

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (3): 592.doi: 10.7503/cjcu20180408
• Polymer Chemistry • Previous Articles Next Articles
CHEN Dongdong2, SONG Wenzhi3, LI Hui3, HE Dan1, SUN Junqi2, YIN Wanzhong1,*( )
)
Received:2019-06-04
Online:2019-03-10
Published:2019-01-24
Contact:
YIN Wanzhong
E-mail:yinwz@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
CHEN Dongdong, SONG Wenzhi, LI Hui, HE Dan, SUN Junqi, YIN Wanzhong. Layer-by-layer Assembled Polymeric Complexes Films for High Loading and Differential Release of Macromolecular and Small Molecular Drugs†[J]. Chem. J. Chinese Universities, 2019, 40(3): 592.
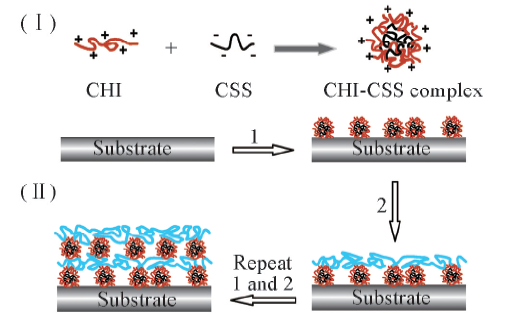
Scheme 1 Schematic illustration of the formation of CHI-CSS polyelectrolyte complexes(Ⅰ), and the layer-by-layer assembly process for the fabrication of CHI-CSS/HA polymeric films(Ⅱ)
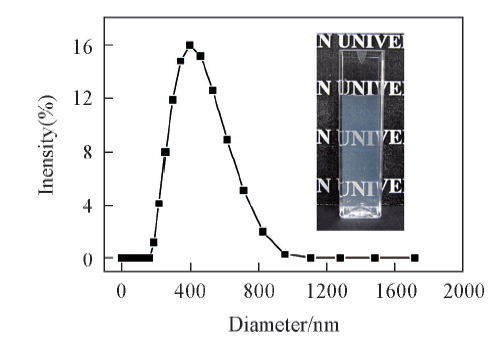
Fig.1 Hydrodynamic diameter distribution curve of the aqueous CHI-CSS complex dispersion at pH=6.0Inset is the photograph of the aqueous CHI-CSS complex dispersion.
