

Chem. J. Chinese Universities ›› 2022, Vol. 43 ›› Issue (2): 20210516.doi: 10.7503/cjcu20210516
• Physical Chemistry • Previous Articles Next Articles
JIN Keyan1, BAI Pu1,4, LI Xiaolong2( ), ZHANG Jianan3, YAN Wenfu1(
), ZHANG Jianan3, YAN Wenfu1( )
)
Received:2021-07-19
Online:2022-02-10
Published:2021-09-16
Contact:
LI Xiaolong,YAN Wenfu
E-mail:free123orange@163.com;yanw@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
JIN Keyan, BAI Pu, LI Xiaolong, ZHANG Jianan, YAN Wenfu. New Mg-Al Type Sorbent for Efficient Removal of Boron from Waste Water Containing High-concentration of Boron from Pressurized Water Reactor Nuclear Power Plants[J]. Chem. J. Chinese Universities, 2022, 43(2): 20210516.
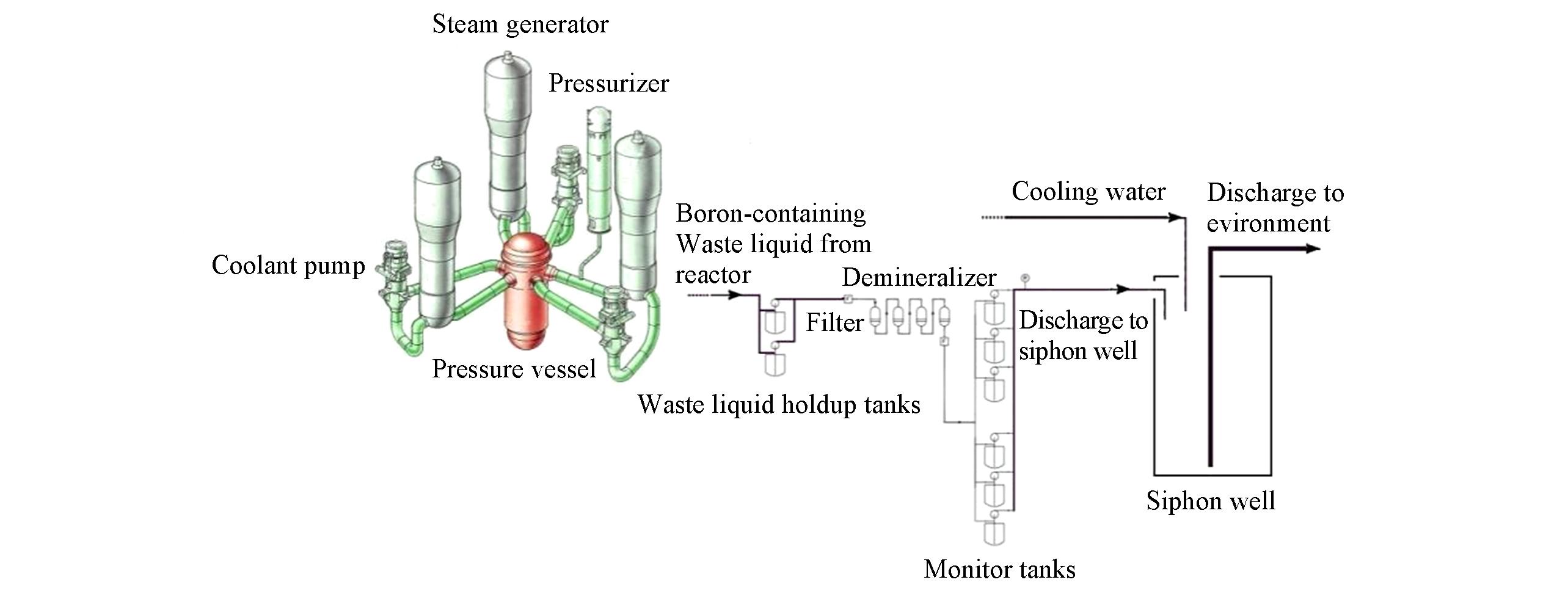
