

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (11): 2053.doi: 10.7503/cjcu20170321
• Physical Chemistry • Previous Articles Next Articles
FAN Jianxun1,2, JI Lifei1, REN Aimin1,*( )
)
Received:2017-05-23
Online:2017-11-10
Published:2017-10-30
Contact:
REN Aimin
E-mail:aimin_ren@yahoo. com
Supported by:CLC Number:
TrendMD:
FAN Jianxun, JI Lifei, REN Aimin. Theoretical Study on Charge Transport Properties of Copolymers of Diketopyrrolopyrrole and Oligo-thiophene†[J]. Chem. J. Chinese Universities, 2017, 38(11): 2053.
| Copolymer | Energy | m=1 | m=2 | m=3 | m=4 | m=∞ | Exp. |
|---|---|---|---|---|---|---|---|
| PDPP-2T | EHOMO/eV | -4.97 | -4.82 | -4.76 | -4.73 | -4.67 | -5.40[ |
| ELUMO/eV | -2.51 | -2.98 | -3.15 | -3.24 | -3.47 | -4.20[ | |
| Eg/eV | 2.46 | 1.84 | 1.61 | 1.49 | 1.20 | 1.20 | |
| PDPP-3T | EHOMO/eV | -4.86 | -4.74 | -4.70 | -4.68 | -4.62 | -5.30[ |
| ELUMO/eV | -2.60 | -2.94 | -3.06 | -3.12 | -3.29 | -4.00[ | |
| Eg/eV | 2.26 | 1.80 | 1.64 | 1.56 | 1.33 | 1.30 | |
| PDPP-4T | EHOMO/eV | -4.81 | -4.70 | -4.67 | -4.66 | -4.60 | -5.20[ |
| ELUMO/eV | -2.65 | -2.91 | -3.01 | -3.05 | -3.18 | -4.00[ | |
| Eg/eV | 2.16 | 1.79 | 1.66 | 1.61 | 1.42 | 1.20 | |
| PDPP-5T | EHOMO/eV | -4.78 | -4.67 | -4.65 | -4.64 | -4.59 | -5.12[ |
| ELUMO/eV | -2.68 | -2.89 | -2.96 | -2.99 | -3.10 | -3.55[ | |
| Eg/eV | 2.10 | 1.78 | 1.69 | 1.65 | 1.49 | 1.57 | |
| PDPP-6T | EHOMO/eV | -4.76 | -4.65 | -4.64 | -4.63 | -4.58 | -5.10[ |
| ELUMO/eV | -2.70 | -2.88 | -2.93 | -2.96 | -3.05 | -3.40[ | |
| Eg/eV | 2.06 | 1.77 | 1.71 | 1.67 | 1.55 | 1.70 |
Table 1 Calculated HOMO and LUMO levels and energy gaps at B3LYP-D3(BJ)/6-31G(d,p) level
| Copolymer | Energy | m=1 | m=2 | m=3 | m=4 | m=∞ | Exp. |
|---|---|---|---|---|---|---|---|
| PDPP-2T | EHOMO/eV | -4.97 | -4.82 | -4.76 | -4.73 | -4.67 | -5.40[ |
| ELUMO/eV | -2.51 | -2.98 | -3.15 | -3.24 | -3.47 | -4.20[ | |
| Eg/eV | 2.46 | 1.84 | 1.61 | 1.49 | 1.20 | 1.20 | |
| PDPP-3T | EHOMO/eV | -4.86 | -4.74 | -4.70 | -4.68 | -4.62 | -5.30[ |
| ELUMO/eV | -2.60 | -2.94 | -3.06 | -3.12 | -3.29 | -4.00[ | |
| Eg/eV | 2.26 | 1.80 | 1.64 | 1.56 | 1.33 | 1.30 | |
| PDPP-4T | EHOMO/eV | -4.81 | -4.70 | -4.67 | -4.66 | -4.60 | -5.20[ |
| ELUMO/eV | -2.65 | -2.91 | -3.01 | -3.05 | -3.18 | -4.00[ | |
| Eg/eV | 2.16 | 1.79 | 1.66 | 1.61 | 1.42 | 1.20 | |
