

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (7): 1378.doi: 10.7503/cjcu20150012
• Physical Chemistry • Previous Articles Next Articles
SONG Yukun1,2, LÜ Hong1,2,*( ), HAO Chuanpu1,2, MI Cangen1,3
), HAO Chuanpu1,2, MI Cangen1,3
Received:2015-01-07
Online:2015-07-10
Published:2015-06-17
Contact:
LÜ Hong
E-mail:lvhong@tongji.edu.cn
Supported by:CLC Number:
TrendMD:
SONG Yukun, LÜ Hong, HAO Chuanpu, MI Cangen. Effect of IrO2 Modification of Ti Mesh on Electrochemical Performance of Collector Layer for Solid Polymer Electrolyte Electrolyzer†[J]. Chem. J. Chinese Universities, 2015, 36(7): 1378.
| Ti mesh | Mass before modification/mg | Mass after modification/mg | IrO2 loading/mg | IrO2 loading ratio(%) |
|---|---|---|---|---|
| 50 mesh Adams | 80.53 | 81.92 | 1.39 | 1.70 |
| 100 mesh Adams-1 | 45.98 | 46.19 | 0.21 | 0.45 |
| 100 mesh Adams-2 | 48.71 | 49.39 | 0.68 | 1.38 |
| 100 mesh Adams-3 | 47.34 | 48.63 | 1.29 | 2.65 |
Table 1 Modified Ti mesh specimens
| Ti mesh | Mass before modification/mg | Mass after modification/mg | IrO2 loading/mg | IrO2 loading ratio(%) |
|---|---|---|---|---|
| 50 mesh Adams | 80.53 | 81.92 | 1.39 | 1.70 |
| 100 mesh Adams-1 | 45.98 | 46.19 | 0.21 | 0.45 |
| 100 mesh Adams-2 | 48.71 | 49.39 | 0.68 | 1.38 |
| 100 mesh Adams-3 | 47.34 | 48.63 | 1.29 | 2.65 |

Fig.2 SEM images of Ti meshes before(A, B) and after(C, D) modification at low(A, C) and high(B, D) magnifications(A), (B) 50 mesh Ti mesh; (C), (D) 50 mesh Adams.
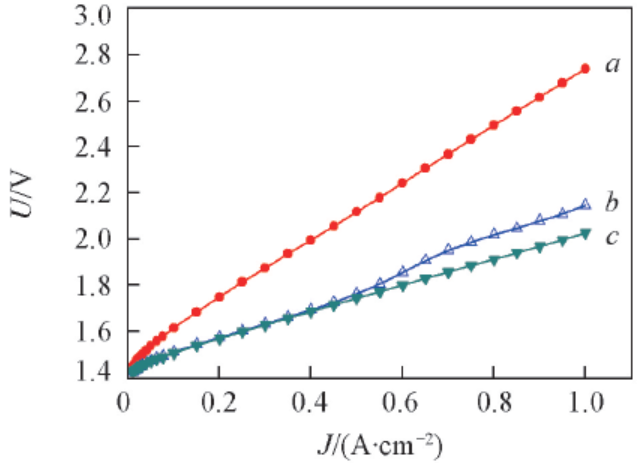
Fig.5 Polarization curves of water electrolyzers employing different anode electrode collector layersa. 50 mesh non-modified; b. 50 mesh Adams; c. 100 mesh Adams-2.
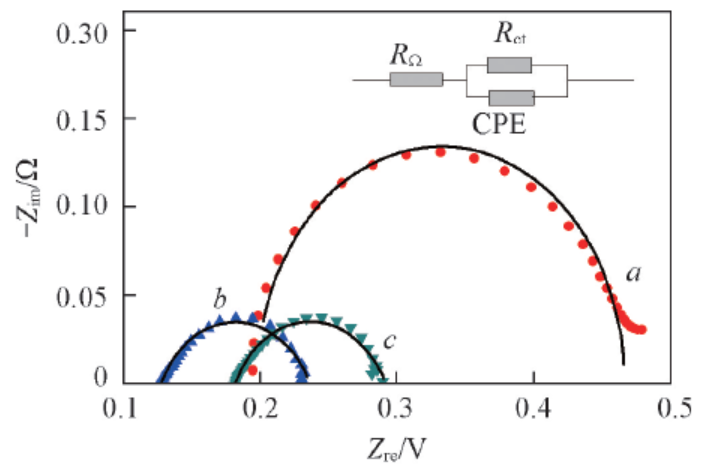
Fig.6 EIS of water electrolyzers employing diffe-rent anode electrode collector layers at 0.05 A/cm2a. 50 mesh non-modified; b. 50 mesh Adams; c. 100 mesh Adams-2.