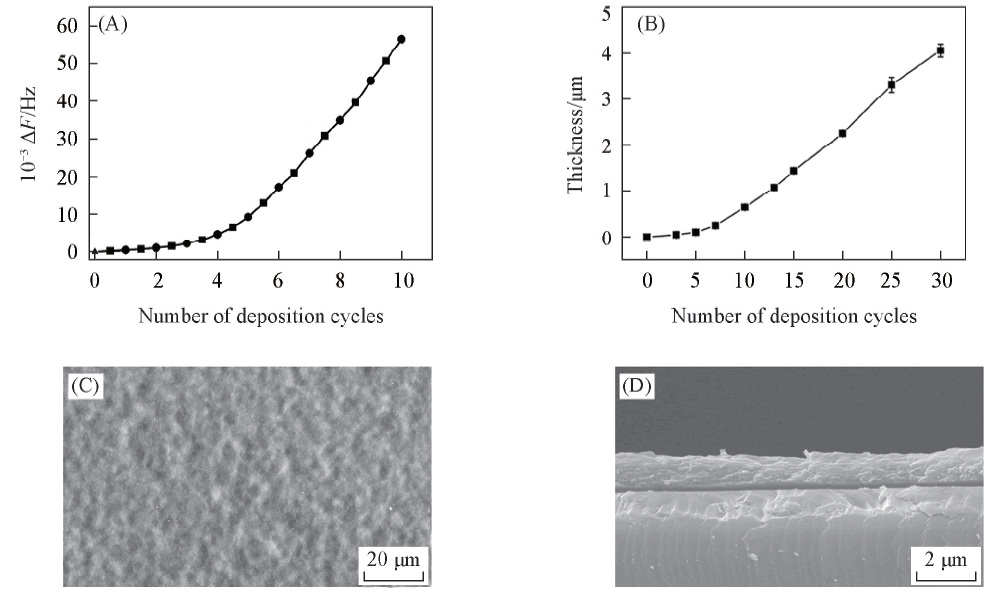
Fig.2 Typical QCM frequency decreases for the alternate deposition of CHI-CSS complexes(■) and HA(●)(A), thickness of CHI-CSS/HA films as a function of the number of deposition cycles(B) and top view(C) and cross-sectional(D) SEM images of (CHI-CSS/HA)×13 films
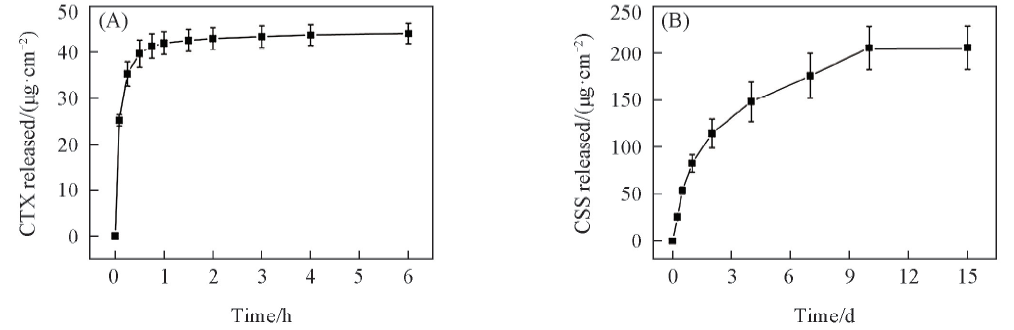
Fig.3 Time-depended release profile of CTX(A) and CSS(B) from (CHI-CSS/HA)×13 films in PBS(0.01 mol/L) containing lysozyme(2 mg/mL) at pH=7.4 and 37 ℃
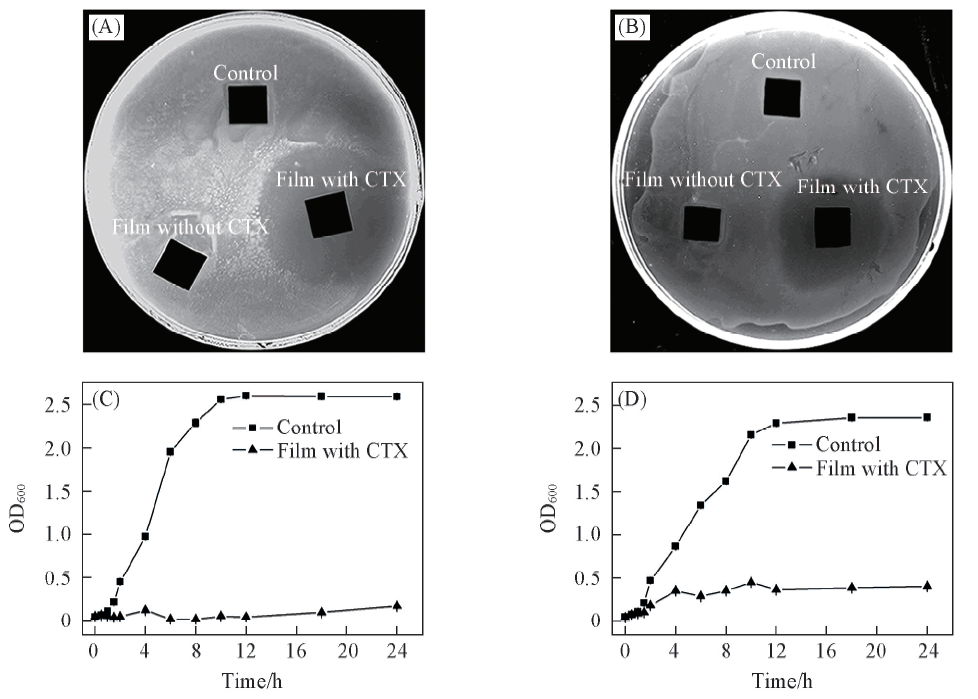
Fig.4 Inhibition of E. coli(A, C) and B. subtilis(B, D) growth by release of CTX from (CHI-CSS/HA)×13 films loaded with CTXAgar plates of E. coli.(A) and B. subtilis(B) treated with the control sample(bare silicon substrate) and (CHI-CSS/HA)×13 films with and without CTX. The growth curves of E. coli(C) and B. coli(D) incubated for 24 h with the control sample(bare silicon substrate) and (CHI-CSS/HA)×13 films with CTX.
