

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (8): 1647.doi: 10.7503/cjcu20170806
• Analytical Chemistry • Previous Articles Next Articles
XIA Yuting, JIANG Bo, WU Qing, HU Qinghong, YUAN Zeli*( )
)
Received:2017-12-11
Online:2018-08-10
Published:2018-06-04
Contact:
YUAN Zeli
E-mail:zlyuan2002@126.com
Supported by:CLC Number:
TrendMD:
XIA Yuting, JIANG Bo, WU Qing, HU Qinghong, YUAN Zeli. Synthesis and Application of Colorimetric Probe for the Simultaneous Detection of Sulphion and Homocysteine/Cysteine in the Serum Samples†[J]. Chem. J. Chinese Universities, 2018, 39(8): 1647.
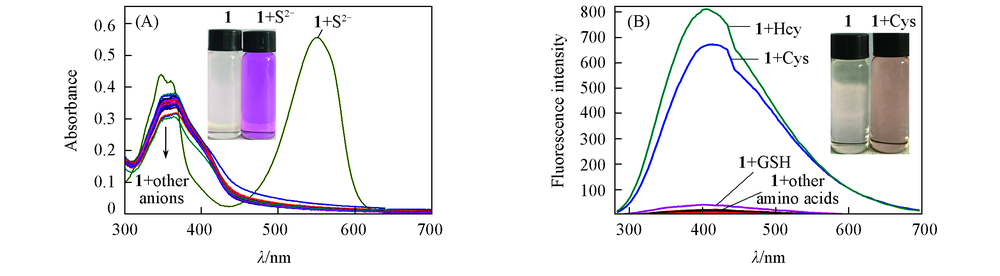
Fig.1 Absorbance spectra change of probe 1(20 μmol/L) in Tris-HCl(10 mmol/L, pH=7.4, 50% ethanol) upon individual addition of 100 μmol/L different anions(A) and fluorescence spectra change of probe 1(20 μmol/L) in Tris-HCl(10 mmol/L, pH=7.4, 50% ethanol) upon individual addition of 100 μmol/L different amino acids(B)Inset: color changes of probe 1 solution before(left) and after(right) the addition of S2-(A) and Cys(B). (A) Other anions include F-, Cl-, Br-, I-, NO3-, CN-, PO43-, HPO42-, H2PO4-, SCN-, SO42-, HSO4-, CO32-, HCO3-, ClO3-, IO4-, CH3COO-; (B) other amino acids include glycine, serine, aspartic acid, glutathione, tyrosine, glutamic acid, arginine, tryptophan, valine, histidine, methionine, phenylalanine, phenylglycine, alanine, leucine, methionine.
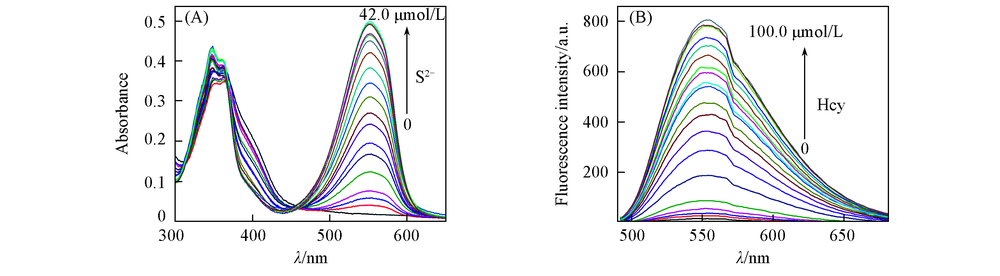
Fig.2 Absorbance spectra of probe 1(20 μmol/L) in the presence of S2-(0—42 μmol/L)(A) and luorescece spectra of probe 1(20 μmol/L) in the presence of Hcy(0—100 μmol/L)(B)
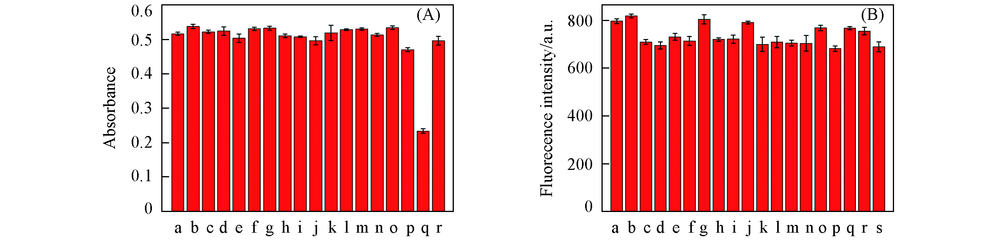
