

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (7): 1335.doi: 10.7503/cjcu20150863
• Organic Chemistry • Previous Articles Next Articles
ZHANG Xiao, SHAN Xindi, ZHAO Xiaoliang, LI Guoyun, WANG Xiaojiang, CAI Chao, HAO Jiejie, YU Guangli*( )
)
Received:2015-11-11
Online:2016-07-10
Published:2016-06-06
Contact:
YU Guangli
E-mail:glyu@ouc.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Xiao, SHAN Xindi, ZHAO Xiaoliang, LI Guoyun, WANG Xiaojiang, CAI Chao, HAO Jiejie, YU Guangli. Preparation, Characterization and Immunological Activity Evaluation of Low Anticoagulant Heparin Oligosaccharides†[J]. Chem. J. Chinese Universities, 2016, 37(7): 1335.
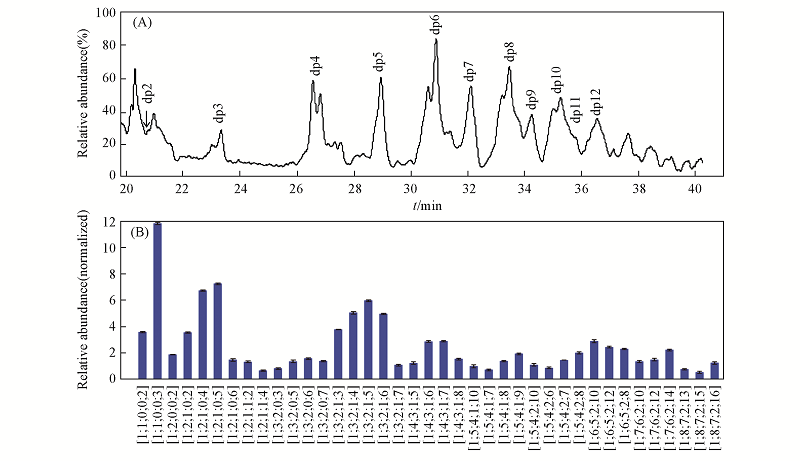
Fig.2 HILIC-FTMS total ion chromatogram(TIC) and major oligosaccharides quantified from BE-LAH (A) TIC of BE-LAH; (B) typical unsaturated oligosaccharide products quantification comparison of BE-LAH. Oligosaccharide compositions are given as △HexA, GlcN, HexA, Ac and SO3.
| Sample | Composition(%) | ||||
|---|---|---|---|---|---|
| Unsulfated disaccharides | Monosulfated disaccharides | Disulfated disaccharides | Trisulfated disaccharides | Total 3-O-sulfo group containing tetrasaccharides | |
| HP-STD | 1.69 | 5.17 | 10.98 | 75.96 | 6.20 |
| ES-STD | 2.07 | 9.02 | 11.81 | 71.28 | 5.82 |
| LAH | 4.30 | 28.92 | 17.07 | 49.13 | 0.57 |
| BE-LAH | 4.52 | 30.63 | 20.83 | 44.02 | |
| HS-STD | 43.96 | 32.19 | 14.35 | 9.50 | |
Table 1 Compositional analysis of BE-LAH, LAH and standard HP, HS, ES*
| Sample | Composition(%) | ||||
|---|---|---|---|---|---|
| Unsulfated disaccharides | Monosulfated disaccharides | Disulfated disaccharides | Trisulfated disaccharides | Total 3-O-sulfo group containing tetrasaccharides | |
| HP-STD | 1.69 | 5.17 | 10.98 | 75.96 | 6.20 |
| ES-STD | 2.07 | 9.02 | 11.81 | 71.28 | 5.82 |
| LAH | 4.30 | 28.92 | 17.07 | 49.13 | 0.57 |
| BE-LAH | 4.52 | 30.63 | 20.83 | 44.02 | |
| HS-STD | 43.96 | 32.19 | 14.35 | 9.50 | |
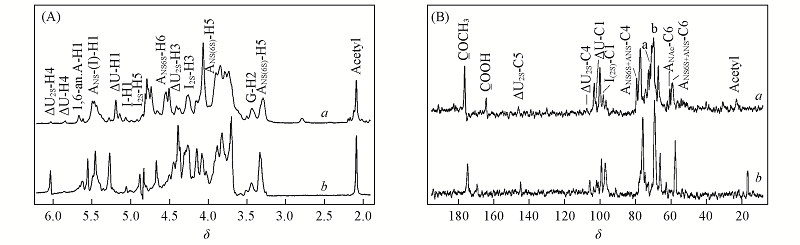
Fig.3 1H NMR(A), 13C NMR(B) of BE-LAH(a) and USP ES(b) A: glucosamine; M: mannosamine; G: glucuronic acid; I: iduronic acid; △U: 4-deoxy-α-L-threo-hex-4-enopyranosil uronic acid; peak a: ANS6S+ANS-C3; peak b: I2S-C3.