| PDPP-5T | EHOMO/eV | -4.78 | -4.67 | -4.65 | -4.64 | -4.59 | -5.12[ |
| ELUMO/eV | -2.68 | -2.89 | -2.96 | -2.99 | -3.10 | -3.55[ | |
| Eg/eV | 2.10 | 1.78 | 1.69 | 1.65 | 1.49 | 1.57 | |
| PDPP-6T | EHOMO/eV | -4.76 | -4.65 | -4.64 | -4.63 | -4.58 | -5.10[ |
| ELUMO/eV | -2.70 | -2.88 | -2.93 | -2.96 | -3.05 | -3.40[ | |
| Eg/eV | 2.06 | 1.77 | 1.71 | 1.67 | 1.55 | 1.70 |
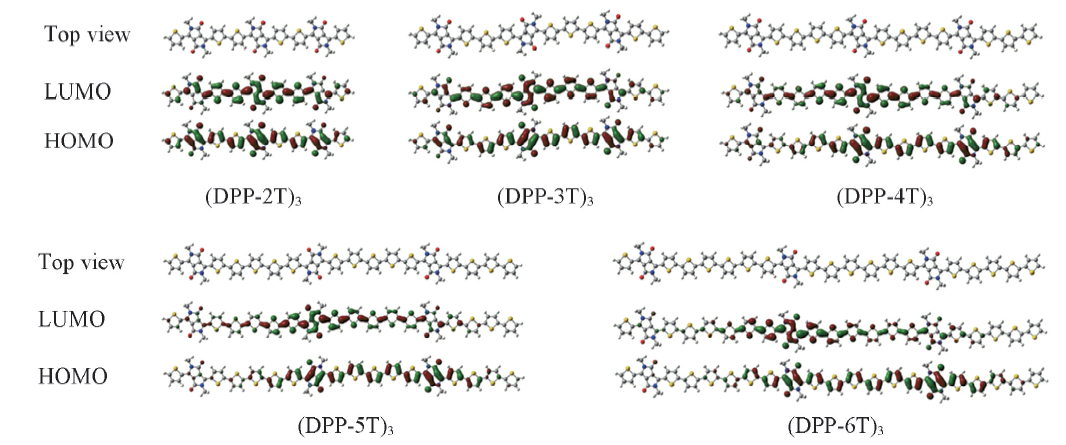
Fig.4 Top-view of the energy-minimized structure of trimers investigated by the B3LYP-D3(BJ)/6-31G(d,p) method with a visualization of the LUMO and HOMO molecular orbitals
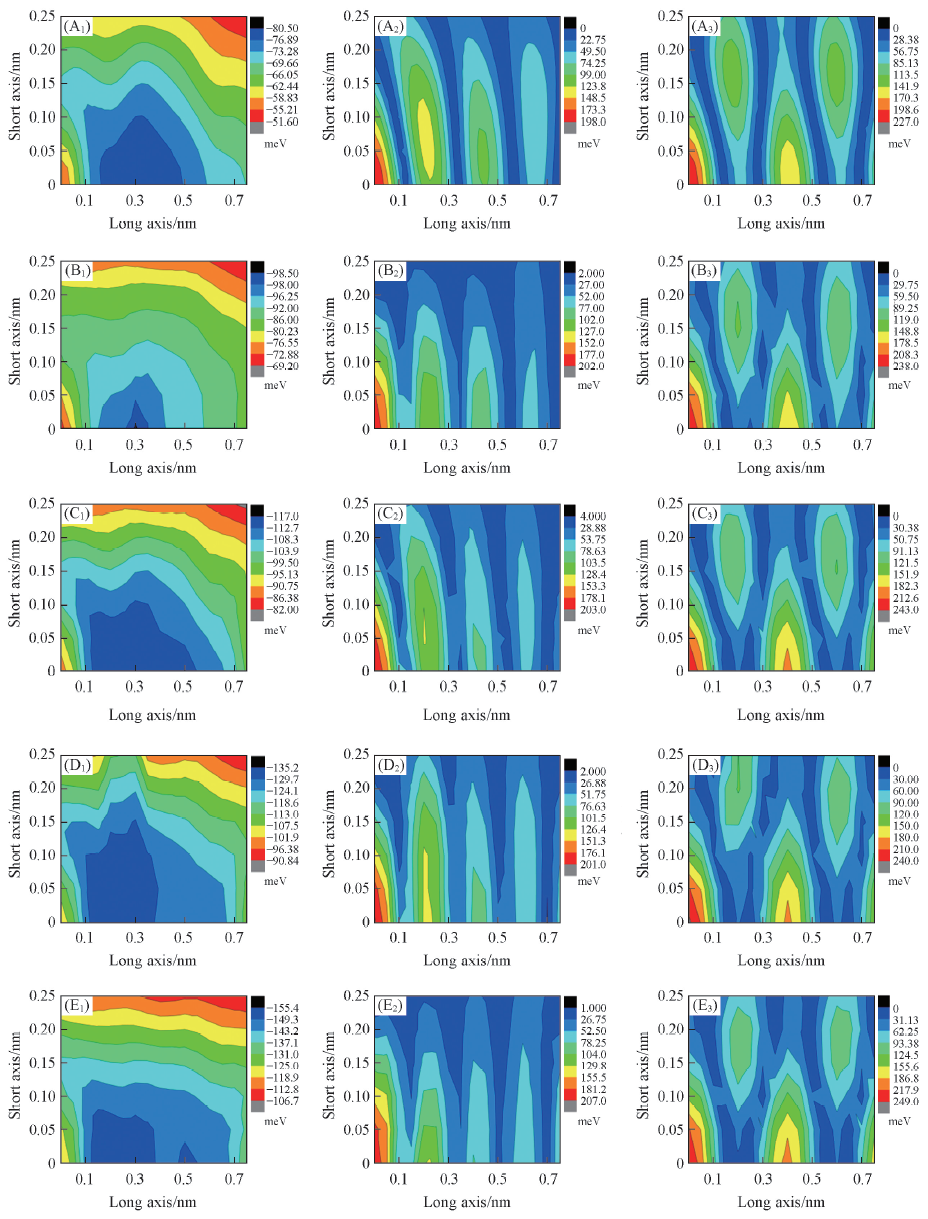
Fig.7 Interaction energies(A1—E1) and the hole(A2—E2) and electron(A3—E3) transfer integrals for (DPP-nT)3 dimers as a function of the long-axis shifts and short-axis shifts with an interplanar distance of 0.365 nm(A1)—(A3) (DPP-2T)3; (B1)—(B3) (DPP-3T)3; (C1)—(C3) (DPP-4T)3; (D1)—(D3) (DPP-5T)3;(E1)—(E3) (DPP-6T)3. Each scanning step is set to be 0.05 nm.
| Compound | r/nm | λh/eV | λe/eV | Vh/eV | Ve/eV | μh/ (cm2·V-1·s-1) | μe/ (cm2·V-1·s-1) | μe/μh |
|---|---|---|---|---|---|---|---|---|
| (DPP-2T)3 | 0.499 | 0.187 | 0.169 | 0.001 | 0.100 | 0.0005 | 3.787 | 7136.3 |
| (DPP-3T)3 | 0.473 | 0.151 | 0.135 | 0.059 | 0.057 | 1.515 | 1.709 | 1.13 |
| (DPP-4T)3 | 0.469 | 0.122 | 0.102 | 0.064 | 0.065 | 2.548 | 3.544 | 1.39 |
| (DPP-5T)3 | 0.403 | 0.102 | 0.077 | 0.098 | 0.016 | 5.888 | 0.237 | 0.04 |
| (DPP-6T)3 | 0.405 | 0.090 | 0.066 | 0.104 | 0.014 | 8.072 | 0.216 | 0.03 |
Table 2 Summary of the hole and electron reorganization energies(λh/λe), the hole and electron transfer integrals(Vh/Ve) and the hole and electron mobilities(μe/μh) based on the one-dimensional π-stacking model
| Compound | r/nm | λh/eV | λe/eV | Vh/eV | Ve/eV | μh/ (cm2·V-1·s-1) | μe/ (cm2·V-1·s-1) | μe/μh |
|---|---|---|---|---|---|---|---|---|
| (DPP-2T)3 | 0.499 | 0.187 | 0.169 | 0.001 | 0.100 | 0.0005 | 3.787 | 7136.3 |
| (DPP-3T)3 | 0.473 | 0.151 | 0.135 | 0.059 | 0.057 | 1.515 | 1.709 | 1.13 |
| (DPP-4T)3 | 0.469 | 0.122 | 0.102 | 0.064 | 0.065 | 2.548 | 3.544 | 1.39 |
| (DPP-5T)3 | 0.403 | 0.102 | 0.077 | 0.098 | 0.016 | 5.888 | 0.237 | 0.04 |
| (DPP-6T)3 | 0.405 | 0.090 | 0.066 | 0.104 | 0.014 | 8.072 | 0.216 | 0.03 |
| [1] | Tsumura A., Koezuka H., Ando T., Appl. Phys. Lett., 1986, 49(18), 1210-1212 |
