

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (3): 477.doi: 10.7503/cjcu20140819
• Organic Chemistry • Previous Articles Next Articles
ZHUO Jibin2, WAN Qian2, YAN Xiquan2, XIE Lili2, YUAN Yaofeng1,2,*( )
)
Received:2014-09-10
Online:2015-03-10
Published:2015-01-30
Contact:
YUAN Yaofeng
E-mail:yaofeng_yuan@fzu.edu.cn
Supported by:CLC Number:
TrendMD:
ZHUO Jibin, WAN Qian, YAN Xiquan, XIE Lili, YUAN Yaofeng. Synthesis and Anion Recognition of Trinuclear Ferrocene-based Imidazole Receptors†[J]. Chem. J. Chinese Universities, 2015, 36(3): 477.
| Compd. | Appearance | Yielda(%) | m. p./℃ | LC-MS,m/z | IR(KBr), | Elemental analysis(%, calcd.) | ||
|---|---|---|---|---|---|---|---|---|
| C | H | N | ||||||
| 1a | White solid | 22.2 | 101—103 | 445.44b | 3063, 3041, 2970, 1601, 1566, 1471, 1245, 1073, 576 | 32.36 (32.25) | 3.48 (3.38) | |
| 1b | White solid | 32.1 | 40—42 | 487.62b | 3060, 3021, 2970, 1471, 1456, 1443, 1387, 996, 582 | 37.02 (36.84) | 4.33 (4.33) | |
| 1c | White solid | 38.2 | 36—37 | 528.91b | 3063, 3041, 1601, 1588, 1497, 1471, 1377, 1274, 564 | 40.91 (40.71) | 5.21 (5.12) | |
| 2a | Yellow solid | 32.2 | 93—94 | 1294.81c | 3448, 3368, 3034,2940, 1566, 1489, 1439, 1376, 1325, 1015, 836, 497 | 45.29 (45.02) | 4.01 (3.99) | 5.72 (5.83) |
| 2b | Yellow solid | 36.4 | 77—78 | 1338.88c | 3418, 3097, 2953, 2867, 1617, 1552, 1479, 1433, 1376, 831, 491 | 46.27 (46.18) | 4.33 (4.28) | 5.80 (5.67) |
| 2c | Yellow solid | 46.2 | 72—73 | 1378.83c | 3437, 3149, 3090, 2966, 2920, 1552, 1453, 1315, 1112, 831, 493 | 47.49 (47.27) | 4.72 (4.56) | 5.69 (5.51) |
Table 1 Appearance, yields, melting points, LC-MS, IR data and elemental analysis for compounds 1a—1c and 2a—2c
| Compd. | Appearance | Yielda(%) | m. p./℃ | LC-MS,m/z | IR(KBr), | Elemental analysis(%, calcd.) | ||
|---|---|---|---|---|---|---|---|---|
| C | H | N | ||||||
| 1a | White solid | 22.2 | 101—103 | 445.44b | 3063, 3041, 2970, 1601, 1566, 1471, 1245, 1073, 576 | 32.36 (32.25) | 3.48 (3.38) | |
| 1b | White solid | 32.1 | 40—42 | 487.62b | 3060, 3021, 2970, 1471, 1456, 1443, 1387, 996, 582 | 37.02 (36.84) | 4.33 (4.33) | |
| 1c | White solid | 38.2 | 36—37 | 528.91b | 3063, 3041, 1601, 1588, 1497, 1471, 1377, 1274, 564 | 40.91 (40.71) | 5.21 (5.12) | |
| 2a | Yellow solid | 32.2 | 93—94 | 1294.81c | 3448, 3368, 3034,2940, 1566, 1489, 1439, 1376, 1325, 1015, 836, 497 | 45.29 (45.02) | 4.01 (3.99) | 5.72 (5.83) |