Fig.3 Absorbance(552 nm) changes of probe 1(20 μmol/L) in Tris-HCl(10 mmol/L, pH=7.40, 50% ethanol) solution upon individual addition of 44 μmol/L miscellaneous anions and followed by further addition of 44 μmol/L S2-(A) and fluorescence intensity(552 nm) changes of probe 1(20 μmol/L) in Tris-HCl(10 mmol/L, pH=7.40, 50% ethanol) solution upon individual addition of 100 μmol/L amino acids and followed by further addition of 100 μmol/L of Hcy(A) a. S2-; b. S2-+F-; c. S2++SCN-; d. S2-+Cl-; e. S2-+SO42-; f. S2-+Br-; g. S2-+I-; h. S2-+HSO3-; i. S2-+NO3-; j. S2-+CN-; k. S2-+CO32-; l. S2-+PO43-; m. S2-+HCO3-; n. S2-+HPO42-; o. S2-+ClO3-; p. S2-+IO4-; q. S2-+H2PO4-; r. S2-+Ac-. (B) a. Hcy; b. Hcy+GSH; c. Hcy+Try; d. Hcy+Glu; e. Hcy+Arg; f. Hcy+Gly; g. Hcy+Trp; h. Hcy+Asn; i. Hcy+Val; j. Hcy+Hcy; k. Hcy+His; l. Hcy+Met; m. Hcy+Ser; n. Hcy+Phe; o. Hcy+Phg; p. Hcy+Cys; q. Hcy+Ala; r. Hcy+Leu; s. Hcy+Mts.
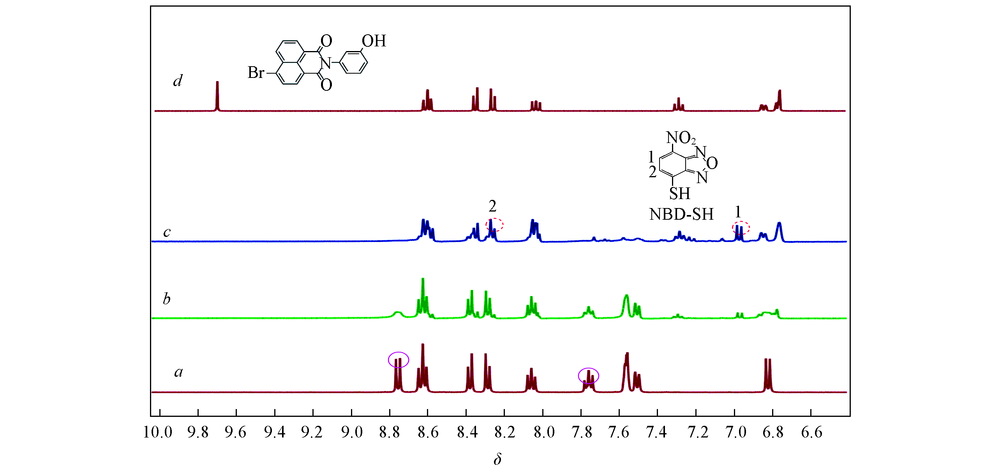
Fig.4 Partial 1H NMR spectra of probe 1(a), product of probe 1 with 0.835 mol/L(b) and 1.67 mol/L(c) S2- in DMSO-d6/D2O(volume ratio 9:1) and the intermidate(d)

Fig.6 Color changes of the filter paper coated with probe 1(20 μmol/L) used for the detection of S2-(A) and Cys(B)(A) a. Only probe 1; b. 1+F-; c. 1+Cl-.; d. 1+Br-; e. 1+ I-; f. 1+NO3-; g. 1+S2-; h. 1+SO42-; i. 1+HSO4-; j. 1+CO32-; k.1+HCO3-; l. 1+ClO32-; m. 1+PO43-; n. 1+H2PO4-; o. 1+HPO42-; p. 1+SCN-; q. 1+IO4-; r. 1+CN-; s. 1+AcO-. (B) a. Only probe 1; b. 1+Hcy; c. 1+Met; d. 1+Arg; e. 1+Gly; f. 1+Trp; g. 1+GSH; h. 1+Ser; i. 1+His; j. 1+Lys; k. 1+Val; l. 1+Phg; m. 1+Phe; n. 1+Ala; o. 1+cys; p. 1+Tyr; q. 1+Asn; r. 1+Leu; s. 1+Glu.

