

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (1): 19.doi: 10.7503/cjcu20120521
• Analytical Chemistry • Previous Articles Next Articles
NONG Ruiyu1, KONG Jiao1, ZHANG Junhui2, CHEN Ling1, TANG Bo1, XIE Shengming1, YUAN Liming1,*( )
)
Received:2015-07-06
Online:2016-01-10
Published:2015-12-20
Contact:
YUAN Liming
E-mail:yuan_limingpd@126.com
Supported by:CLC Number:
TrendMD:
NONG Ruiyu, KONG Jiao, ZHANG Junhui, CHEN Ling, TANG Bo, XIE Shengming, YUAN Liming. Chiral Metal-organic Framework{[Co(L-trp)(bpe)(H2O)]·H2O·NO3}n Used for High Performance Liquid Chromatographic Separation†[J]. Chem. J. Chinese Universities, 2016, 37(1): 19.
| Positional isomer | Mobile phase V(Hexane)/V(Isopropanol) | Retention factor, k' | Separation factor, α | |||
|---|---|---|---|---|---|---|
| o- | m- | p- | ||||
| o,m,p-Nitroaniline | 9∶1 | 1.75 | 2.07 | 5.60 | 1.11(m-/o-) | 2.15(p-/m-) |
| o,m,p-Phenylenediamine | 98∶2 | 0.97 | 1.11 | 7.39 | 1.12(m-/o-) | 3.47(p-/m-) |
| o,m,p-Benzenediol | 98∶2 | 2.02 | 1.03 | 3.77 | 2.87(o-/m-) | 1.96(p-/o-) |
Table 1 Separations of the positional isomers on the Co-Trp-MOF-column
| Positional isomer | Mobile phase V(Hexane)/V(Isopropanol) | Retention factor, k' | Separation factor, α | |||
|---|---|---|---|---|---|---|
| o- | m- | p- | ||||
| o,m,p-Nitroaniline | 9∶1 | 1.75 | 2.07 | 5.60 | 1.11(m-/o-) | 2.15(p-/m-) |
| o,m,p-Phenylenediamine | 98∶2 | 0.97 | 1.11 | 7.39 | 1.12(m-/o-) | 3.47(p-/m-) |
| o,m,p-Benzenediol | 98∶2 | 2.02 | 1.03 | 3.77 | 2.87(o-/m-) | 1.96(p-/o-) |
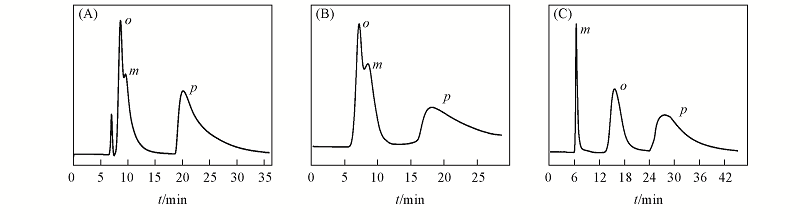
Fig.4 HPLC chromatograms on the packed Co-Trp-MOF-column(250 mm×2.0 mm i.d.) for the separation of positional isomers^(A) o,m,p-Nitroaniline; (B) o,m,p-phenylenediamine; (C) o,m,p-benzenediol. Mobile phase: hexane and isopropanol; flow rate: 0.1 mL/min; UV: 254 nm.