| 2b | Yellow solid | 36.4 | 77—78 | 1338.88c | 3418, 3097, 2953, 2867, 1617, 1552, 1479, 1433, 1376, 831, 491 | 46.27 (46.18) | 4.33 (4.28) | 5.80 (5.67) |
| 2c | Yellow solid | 46.2 | 72—73 | 1378.83c | 3437, 3149, 3090, 2966, 2920, 1552, 1453, 1315, 1112, 831, 493 | 47.49 (47.27) | 4.72 (4.56) | 5.69 (5.51) |
| Compd. | 1H NMR(400 MHz, DMSO-d6), δ | 13C NMR(100 MHz, DMSO-d6), δ |
|---|---|---|
| 1a | 6.15(s, 3H, PhH), 4.28(t, J=6.2 Hz, 6H, CH2), 3.64(t, J=6.2 Hz, 6H, CH2) | 160.04, 96.08, 68.00, 29.04 |
| 1b | 6.13(s, 3H, PhH), 4.09(t, J=5.6 Hz, 6H, CH2), 3.62(t, J=6.8 Hz, 6H, CH2), 2.32(p, J=6.8, 5.6 Hz, 6H, CH2) | 160.59, 94.35, 65.41, 32.38, 29.97 |
| 1c | 6.08(s, 3H, PhH), 3.98(t, J=5.6 Hz, 6H, CH2), 3.50(t, J=6.8 Hz, 6H, CH2), 2.08(p, J=7.6, 5.6 Hz, 6H, CH2), 1.95(p, J=7.6, 6.8 Hz, 6H, CH2) | 160.59, 94.35, 65.41, 32.38, 29.97 |
| 2a | 9.21(s, 3H, NCHN), 7.80(s, 3H, NCH), 7.76(s, 3H, NCH), 6.13(s, 3H, PhH), 5.19(s, 6H, CH2), 4.59(t, 6H, J=4.8 Hz, CH2), 4.43(t, 6H, J=4.8 Hz, CH2), 4.25(m, 12H, PhH), 4.23(s, 15H, CpH) | 159.85, 136.42, 123.30, 122.72, 95.17, 81.48, 69.36, 69.34, 69.22, 66.16, 48.88 |
| Compd. | 1H NMR(400 MHz, DMSO-d6), δ | 13C NMR(100 MHz, DMSO-d6), δ |
| 2b | 9.18(s, 3H, NCHN), 7.78(s, 6H, NCH), 6.04(s, 3H, PhH), 5.18(s, 6H, CH2), 4.43(s, 6H, CpH), 4.31(t, J=7.0 Hz, 6H, CH2), 4.25(d, 6H, CpH), 4.24(s, 15H, CpH), 4.96(t, J=5.6 Hz, 6H, CH2), 2.20(dt, J=5.6, 7.0 Hz, 6H, CH2) | 160.44, 136.01, 123.03, 122.72, 94.46, 81.31, 69.37, 69.36, 69.20, 64.92, 48.85, 46.88, 29.44 |
| 2c | 9.17(s, 3H, NCHN), 7.77(s, 6H, NCH), 6.06(s, 3H, PhH), 5.17(s, 6H, CH2), 4.46(s, 6H, CpH), 4.26(s, 6H, CpH), 4.24(s, 15H, CpH), 4.23(t, J=7.6 Hz, 6H), 3.92(t, J=6.0 Hz, 6H), 1.95(dd, J=6.0, 5.6 Hz, 6H), 1.66(dd, J=6.0, 5.6 Hz, 6H) | 160.77, 135.88, 122.87, 122.80, 94.35, 81.40, 69.36, 69.20, 67.24, 49.15, 48.89, 26.82, 25.87 |
Table 2 1H NMR and 13C NMR data for compounds 1a—1c and 2a—2c
| Compd. | 1H NMR(400 MHz, DMSO-d6), δ | 13C NMR(100 MHz, DMSO-d6), δ |
|---|---|---|
| 1a | 6.15(s, 3H, PhH), 4.28(t, J=6.2 Hz, 6H, CH2), 3.64(t, J=6.2 Hz, 6H, CH2) | 160.04, 96.08, 68.00, 29.04 |
| 1b | 6.13(s, 3H, PhH), 4.09(t, J=5.6 Hz, 6H, CH2), 3.62(t, J=6.8 Hz, 6H, CH2), 2.32(p, J=6.8, 5.6 Hz, 6H, CH2) | 160.59, 94.35, 65.41, 32.38, 29.97 |
| 1c | 6.08(s, 3H, PhH), 3.98(t, J=5.6 Hz, 6H, CH2), 3.50(t, J=6.8 Hz, 6H, CH2), 2.08(p, J=7.6, 5.6 Hz, 6H, CH2), 1.95(p, J=7.6, 6.8 Hz, 6H, CH2) | 160.59, 94.35, 65.41, 32.38, 29.97 |