| [2] | Virkar A. A., Mannsfeld S., Bao Z., Stingelin N., Adv. Mater., 2010, 22(34), 3857-3875 |
| [3] | Qu S. Y., Tian H., Chem. Commun., 2012, 48(25), 3039-3051 |
| [4] | Biniek L., Schroeder B. C., Nielsen C. B., McCulloch I., J. Mater. Chem., 2012, 22(30), 14803-14813 |
| [5] | Nielsen C. B., Turbiez M., McCulloch I., Adv. Mater., 2013, 25(13), 1859-1880 |
| [6] | Li Y. N., Sonar P., Murphy L., Hong W., Energy Environ. Sci., 2013, 6(6), 1684-1710 |
| [7] | Zhao Y., Guo Y. L., Liu Y. Q., Adv. Mater., 2013, 25(38), 5372-5391 |
| [8] | Turbiez M. G., Janssen R. A. J., Wienk M. M., Kirner H. J., Düggeli M., Tieke B., Zhu Y., Diketopyrrolopyrrole Polymers as Organic Semiconductors, WO 2008000664,2008-01-03 |
| [9] | Wienk M. M., Turbiez M., Gilot J., Janssen R. A. J., Adv. Mater., 2008, 20(13), 2556-2560 |
| [10] | Burgi L., Turbiez M., Pfeiffer R., Bienewald F., Kirner H. J., Winnewisser C., Adv. Mater., 2008, 20(11), 2217-2224 |
| [11] | Zoombelt A. P., Mathijssen S. G. J., Turbiez M. G. R., Wienk M. M., Janssen R. A. J., J. Mater. Chem., 2010, 20(11), 2240-2246 |
| [12] | Li Y. N., Sun B., Sonar P., Singh S. P., Org. Electron., 2012, 13(9), 1606-1613 |
| [13] | Kanimozhi C., Yaacobi-Gross N., Chou K. W., Amassian A., Anthopoulos T. D., Patil S., J. Am. Chem. Soc., 2012, 134(40), 16532-16535 |
| [14] | Bijleveld J. C., Zoombelt A. P., Mathijssen S. G. J., Wienk M. M., Turbiez M., de Leeuw D. M., Janssen R. A. J., J. Am. Chem. Soc., 2009, 131(46), 16616-16617 |
| [15] | Lee J. S., Son S. K., Song S., Kim H., Lee D. R., Kim K., Ko M. J., Choi D. H., Kim B., Cho J. H., Chem. Mater., 2012, 24(7), 1316-1323 |
| [16] | Li Y. N., Sonar P., Singh S. P., Soh M. S., van Meurs M., Tan J., J. Am. Chem. Soc., 2011, 133(7), 2198-2204 |
| [17] | Yi Z. R., Sun X. N., Zhao Y., Guo Y. L., Chen X. G., Qin J. G., Yu G., Liu Y. Q., Chem. Mater., 2012, 24(22), 4350-4356 |
| [18] | Yi Z. R., Ma L. C., Chen B., Chen D. G., Chen X. G., Qin J. G., Zhan X. W., Liu Y. Q., Ong W. J., Li J., Chem. Mater., 2013, 25(21), 4290-4296 |
| [19] | Shahid M., McCarthy-Ward T., Labram J., Rossbauer S., Domingo E. B., Watkins S. E., Stingelin N., Anthopoulos T. D., Heeney M., Chem. Sci., 2012, 3(1), 181-185 |
| [20] | Marcus R. A., Rev. Mod. Phys., 1993, 65(3), 599-610 |