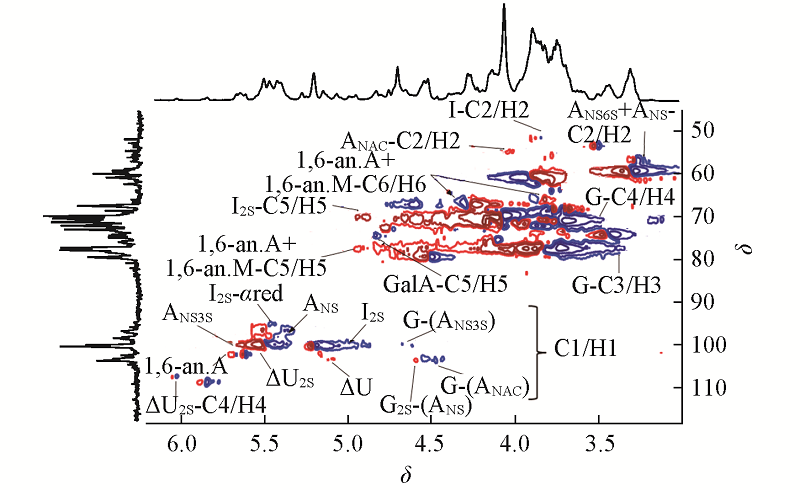
Fig.4 1H-13C HSQC spectrum of BE-LAH A: glucosamine; M: mannosamine; G: glucuronic acid; I: iduronic acid; △U: 4-deoxy-α-L-threo-hex-4-enopyranosil uronic acid; 1,6-an. A: 1,6-anhydro-glucosamine; 1,6-an. M: 1,6-anhydro-mannosamine; α/β red: terminal reducing residue with α/β configuration.
| GAGs | Anti-Xa activity/(U·mg-1) | Anti-Ⅱa activity/(U·mg-1) | Anti-Xa/anti-Ⅱa(ratio) |
|---|---|---|---|
| HP | 180 | 180 | 1.0 |
| ES | 160±5 | 49±4 | 3.3 |
| HS | 11±2 | 11±2 | 1.0 |
| LAH | 24±3 | 25±3 | 1.0 |
| BE-LAH | 9±1 | 7±1 | 1.3 |
Table 2 Anti-Xa and anti-Ⅱa activities of samples
| GAGs | Anti-Xa activity/(U·mg-1) | Anti-Ⅱa activity/(U·mg-1) | Anti-Xa/anti-Ⅱa(ratio) |
|---|---|---|---|
| HP | 180 | 180 | 1.0 |
| ES | 160±5 | 49±4 | 3.3 |
| HS | 11±2 | 11±2 | 1.0 |
| LAH | 24±3 | 25±3 | 1.0 |
| BE-LAH | 9±1 | 7±1 | 1.3 |
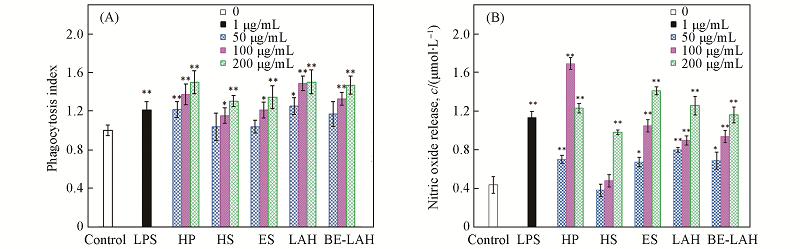
Fig.5 Effects of different doses of GAGs on the phagocytosis index(A) and NO release(B) of the mouse macrophage cell line RAW 264.7Results are expressed as means ±S.D. * P<0.05, ** P<0.01, compared with blank control, as determined by unpaired student’s t-test.