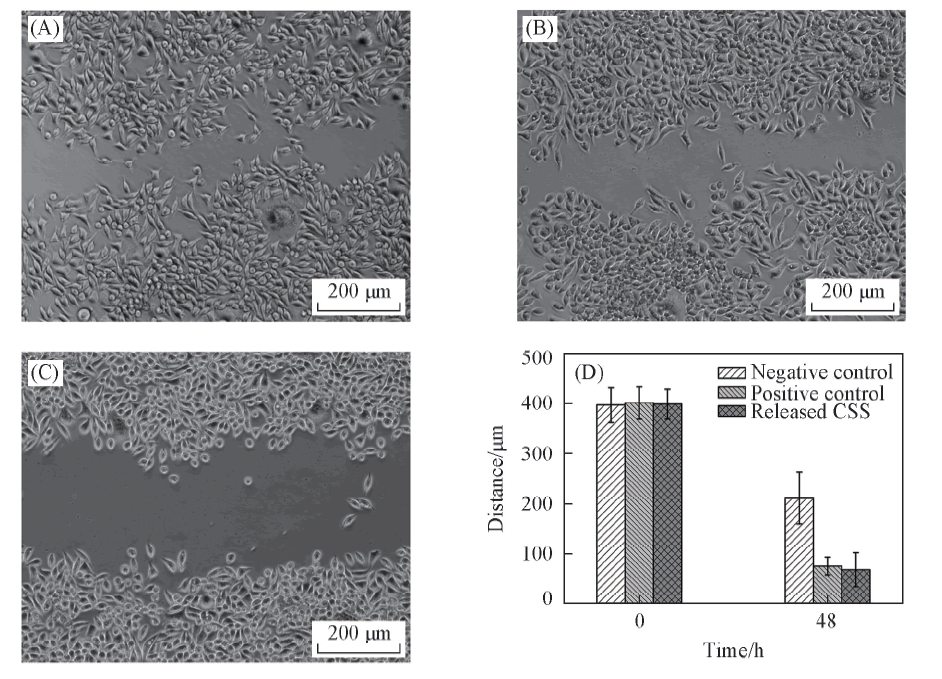
Fig.5 Bioactivity of released CSS from (CHI-CSS/HA)×13 films on wound healing in vitroThe mouse fibroblast L929 cells with wound gap incubated for 48 h with growth medium containing released CSS(A), free CSS(B) and blank PBS(C). (D) Statistical analysis of wound healing for L929 cells incubated with blank PBS(negative control), free CSS(positive control) and released CSS.
| [1] | Zhang H., Wang G., Yang H., Expert Opin. Drug Deliv., 2011, 8(2), 171—190 |
| [2] | Shukla A., Fuller R.C.., Hammond P. T.,J. Control Release, 2011, 155(2), 159—166 |
| [3] | Gilmore K. A., Lampley M. W., Boyer C., Harth E., Adv. Drug Dev. Rev., 2016, 98, 77—85 |
| [4] | He C., Tang Z., Tian H., Chen X., Adv. Drug Dev.Rev., 2016, 98, 64—76 |
| [5] | Teo P. Y., Cheng W., Hedrick J. L., Yang Y. Y., Adv. Drug Dev. Rev., 2016, 98, 41—63 |
| [6] | Pugliese E., Coentro J. Q., Zeugolis D. I., Adv. Mater., 2019, 30(13), 1704324 |
| [7] | Layman H., Sacasa M., Murphy A. E., Murphy A. M., Pham S. M., Andreopoulos F. M., Acta Biomater., 2009, 5(1), 230—239 |
| [8] | Wei L., Cai C., Lin J., Chen T., Biomaterials, 2009, 30(13), 2606—2613 |
| [9] | Cao Z., Zhou X., Wang G., ACS Appl.Mater. Interfaces, 2016, 8(42), 28888—28896 |
| [10] | Wu X., He C., Wu Y., Chen X., Biomaterials, 2016, 75, 148—162 |
| [11] | Cheng D., Sefton M. V., Tissue Engineering Part A, 2009, 15(8), 1929—1939 |
| [12] | Manna U., Patil S., Langmuir, 2009, 25(18), 10515—10522 |
| [13] | Kim C. S., Mout R., Zhao Y., Yeh Y. C., Tang R., Jeong Y., Duncan B., Hardy J. A., Rotello V. M., Bioconjugate Chem., 2015, 26(5), 950—954 |
| [14] | Fang J. H., Lai Y. H., Chiu T. L., Chen Y. Y., Hu S. H., Chen S. Y., Adv. Heathcare Mater., 2014, 3(8), 1250—1260 |
| [15] | Ke X. Y., Ng V. W. L., Gao S. J., Tong Y. W., Hedrick J. L., Yang Y. Y., Biomaterials, 2014, 35(3), 1096—1108 |
| [16] | Scarano W., de Souza P., Stenzel M. H., Biomater. Sci., 2015, 3(1), 163—174 |
| [17] | Chen A. M., Zhang M., Wei D., Stueber D., Taratula O., Minko T., He H., Small, 2009, 5(23), 2673—2677 |
| [18] | Wang H., Zhao Y., Wu Y., Hu Y. L., Nan K., Nie G., Chen H., Biomaterials, 2011, 32(32), 8281—8290 |
| [19] | Mi Y., Zhao J., Feng S. S., J. Control Release, 2013, 169(3), 185—192 |
| [20] | Mo R., Jiang T., Gu Z., Nanomedicine, 2014, 9(8), 1117—1120 |
| [21] | Assanhou A. G., Li W., Zhang L., Xue L., Kong L., Sun H., Mo R., Zhang C., Biomaterials, 2015, 73, 284—295 |
| [22] | Saurer E. M., Flessner R. M., Sullivan S. P., Prausnitz M. R., Lynn D. M., Biomacromolecules, 2010, 11(11), 3136—3143 |
| [23] | Shah N. J., Macdonald M. L., Beben Y. M., Padera R. F., Samuel R. E., Hammond P. T., Biomaterials, 2011, 32(26), 6183—6193 |
| [24] | Sun J. K., Wang X. F., Prog. Chem., 2009, 21, 2682—2688 |
| [25] | Smith R. C., Riollano M., Leung A., Hammond P. T., Angew. Chem. Int. Ed., 2009, 48(47), 8974—8977 |