| [21] | Schein L. B., McGhie A. R., Chem. J. Chinese Universities,2010, 31(4), 1631-1639 |
| [22] | Te Velde G., Bickelhaupt F. M., Baerends E. J., Fonseca Guerra C., van Gisbergen S. J. A., Snijders J. G., Ziegler T., J. Comput. Chem., 2001, 22(9), 931-967 |
| [23] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas Ö., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision E.01, Gaussian Inc., Wallingford CT, 2013 |
| [24] | Yi Z. R., Wang S., Liu Y. Q., Adv. Mater., 2015, 27(24), 3589-3606 |
| [25] | Yun H. J., Choi H. H., Kwon S. K., Kim Y. H., Cho K., Chem. Mater., 2014, 26(13), 3928-3937 |
| [26] | Kanimozhi C., Yaacobi-Gross N., Burnett E. K., Briseno A. L., Anthopoulos T. D., Salzner U., Patil S., Phys. Chem. Chem. Phys., 2014, 16(32), 17253-17265 |
| [27] | Coropceanu V., Cornil J., da Silva D. A., Olivier Y., Silbey R., Bredas J. L., Chem. Rev., 2007, 107(4), 926-952 |
| [1] | ZHANG Yichao, ZHAO Fulai, WANG Yu, WANG Yaling, SHEN Yongtao, FENG Yiyu, FENG Wei. Experimental Optimization and Theoretical Simulation of High Performance Field-effect Transistors Based on Multilayer Tungsten Diselenide [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220113. |
| [2] | XIE Ziyi, LIU Dan, ZHANG Yihan, LIU Qingqing, DONG Huanli, HU Wenping. Recent Advances on High Mobility Emissive Anthracene-derived Organic Semiconductors † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1179. |
| [3] | LIU Tianshuo, LONG Shichuan, YAO Zhiyi, SHI Jia, YANG Yang, HONG Wenjing. Progress of Charge Transport Through Self-assembled Monolayers by Employing Eutectic Gallium-Indium Technique [J]. Chem. J. Chinese Universities, 2020, 41(12): 2629. |
| [4] | HAN Bin,YU Xi,HU Wenping. Study on the Mechanism of Charge Tunneling and Hopping Transport in Ferrocene Self-Assembled Molecular Junctions† [J]. Chem. J. Chinese Universities, 2019, 40(2): 298. |
| [5] | CHENG Yingying,LIU Haiying,TIAN Yigeng,LIU Zhongqi,LI Qingxin. Theoretical Study on Enhancement Effect of Amino Modification of Adenine on Conductivity of DNA† [J]. Chem. J. Chinese Universities, 2019, 40(2): 279. |
| [6] | LAI Ying, LIN Rui, DONG Qingmu, HUANG Zongping, TU Xingpeng, LIN Weijing, SUN Weijia. Determination of the Migration of Methyl Methacrylate in Food Contact Materials by High Field Asymmetric Waveform Ion Mobility Spectrometry Technology† [J]. Chem. J. Chinese Universities, 2018, 39(4): 660. |