| Racemate | Mobile phase V(Hexane)/V(Isopropanol) | Retention factor, k | Separation factor, α | |||
|---|---|---|---|---|---|---|
| Column A | Column B | Column C | Column D | |||
| 3-Benzylexy-1,2-propanediol | 98∶2 | 1.07 | 4.68 | 3.72 | 1.36 | 2.69 |
| 3,5-Dinitro-N-(1-phenylethyl)benzamide | 95∶5 | 5.08 | 1.66 | 1.49 | 2.26 | |
| Propranolol hydrochloride | 95∶5 | 0.84 | 1.38 | |||
| 1-(9-Anthryl)-2,2,2-trifluoroethanol | 90∶10 | 1.01 | 1.37 | 2.60 | ||
| Pindolol | 90∶10 | 0.78 | 1.47 | |||
| DNB-(R,S)-Leucine | 90∶10 | 0.82 | 2.12 | |||
| Hydrobenzoin | 80∶10 | 1.02 | 1.77 | 1.62 | ||
| Warfarin sodium | 80∶10 | 4.36 | 1.74 | 1.35 | ||
| Omeprazole | 80∶20 | 2.26 | 1.85 | |||
| Chlorpheniramine maleate | 80∶20 | 1.27 | 1.58 | |||
| Alprenolol | 80∶20 | 1.28 | 2.69 | |||
Table 2 Separations of racemates on columns Aa, Bb, Cc and Dd
| Racemate | Mobile phase V(Hexane)/V(Isopropanol) | Retention factor, k | Separation factor, α | |||
|---|---|---|---|---|---|---|
| Column A | Column B | Column C | Column D | |||
| 3-Benzylexy-1,2-propanediol | 98∶2 | 1.07 | 4.68 | 3.72 | 1.36 | 2.69 |
| 3,5-Dinitro-N-(1-phenylethyl)benzamide | 95∶5 | 5.08 | 1.66 | 1.49 | 2.26 | |
| Propranolol hydrochloride | 95∶5 | 0.84 | 1.38 | |||
| 1-(9-Anthryl)-2,2,2-trifluoroethanol | 90∶10 | 1.01 | 1.37 | 2.60 | ||
| Pindolol | 90∶10 | 0.78 | 1.47 | |||
| DNB-(R,S)-Leucine | 90∶10 | 0.82 | 2.12 | |||
| Hydrobenzoin | 80∶10 | 1.02 | 1.77 | 1.62 | ||
| Warfarin sodium | 80∶10 | 4.36 | 1.74 | 1.35 | ||
| Omeprazole | 80∶20 | 2.26 | 1.85 | |||
| Chlorpheniramine maleate | 80∶20 | 1.27 | 1.58 | |||
| Alprenolol | 80∶20 | 1.28 | 2.69 | |||
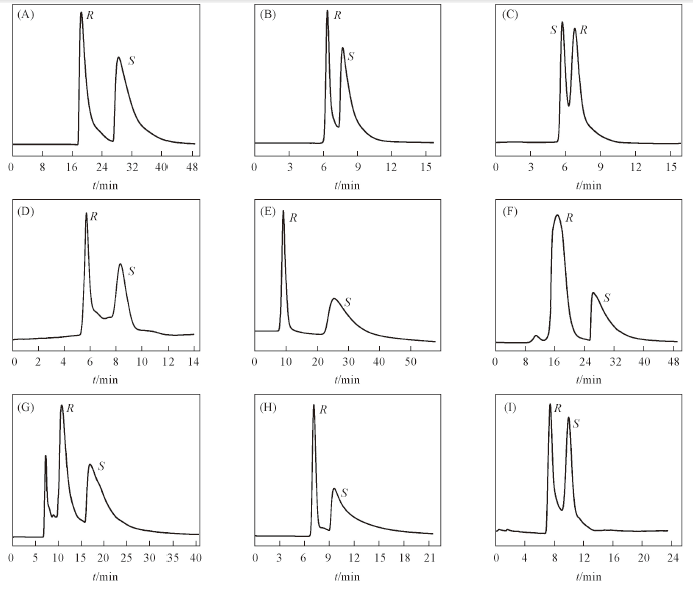
Fig.5 HPLC chromatograms on the Co-Trp-MOF-column(250 mm× 2.0 mm i.d.) for the separation of racemates^(A) 3,5-Dinitro-N-(1-phenylethyl)benzamide; (B) 1-(9-anthryl)-2,2,2-trifluoroethanol; (C) hydrobenzoin; (D) pindolol; (E) 3-benzylexy-1,2-propanediol; (F) warfarin sodium; (G) omeprazole; (H) chlorpheniramine maleate; (I) alprenolol. Mobile phase: hexane and isopropanol(Table 2); flow rate: 0.1 mL/min; UV: 254 nm.
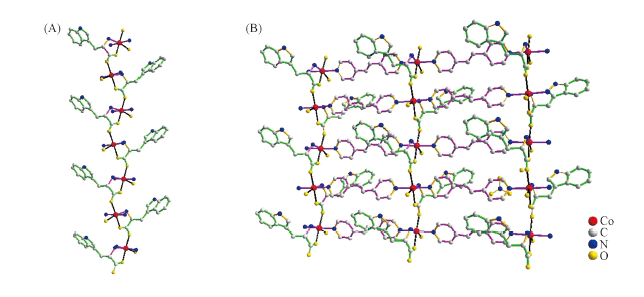
Fig.6 Structure of {[Co(L-trp)(bpe)(H2O)]·H2O·NO3}n^(A) 1D chiral chain having L-trp units arranged in a syn-anti mode; (B) 2D homochiral metal-tryptophanate coordination (4,4)-layer structure along the a axis.