| 2a | 9.21(s, 3H, NCHN), 7.80(s, 3H, NCH), 7.76(s, 3H, NCH), 6.13(s, 3H, PhH), 5.19(s, 6H, CH2), 4.59(t, 6H, J=4.8 Hz, CH2), 4.43(t, 6H, J=4.8 Hz, CH2), 4.25(m, 12H, PhH), 4.23(s, 15H, CpH) | 159.85, 136.42, 123.30, 122.72, 95.17, 81.48, 69.36, 69.34, 69.22, 66.16, 48.88 |
| Compd. | 1H NMR(400 MHz, DMSO-d6), δ | 13C NMR(100 MHz, DMSO-d6), δ |
| 2b | 9.18(s, 3H, NCHN), 7.78(s, 6H, NCH), 6.04(s, 3H, PhH), 5.18(s, 6H, CH2), 4.43(s, 6H, CpH), 4.31(t, J=7.0 Hz, 6H, CH2), 4.25(d, 6H, CpH), 4.24(s, 15H, CpH), 4.96(t, J=5.6 Hz, 6H, CH2), 2.20(dt, J=5.6, 7.0 Hz, 6H, CH2) | 160.44, 136.01, 123.03, 122.72, 94.46, 81.31, 69.37, 69.36, 69.20, 64.92, 48.85, 46.88, 29.44 |
| 2c | 9.17(s, 3H, NCHN), 7.77(s, 6H, NCH), 6.06(s, 3H, PhH), 5.17(s, 6H, CH2), 4.46(s, 6H, CpH), 4.26(s, 6H, CpH), 4.24(s, 15H, CpH), 4.23(t, J=7.6 Hz, 6H), 3.92(t, J=6.0 Hz, 6H), 1.95(dd, J=6.0, 5.6 Hz, 6H), 1.66(dd, J=6.0, 5.6 Hz, 6H) | 160.77, 135.88, 122.87, 122.80, 94.35, 81.40, 69.36, 69.20, 67.24, 49.15, 48.89, 26.82, 25.87 |
| Compd. | Epa(free) | Epc(free) | ︳Epa- Epc︳ | ipa/ | Ec |
|---|---|---|---|---|---|
| Ferrocene | 496 | 429 | 67 | 1.03 | 458 |
| 2a | 659 | 587 | 72 | 1.04 | 623 |
| 2b | 653 | 585 | 68 | 1.03 | 619 |
| 2c | 658 | 583 | 75 | 1.02 | 620 |
Table 3 Electrochemical data for ferrocene and 2a—2ca
| Compd. | Epa(free) | Epc(free) | ︳Epa- Epc︳ | ipa/ | Ec |
|---|---|---|---|---|---|
| Ferrocene | 496 | 429 | 67 | 1.03 | 458 |
| 2a | 659 | 587 | 72 | 1.04 | 623 |
| 2b | 653 | 585 | 68 | 1.03 | 619 |
| 2c | 658 | 583 | 75 | 1.02 | 620 |
| Receptor | ΔEp/mV | |||||
|---|---|---|---|---|---|---|
| F- | Cl- | Br- | I- | AcO- | HS | |
| 2a | -152 | -27 | -8 | -6 | -34 | -76 |
| 2b | -178 | -16 | -8 | -4 | -20 | -98 |
| 2c | -160 | -20 | -18 | -19 | -16 | -130 |
Table 4 Electrochemical data for receptors 2a—2c*
| Receptor | ΔEp/mV | |||||
|---|---|---|---|---|---|---|
| F- | Cl- | Br- | I- | AcO- | HS | |
| 2a | -152 | -27 | -8 | -6 | -34 | -76 |
| 2b | -178 | -16 | -8 | -4 | -20 | -98 |
| 2c | -160 | -20 | -18 | -19 | -16 | -130 |
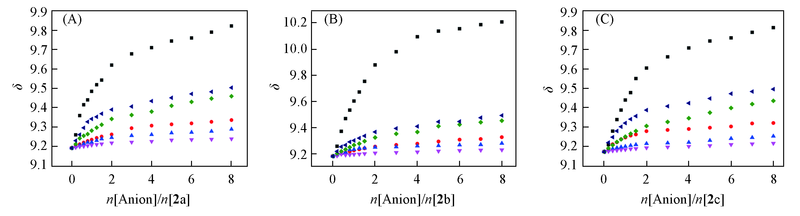
Fig.1 1H NMR titration curves of receptor 2a(A), 2b(B) and 2c(C) in DMSO-d6 by monitoring the C2 proton of imidazolium moieties ■ F-; ● Cl-; ▲ Br-; ▼ I-; ◆ AcO-; ? HSO4-.
| Receptor | Anionb | -Δ | |
|---|---|---|---|
