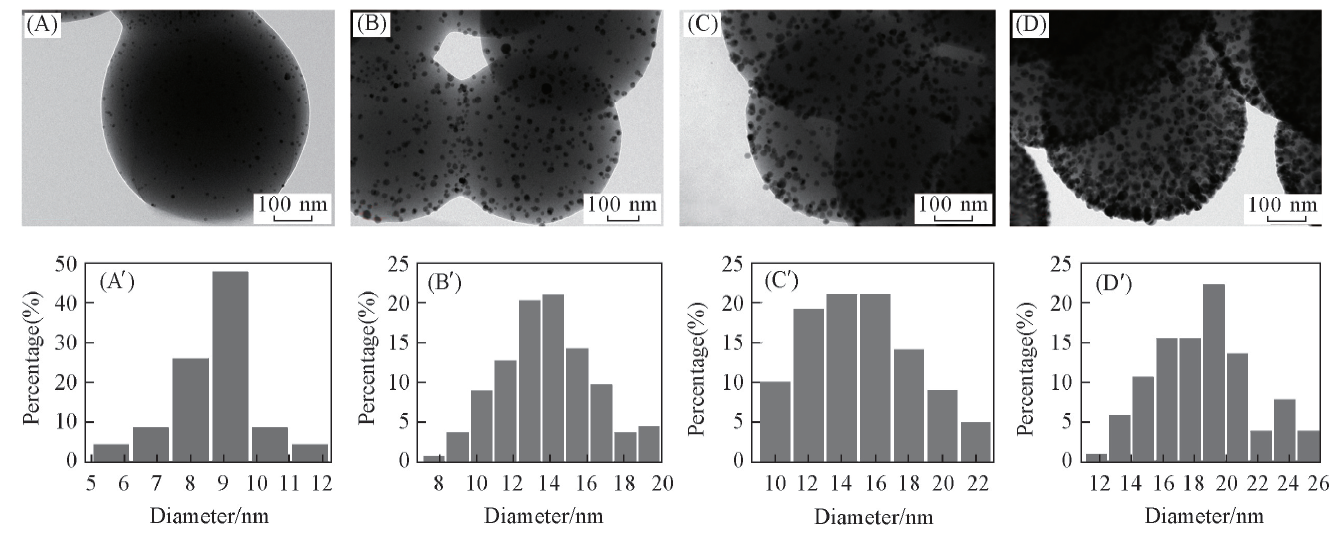
Fig.4 TEM images(A—D) for the PS/Ag NPs composite spheres prepared from 0.29 mol/L(A, A'), 0.58 mol/L(B, B'), 0.87 mol/L(C, C') and 1.16 mol/L(D, D') [Ag(NH3)2]+ solutions and corresponding size distributions(A'—D') of Ag NPs on the PS spheres
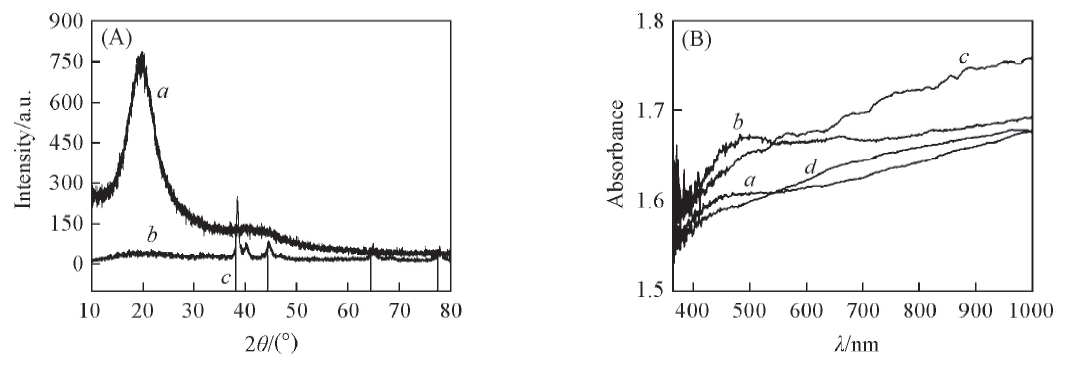
Fig.5 XRD patterns(A) for PS spheres(a), PS/Ag NPs composite spheres(b) and standard pattern for Ag(c) and UV-Vis spectra(B) of PS/Ag NPs composite crospheres prepared from 0.29 mol/L(a), 0.58 mol/L(b), 0.87 mol/L(c) and 1.16 mol/L(d) [Ag(NH3)2]+ solutions
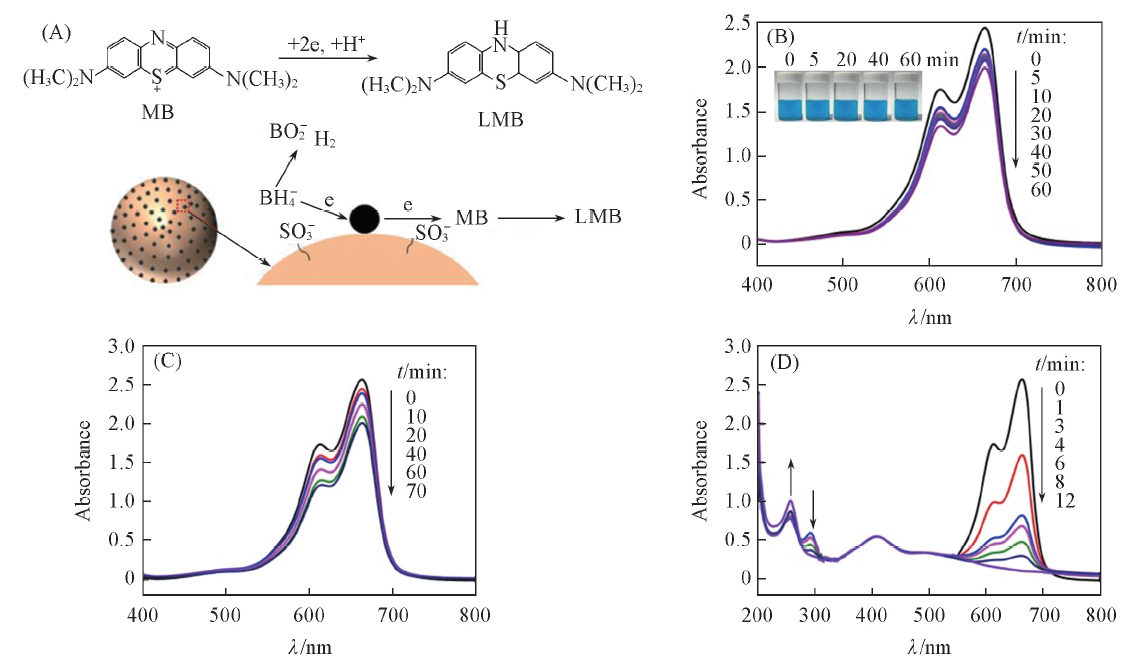
Fig.6 Schematic showing the reduction mechanism of MB on the PS/Ag NPs composite spheres(A) and time-dependent UV-Vis spectra for MB solution(0.04 mmol/L) without catalyst(B) and in the presence of sulfonated PS spheres(C) and pure Ag NPs(D)
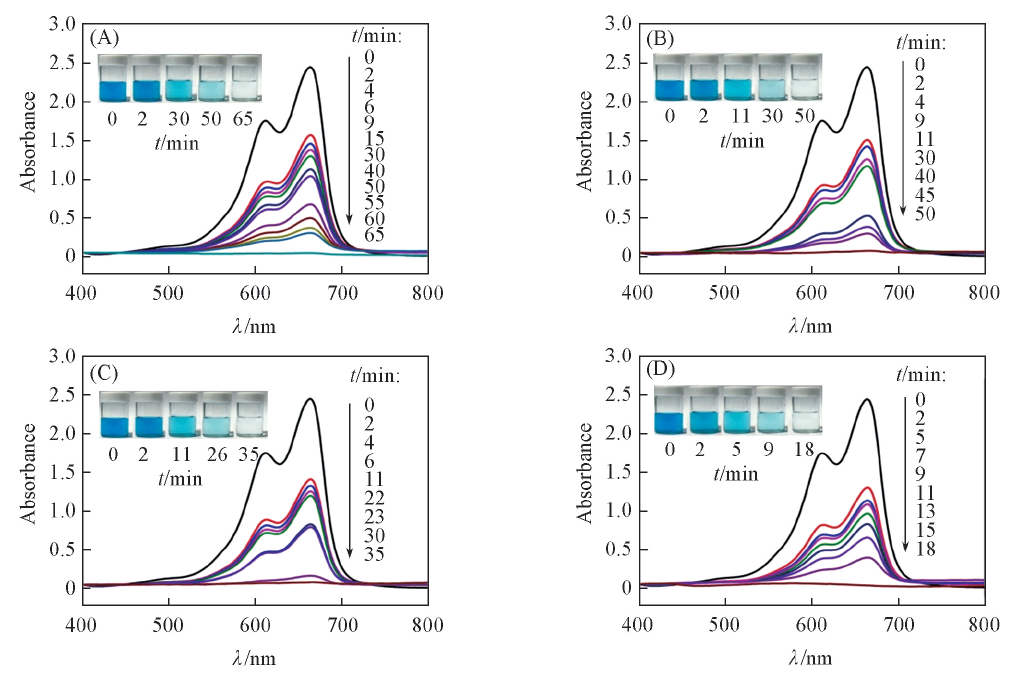
Fig.7 Time-dependent UV-Vis spectra for MB solution(0.04 mmol/L) in the presence of PS/Ag NPs composite spheres prepared from 0.29(A), 0.58(B), 0.87(C) and 1.16 mol/L(D) [Ag(NH3)2]+ solutions
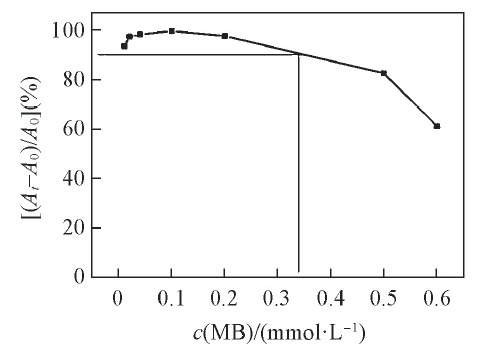
Fig.8 Conversion efficiency of MB solution with different initial concentration in the presence of PS/Ag NPs composite(1.1 mg, prepared from 1.16 mol/L [Ag(NH3)2]+ solution)
| Material | Temperature/℃ | Ag NPs diameter/nm | k/(s-1·mg-1) | Ref. |
|---|---|---|---|---|
| Poly(S-co-HEA)/Ag NPs | r. t. | 23.0 | 6.60×10-4 | [ |
| Fe3O4@polydopamine-Ag microspheres | 25 | 25.0 | 1.43×10-3 | [ |
| Ag NPs on Fe3O4@C nanocomposites | 25 | 10.0 | 5.67×10-4 | [ |
| LCPS/Ag NPs | 25 | 40.0 | 1.62×10-3 | [ |
| P(St-NaSS)/Ag NPs | 25 | 19.2 | 2.60×10-3 | [ |
| PS/Ag NPs composite spheres | 25 | 18.5 | 2.42×10-3 | This work |