Scheme 1 Schematic diagram of the treatment and discharge of the boron?containing waste water generated from pressurized water reactor nuclear power plant
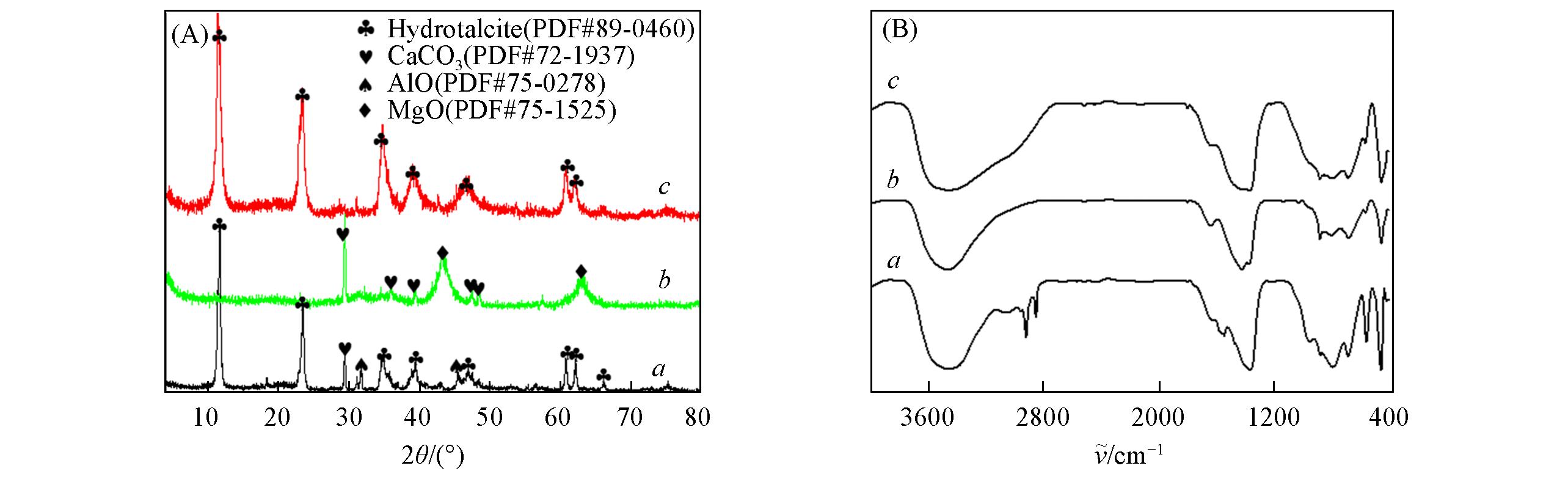
Fig.1 Experimental XRD patterns(A) and FTIR spectra(B) of raw Mg?Al LDH(a), intermediate oxide from the calcination of LDH(b), and layered double hydroxide after B?anions adsorption(c)

Fig.2 SEM images of the raw Mg?Al LDH(A), intermediate oxide from the calcination of LDH(B), and layered double hydroxide after B?anions adsorption(C)
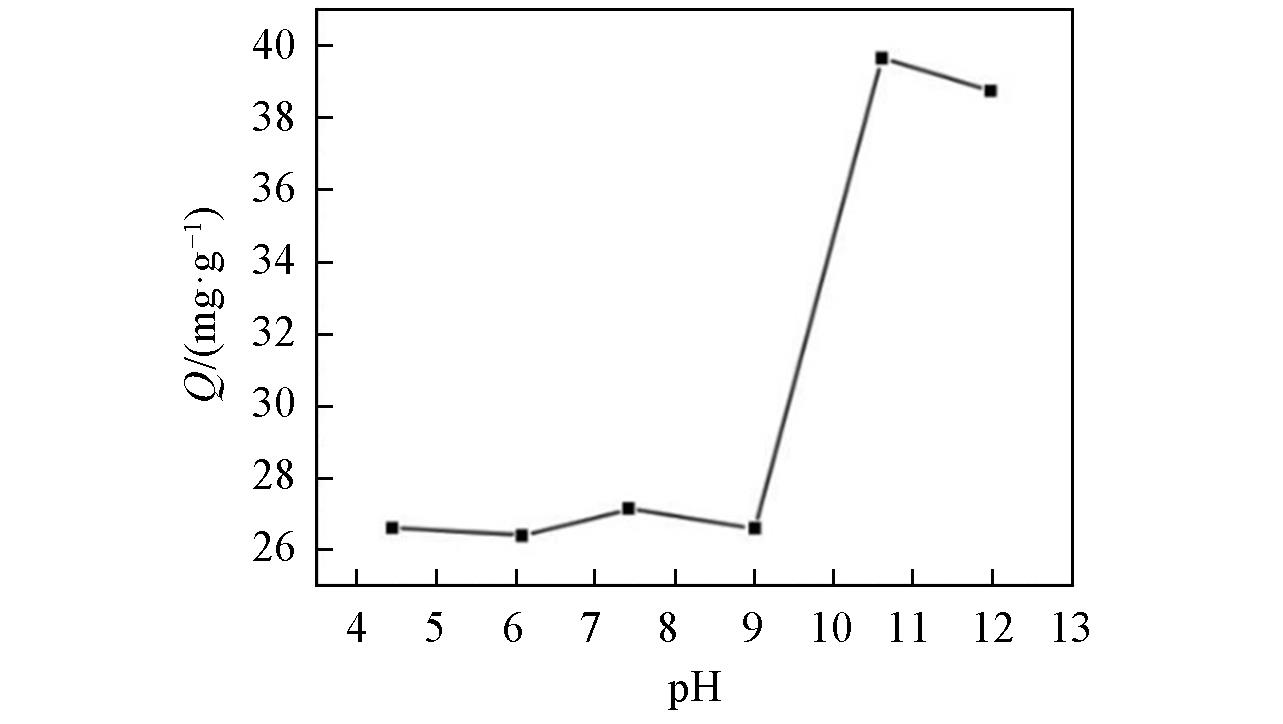
Fig.3 Influence of pH value on the removal of boron at an initial boron concentration of 2000 mg/LAdsorbent dose/solution volume ratio: 1/40 g/mL; temperature: 20 ℃; reaction time: 24 h.