| [26] | Boudou T., Crouzier T., Ren K., Blin G., Picart C., Adv. Mater., 2010, 22(4), 441—467 |
| [27] | Wohl B.M.., Engbersen J. F.,J. Control Release, 2012, 158(1), 2—14 |
| [28] | Zelikin A. N., ACS Nano, 2010, 4(5), 2494—2509 |
| [29] | Ariga K., Lvov Y. M., Kawakami K., Ji Q., Hill J. P., Adv. Drug Dev. Rev., 2011, 63(9), 762—771 |
| [30] | Fujie T., Okamura Y., Takeoka S., Adv. Mater., 2007, 19(21), 3549—3553 |
| [31] | Chen D., Wu M., Chen J., Zhang C., Pan T., Zhang B., Tian H., Chen X., Sun J., Langmuir, 2014, 30(46), 13898—13906 |
| [32] | Wang L., Chen D., Sun J., Langmuir, 2009, 25(14), 7990—7994 |
| [33] | Shukla A., Avadhany S. N., Fang J. C., Hammond P. T., Small, 2010, 6(21), 2392—2404 |
| [34] | Macdonald M. L., Samuel R. E., Shah N. J., Padera R. F., Beben Y. M., Hammond P. T., Biomaterials, 2011, 32(5), 1446—1453 |
| [35] | Anandhakumar S., Raichur A. M., Acta Biomater., 2013, 9(11), 8864—8874 |
| [36] | Chen D. D., Chen J., Tian H. Y., Chen X. S., Sun J. Q., Chem. J. Chinese Universities, 2015, 36(11), 2342—2348 |
| (陈栋栋, 陈杰, 田华雨, 陈学思, 孙俊奇. 高等学校化学学报, 2015, 36(11), 2342—2348) | |
| [37] | Mandapalli P. K., Labala S., Jose A., Bhatnagar S., Janupally R., Sriram D., Venuganti V. V. K., AAPS Pharm. Sci. Tech., 2017, 18(3), 809—820 |
| [38] | Zhang L., Sun J., Chem. Commun., 2009, (26), 3901—3903 |
| [39] | Guo Y., Geng W., Sun J., Langmuir, 2008, 25(2), 1004—1010 |
| [40] | Liu X., Dai B., Zhou L., Sun J., J. Mater. Chem., 2009, 19(4), 497—504 |
| [41] | Liu F., Wang X., Ye K. Q., Qiu L. Y., Shen J. C., Chem. J. Chinese Universities, 2011, 32(4), 990—994 |
| (刘峰, 王旭, 叶开其, 裘令瑛, 沈家骢. 高等学校化学学报, 2011, 32(4), 990—994) | |
| [42] | Zou X., Foong W., Cao T., Bay B., Ouyang H., Yip G., J. Dental Res., 2004, 83(11), 880—885 |
| [43] | Liu X., Zhou L., Geng W., Sun J., Langmuir, 2008, 24(22), 12986—12989 |
| [44] | Zhang Y., Sun J., ACS Nano, 2015, 9(7), 7124—7132 |
| [45] | Nordtveit R. J., Vårum K. M., Smidsrød O., Carbohydr. Polym., 1994, 23(4), 253—260 |
| [46] | Gilbert M. E., Kirker K. R., Gray S. D., Ward P. D., Szakacs J. G., Prestwich G. D., Orlandi R. R., The Laryngoscope, 2004, 114(8), 1406—1409 |
| [47] | Im A. R., Park Y., Kim Y. S., Biol. Pharm. Bull., 2010, 33(8), 1268—1273 |
| [1] | Jinhan Sheng, Qizhen Zheng, Ming Wang. Non-viral delivery of CRISPR/Cas9 Genome Editing [J]. Chem. J. Chinese Universities, 2022, 43(Album-4): 20220344. |
| [2] | WU Yushuai, SHANG Yingxu, JIANG Qiao, DING Baoquan. Research Progress of Controllable Self-assembled DNA Origami Structure as Drug Carrier [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220179. |
| [3] | REN Yushuang, GUO Yuanyuan, LIU Xueyi, SONG Jie, ZHANG Chuan. Platinum(Ⅳ) Prodrug-grafted Phosphorothioate DNA and Its Self-assembled Nanostructure for Targeted Drug Delivery [J]. Chem. J. Chinese Universities, 2020, 41(8): 1721. |