| [1] | Wei W. Y., Fang J., Kong H. N., Han J. Y., Chang H. Y., Progress in Chemistry, 2005, 17(6), 1110—1114 |
| (魏文英, 方键, 孔海宁, 韩金玉, 常贺英. 化学进展, 2005, 17(6), 1110—1114) | |
| [2] | Xie S.M., Yuan L. M.,Progress in Chemistry, 2013, 25(10), 1763—1770 |
| (谢生明, 袁黎明. 化学进展, 2013, 25(10), 1763—1770) | |
| [3] | Ma S. Q., Sun D. F., Simmons J. M., Collier C. D., Yuan D. Q., Zhou H. C., J. Am. Chem. Soc., 2008, 130(3), 1012—1016 |
| [4] | Allendorf M. D., Houk R. J. T., Andruszkiewicz L., Talin A. A., Pikarsky J., Choudhury A., Gall K. A., Hesketh P. J., J. Am. Chem. Soc., 2008, 130(44), 14404—14405 |
| [5] | Yang C. X., Chen Y. J., Wang H. F., Yan X. P., Chem. Eur. J., 2011, 17(42), 1734—11737 |
| [6] | Liu S. S., Yang C. X., Wang S. W., Yan X. P., Analyst,2012, 137(4), 816—818 |
| [7] | Fu Y. Y., Yang C. X., Yan X. P., J. Chromatogr. A, 2013, 1274, 137—144 |
| [8] | Yang B. C., Jiang Y. D., Qin X. J., Chen Z. L., Ren F., Chem. J. Chinese Universities, 2012, 33(1), 26—31 |
| (杨宝春, 姜耀东, 秦雪娟, 陈志良, 任非. 高等学校化学学报, 2012, 33(1), 26—31) | |
| [9] | Li Z. Q., Wang A., Guo C. Y., Hu W. N., Tai Y. F., Chem. J. Chinese Universities, 2013, 34(11), 2470—2477 |
| (李宗群, 汪艾, 郭春燕, 胡文娜, 邰燕芳. 高等学校化学学报, 2013, 34(11), 2470—2477) | |
| [10] | Aijaz A., Akita T., Tsumori N., Xu Q., J. Am. Chem. Soc., 2013, 135(44), 6356—16359 |
| [11] | Li Y. X., Xue M., Guo L. J., Huang L., Chen S. R., Qiu S. L., Chem. Res. Chinese Universities, 2013, 29(2), 196—200 |
| [12] | Yang C. X., Yan X. P., Chin. J. Anal. Chem., 2013, 41(9), 1297—1301 |
| (杨成雄, 严秀平. 分析化学, 2013, 41(9), 1297—1301) | |
| [13] | Cheng B. P., Li L. S., Zhou R. D., Li L., Zhang H. F., Chem. J. Chinese Universities, 2015, 36(5), 872—880 |
| (程彪平, 李来生, 周仁丹, 李良, 张宏福. 高等学校化学学报, , 2015, 36(5), 872—880 | |
| [14] | Ezuhara T., Endo K., Aoyama Y., J. Am. Chem. Soc., 1999, 121, 3279—3283 |
| [15] | Chelucci G., Thummel R. P., Chem. Rev., 2002, 102, 3129—3170 |
| [16] | Bu X. H., Tong M. L., Kitagawa H. C. S., Batten S. R., Angew. Chem. Int. Ed., 2004, 43, 192—195 |
| [17] | Cai Y. G., Bernasek S. L., Phys J. Chem. B, 2005, 109(10), 4514—4519 |
| [18] | Dybtsev D. N., Nuzhdin A. L., Chun H., Bryliakov K. P., Talsi E. P., Fedin V. P., Kim K., Angew. Chem. Int. Ed., 2006, 45(6), 916—920 |
| [19] | Zhang J., Chen S. M., Wu T., Feng P., Bu X. H., J. Am. Chem. Soc., 2008, 130, 12882—12883 |
| [20] | Ma L. Q., Abney C., Lin W. B., Chem. Soc. Rev., 2009, 38(5), 1248—1256 |
| [21] | Jiang W. J., Babarao R., Hu Z. Q., Chem. Soc. Rev., 2011, 40(7), 3599—3612 |
| [22] | Nuzhdin A. L., Dybtsev D. N., Bryliakov K. P., Talsi E. P., Fedin V. P., J. Am. Chem. Soc., 2007, 129, 12958—12959 |
| [23] | Padmanaban M., Muller P., Lieder C., Gedrich K., Grunker R., Bon V., Senkovska I., Baumgartner S., Opelt S., Paasch S., Brunner E., Glorius F., Klemm E., Kaskel S., Chem. Commun., 2011, 47, 12089—12091 |
| [24] | Tanaka K., Muraoka T., Hirayama D., Ohnish A., Chem. Commun., 2012, 48, 8577—8579 |
| [25] | Kuang X., Ma Y., Su H., Zhang J., Dong Y. B., Tang B., Anal. Chem., 2014, 86, 1277—1281 |
| [26] | Zhang M., Pu Z. J., Chen X. L., Gong X. L., Zhu A. X., Yuan L. M., Chem. Commun., 2013, 49(45), 5201—5203 |
| [27] | Zhang M., Zhang J. H., Zhang Y., Wang B. J., Xie S. M., Yuan L. M., J. Chromatogr. A, 2014, 1325, 163—170 |
| [28] | Kong J., Zhang M., Duan A. H., Zhang J. H., Yang R., Yuan L. M., J. Sep. Sci., 2015, 38, 556—561 |
| [29] | Mendiratta S., Usman M., Luo T. T., Chang B. C., Lee S. F., Lin Y. C., Lu K. L., Cryst. Growth Des., 2014, 14, 1572—1579 |