| 2a | F- | 1.12 × 104 | -23.1 |
| Cl- | 2.86 × 103 | -19.7 | |
| Br- | 1.69 × 103 | -18.4 | |
| I- | 8.01 × 102 | -16.6 | |
| AcO- | 4.73 × 103 | -21.0 | |
| HS | 5.52 × 103 | -21.3 | |
| 2b | F- | 1.83 × 104 | -24.3 |
| Cl- | 2.55 × 103 | -19.4 | |
| Br- | 1.71 × 103 | -18.4 | |
| I- | 8.18 × 102 | -16.6 | |
| AcO- | 4.80 × 103 | -21.0 | |
| HS | 5.34 × 103 | -21.2 | |
| 2c | F- | 1.14 × 104 | -23.1 |
| Cl- | 2.62 × 103 | -19.5 | |
| Br- | 1.41 × 103 | -17.9 | |
| I- | 7.47 × 102 | -16.4 | |
| AcO- | 4.66 × 103 | -20.9 | |
| HS | 5.75 × 103 | -21.4 |
Table 5 Association constants(Ka) and binding free energies(ΔG 0—) for 1:1 complexes of receptors 2a—2c with anions in DMSO-d6 at 298 Ka
| Receptor | Anionb | -Δ | |
|---|---|---|---|
| 2a | F- | 1.12 × 104 | -23.1 |
| Cl- | 2.86 × 103 | -19.7 | |
| Br- | 1.69 × 103 | -18.4 | |
| I- | 8.01 × 102 | -16.6 | |
| AcO- | 4.73 × 103 | -21.0 | |
| HS | 5.52 × 103 | -21.3 | |
| 2b | F- | 1.83 × 104 | -24.3 |
| Cl- | 2.55 × 103 | -19.4 | |
| Br- | 1.71 × 103 | -18.4 | |
| I- | 8.18 × 102 | -16.6 | |
| AcO- | 4.80 × 103 | -21.0 | |
| HS | 5.34 × 103 | -21.2 | |
| 2c | F- | 1.14 × 104 | -23.1 |
| Cl- | 2.62 × 103 | -19.5 | |
| Br- | 1.41 × 103 | -17.9 | |
| I- | 7.47 × 102 | -16.4 | |
| AcO- | 4.66 × 103 | -20.9 | |
| HS | 5.75 × 103 | -21.4 |
| [1] | Wade C.R., Broomsgrove A. E., Aldridge S., Gabbai F. P.,Chem. Rev., 2010, 110(7), 3958—3985 |
| [2] | Bai Y., Zhang B.G., Duan C. Y., Dang D. B., Meng Q. J.,New J. Chem., 2005, 29, 777—779 |
| [3] | Dong Z.Y., Jiang X. Z., Zhang D. W., Gao G. H.,Chem. J. Chinese Universities, 2012, 33(10), 2256—2262 |
| (董智云, 江小枝, 张大卫, 高国华. 高等学校化学学报, 2012, 33(10), 2256—2262) | |
| [4] | Liu W.X., Jiang Y. B.,J. Org. Chem., 2007, 73(3), 1124—1127 |
| [5] | Nishiyabu R., Palacios M.A., Dehaen W., Anzenbacher P.,J. Am. Chem. Soc., 2006, 128(35), 11496—11504 |
| [6] | Hossain M.A., Kang S. O., Powell D., Bowman-James K.,Inorg. Chem., 2003, 42(5), 1397—1399 |
| [7] | Bates G.W., Gale P. A., Light M. E.,Chem. Commun., 2007, 21, 2121—2123 |
| [8] | Kong D., Weng T., He W., Liu B., Jin S., Hao X., Liu S.,J. Organomet. Chem., 2013, 72, 19—27 |
| [9] | Cao Q.Y., Lee M. H., Zhang J. F., Ren W. X., Kim J. S.,Tetrahedron Lett., 2011, 52, 2786—2789 |
| [10] | Thomas J.L., Howarth J., Hanlon K., McGuirk D.,Tetrahedron Lett., 2000, 41, 413—416 |
| [11] | Thomas J.L., Howarth J., Kennedy A.,Molecules, 2002, 7, 861—866 |