Table 1 Comparison of normalized kinetic constant of different catalysts used for the conversion of MB
| Material | Temperature/℃ | Ag NPs diameter/nm | k/(s-1·mg-1) | Ref. |
|---|---|---|---|---|
| Poly(S-co-HEA)/Ag NPs | r. t. | 23.0 | 6.60×10-4 | [ |
| Fe3O4@polydopamine-Ag microspheres | 25 | 25.0 | 1.43×10-3 | [ |
| Ag NPs on Fe3O4@C nanocomposites | 25 | 10.0 | 5.67×10-4 | [ |
| LCPS/Ag NPs | 25 | 40.0 | 1.62×10-3 | [ |
| P(St-NaSS)/Ag NPs | 25 | 19.2 | 2.60×10-3 | [ |
| PS/Ag NPs composite spheres | 25 | 18.5 | 2.42×10-3 | This work |
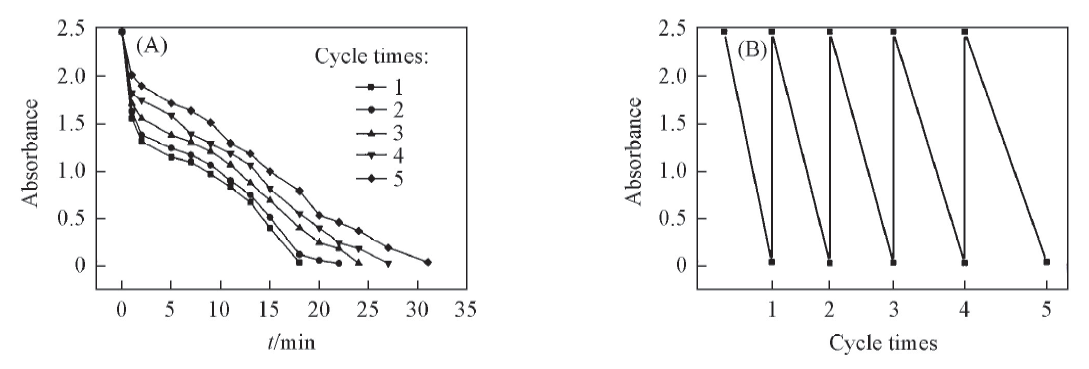
Fig.10 Covertic degradation kinetics for MB solution in five cycles with the presence of PS/Ag NPs composite spheres(prepared from 1.16 mol/L [Ag(NH3)2]+ solution)(A) and absorbance intensity variation for each cycle(B)
| [1] | Liu X. H., Cao Y. Y., Peng H. Y., Qian H. S., Yang X. Z., Zhang H. B., Cryst. Eng. Comm., 2014, 16(12), 2365 |
| [2] | Teng X. B., Zhang C. X., Zhang Y., Chem. J. Chinese Universities,2010, 31(8), 1509-1520 |
| (滕小波, 张春霞, 张颖. 高等学校化学学报, 2016, 37(8), 1509-1520 | |
| [3] | Tian C., Wang E., Kang Z., Mao B., Zhang C., Lan Y., Wang C., Song Y., J. Solid State Chem., 2006, 179(11), 3270-3276 |
| [4] | Zhang N., Yu X., Hu J., Xue F., Ding E., RSC Adv., 2013, 3(33), 13740 |
| [5] | Kwan K, Lee H. B., Han K. P., Ksed S. S., J. Colloid Interface Sci., 2008, 318, 195-201 |
| [6] | Ma Y., Zhang Q., Appl. Surf. Sci., 2012, 258(19), 7774-7780 |
| [7] | Cai W., Wang W., Yang Y., Ren G., Chen T., RSC Adv., 2014, 4(5), 2295-2299 |
| [8] | Cassagneau T., Caruso F., Adv. Mater., 2002, 10, 732-736 |
| [9] | Siiman O., Burshteyn A., Chem. J. Chinese Universities,2010, 31, 9795-9810 |