| Run | c0/(mg·L-1) | ce/(mg·L-1) | Removal efficiency(%) | Qe/(mg·g-1) |
|---|---|---|---|---|
| 1 | 50 | 11.07 | 77.86 | 1.56 |
| 2 | 200 | 36.46 | 81.77 | 6.54 |
| 3 | 500 | 69.01 | 86.20 | 17.24 |
| 4 | 1000 | 453.4 | 54.66 | 21.86 |
| 5 | 2000 | 1277.4 | 54.66 | 28.90 |
Table 1 Boron adsorption on intermediate oxide from aqueous solution with various initial B concentrations(Adsorbent dose/solution volume ratio: 1/40 g/mL, pH=7.43, contact time: 24 h)
| Run | c0/(mg·L-1) | ce/(mg·L-1) | Removal efficiency(%) | Qe/(mg·g-1) |
|---|---|---|---|---|
| 1 | 50 | 11.07 | 77.86 | 1.56 |
| 2 | 200 | 36.46 | 81.77 | 6.54 |
| 3 | 500 | 69.01 | 86.20 | 17.24 |
| 4 | 1000 | 453.4 | 54.66 | 21.86 |
| 5 | 2000 | 1277.4 | 54.66 | 28.90 |
| Material | qm,exp/(mg·g-1) | Langmuir model | Freundlich model | |||||
|---|---|---|---|---|---|---|---|---|
| qm/(mg·g-1) | b/(L·mg-1) | R2 | kF/(mg·g-1) | 1/n | R2 | |||
| Intermediate oxide | 28.90 | 32.01 | 0.0065 | 0.981 | 0.756 | 0.554 | 0.735 | |
Table 2 Langmuir and Freundlich isotherm parameters of B adsorption on intermediate oxide
| Material | qm,exp/(mg·g-1) | Langmuir model | Freundlich model | |||||
|---|---|---|---|---|---|---|---|---|
| qm/(mg·g-1) | b/(L·mg-1) | R2 | kF/(mg·g-1) | 1/n | R2 | |||
| Intermediate oxide | 28.90 | 32.01 | 0.0065 | 0.981 | 0.756 | 0.554 | 0.735 | |
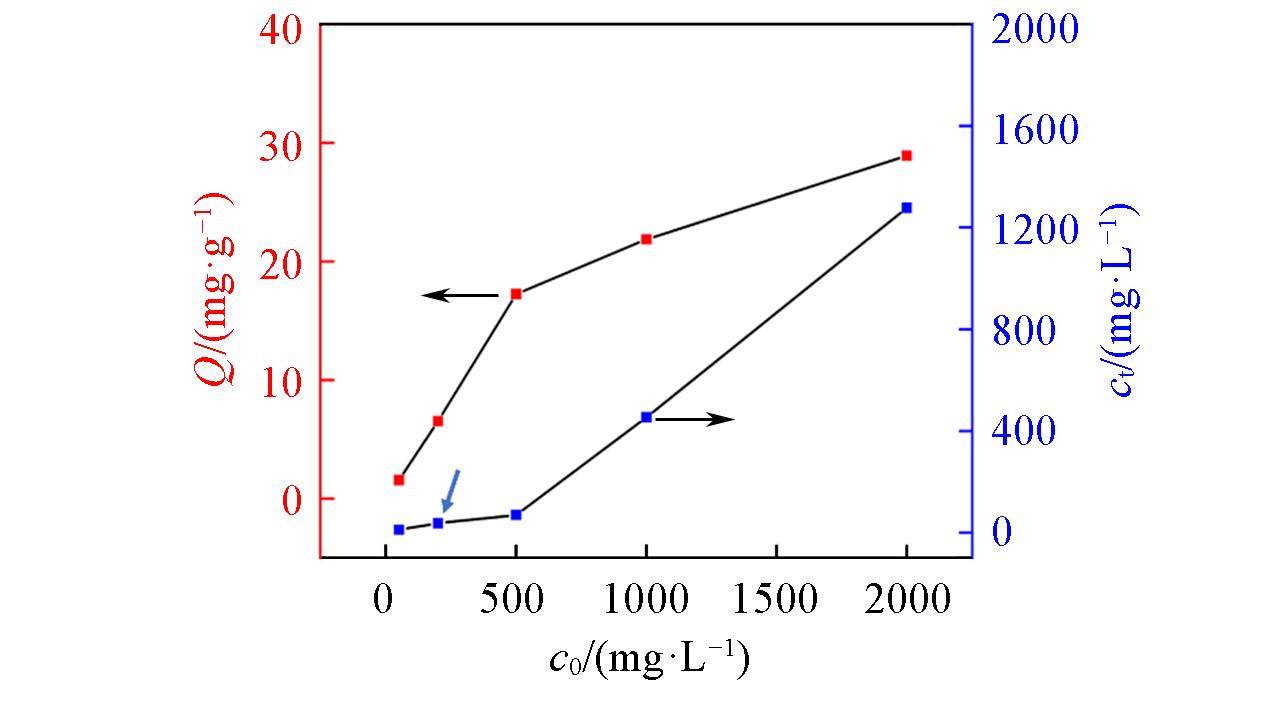
Fig.5 Influence of initial boron concentration(c0) on the removal of boron at pH of 7.43Adsorbent dose/solution volume ratio: 1/40 g/mL;temperature: 20 °C; reaction time: 24 h.