Fig.7 Color changes of the filter paper coated with probe 1(20 μmol/L) used for the detection of S2-(A) and Cys(B)(A)c(S2-)/(μmol·L-1): a. 0; b. 6.0; c. 12.0; d. 18.0; e. 24.0; f. 30.0; g. 40.0. (B) c(Cys)/(μmol·L-1): a. 0; b. 10.0; c. 30.0; d. 40.0; e. 50.0; f. 60.0; g. 80.0.
| Analyte | c(Spiked)/(μmol·L-1) | Quantities measured/(μmol·L-1) | Average recovery(%) | RSD(%)(n=5) |
|---|---|---|---|---|
| 4 | 3.9 | 96.4 | 2.0 | |
| 6 | 6.3 | 105.8 | 7.6 | |
| 8 | 7.6 | 95.3 | 2.2 | |
| Cysb | 14 | 13.8 | 98.2 | 2.6 |
| 16 | 15.9 | 99.5 | 3.6 | |
| 18 | 17.7 | 98.3 | 2.3 | |
| Hcyb | 16 | 15.9 | 99.3 | 5.3 |
| 18 | 17.9 | 99.4 | 4.9 | |
| 20 | 19.4 | 97.2 | 6.7 |
Table 1 Determination of S2-, Hcy and Cys in the serum samples
| Analyte | c(Spiked)/(μmol·L-1) | Quantities measured/(μmol·L-1) | Average recovery(%) | RSD(%)(n=5) |
|---|---|---|---|---|
| 4 | 3.9 | 96.4 | 2.0 | |
| 6 | 6.3 | 105.8 | 7.6 | |
| 8 | 7.6 | 95.3 | 2.2 | |
| Cysb | 14 | 13.8 | 98.2 | 2.6 |
| 16 | 15.9 | 99.5 | 3.6 | |
| 18 | 17.7 | 98.3 | 2.3 | |
| Hcyb | 16 | 15.9 | 99.3 | 5.3 |
| 18 | 17.9 | 99.4 | 4.9 | |
| 20 | 19.4 | 97.2 | 6.7 |
| [1] | Erinn C.H., James R. H., Alison B., Chris R. R., Helmut B., Rory W., Wenying Y., José M. G., Kimberly M., Samantha B. J., Ronald P. K., William B. W., Mary A. M., Science, 2006, 314, 649—652 |
| [2] | Cheng X.H., Zhong Z. C., Li W. N., Chin. J. Luminescience, 2017, 38, 768—779 |
| (程晓红, 钟志成, 李望南. 发光学报, 2017, 38, 768—779) | |
| [3] | Wen X., Yuan Z.L., Hu Q. H., Jiang B., Chin. J. Mol. Science, 2015, 31, 509—515 |
| (文旭, 袁泽利, 胡庆红, 江波. 分子科学学报, 2015, 31, 509—515) | |
| [4] | Patnaik P.A., J. Haz. Mat., 2007, 149, 532 |
| [5] | Wang K., Liu Z.L., Jiang K., Acta Chim. Sinica, 2014, 72, 590—594 |
| (王魁, 刘自力, 蒋凯. 化学学报, 2014, 72, 590—594) | |
| [6] | Yue Y.K., Yin C. X., Huo F. J., Chao J. B., Zhang Y. B., Sensor. Actuat. B, 2016 , 223, 496—500 |
| [7] | Yang J.J., Yu Y. W., Wang B. X., Jiang Y. L., Chem. J. Chinese Universities, 2017, 38(7), 1198—1202 |
| (杨敬敬, 俞岳文, 王炳祥, 江玉亮. 高等学校化学学报, 2017, 38(7), 1198—1202) | |
| [8] | Wang Q.Q., Wang H., Huang J. X., Li N., Gu Y. Q., Wang P., Sensor. Actuat. B, 2017, 253, 400—406 |
| [9] | Sanskriti I., Upadhyay K.K., New J. Chem., 2017, 41, 4316—4321 |
| [10] | Burford N., Eelman D.M., Mahony D. E., Morash M., Chem. Commun., 2003, (1), 146—147 |