| [10] | Wang W., Cai W., Yang Y., Li H., Cong M., Chen T., Mater. Chem. Phys., 2013, 142(2/3), 756-762 |
| [11] | Chen H. Y., Shen H. P., Wu H. C., Wang M. S., Lee C. F., Chiu W. Y., Chen W. C., Chem. J. Chinese Universities,2010, 31(14), 3318-3328 |
| [12] | Dong A.G., Wang Y. J., Tang Y., Ren N., Yang W. L., Gao Z.,Chem. Commun., 2002, (4), 350-351 |
| [13] | Hoppe C. E., Lazzari M., Pardinas-Blanco I., López-Quintela M. A., Langmuir,2006, 22, 7027-7034 |
| [14] | Zhang J., Chen Z., Wang Z., Zhang W., Ming N., Mater. Lett., 2003, 57(28), 4466-4470 |
| [15] | Xu D., Shen X. H., Li H. H., Du Y. C., Hu J. W., Chem. J. Chinese Universities,2010, 31(12), 2511-2515 |
| (徐蝶, 申晓华, 李焕焕, 杜元春, 胡家文. 高等学校化学学报, 2015, 36(12), 2511-2515 | |
| [16] | Hazarika M., Malkappa K., Jana T., Polym. Int., 2012, 61(9), 1425-1432 |
| [17] | Bastús N. G., Merkoçi F., Piella J., Puntes V., Chem. Mater., 2014, 26(9), 2836-2846 |
| [18] | Wang W., Zhang A., Liu L., Tian M., Zhang L., J. Electrochem. Soc., 2011, 158(4), 228 |
| [19] | Feng X. L., Ma H. Y., Huang S. X., Pan W., Zhang X. K., Tian F., Gao C. X., Cheng Y. W., Luo A. J., Chem. J. Chinese Universities,2010, 31, 12311-12317 |
| [20] | Tian C. G., Wang E. B., Kang Z. H., Song Y. L., Wang C. L., Song L., Chem. J. Chinese Universities,2010, 31, 3651-3657 |
| [21] | Huang Y. G., Zhao T., Zhang P. L., Song R., Colloid. Polym. Sci., 2011, 290(5), 401-409 |
| [22] | Ai L., Yue H., Jiang J., J. Mater. Chem., 2012, 22(44), 23447 |
| [23] | Ansari S. A., Khan M. M., Ansari M. O., Lee J., Cho M. H., Chem. J. Chinese Universities,2010, 31(51), 27023-27030 |
| [24] | Xie Y., Yan B., Xu H., Chen J., Liu Q., Deng Y. Zeng H., Appl. Mater. Int., 2014, 6(11), 8845-8852 |
| [25] | Chao Z., Wang L., Song L., Zhou Y., Nie W. Chen P., Appl. Surf. Sci., 2015, 329, 158-164 |