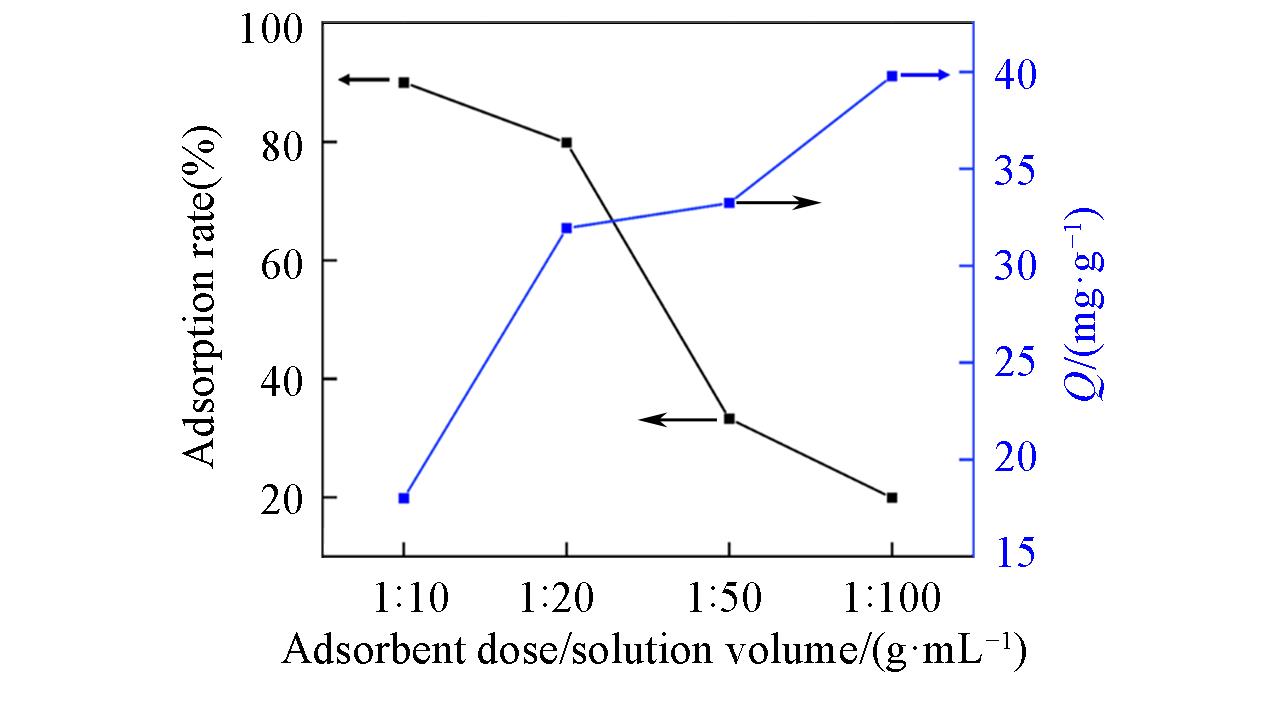
Fig.6 Influence of adsorbent dose/solution volume ratio on the removal of boron at an initial boron concentration of 2000 mg/L, pH=7.43, temperature of 20 ℃, and reaction time of 24 h
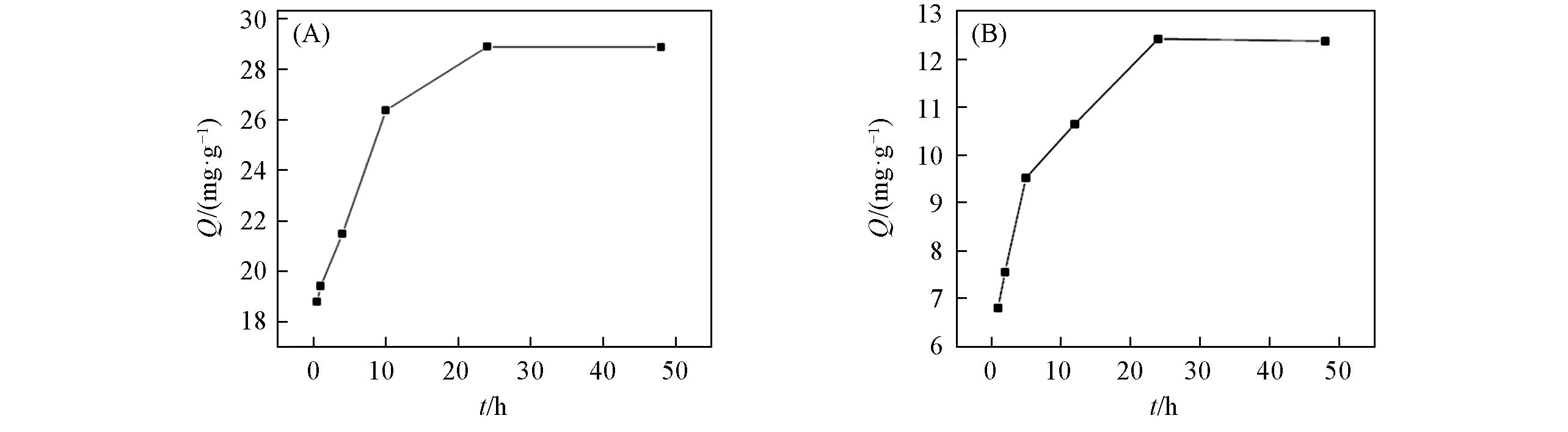
Fig.7 Influence of agitation time on the removal of boron of calcined Mg?Al LDH(A) and un?calcined Mg?Al LDH(B)Initial boron concentration: 2000 mg/L, pH=7.43, adsorbent dose: 1/40 g/mL for calcined Mg?Al LDH, 1/100 g/mL for un?calcined Mg?Al LDH, temperature: 20 ℃.
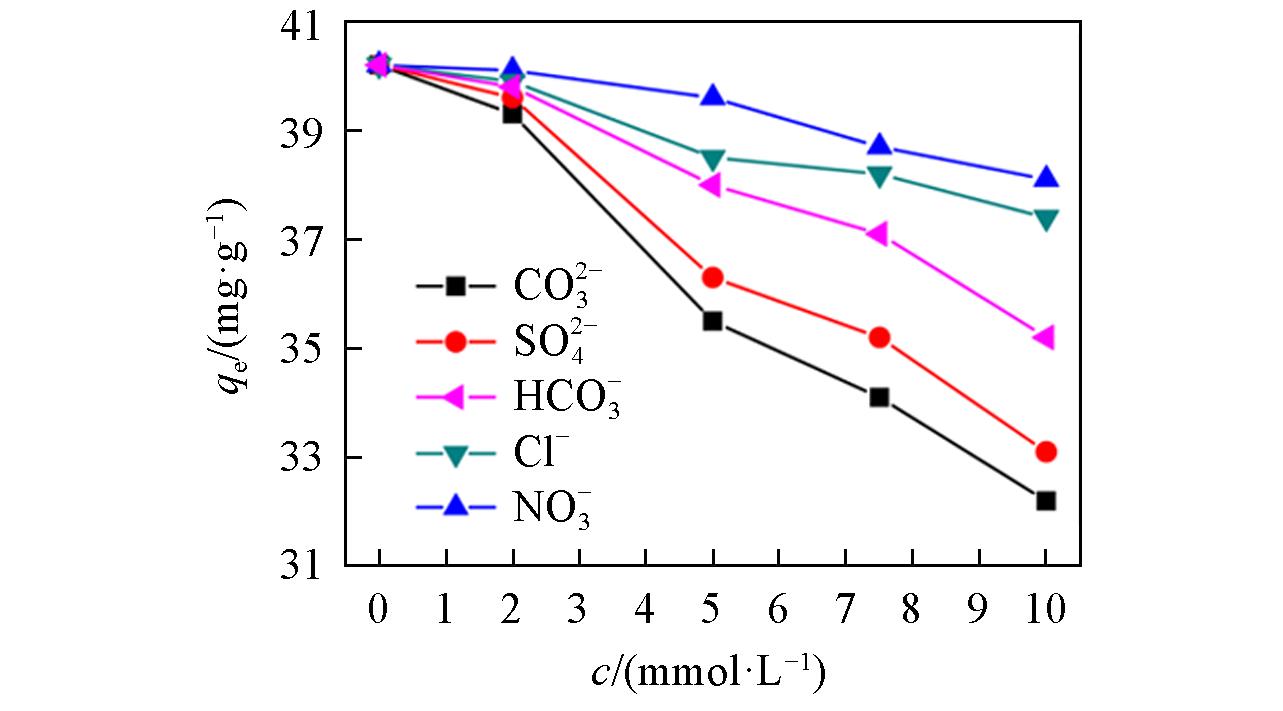
Fig.8 Influence of competition ion on the removal of boron at an initial boron concentration of 2000 mg/L, pH of 7.43, adsorbent dose/solution volume of 1/100 g/mL, and temperature of 20 ℃
| 1 | Ozturk N., Kavak D., J. Hazard. Mater., 2005, 127(1—3), 81—88 |
| 2 | Şahin S., Desalination, 2002, 143(1), 35—43 |
| 3 | Li G. Q., Yu J. S., Li G. W., Chem. J. Chinese Universities, 2011, 32(3), 793—796(李广全, 于景生, 李国文. 高等学校化学学报, 2011, 32(3), 793—796) |
| 4 | Fang F., Xu C. Y., Liu X. H., Wu H., Radiation Protection, 2012, 32(1), 8—14, 20(方岚, 徐春艳, 刘新华, 吴浩. 辐射防护, 2012, 32(1), 8—14, 20) |
| 5 | Zerze H., Özbelge H. Ö., Bıçak N., Yılmaz L., Sep. Sci. Technol., 2015, 50(13), 2004—2014 |
| 6 | Liu P., Hu W., Wu G. P., Wei F. S., World Sci⁃Tech R&D, 2003, 25(6), 6—11(刘平, 胡伟, 吴国平, 魏复盛. 世界科技研究与发展, 2003, 25(6), 6—11) |
| 7 | Kluczka J., Int. J. Environ. Res., 2015, 9(2), 711—720 |
| 8 | Ping Q., Abu⁃Reesh I. M., He Z., Desalination, 2015, 376, 55—61 |
| 9 | Zheng Z. Q., Bian S. P., Zheng J. M., Shang S. Y., Li B. J., Liu X. L., Environ. Sci., 1983, 4(3), 16—20(郑泽群, 边淑萍, 郑建民, 商寿岩, 李炳钧, 刘秀兰. 环境科学, 1983, 4(3), 16—20) |
| 10 | Rivas B. L., Sanchez J., Macromol. Symp., 2015, 351(1), 37—45 |
| 11 | Fang L., Liu X. H.,Wu H., Zhang Z. Y., Chin. J. Nucl. Sci. Eng., 2011, 31(1), 86—92(方岚, 刘新华, 吴浩, 张志银. 核科学与工程, 2011, 31(1), 86—92) |
| 12 | Wei X. Y., Ma H. B., Wang P., Yang D. J., Shang Z. R., Radiat. Prot. Bull., 2012, 32(5), 32—36(魏新渝, 马鸿宾, 汪萍, 杨端节, 商照荣. 辐射防护通讯, 2012, 32(5), 32—36) |