| [1] | Lindahl U., Bäckström G., Thunberg L., Leder I. G., Proc. Natl. Acad. Sci. USA,1980, 77(11), 6551—6555 |
| [2] | Ishihara M., Guo Y., Wei Z., Yang Z., Swiedler S. J., Orellana A., Hirschberg C. B., J. Biol. Chem., 1993, 268(27), 20091—20095 |
| [3] | Linhardt R. J., J. Med. Chem., 2003, 46(13), 2551—2564 |
| [4] | Spinler S. A., Inverso S. M., Cohen M., Goodman S. G., Stringer K. A., Antman E. M., Am. Heart J., 2003, 146(1), 33—41 |
| [5] | Büller H. R., Davidson B. L., Decousus H., Gallus A., Gent M., Piovella F., Prins M. H., Raskob G., Segers A. E., Cariou R., Leeuwenkamp O., Lensing A. W., Ann. Intern. Med., 2004, 140(11), 867—873 |
| [6] | Rao N. V., Argyle B., Xu X., Reynolds P. R., Walenga J. M., Prechel M., Prestwich G. D., MacArthur R. B., Walters B. B., Hoidal J. R., Kennedy T. P., Am. J. Physiol. Cell. Physiol., 2010, 299(1), C97—C110 |
| [7] | Fryer A., Huang Y. C., Rao G., Jacoby D., Mancilla E., Whorton R., Piantadosi C. A., Kennedy T., Hoidal J., J. Pharmacol. Exp. Ther., 1997, 282(1), 208—219 |
| [8] | Fransson L. A., Nieduszynski L. A., Sheehan J. K., Biochim. Biophys. Acta,1980, 630(2), 287—230 |
| [9] | Gonzales J. N., Kim K. M., Zemskova M. A., Rafikov R., Heeke B., Varn M. N., Black S., Kennedy T. P., Verin A. D., Zemskov E. A., Vascular. Pharmacology,2014, 62(2), 63—71 |
| [10] | Vogt A. M., Pettersson F., Moll K., Jonsson C., Normark J., Ribacke U., Egwang T. G., Ekre H. P., Spillmann D., Chen Q., Wahlgren M., PLoS. Pathog., 2006, 2(9), e100 |
| [11] | Leitgeb A. M., Blomqvist K., Cho-Ngwa F., Samje M., Nde P., Titanji V., Wahlgren M., Am. J. Trop. Med. Hyg., 2011, 84(3), 390—396 |
| [12] | Balazs E. A., Berntsen K. O., Karossa J., Swann D. A., Anal. Biochem., 1965, 12(3), 547—558 |
| [13] | Hintze V., Miron A., Möller S., Schnabelrauch M., Heinemann S., Worch H., Scharnweber D., J. Tissue. Eng. Regen. Med., 2014, 8(4), 314—324 |
| [14] | Li L., Zhang F., Zaia J., Linhardt R. J., Anal. Chem., 2012, 84(20), 8822—8829 |
| [15] | Maxwell E., Tan Y., Tan Y., Hu H., Benson G., Aizikov K., Conley S., Staples G. O., Slysz G. W., Smith R. D., Zaia J., PLoS. One,2012, 7(9), e45474 |
| [16] | Li G., Steppich J., Wang Z., Sun Y., Xue C., Linhardt R. J., Li L., Anal. Chem., 2014, 86(13), 6626—6632 |
| [17] | Fu L., Zhang F., Li G., Onishi A., Bhaskar U., Sun P., Linhardt R. J., J. Pharm. Sci., 2014, 103(5), 1375—1383 |
| [18] | Zhao Y. X., Shangguan L. H., Zhao R., Liu G. Q., Chem. J. Chinese Universities,2002, 23(2), 203—206(赵莹歆, 上官隶华, 赵睿, 刘国诠. 高等学校化学学报, 2002, 23(2), 203—206) |
