

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (11): 2294.doi: 10.7503/cjcu20190379
• Analytical Chemistry • Previous Articles Next Articles
YANG Weiqiang1,ZHANG Guiyun2,LIN Hua1,NI Jiancong1,*( ),HUANG Lingfeng1
),HUANG Lingfeng1
Received:2019-07-07
Online:2019-11-10
Published:2019-10-15
Contact:
NI Jiancong
E-mail:nijiancong@foxmail.com
Supported by:CLC Number:
TrendMD:
YANG Weiqiang, ZHANG Guiyun, LIN Hua, NI Jiancong, HUANG Lingfeng. Ratiometric Fluorescence Assay for Visual Detection of Tetracycline Residues based on the Complex of Carbon Dots and Europium †[J]. Chem. J. Chinese Universities, 2019, 40(11): 2294.
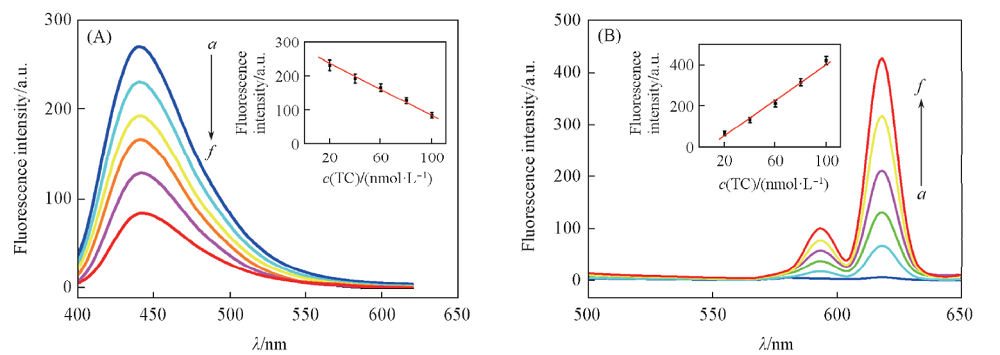
Fig.1 Fluorescence quenching effect of tetracycline on CDs(A) and fluorescence enhancement effect of tetracycline on Eu3+ (B) c(Tetracycline)/(nmol·L-1): a. 0; b. 20; c. 40; d. 60; e. 80; f. 100. ρ(CDs)=1 μg/mL, c(Eu3+)=20 μmol/L. The NaOH-H3BO3 buffer solution concentration was 20 mmol/L with pH=9.0. Insets are the linear relationships between the fluorescence intensities of CDs(A) and Eu-TC(B) and the concentration of tetracycline, respectively.
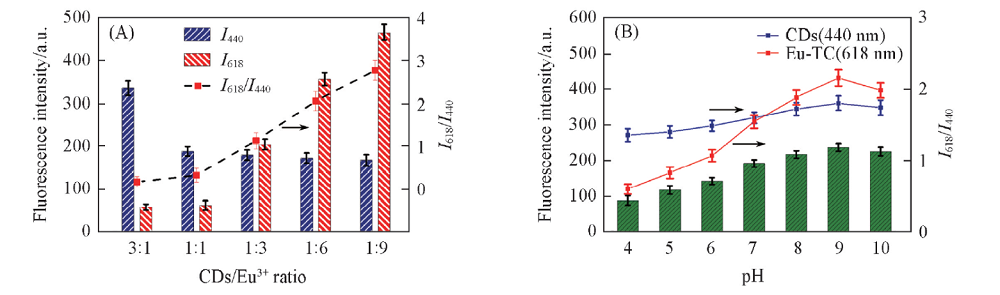
Fig.3 Effect of mass concentration ratio of CDs/Eu3+ on the fluorescence assay(A) and effect of pH on the fluorescence of CDs and Eu-TC(B) (A) c(Tetracycline)=50 nmol/L, ρ(CDs)=1—3 μg/mL, c(Eu3+)=6—60 μmol/L. The NaOH-H3BO3 buffer solution concentration was 20 mmol/L(pH=9.0); (B) c(tetracycline)=100 nmol/L, ρ(CDs)=1 μg/mL, c(Eu3+)=20 μmol/L. The NaOH-H3BO3 buffer solution concentration was 20 mmol/L.