| 13 | Chang R. R., Wu J. Q., Wang G. Z., Liang L., Guo L. M., Appl. Chem. Ind., 2020, 49(11), 2718—2721(常瑞瑞, 吴家全, 王桂珠, 梁利, 郭丽梅. 应用化工, 2020, 49(11), 2718—2721) |
| 14 | Yoshikawa E., Sasaki A., Endo M., J. Hazard. Mater., 2012, 237(30), 277—282 |
| 15 | Goldberg S., Forster H. S., Lesch S. M., Heick E. L., Soil Sci., 1996, 161(2), 99—103 |
| 16 | Ozturk N., Kavak D., Fresenius Environ. Bull., 2003, 12(12), 1450—1456 |
| 17 | Xu X. Y., Liao Y. Q., Sun J. C., Wang X. H., Chen S. Q., Lv Z., Song J. Q., Acta Phys. ⁃Chim. Sin., 2019, 35(3), 317—326 |
| 18 | Gong F. F., Wang Y., Chen S. H., Zhang Y. Q., Liu Y. D., Meng T. Y., Lou Y. J., Li Y. Y., J. Nucl. Agric. Sci., 2020, 34(8), 1763—1769(龚芳芳, 王晔, 陈淑敏, 张玉琦, 刘英丹, 孟天宇, 娄永江, 李勇勇. 核农学报, 2020, 34(8), 1763—1769) |
| 19 | Wu X. X., Li Q. Q., Li M., Tang H., Hu B., Light Met., 1995, (7), 35—38(吴贤熙, 李琴琴, 黎曼, 汤辉, 胡波. 轻金属, 1995, (7), 35—38) |
| 20 | Gao C. K., Li R. H., Liu L. F., Song X. X., Xie X., Zhang X., Chem. J. Chinese Universities, 2019, 40(9), 2033—2040(高从堦, 李蕊含, 刘立芬, 宋潇潇, 谢欣, 张潇. 高等学校化学学报, 2019, 40(9), 2033—2040) |
| 21 | Wang J. Q., Zhou M. S., Lu D. N., Fei W. Y., Wu J. Z., Green Energy Environ., 2020, 5(3), 364—373 |
| 22 | Wang N., Wei R. Q., Cao F. T., Liu X. N., Chem. J. Chinese Universities, 2012, 33(12), 2795—2800(王楠, 魏荣卿, 曹飞婷, 刘晓宁. 高等学校化学学报, 2012, 33(12), 2795—2800) |
| 23 | Kluczka J., Ciba J., Trojanowska J., Zolotajkin M., Turek M., Dydo P., Environ. Prog., 2007, 26(1), 71—77 |
| 24 | Kluczka J., Trojanowska J., Zolotajkin M., Ciba J., Turek M., Dydo P., Environ. Technol., 2007, 28(1), 105—113 |
| 25 | Celik Z. C., Can B. Z., Kocakerim M. M., J. Hazard. Mater., 2008, 152(1), 415—422 |
| 26 | Halim A. A., Roslan N. A., Yaacub N. S., Latif M. T., Sains Malays., 2013, 42(9), 1293—1300 |
| 27 | Zohdi N., Mahdavi F., Abdullah L. C., Choong T. S. Y., J. Environ. Health Sci. Eng., 2014, 12, 3 |
| 28 | Rajaković L. V., Ristić M. D., Carbon, 1996, 34(6), 769—774 |
| 29 | Seyhan S., Seki Y., Yurdakoc M., Merdivan M., J. Hazard. Mater., 2007, 146(1/2), 180—185 |
| 30 | Cengeloglu Y., Tor A., Arslan G., Ersoz M., Gezgin S., J. Hazard. Mater., 2007, 142(1/2), 412—417 |