| [19] | Wang J. H., Zha X. Q., Pan L. H., Luo J. P., Chem. J. Chinese Universities,2013, 34(4), 881—885(王军辉, 查学强, 潘利华, 罗建平. 高等学校化学学报, 2013, 34(4), 881—885) |
| [20] | Jeske W. P., Neville B., Ma Q., Hoppensteadt D. A., Fareed J., Blood(ASH Annual Meeting Abstracts), 2004, 104, 1868 |
| [21] | Galeotti F., Volpi N., J. Chromatogr. A., 2013, 1284(1), 141—147 |
| [22] | Li G., Cai C., Li L., Fu L., Chang Y., Zhang F., Toida T., Xue C., Linhardt R. J., Anal. Chem., 2014, 86(1), 326—330 |
| [23] | Vismara E., Pierini M., Guglieri S., Liverani L., Mascellani G., Torri G., Semin. Thromb. Hemost., 2007, 33(5), 466—477 |
| [24] | Guerrini M., Guglieri S., Naggi A., Sasisekharan R., Torri G., Semin. Thromb. Hemost., 2007, 33(5), 478—487 |
| [25] | Limtiaco J. F., Jones C. J., Larive C. K., Anal. Chem., 2009, 81(24), 10116—10123 |
| [26] | Keire D. A., Trehy M. L., Reepmeyer J. C., Kolinski R. E., Ye W., Dunn J., Westenberger B. J., Buhse L. F., J. Pharm. Biomed. Anal., 2010, 51(4), 921—926 |
| [27] | Sudo M., Sato K., Chaidedgumjorn A., Toyoda H., Toida T., Imanari T., Anal. Biochem., 2001, 297(1), 42—51 |
| [28] | Bianchini P., Liverani L., Spelta F., Mascellani G., Parma B., Semin. Thromb. Hemost., 2007, 33(5), 496—502 |
| [1] | XU Yongbin, FENG Shuaixia, CHEN Jie, GONG Hua, SHI Songshan, WANG Huijun, WANG Shunchun. Structural Characterization of a Homogeneous Polysaccharide Isolated From the Flower of Carthamus tinctorius L. and Its Inhibitory Activity on HepG2 Proliferation [J]. Chem. J. Chinese Universities, 0, (): 20220600. |
| [2] | ZHU Kai, LI Jie, WU Xiaoyi, HU Weiwei, WU Dongmei, YU Chengxiao, GE Zhiwei, YE Xingqian, CHEN Shiguo. Combined PGC-Triple-Tof-MS Enables the Separation, Identification of Sugar Beet Pectin Derived Oligomers [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220023. |
| [3] | HU Haocheng, LI Wenli, ZHANG Jianing, LIU Yubo. Extraction, Structure Characterization and Biological Activities of Oligosaccharides from Auricularia heimuer [J]. Chem. J. Chinese Universities, 2021, 42(8): 2465. |
| [4] | HU Zhifei, XU Peng, WEI Bingcheng, YU Biao. Total Synthesis of Phenylpropanoid Glycosides,Acteoside, Isoacteoside and Ligupurpuroside J [J]. Chem. J. Chinese Universities, 2020, 41(8): 1708. |
| [5] | ZHANG Yanyan, HAO Kaifeng, ZHANG Guoqiang, ZHAO Wei. β-Stereoselectivity of Glucuronic Acid Analogues Mediated by 2-Cyanobenzyl Group † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1904. |