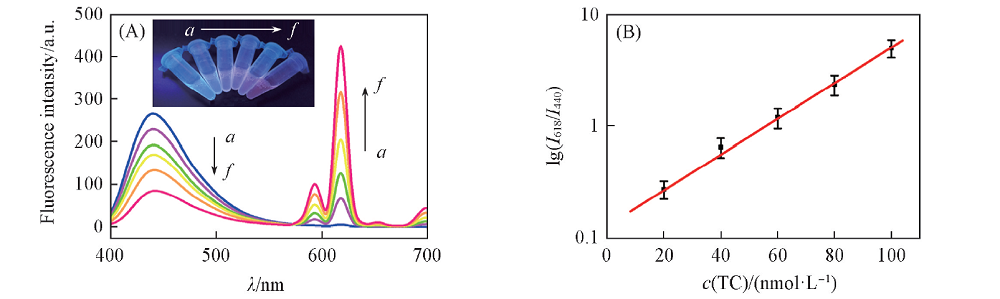
Fig.4 Fluorescence spectrum of CDs-Eu3+ probe varying with the concentration of tetracycline(A) and linear relationship between the lg(I618/I440) and the concentration of tetracycline(B) (A) The inset was the fluorescence color change of the solution. c(Tetracycline)/(nmol·L-1): a. 0; b. 20; c. 40; d. 60; e. 80; f. 100. ρ(CDs)=1 μg/mL; ρ(Eu3+)=20 μmol/L. The NaOH-H3BO3 buffer solution concentration was 20 mmol/L with pH=9.0.
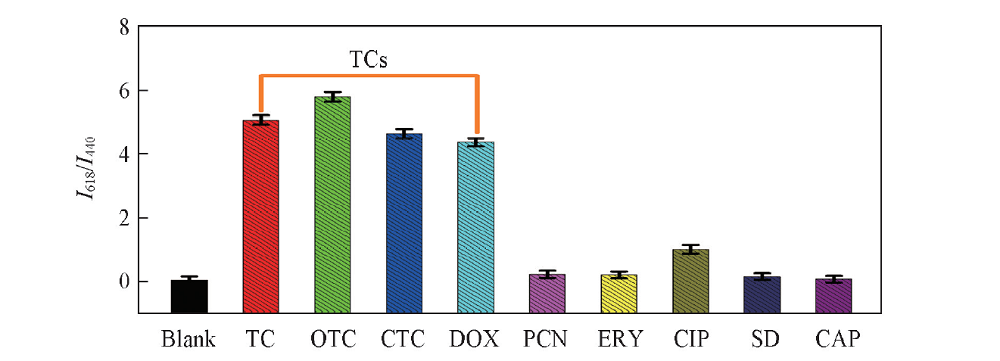
Fig.5 Fluorescence response of CDs-Eu3+ probe to different antibiotics The concentration of tetracycline(TC), oxytetracycline(OTC), chlorotetracycline(CTC) and doxycycline(DOX) were 100 nmol/L; and the concentration of penicillin(PCN), erythromycin(ERY), ciprofloxacin(CIP), sulfadiazine(SD) and chloramphenicol(CAP) were 1 μmol/L.