| 31 | Kameda T., Oba J., Yoshioka T., J. Hazard. Mater., 2015, 293, 54—63 |
| 32 | Yan C. Y., Yi W. T., Ma P. H., Li F. Q., Ion Exch. Adsorpt., 2009, 25(3), 233—240(闫春燕, 伊文涛, 马培华, 李法强. 离子交换与吸附. 2009, 25(3), 233—240) |
| 33 | Ferreira O. P., de Moraes S. G., Duran N., Cornejo L., Alves O. L., Chemosphere, 2006, 62(1), 80—88 |
| 34 | Kentjono L., Liu J. C., Chang W. C., Irawan C., Desalination, 2010, 262(1—3), 280—283 |
| 35 | Delazare T., Ferreira L. P., Ribeiro N. F. P., Souza M. M. V. M., Campos J. C., Yokoyama L., Environ. Eng., 2014, 49(8), 923—932 |
| 36 | Jiang J. Q., Xu Y., Quill K., Simon J., Shettle K., Ind. Eng. Chem. Res., 2007, 46(13), 4577—4583 |
| 37 | Liu J., Guo X. L., Yuan J. S., Desalin. Water Treat., 2014, 52(10—12), 1919—1927 |
| 38 | Ay A. N., Zümreoglu⁃Karan B., Temel A., Microporous Mesoporous Mater., 2007, 98(1—3), 1—5 |
| 39 | Yan C. Y., Yi W. T., Environ. Prot. Chem. Ind., 2010, 30(2), 172—175(闫春燕, 伊文涛. 化工环保, 2010, 30(2), 172—175) |
| 40 | Qiu X. H., Sasaki K., Osseo⁃Asare K., Hirajima T., Ideta K., Miyawaki J., J. Colloid Interface Sci., 2015, 445, 183—194 |
| 41 | Koilraj P., Srinivasan K., Ind. Eng. Chem. Res., 2011, 50(11), 6943—6951 |
| 42 | García S. M. M. F., Muñoz C. E., Sep. Purif. Technol., 2006, 48(1), 36—44 |
| 43 | García S. M. M. F., Muñoz C. E., Desalination, 2009, 249(2), 626—634 |
| 44 | Dionisiou N., Matsi T., Misopolinos N. D., J. Environ. Qual., 2006, 35(6), 2222—2228 |
| 45 | Seki Y., Seyhan S., Yurdakoc M., J. Hazard. Mater., 2006, 138(1), 60—66 |
| 46 | Bouguerra W., Mnif A., Hamrouni B., Dhahbi M., Desalination, 2008, 223(1), 31—37 |
| 47 | Zhang G. F., Sun G. Q., Mar. Sci., 1995, (2), 58—61(张国防, 孙国清. 海洋科学. 1995, (2), 58—61) |
| 48 | Qiu X. H., Sasaki K., Osseo⁃Asare K., Hirajima T., Ideta K., Jin M., J. Colloid Interface Sci., 2015, 445, 183—194 |
| 49 | Fan H. L., He W., Jiang Y. Y., Bao G. H., Ge T. J., Eng. Plast. Appl., 2011, 39(4), 10—14(范惠琳, 何伟, 姜莹莹, 保功辉, 葛铁军. 工程塑料应用, 2011, 39(4), 10—14) |
| 50 | Ma Z. Y., Spectrosc. Spectral Anal., 2012, 32(5), 1255—1258(马赵扬. 光谱学与光谱分析, 2012, 32(5), 1255—1258) |