| [26] | Zhu M., Wang C., Meng D., Diao G., Chem. J. Chinese Universities,2010, 31(6), 2118-2125 |
| [27] | Hu Y., Zhao T., Zhu P., Liang X., Sun R., Wong C. P., RSC Adv., 2015, 5(1), 58-67 |
| [28] | Tian Q., Yu X., Zhang L., Yu D., J. Colloid Interface Sci., 2017, 491, 294-304 |
| [1] | TENG Zhenyuan, ZHANG Qitao, SU Chenliang. Charge Separation and Surface Reaction Mechanisms for Polymeric Single-atom Photocatalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220325. |
| [2] | YANG Jingyi, SHI Siqi, PENG Huaitao, YANG Qihao, CHEN Liang. Integration of Atomically Dispersed Ga Sites with C3N4 Nanosheets for Efficient Photo-driven CO2 Cycloaddition [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220349. |
| [3] | WANG Ruyue, WEI Hehe, HUANG Kai, WU Hui. Freezing Synthesis for Single Atom Materials [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220428. |
| [4] | WANG Xintian, LI Pan, CAO Yue, HONG Wenhao, GENG Zhongxuan, AN Zhiyang, WANG Haoyu, WANG Hua, SUN Bin, ZHU Wenlei, ZHOU Yang. Techno-economic Analysis and Industrial Application Prospects of Single-atom Materials in CO2 Catalysis [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220347. |
| [5] | YANG Jingyi, LI Qinghe, QIAO Botao. Synergistic Catalysis Between Ir Single Atoms and Nanoparticles for N2O Decomposition [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220388. |
| [6] | LIN Gaoxin, WANG Jiacheng. Progress and Perspective on Molybdenum Disulfide with Single-atom Doping Toward Hydrogen Evolution [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220321. |
| [7] | WANG Sicong, PANG Beibei, LIU Xiaokang, DING Tao, YAO Tao. Application of XAFS Technique in Single-atom Electrocatalysis [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220487. |