| [12] | Bai Y., Zhang B.G., Duan C. Y., Dang D. B., Meng Q. J.,New J. Chem., 2006, 30, 266—271 |
| [13] | Niu H.T., Yin Z., Su D., Niu D., He J., Cheng J. P.,Dalton Trans., 2008, 3694—3700 |
| [14] | Niu H.T., Yin Z., Su D., Niu D., Ao Y., He J., Cheng J. P.,Tetrahedron, 2008, 64, 6300—6306 |
| [15] | Liu Q.X., Yao Z. Q., Zhao X. J., Chen A. H., Yang X. Q., Liu S. W., Wang X. G.,Organometallics, 2011, 30, 3732—3739 |
| [16] | Zhuo J. B., Zhang C. Y., Lin C. X., Bai S., Xie L. L., Yuan Y. F., J. Organomet. Chem., 2014, 34—43, 763/764 |
| [17] | Zhuo J.B., Zhu X. X., Lin C. X., Bai S., Xie L. L., Yuan Y. F.,J. Organomet. Chem., 2014, 770, 85—93 |
| [18] | Beer P. D., Gale P. A., Chen G. Z., Coord. Chem. Rev., 1999, 185/186, 3—36 |
| [19] | Beer P.D., Chen Z., Ogden M. I., J. Chem. Soc.,Faraday Trans., 1995, 91, 295—302 |
| [20] | Beer P.D., Graydon A. R., Sutton L. R.,Polyhedron, 1996, 15, 2457—2461 |
| [21] | Boiocchi M., Del Boca L., Gómez D.E., Fabbrizzi L., Licchelli M., Monzani E.,J. Am. Chem. Soc., 2004, 126, 16507—16514 |
| [22] | Amendola V., Boiocchi M., Fabbrizzi L., Palchetti A.,Chem. Eur. J., 2005, 11, 5648—5660 |
| [23] | Kumar R., Guchhait T., Mani G.,Inorg. Chem., 2012, 51, 9029—9038 |
| [24] | Thordarson P.,Chem. Soc. Rev., 2011, 40, 1305—1323 |
| [1] | FAN Jianling, TANG Hao, QIN Fengjuan, XU Wenjing, GU Hongfei, PEI Jiajing, CEHN Wenxing. Nitrogen Doped Ultra-thin Carbon Nanosheet Composited Platinum-ruthenium Single Atom Alloy Catalyst for Promoting Electrochemical Hydrogen Evolution Process [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220366. |
| [2] | JIANG Bowen, CHEN Jingxuan, CHENG Yonghua, SANG Wei, KOU Zongkui. Recent Progress of Single-atom Materials in Electrochemical Biosensing [J]. Chem. J. Chinese Universities, 2022, 43(9): 20220334. |
| [3] | LI Yulong, XIE Fating, GUAN Yan, LIU Jiali, ZHANG Guiqun, YAO Chao, YANG Tong, YANG Yunhui, HU Rong. A Ratiometric Electrochemical Sensor Based on Silver Ion Interaction with DNA for the Detection of Silver Ion [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220202. |
| [4] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [5] | WANG Lijun, LI Xin, HONG Song, ZHAN Xinyu, WANG Di, HAO Leiduan, SUN Zhenyu. Efficient Electrocatalytic CO2 Reduction to CO by Tuning CdO-Carbon Black Interface [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220317. |