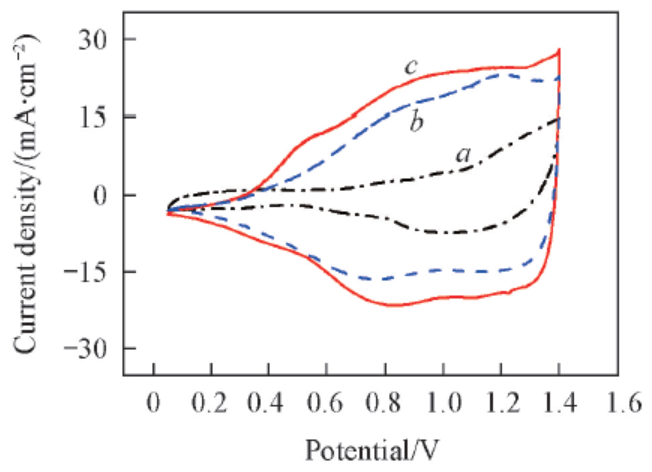
Fig.7 Repetitive cyclic voltammetry of water electrolyzers employing different anode electrode collector layersa. 50 mesh non-modified; b. 50 mesh Adams; c. 100 mesh Adams-2.
| Ti mesh | RΩ/Ω | Rct/Ω |
|---|---|---|
| 50 mesh non-modified | 0.195 | 0.272 |
| 50 mesh Adams | 0.129 | 0.105 |
| 100 mesh Adams-2 | 0.186 | 0.102 |
Table 2 Rct and RΩ of water electrolyzers employing different anode electrode collector layers
| Ti mesh | RΩ/Ω | Rct/Ω |
|---|---|---|
| 50 mesh non-modified | 0.195 | 0.272 |
| 50 mesh Adams | 0.129 | 0.105 |
| 100 mesh Adams-2 | 0.186 | 0.102 |
| Ti mesh | RΩ/Ω | Rct/Ω |
|---|---|---|
| 100 mesh Adams-1 | 0.217 | 0.110 |
| 100 mesh Adams-2 | 0.186 | 0.102 |
| 100 mesh Adams-3 | 0.211 | 0.098 |
Table 3 Rct and RΩ of water electrolyzers employing modified Ti meshes with different IrO2 loadings
| Ti mesh | RΩ/Ω | Rct/Ω |
|---|---|---|
| 100 mesh Adams-1 | 0.217 | 0.110 |
| 100 mesh Adams-2 | 0.186 | 0.102 |
| 100 mesh Adams-3 | 0.211 | 0.098 |
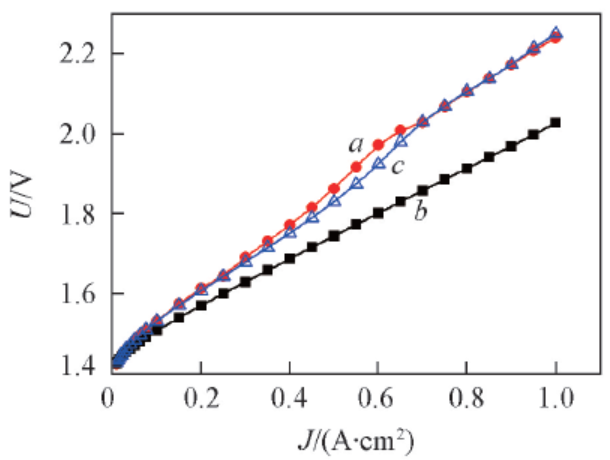
Fig.9 Polarization curves of water electrolyzers employing modified Ti meshes with diffe-rent IrO2 loadingsw(IrO2): a. 0.45%(100 mesh Adams-1); b. 1.38%(100 mesh Adams-2); c. 2.65%(100 mesh Adams-3).