| 51 | Wang X., Preparation of Hydrotalcite by Mechano⁃chemical Method and Study the Memory Effect of Structure, Dalian Jiaotong University, Dalian, 2014(王旭. 机械力化学法制备镁铝水滑石的结构记忆效应研究, 大连: 大连交通大学, 2014) |
| 52 | Sato T., Wakabayashi T., Shimada M., Ind. Eng. Chem. Prod. Res. Dev., 1986, 25(1), 89—92 |
| 53 | Sasaki K., Qiu X. H., Moriyama S., Tokoro C., Ideta K., Miyawaki J., Mater. Trans., 2013, 54(9), 1809—1817 |
| 54 | Goh K. H., Lim T. T., Dong Z. L., Water Res., 2008, 42(6/7), 1343—1368 |
| 55 | Iyi N., Matsumoto T., Kaneko Y., Kitamura K., Chem. Lett., 2004, 33(9), 1122—1123 |
| 56 | Li L., Ma R. Z., Ebina Y., Iyi N., Sasaki T., Chem. Mater., 2005, 17(17), 4386—4391 |
| 57 | Zhang T., Li Q. R., Xiao H. Y., Lu H. X., Zhou Y. M., Ind. Eng. Chem. Res., 2012, 51(35), 11490—11498 |
| [1] | REN Shijie, QIAO Sicong, LIU Chongjing, ZHANG Wenhua, SONG Li. Synchrotron Radiation X-Ray Absorption Spectroscopy Research Progress on Platinum Single-atom Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220466. |
| [2] | WANG Sicong, PANG Beibei, LIU Xiaokang, DING Tao, YAO Tao. Application of XAFS Technique in Single-atom Electrocatalysis [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220487. |
| [3] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [4] | JIANG Hongbin, DAI Wenchen, ZHANG Rao, XU Xiaochen, CHEN Jie, YANG Guang, YANG Fenglin. Research on Co3O4/UiO-66@α-Al2O3 Ceramic Membrane Separation and Catalytic Spraying Industry VOCs Waste Gas [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220025. |
| [5] | HAO Honglei, MENG Fanyu, LI Ruoyu, LI Yingqiu, JIA Mingjun, ZHANG Wenxiang, YUAN Xiaoling. Biomass Derived Nitrogen Doped Porous Carbon Materials as Adsorbents for Removal of Methylene Blue in Water [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220055. |
| [6] | WANG Hongning, HUANG Li, QING Jiang, MA Tengzhou, JIANG Wei, HUANG Weiqiu, CHEN Ruoyu. Activation of Biochar from Cattail and the VOCs Adsorption Application [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210824. |
| [7] | LI Qiao, ZHAO Yang, WANG Enju. Moisture Absorption Reaction and Fluorescence Property of Highly Active Michael System Based on Arylidenemalononitrile [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210690. |