| [8] | TANG Quanjun, LIU Yingxin, MENG Rongwei, ZHANG Ruotian, LING Guowei, ZHANG Chen. Application of Single-atom Catalysis in Marine Energy [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220324. |
| [9] | QIN Yongji, LUO Jun. Applications of Single-atom Catalysts in CO2 Conversion [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220300. |
| [10] | YAO Qing, YU Zhiyong, HUANG Xiaoqing. Progress in Synthesis and Energy-related Electrocatalysis of Single-atom Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220323. |
| [11] | LIN Zhi, PENG Zhiming, HE Weiqing, SHEN Shaohua. Single-atom and Cluster Photocatalysis: Competition and Cooperation [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220312. |
| [12] | HUANG Qiuhong, LI Wenjun, LI Xin. Organocatalytic Enantioselective Mannich-type Addition of 5H-Oxazol-4-ones to Isatin Derived Ketimines [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220131. |
| [13] | TAN Yan, YU Shen, LYU Jiamin, LIU Zhan, SUN Minghui, CHEN Lihua, SU Baolian. Efficient Preparation of Mesoporous γ-Al2O3 Microspheres and Performance of Pd-loaded Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220133. |
| [14] | HAN Fuchao, LI Fujin, CHEN Liang, HE Leiyi, JIANG Yunan, XU Shoudong, ZHANG Ding, QI Lu. Enhance of CoSe2/C Composites Modified Separator on Electrochemical Performance of Li-S Batteries at High Sulfur Loading [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220163. |
| [15] | ZHOU Zixuan, YANG Haiyan, SUN Yuhan, GAO Peng. Recent Progress in Heterogeneous Catalysts for the Hydrogenation of Carbon Dioxide to Methanol [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220235. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