| [4] | ZHANG Kaixiang, LIU Junjie, SONG Qiaoli, WANG Danyu, SHI Jinjin, ZHANG Haiyue, LI Jinghong. Multifunctional DNA Nanoflowers for Autophagy Inhibition and Enhanced Antitumor Chemotherapy† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1461. |
| [5] | HOU Chunxi, LI Yijia, WANG Tingting, LIU Shengda, YAN Tengfei, LIU Junqiu. Application of Elastin-like Polypeptides in Supramolecular Assembly † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1163. |
| [6] | ZHAO Yu, CAO Wanqing, LIU Yang. Recent Advances in Polymeric Nano-sized Carrier Systems † [J]. Chem. J. Chinese Universities, 2020, 41(5): 909. |
| [7] | FAN Ye, LI Qian, FANG Yun, XIA Yongmei. Fabrication of Lamellar Liquid Crystals of Conjugated Linoleic Acid as Drug Delivery Systems † [J]. Chem. J. Chinese Universities, 2020, 41(4): 750. |
| [8] | WANG Xinghuo,TANG Jun,YANG Yingwei. Mesoporous Silica Nanoparticles-Based Stimuli-Responsive Drug Delivery Systems Gated by Polymers † [J]. Chem. J. Chinese Universities, 2020, 41(1): 28. |
| [9] | LIU Xiaozhou, WANG Yujie, LIU Yaozu, LI Zonglong, LI Hui, FANG Qianrong, JIN Yongri. A Covalent Organic Framework with High Surface Area for Drug Delivery † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1813. |
| [10] | SHI Mai,JIANG Rui,CUI Xinxia,ZHANG Xin,SHEN Shigang,DING Liang,PAN Xuefeng. Preparation, Structure and Pharmaceutical Analysis of Protamine-siRNA Complexes† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1164. |
| [11] | ZHAO Junqiang, YAN Caixia, CHEN Ze, YANG Ning, FENG Xia, ZHAO Yiping, CHEN Li. Synthesis and Self-assembly Properties of Intracellular Redox Bioresponsive Block Copolymers with Hepatoma-targeting Groups† [J]. Chem. J. Chinese Universities, 2018, 39(7): 1592. |
| [12] | GAO Yan, ZHANG Hua, ZHANG Wen, LI Xiangpeng, YUE Pan, LI Wei. Preparation of Micro/Nano-fibers Membranes Encapsulated with Dual Drugs by Emulsion Electrospun and Controlled Release of Hydrophilic and Hydrophobic Drugs† [J]. Chem. J. Chinese Universities, 2018, 39(3): 575. |
| [13] | YANG Zechuan, LI Fan, HUANG Qingrong, ZHANG Guo, SHI Tongfei. Synthesis and Properties of the Amino Acid Functionalized Curcumin/His-Pectin Colloidal Particles† [J]. Chem. J. Chinese Universities, 2016, 37(2): 381. |
| [14] | LUO Zewei, WANG Yimin, LIU Kunping, WEI Fujing, LI Yu, DUAN Yixiang. Preparation of a Functionalized Graphene and Its Role as Delivery Carrier for Anti-cancer Drug† [J]. Chem. J. Chinese Universities, 2016, 37(10): 1900. |
| [15] | ZHANG Fada, LIU Yi, XU Jingcheng, LI Shengjuan, WANG Xiunan, SUN Yue, ZHAO Xinluo. Molecular Dynamics Study on Binding Strength and Conformation of Dendrimer-based Drug Delivery Systems† [J]. Chem. J. Chinese Universities, 2015, 36(6): 1156. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||