| [6] | GONG Yanxi, WANG Jianbing, CHAI Buyu, HAN Yuanchun, MA Yunfei, JIA Chaomin. Preparation of Potassium Doped g-C3N4 Thin Film Photoanode and Its Application in Photoelectrocatalytic Oxidation of Diclofenac Sodium in Water [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220005. |
| [7] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [8] | LIU Jiaqi, LI Tianbao. Preparation and Photoelectrochemical Performance of BiVO4/CuBi2O4 Thin Film Photoanodes [J]. Chem. J. Chinese Universities, 2022, 43(4): 20220017. |
| [9] | CHEN Wangsong, LUO Lan, LIU Yuguang, ZHOU Hua, KONG Xianggui, LI Zhenhua, DUAN Haohong. Recent Progress in Photoelectrochemical H2 Production Coupled with Biomass-derived Alcohol/aldehyde Oxidation [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210683. |
| [10] | ZHU Haotian, JIN Meixiu, TANG Wensi, SU Fang, LI Yangguang. Properties of Transition Metal-biimidazole-Dawson-type Tungstophosphate Hybrid Compounds as Supports for Enzyme Immobilization [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220328. |
| [11] | HOU Congcong, WANG Huiying, LI Tingting, ZHANG Zhiming, CHANG Chunrui, AN Libao. Preparation and Electrochemical Properties of N-CNTs/NiCo-LDH Composite [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220351. |
| [12] | SHI Xiaofan, ZHU Jian, BAI Tianyu, FU Zixuan, ZHANG Jijie, BU Xianhe. Research Status and Progress of MOFs with Application in Photoelectrochemical Water-splitting [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210613. |
| [13] | MA Jianxin, LIU Xiaodong, XU Na, LIU Guocheng, WANG Xiuli. A Multi-functional Zn(II) Coordination Polymer with Luminescence Sensing, Amperometric Sensing, and Dye Adsorption Performance [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210585. |
| [14] | WEI Chuangyu, CHEN Yanli, JIANG Jianzhuang. Fabrication of Electrochemical Sensor for Dopamine and Uric Acid Based on a Novel Dimeric Phthalocyanine-involved Quintuple-decker Modified Indium Tin Oxide Electrode [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210582. |
| [15] | JIANG Jun, GONG Tiantian, ZHANG Chengpeng, LIU Xiaoqian, ZHAO Junwei. Synthesis and Electrochemical Biosensing Properties of Pyridine Dicarboxylic Acid Decorated Rare-earth-incorporatedTellurotungstates [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210561. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||