| Interference | Concentration of interference/(μmol·L-1) | Error(%) | Interference | Concentration of interference/(μmol·L-1) | Error(%) |
|---|---|---|---|---|---|
| LAS | 50.0 | 3.58 | Al3+ | 5.0 | -2.67 |
| CPC | 50.0 | 2.89 | Fe3+ | 5.0 | 3.16 |
| Tween-80 | 20.0 | 3.77 | Cu2+ | 10.0 | 2.53 |
| Ca2+ | 50.0 | -0.83 | Zn2+ | 50.0 | 1.12 |
| Mg2+ | 50.0 | -0.15 | Pb2+ | 20.0 | 0.45 |
| Interference | Concentration of interference/(μmol·L-1) | Error(%) | Interference | Concentration of interference/(μmol·L-1) | Error(%) |
|---|---|---|---|---|---|
| LAS | 50.0 | 3.58 | Al3+ | 5.0 | -2.67 |
| CPC | 50.0 | 2.89 | Fe3+ | 5.0 | 3.16 |
| Tween-80 | 20.0 | 3.77 | Cu2+ | 10.0 | 2.53 |
| Ca2+ | 50.0 | -0.83 | Zn2+ | 50.0 | 1.12 |
| Mg2+ | 50.0 | -0.15 | Pb2+ | 20.0 | 0.45 |
| Sample | Add/(nmol·L-1) | Found*/(nmol·L-1) | Recovery(%) | RSD(%, n=3) |
|---|---|---|---|---|
| 1 | 5.0 | 5.33 | 106.6 | 4.7 |
| 2 | 20.0 | 20.33 | 101.7 | 3.9 |
| 3 | 80.0 | 78.21 | 97.8 | 2.5 |
| Sample | Add/(nmol·L-1) | Found*/(nmol·L-1) | Recovery(%) | RSD(%, n=3) |
|---|---|---|---|---|
| 1 | 5.0 | 5.33 | 106.6 | 4.7 |
| 2 | 20.0 | 20.33 | 101.7 | 3.9 |
| 3 | 80.0 | 78.21 | 97.8 | 2.5 |
| [1] | Chopra I., Roberts M., Microbiol. Mol.Biol.Rev., 2001,65, 232— 260 |
| [2] | Zhang X., Chen Z., Deng H., Yang K., Gong S., Lu W., Lan H., Asian J . Ecotoxicol., 2016,11, 44— 52 |
| [3] | Liu X., Huang D., Lai C., Zeng G., Qin L., Zhang C., Yi H., Li B., Deng R., Liu S., Zhang Y., Trends Anal.Chem., 2018,109, 260— 274 |
| [4] | Virolainen N. E., Pikkemaat M. G., Elferink J. W. A., Karp M. T., J. Agric. Food Chem., 2008,56, 11065— 11070 |
| [5] | Chen Y., Kong D., Liu L., Song S., Hua K., Xu C., Food Anal. Method, 2015,9, 1— 10 |
| [6] | Önal A., ., Food Chem 2011,127, 197— 203 |
| [7] | Yang X., Zhu S., Dou Y., Zhuo Y., Luo Y., Feng Y ., Talanta, 2014,122, 36— 42 |
| [8] | Sun X., Lei Y., Trends Anal.Chem., 2017,89, 163— 180 |
| [9] | Gao X., Du C., Zhuang Z., Chen W ., J. Mater. Chem. C, 2016,4, 6927— 6945 |
| [10] | Li Y. K., Yang T., Chen M. L., Wang J. H ., Talanta, 2018,180, 18— 24 |
| [11] | Shamsipur M., Molaei K., Molaabasi F., Hosseinkhani S., Alizadeh N., Alipour M., Moassess S ., . Sens Actuators B, 2018,257, 772— 782 |
| [12] | Jin H., Gui R., Sun J., Wang Y ., Talanta, 2018,176, 277— 283 |
| [13] | Yan X., Song Y., Zhu C., Li H., Du D., Su X., Lin Y., Anal.Chem., 2018,90, 2618— 2624 |
| [14] | Shi X., Wei W., Fu Z., Gao W., Zhang C., Zhao Q., Deng F., Lu X ., Talanta, 2019,194, 809— 821 |
| [15] | Chen C., Zhao J., Lu Y., Sun J., Yang X., Anal. Chem., 2018,90, 3505— 3511 |