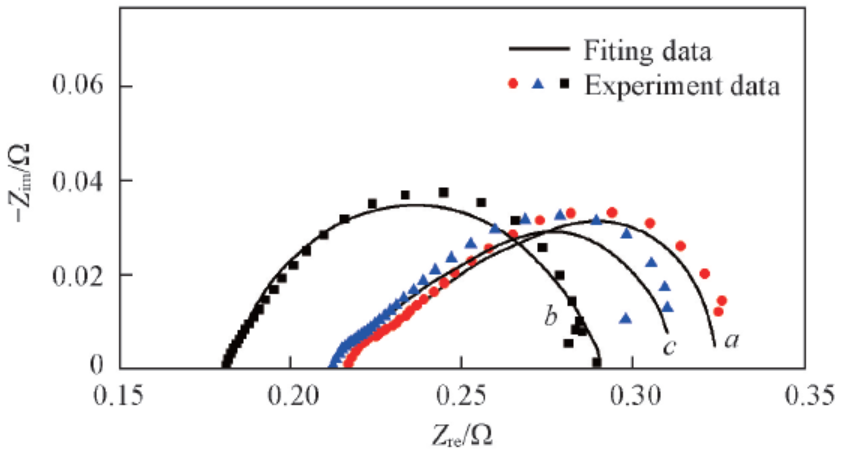
Fig.10 EIS of water electrolyzers employing modified Ti meshes with different IrO2 loadings at 0.05 A/cm2w(IrO2): a. 0.45%(100 mesh Adams-1); b. 1.38%(100 mesh Adams-2); c. 2.65%(100 mesh Adams-3).
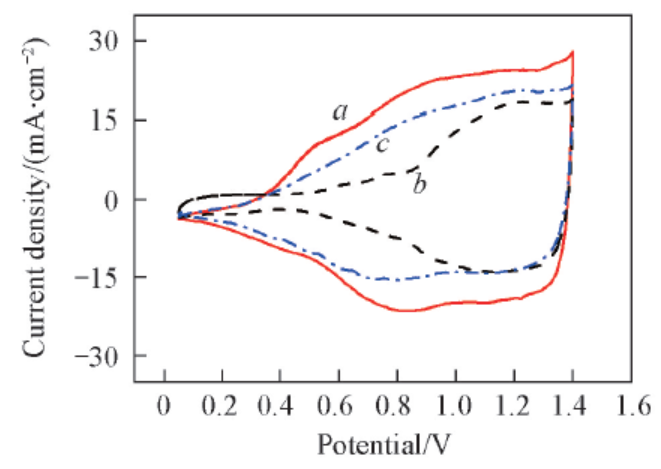
Fig.11 Repetitive cyclic voltammetry of water electrolyzers employing modified Ti meshes with different catalyst loadingsw(IrO2): a. 1.38%(100 mesh Adams-2); b. 0.45%(100 mesh Adams-1); c. 2.65%(100 mesh Adams-3).

Fig.12 SEM images of 100 mesh Ti meshes with different IrO2 loadingsw(IrO2): a. 0.45%(100 mesh Adams-1); b. 1.38%(100 mesh Adams-2); c. 2.65%(100 mesh Adams-3).