| [1] | ZHAO Yingzhe, ZHANG Jianling. Applications of Metal-organic Framework-based Material in Carbon Dioxide Photocatalytic Conversion [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220223. |
| [2] | LU Cong, LI Zhenhua, LIU Jinlu, HUA Jia, LI Guanghua, SHI Zhan, FENG Shouhua. Synthesis, Structure and Fluorescence Detection Properties of a New Lanthanide Metal-Organic Framework Material [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220037. |
| [3] | TIAN Xueqin, MO Zheng, DING Xin, WU Pengyan, WANG Yu, WANG Jian. A Squaramide-containing Luminescent Metal-organic Framework as a High Selective Sensor for Histidine [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210589. |
| [4] | XING Peiqi, LU Tong, LI Guanghua, WANG Liyan. Controllable Syntheses of Two Cd(II) Metal-organic Frameworks Possessing Related Structures [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220218. |
| [5] | HAN Zongsu, YU Xiaoyong, MIN Hui, SHI Wei, CHENG Peng. A Rare Earth Metal-Organic Framework with H6TTAB Ligand [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210342. |
| [6] | MO Zongwen, ZHANG Xuewen, ZHOU Haolong, ZHOU Dongdong, ZHANG Jiepeng. Guest-responses of A Porous Coordination Polymer Based on Synergistic Hydrogen Bonds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210576. |
| [7] | LIU Xueguang, YANG Xiaoshan, MA Jingjing, LIU Weisheng. Separating Methyl Blue Selectively from the Mixture of Dyes by Europium Metal-organic Frameworks [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210715. |
| [8] | SHI Xiaofan, ZHU Jian, BAI Tianyu, FU Zixuan, ZHANG Jijie, BU Xianhe. Research Status and Progress of MOFs with Application in Photoelectrochemical Water-splitting [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210613. |
| [9] | WU Ji, ZHANG Hao, LUO Yuhui, GENG Wuyue, LAN Yaqian. A Microporous Cationic Ga(III)-MOF with Fluorescence Properties for Selective sensing Fe3+ Ion and Nitroaromatic Compounds [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210617. |
| [10] | LI Wen, QIAO Junyi, LIU Xinyao, LIU Yunling. Zirconium-based Metal-Organic Framework with Naphthalene for Fluorescent Detection of Nitroaromatic Explosives in Water [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210654. |
| [11] | WANG Jie, HUO Haiyan, WANG Yang, ZHANG Zhong, LIU Shuxia. General Strategy for In situ Synthesis of NENU-n Series Polyoxometalate-based MOFs on Copper Foil [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210557. |
| [12] | LI Shurong, WANG Lin, CHEN Yuzhen, JIANG Hailong. Research Progress of Metal⁃organic Frameworks on Liquid Phase Catalytic Chemical Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210575. |
| [13] | ZHANG Chi, SUN Fuxing, ZHU Guangshan. Synthesis, N2 Adsorption and Mixed-matrix Membrane Performance of Bimetal Isostructural CAU-21 [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210578. |
| [14] | SHI Ge, XU Qian, DAI Xiao, ZHANG Jie, SHEN Jun, WAN Xinhua. Effect of Aromatic Substituent on Chiral Recognition of Helical Polyacetylene-based Chiral Stationary Phases for High-Performance Liquid Chromatography [J]. Chem. J. Chinese Universities, 2021, 42(8): 2673. |
| [15] | ZHAO Yangyang, LIU Qiyong, CHEN Boxin, ZHAO Bin, ZHOU Haimei, LI Xinxin, ZHENG Dan, FENG Fei. Silicon-based Micro Gas Chromatographic Column Using Metal-Organic Framework Material ZIF-8 as Stationary Phase [J]. Chem. J. Chinese Universities, 2021, 42(6): 1736. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||