| [11] | Lee P.T., Lowinsohn D., Compton R. G., Electroanalysis, 2014, 26, 1488—1496 |
| [12] | Zhang X., Yan Y.C., Hang Y. D., Wang J., Hua J. L., Tian H, Chem. Commun., 2017, 53, 5760—5763 |
| [13] | Liu C.H., Li Y. H., Qi F. P., Long L. P., Yang R. H, Sci. Sin. Chim., 2017, 47, 1015—1021 |
| (刘长辉, 李银辉, 齐风佩, 龙立平, 杨荣华. 中国科学: 化学, 2017, 47, 1015—1021) | |
| [14] | Liu K.Y., Shang H. M., Kong X. Q., Lin W. Y., J. Mater. Chem. B, 2017, 5, 3836—3841 |
| [15] | Yuan Z.L., Wu Q., Yang X. B., Hu Q. H., Zhang M. Q., Chin. J. Org. Chem., 2011, 31(10), 1698—1702 |
| (袁泽利, 吴庆, 杨兴变, 胡庆红, 张铭钦. 有机化学, 2011, 31(10), 1698—1702) | |
| [16] | Cao S., Fang A.L., Huang Z. M., Zhang Z., Chem. J. Chinese Universities, 1997, 18(6), 894—897 |
| (曹松, 樊爱龙, 黄自明, 张正. 高等学校化学学报, 1997, 18(6), 894—897) | |
| [17] | Yu G.Q., Cao Y. P., Liu H. M., Wu Q., Hu Q. H., Jiang B., Yuan Z. L., Sensor. Actuat. B, 2017, 245, 803—814 |
| [18] | Yang J., Yuan Z.L., Yu G. Q., He S. L., Wu Q., Hu Q. H., Jiang B., Wei G., J. Fluorescence, 2016, 26, 43—51 |
| [19] | Leticia A.M., Taylor F. P., Ryan J. H., Lev N. Z., Michael D. P., J. Org. Chem., 2013, 78, 6550—6557 |
| [20] | Bae J., Choi J., Park T.J., Chang S. K., Tetrahedron Lett., 2014, 55, 1171—1174 |
| [21] | Ding S.S., Feng W. Y., Feng G. Q., Sensor. Actuat. B, 2017, 238, 619—625 |
| [1] | LI Zhiguang, QI Guodong, XU Jun, DENG Feng. Role of Catalyst Acidity in Glucose Conversion over Sn-Al-β Zeolite as Studied by Solid-state NMR [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220138. |
| [2] | ZHAO Yongmei, MU Yeshu, HONG Chen, LUO Wen, TIAN Zhiyong. Bis-naphthalimide Derivatives for Picronitric Acid Detection in Aqueous Solution [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210765. |
| [3] | TANG Qian, DAN Feijun, GUO Tao, LAN Haichuang. Synthesis and Application of Quinolinone-coumarin-based Colorimetric Fluorescent Probe for Recognition of Hg2+ [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210660. |
| [4] | WANG Di, ZHONG Keli, TANG Lijun, HOU Shuhua, LYU Chunxin. Synthesis of Schiff-based Covalent Organic Framework and Its Recognition of I ‒ [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220115. |
| [5] | HUANG Shan, YAO Jiandong, NING Gan, XIAO Qi, LIU Yi. Efficient Determination of Alkaline Phosphatase Activity Based on Graphene Quantum Dots Fluorescent Probes [J]. Chem. J. Chinese Universities, 2021, 42(8): 2412. |