| [1] | Nehrir M. H., Wang C., Strunz K., Aki H., Ramakumar R., Bing J., Miao Z., Salameh Z., Ieee Transactions on Sustainable Energy, 2011, 2(4), 392—403 |
| [2] | Carmo M., Fritz D. L., Merge J., Stolten D., Int. J. Hydrogen Energy, 2013, 38(12), 4901—4934 |
| [3] | Ursua A., Gandia L. M., Sanchis P., Proceedings of the Ieee, 2012, 100(2), 410—426 |
| [4] | Ito H., Maeda T., Nakano A., Takenaka H., Int. J. Hydrogen Energy, 2011, 36(17), 10527—10540 |
| [5] | Grigoriev S. A., Millet P., Volobuev S. A., Fateev V. N., Int. J. Hydrogen Energy, 2009, 34(11), 4968—4973 |
| [6] | Liu M., Guo X. F., Wang J. M., Jiang L., Acta Phys. Chim. Sin., 2012, 28(12), 2931—2938 |
| (刘萌, 郭向飞, 王景明, 江雷. 物理化学学报,2012, 28(12), 2931—2938) | |
| [7] | Goni-Urtiaga A., Presvytes D., Scott K., Int. J. Hydrogen Energy, 2012, 37(4), 3358—3372 |
| [8] | Hu J. M., Meng H. M., Zhang J. Q., Cao C. N., Acta. Phys. Chim. Sin., 2002, 18(1), 14—20 |
| (胡吉明, 孟惠民, 张鉴清, 曹楚南. 物理化学学报,2002, 18(1), 14—20) | |
| [9] | Ren L.B., Xu Z. B., Li Y. H., Yi W., Zhang J., Liu X. J.,Chinese Journal of Power Sources, 2009, (4), 326—328 |
| (任丽彬, 徐志彬, 李勇辉, 易炜, 张军, 刘兴江. 电源技术, 2009, (4), 326—328) | |
| [10] | Jiang J. M., Liu G. Y., Xu J. Y., Wang X. D., Battery Bimonthly, 2014, 44(2), 64—67 |
| (蒋钜明, 刘高阳, 许军元, 王新东. 电池,2014, 44(2), 64—67) | |
| [11] | Adams R., Shriner R., J. Am. Chem. Soc., 1923, 45(9), 2171—2179 |
| [12] | Li G., Yu H., Song W., Dou M., Li Y., Shao Z., Yi B., Chem. Sus. Chem., 2012, 5(5), 858—861 |
| [13] | Kokoh K. B., Mayousse E., Napporn T. W., Servat K., Guillet N., Soyez E., Grosjean A., Rakotondrainibe A., Paul-Joseph J., Int. J. Hydrogen Energy, 2014, 39(5), 1924—1931 |
| [14] | Marshall A., Børresen B., Hagen G., Tsypkin M., Tunold R., Mater. Chem. Phys., 2005, 94(2), 226—232 |
| [15] | Polonsky J., Petrushina I. M., Christensen E., Bouzek K., Prag C. B., Andersen J. E. T., Bjerrum N. J., Int. J. Hydrogen. Energy, 2012, 37(3), 2173—2181 |
| [16] | Hao C. P., Lv H., Li B., Xin H. F., Ma J. X., Acta Energiae Solaris Sinica, 2013, 34(8), 1464—1470 |
| (郝传璞, 吕洪, 李冰, 辛海峰, 马建新. 太阳能学报,2013, 34(8), 1464—1470) | |
| [17] | Wang C., Mao Z. Q., Xu J. M., Xie X. F., Yang L. H., Chem. J. Chinese Universities, 2005, 26(1), 125—128 |
| (王诚, 毛宗强, 徐景明, 谢晓峰, 杨立寨. 高等学校化学学报,2005, 26(1), 125—128) | |
| [18] | Cheng H., Dong J. Z., Chao H., Yao J. H., Cao Y. A., Acta. Phys. Chim. Sin., 2012, 28(4), 850—856 |
| (程辉, 董江舟, 巢晖, 姚江宏, 曹亚安. 物理化学学报,2012, 28(4), 850—856) | |
| [19] | Su H., Bladergroen B. J., Linkov V., Pasupathi S., Ji S., Int. J. Hydrogen. Energy, 2011, 36(23), 15081—15088 |
| [20] | Guo J. W., Mao Z. L., Xu J. M., Chem. J. Chinese Universities, 2003, 24(8), 1477—1481 |
| (郭建伟, 毛宗强, 徐景明. 高等学校化学学报,2003, 24(8), 1477—1481) | |
| [21] | Banerjee W., Maikap S., Lai C. S., Chen Y. Y., Tien T. C., Lee H. Y., Chen W. S., Chen F. T., Kao M. J., Tsai M. J., Nanoscale Research Letters, 2012, 7(1), 1—12 |
| [22] | De Oliveira-Sousa A., Da Silva M., Machado S., Avaca L., de Lima-Neto P., Electrochimica Acta, 2000, 45(27), 4467—4473 |
| [23] | Trasatti S., Electrochimica Acta, 1991, 36(2), 225—241 |
| [1] | GUO Biao, ZHAO Chencan, LIU Xinxin, YU Zhou, ZHOU Lijing, YUAN Hongming, ZHAO Zhen. Effects of Surface Hydrothermal Carbon Layer on the Photocatalytic Activity of Magnetic NiFe2O4 Octahedron [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220472. |