| [7] | ZHAO Bing, ZHUANG Xiaoyu, LIU Shu, ZHENG Zhong, LIU Zhiqiang, SONG Fengrui. Interaction Between 5-Furd and Cu,Zn-SOD1 by Ion Mobility Mass Spectrometry and Fluorescence Spectroscopy [J]. Chem. J. Chinese Universities, 2017, 38(8): 1341. |
| [8] | ZHENG Jiawei, JIANG Ling, DING Yong, MO Lie, DING Youcai, HU Linhua, DAI Songyuan. Influence of Au Doping on the Surface States and Charge Transport in TiO2 Films† [J]. Chem. J. Chinese Universities, 2017, 38(11): 2038. |
| [9] | CHEN Jiuju. Theoretical Studies on the of Ambipolar Charge Transport in Terazulene Single Crystal† [J]. Chem. J. Chinese Universities, 2016, 37(1): 121. |
| [10] | LI Juan, ZHAO Weijun, LI Lingfeng, LI Xin, LI Peng, WANG Xiaozhi. Rapid Measurement of Synthetic Pigments by Thermal Desorption-High Field Asymmetric Waveform Ion Mobility Spectrometry(FAIMS) Technology† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1403. |
| [11] | ZHANG Wei-Min, FENG Yu, DIAO Kai-Sheng, SUN Li, HONG Fang, SU Zhi-Xing, HU Yu-Ping. Synthesis and Properties of Poly(indenofluorene-triphenylamine) for Organic Field Effect Transistors [J]. Chem. J. Chinese Universities, 2013, 34(9): 2233. |
| [12] | ZHANG Pan-Pan, ZHU Feng, AI Xi-Cheng, FU Li-Min, XU Dong-Sheng, ZHANG Jian-Ping. Zinc-doping Effects on the Trap State Distribution and Charge Recombination in the Anode of TiO2 Dye-sensitized Solar Cells [J]. Chem. J. Chinese Universities, 2013, 34(2): 418. |
| [13] | TAO Yu-Yu, LI Ling-Feng, LI Xin, LI Peng, WANG Xiao-Zhi. Rapid Determination of Diethanolamine by Field Asymmetric Ion Mobility Spectrometry(FAIMS) Technique [J]. Chem. J. Chinese Universities, 2013, 34(11): 2499. |
| [14] | ZHANG Bing, GENG Yun, TANG Xiao-Dan, DUAN Yu-Ai, WU Yong, SU Zhong-Min. Theoretical Studies on Carrier Transport Properties of Oligofluorenythiophene Compounds [J]. Chem. J. Chinese Universities, 2012, 33(07): 1545. |
| [15] | DUAN Gui-Hua, WANG Li-Juan, ZHANG Hou-Yu*, GU Xin , Chen Jie, MA Yu-Guang*. Theoretical Study of Electronic and Charge Transport Properties of Cruciform π-Conjugated 3,6-Diphenyl-1,2,4,5-(2′,2″-diphenyl)-benzobisazole [J]. Chem. J. Chinese Universities, 2010, 31(5): 1029. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||