| [6] | LI Anran, ZHAO Bing, KAN Wei, SONG Tianshu, KONG Xiangdong, BU Fanqiang, SUN Li, YIN Guangming, WANG Liyan. ON-OFF-ON Double Colorimetric and Fluorescent Probes Based on Phenanthro[9,10-d]imidazole Derivatives and Their Living Cells Imaging [J]. Chem. J. Chinese Universities, 2021, 42(8): 2403. |
| [7] | YANG Xinjie, LAI Yanqiong, LI Qiuyang, ZHANG Yanli, WANG Hongbin, PANG Pengfei, YANG Wenrong. An Enzyme-free and Label-free Fluorescent Probe for Detection of Microcystin-LR Based on Circular DNA-Silver Nanoclusters [J]. Chem. J. Chinese Universities, 2021, 42(12): 3600. |
| [8] | ZHANG Xiaorong, CHEN Lanlan, HU Shanwen. Advances in Bacteria Biosensing Based on Molecular Recognition [J]. Chem. J. Chinese Universities, 2021, 42(11): 3468. |
| [9] | CHEN Weiju, CHEN Shiya, XUE Caoye, LIU Bo, ZHENG Jing. Fluorescent Probe for Hypoxia-triggered Imaging and Cancer Therapy [J]. Chem. J. Chinese Universities, 2021, 42(11): 3433. |
| [10] | WANG Baichun, YUAN Yuxin, YAN Yinghua, DING Chuanfan, TANG Keqi. Glucose-6-phosphate Functionalized Hydrophilic Magnetic Probe: a Dual-purpose Affinity Material for Effective Separation and Enrichment of Glycopeptides/Phosphopeptides [J]. Chem. J. Chinese Universities, 2021, 42(10): 3062. |
| [11] | WANG Mengmeng, LUAN Tianjiao, YANG Mingyan, LYU Jiajia, GAO Jie, LI Hongyu, WEI Gang, YUAN Zeli. Rhodamine Fluorescent Probe for Tumor Targeted Hypoxia-imaging as Intra-operative Navigators [J]. Chem. J. Chinese Universities, 2021, 42(10): 3071. |
| [12] | GONG Shaohua, ZHANG Xia, LI Na, TANG Bo. Recent Progress of Fluorescent Nanoprobes for Organelle pH Detection [J]. Chem. J. Chinese Universities, 2020, 41(9): 1933. |
| [13] | LIU Zhaoyang, SONG Yongxin, WANG Tianshu, SHAN Guiye. Preparation of Histidine Modified Prussian Blue and Its Interaction with Silver Ions† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1492. |
| [14] | HUANG Jialing,LIU Fengjiao,WANG Tingting,LIU Cuie,ZHENG Fengying,WANG Zhenhong,LI Shunxing. Nitrogen and Sulfur co-Doped Carbon Quantum Dots for Accurate Detection of pH in Gastric Juice† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1513. |
| [15] | DONG Qian, LI Zhaoqian, PENG Tianhuan, CHEN Zhuo, TAN Weihong. Progress on Aptamer for Cancer Theranostics [J]. Chem. J. Chinese Universities, 2020, 41(12): 2648. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||