| [16] | Zhong D., Yang K., Wang Y., Yang X ., Talanta, 2017,175, 217— 223 |
| [17] | Ruiz-Palomero C., Benitez-Martinez S., Soriano M. L., Valcarcel M., . Anal. Chim. Acta, 2017,974, 93— 99 |
| [18] | Feng Y., Zhong D., Miao H., Yang X ., Talanta, 2015,140, 128— 133 |
| [19] | Shi W., Guo F., Han M., Yuan S., Guan W., Li H., Huang H., Liu Y., Kang Z ., J. Mater. Chem. B, 2017,5, 3293— 3299 |
| [20] | Qian S Qiao L. n., Xu W., Jiang K., Wang Y., Lin H., ., Talanta, 2019,194, 598— 603 |
| [21] | Heffern M. C., Matosziuk L. M., Meade T. J., Chem. Rev., 2014,114, 4496— 4539 |
| [22] | Tan H., Ma C., Song Y., Xu F., Chen S., Wang L., Biosens. Bioelectron., 2013,50, 447— 452 |
| [23] | Zhou Z., Li X., Gao J., Tang Y., Wang Q ., J. Agric. Food Chem., 2019,67, 3871— 3878 |
| [24] | Li X., Ma H., Deng M., Iqbal A., Liu X., Li B., Liu W., Li J., Qin W ., J. Mater. Chem. C, 2017,5, 2149— 2152 |
| [25] | Shen Z., Zhang C., Yu X., Li J., Wang Z., Zhang Z., Liu B ., J. Mater. Chem. C, 2018,6, 9636— 9641 |
| [26] | Xu J., Shen X., Jia L., Zhou T., Ma T., Xu Z., Cao J., Ge Z., Bi N., Zhu T., Guo S., Li X ., J. Hazard. Mater., 2018,342, 158— 165 |
| [27] | Gui R., Jin H., Bu X., Fu Y., Wang Z., Liu Q., ., Coord. Chem. Rev 2019,383, 82— 103 |
| [28] | Zhou Y., Huang X., Liu C., Zhang R., Gu X., Guan G., Jiang C., Zhang L., Du S., Liu B., Han M. Y., Zhang Z ., Anal. Chem., 2016,88, 6105— 6109 |
| [29] | He W., Gui R., Jin H., Wang B., Bu X., Fu Y ., Talanta, 2018,178, 109— 115 |
| [30] | Li J. Y., Liu Y., Shu Q. W., Liang J. M., Zhang F., Chen X. P., Deng X. Y., Swihart M. T., Tan K. J., Langmuir., 2017,33, 1043— 1050 |
| [31] | Tan H., Chen Y ., Sens. Actuators B, 2012,173, 262— 267 |
| [1] | Yong ZHANG,Cheng SHEN,Zhirong XING,Guiqi CHEN,Zi LU,Zhibing HOU,Xuemei CHEN. Benzimidazole-Derived Fluorescence Enhancement Probe for Visual Detection of HClO † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2480. |
| [2] | ZHANG Chunyan,LUO Jianxin,LI Wenjun,OU Lijuan,YU Guipeng,PAN Chunyue. Preparation and Sensing Properties of Covalent-linked Europium Complex Monodisperse Polystyrene Microspheres† [J]. Chem. J. Chinese Universities, 2019, 40(1): 153. |
| [3] | ZHANG Lutao, ZHOU Guangming, LUO Dan, CHEN Rong. Rapid Detection on Chlortetracycline Residues in Honey by Surface-enhanced Raman Spectroscopy† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1662. |
| [4] | YU Xiaodan, LIN Xinchen, FENG Wei, LI Weiguang. One-step Preparation and UV-Fenton Properties of Fe3O4/TiO2@Bio-carbon Composities† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2500. |
| [5] |
LI Jian,XIE Linxia,LIANG Zupei,GUO Rong,LIU Chenyu,MA Shulan.