| [2] | DONG Yanhong, LU Xinhuan, YANG Lu, SUN Fanqi, DUAN Jingui, GUO Haotian, ZHANG Qinjun, ZHOU Dan, XIA Qinghua. Preparation of Bifunctional Metal-organic Framework Materials and Application in Catalytic Olefins Epoxidation [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220458. |
| [3] | WANG Zumin, MENG Cheng, YU Ranbo. Doping Regulation in Transition Metal Phosphides for Hydrogen Evolution Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220544. |
| [4] | HE Jianyun, JIANG Yunbo, ZHANG Aimin, TANG Zhenyan, LI Hongpeng. Preparation and application of a novel porphyrin-based porous organic polymer COP-180 supported palladium catalyst [J]. Chem. J. Chinese Universities, 0, (): 20220535. |
| [5] | XIA Wenwen, YU Hongjing, WANG Shiye, YAO Li, LI Xiangyuan. Combustion Mechanism Construction Based On Minimized Reaction Network: Combustion of Aromatic hydrocarbon [J]. Chem. J. Chinese Universities, 0, (): 20220616. |
| [6] | LI Huaike, YUE Guichu, XIE Haiyun, LIU Jing, GAO Songwei, HOU Lanlan, LI Shuai, MIAO Beibei, WANG Nü, BAI Jie, CUI Zhimin, ZHAO Yong. Application of Electrospun Hollow Nanofibers in Catalysis [J]. Chem. J. Chinese Universities, 0, (): 20220625. |
| [7] | KUANG Huayi, CHEN Chen. Synthesis methods and electrocatalytic performance of noble-metal nanoframes catalysts [J]. Chem. J. Chinese Universities, 0, (): 20220586. |
| [8] | . Ni-La/SiO2 Catalysts Prepared by Dielectric Barrier Discharge Plasma Applying in the Dry Reforming of Methane [J]. Chem. J. Chinese Universities, 0, (): 20220503. |
| [9] | ZHU Jipeng, LIU Runhui, SONG Gonghua. Application of Bisoxazoline Grafted Amino Acid Polymer as Chiral Catalytic Center in Asymmetric Henry Reaction [J]. Chem. J. Chinese Universities, 0, (): 20220569. |
| [10] | CHENG Yuanyuan, XI Biying. Theoretical Study on the Fragmentation Mechanism of CH3SSCH3 Radical Cation Initiated by OH Radical [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220271. |
| [11] | SUN Jinshi, CHEN Peng, JING Liping, SUN Fuxing, LIU Jia. Synthesis of Hierarchical Porous Aromatic Frameworks for Immobilization of Thiourea Catalyst [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220171. |
| [12] | LI Xueyu, WANG Zhao, CHEN Ya, LI Keke, LI Jianquan, JIN Shunjing, CHEN Lihua, SU Baolian. Enhanced Catalytic Performance of Supported Nano-gold by the Localized Surface Plasmon Resonance for Selective Hydrogenation of Butadiene [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220174. |
| [13] | SONG Jiaxin, CUI Jing, FAN Xiaoqiang, KONG Lian, XIAO Xia, XIE Zean, ZHAO Zhen. Preparation of mesoporous silica supported highly dispersed vanadium catalyst and their catalytic performance for selective oxidation of ethane [J]. Chem. J. Chinese Universities, 0, (): 20220532. |
| [14] | TANG Quanjun, LIU Yingxin, MENG Rongwei, ZHANG Ruotian, LING Guowei, ZHANG Chen. Application of Single-atom Catalysis in Marine Energy [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220324. |
| [15] | LIN Zhi, PENG Zhiming, HE Weiqing, SHEN Shaohua. Single-atom and Cluster Photocatalysis: Competition and Cooperation [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220312. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||