| [8] | MENG Xianglong, YANG Ge, GUO Hailing, LIU Chenguang, CHAI Yongming, WANG Chunzheng, GUO Yongmei. Synthesis of Nano-zeolite and Its Adsorption Performance for Hydrogen Sulfide [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210687. |
| [9] | CHEN Xiaolu, YUAN Zhenyan, ZHONG Yingchun, REN Hao. Preparation of Triphenylamine Based PAF-106s via Mechanical Ball Milling and C2 Hydrocarbons Adsorption Property [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210771. |
| [10] | TAN Lejian, ZHONG Xuanshu, WANG Jin, LIU Zongjian, ZHANG Aiying, YE Lin, FENG Zengguo. Low Critical Dissolution Temperature Behavior of β⁃Cyclodextrin and Its Application in the Preparation of β⁃Cyclodextrin Sheet Crystal with Ordered Nano⁃channel [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220405. |
| [11] | ZHENG Meiqi, MAO Fangqi, KONG Xianggui, DUAN Xue. Layered Double Hydroxides as Sorbent for Remediation of Radioactive Wastewater [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220456. |
| [12] | TIAN Xiaokang, ZHANG Qingsong, YANG Shulin, BAI Jie, CHEN Bingjie, PAN Jie, CHEN Li, WEI Yen. Porous Materials Inspired by Microbial Fermentation: Preparation Method and Application [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220216. |
| [13] | WANG Yuanyue, AN Suosuo, ZHENG Xuming, ZHAO Yanying. Spectroscopic and Theoretical Studies on 5-Mercapto-1,3,4-thiadiazole-2-thione Microsolvation Clusters [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220354. |
| [14] | MA Jianxin, LIU Xiaodong, XU Na, LIU Guocheng, WANG Xiuli. A Multi-functional Zn(II) Coordination Polymer with Luminescence Sensing, Amperometric Sensing, and Dye Adsorption Performance [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210585. |
| [15] | ZHANG Chi, SUN Fuxing, ZHU Guangshan. Synthesis, N2 Adsorption and Mixed-matrix Membrane Performance of Bimetal Isostructural CAU-21 [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210578. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||