Luminescence Property and Detection Capability Towards Hg2+ of LEuH Composites with Mo |
| [6] | HU Xiaoyang, SU Dongping, LI Shimeng, WANG Zhipeng, ZHANG Lirong, LIU Ying, SONG Lianjun, CHEN Zhili, DING Songdong. Extraction of Am(Ⅲ) and Eu(Ⅲ) from Nitric Acid Solution with the Pyridylpyrazole Based Ligand C8-BPP [J]. Chem. J. Chinese Universities, 2017, 38(8): 1324. |
| [7] | DENG Min,FANG Xiaowei,GUO Xiali,HUANG Xueyong,LIU Xingxing,YU Tenghui,LUO Liping. Direct Detection of Tetracycline in Honey by Neutral Desorption-Extractive Electrospray Ionization Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2016, 37(8): 1430. |
| [8] | YANG Shuilan, SONG Pan, SHE Wenjie, YANG Tianlin. Mechanism of the Interaction Between a Phosphorus-containing Tripod Ligand Europium(Ⅲ) Complex and Bovine Serum Albumin† [J]. Chem. J. Chinese Universities, 2015, 36(7): 1254. |
| [9] | HUANG Song, DING Songdong, JIN Yongdong, MA Lijian, XIA Chuanqin, LI Shoujian, WU Yuxuan, HUANG Huang. Extraction of Eu(Ⅲ) and Am(Ⅲ) from Nitric Acid Solution with Bisdiglycolamides† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1369. |
| [10] | ZHOU Ying-Jie, ZHU Hai-E, ZHANG Jun, WU Shui-Lin, YI Chang-Feng, XU Zu-Shun. Preparation and Micellization Behavior of Europium-containing Fluorescent Amphiphilic Graft Copolymer [J]. Chem. J. Chinese Universities, 2013, 34(3): 732. |
| [11] | PAN Qing-Hua, XU Yi-Zhuang, LAI Guo-Qiao, LIU Shao-Xuan, CHEN Jing, YANG Li-Min, ZHANG Yuan-Fu, WENG Shi-Fu, WU Jin-Guang. Application of FT-mid-IR Fiber Optics Technique in the Chemical Reaction [J]. Chem. J. Chinese Universities, 2012, 33(09): 1964. |
| [12] | HU Ji, WANG Jiang-Tao*. Adsorption of Tetracycline on Marine Sediment [J]. Chem. J. Chinese Universities, 2010, 31(2): 320. |
| [13] | LIU Sheng-Gui1,2*, WANG Hui-Hui1, HE Pei1, SHI Jian-Xin1, GONG Meng-Lian1. Synthesis and Luminescence of Eu(Ⅲ) Complexes Based on 4,4′- Bis(4″,4″,4″-trifluoro -1″,3″-dioxobutyl)-biphenyl [J]. Chem. J. Chinese Universities, 2009, 30(12): 2363. |
| [14] | TANG Huai-Jun1, TANG Hao2, ZHANG Zhi-Guo1, YUAN Ji-Bing1,3, ZHANG Ke-Li1*. Synthesis and Luminescent Properties of an Organic Europium(Ⅲ) Complex Containing a Carbazole and an Oxadiazole Fragments as Carrier-transporting Functional Groups [J]. Chem. J. Chinese Universities, 2008, 29(5): 871. |
| [15] |
JIANG Wei1,2, TANG Yu1, LIU Wei-Sheng1, TAN Min-Yu1.
Intercalation Assembly and Luminescent Properties of a Novel Luminescent Supramolecular Composite Material, Eu(Ⅲ) Complex with Tetrapodal LigandMontmorillonite [J]. Chem. J. Chinese Universities, 2006, 27(12): 2243. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||