| [6] | WANG Shanshan, CAI Chao, HAO Jiejie, LI Guoyun, WANG Xueliang, HU Minghua, FAN Luodi, YU Guangli. Preparation and Immunological Activity Evaluation of Water-soluble β-Glucan from Saccharomyces cerevisiae † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1873. |
| [7] | LI Cheng,WANG Chengjian,JIN Wanjun,HAN Jianli,YANG Meifang,GAO Xi,HUANG Linjuan,WANG Zhongfu. Mass Spectrometric Analysis of N-Glycans of Glycoprotein Separated by SDS-PAGE Gel from Ginkgo Seed† [J]. Chem. J. Chinese Universities, 2019, 40(1): 69. |
| [8] | LANG Yinzhi, LIU Shilong, WANG Chen, ZHANG Xiao, LÜ Youjing, CAI Chao, LI Guoyun, YU Guangli. Separation and Structural Sequence Analysis of Sialylated HMOs via Tandem Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2018, 39(4): 645. |
| [9] | ZHAO Xiaoliang, LIU Xi, YANG Yi, CAI Chao, LÜ Youjing, LI Guoyun, HAO Jiejie, YU Guangli. Detection of Animal Tissue Distribution of β-1,3/1,6-Glucan from Durvillaea antarctica by in vivo Imaging [J]. Chem. J. Chinese Universities, 2017, 38(8): 1368. |
| [10] | FENG Jilu, QI Junru, LIU Qianru. Fabrication of Soy Protein Isolate-soluble Soy Polysaccharide Core-shell Nanogels via Maillard Reaction and Self-assembly† [J]. Chem. J. Chinese Universities, 2016, 37(11): 1999. |
| [11] | PENG Yifang, WANG Chengjian, WANG Jingjing, LI Lingmei, JIN Wanjun, QIANG Shan, SHI Hongdan, ZHANG Ying, HUANG Linjuan, WANG Zhongfu. Analysis of Antigenic Determinant Glycans of Peanut Allergy Glycoprotein Ara h1 by Mass Spectrometry† [J]. Chem. J. Chinese Universities, 2016, 37(9): 1622. |
| [12] | SHEN Yanhong, CHEN Linxiao, ZHANG Wenqing, XU Zhizhen, WAN Yanfei, ZHANG Li, XIA Wei. Structural Characterization of Mannatide† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1088. |
| [13] | LIU Yang, YIN Lu, GONG Guiping, PENG Yifang, HUANG Linjuan, WANG Zhongfu. Structural Characterization, Antioxidant Activity and Immunomodulatory Activity of the Polysaccharide LRLP3 from Leaves of Lycium ruthenicum Murra† [J]. Chem. J. Chinese Universities, 2016, 37(2): 261. |
| [14] | YIN Junyi, LIU Xiaoying, NIE Shaoping, XIE Mingyong. Solution, Conformational Properties and Morphology of Psyllium Polysaccharide with Ferulic Acid Removal† [J]. Chem. J. Chinese Universities, 2016, 37(1): 43. |
| [15] | PAN Liying, GU Xiao, WANG Chengjian, QIANG Shan, HUANG Linjuan, ZHANG Ying, WANG Zhongfu. Comparative Analysis of O-glycans from Human Hepatocellular Carcinoma HepG2 and Normal Liver Cells L02† [J]. Chem. J. Chinese Universities, 2015, 36(7): 1275. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||