

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (7): 20230104.doi: 10.7503/cjcu20230104
收稿日期:2023-03-10
出版日期:2023-07-10
发布日期:2023-04-18
通讯作者:
周秋菊,张志国
E-mail:zhouqiuju@xynu.edu.cn;zgzhang@mail.buct.edu.cn
基金资助:
CHEN Hongru1, BAI Yang1, ZHOU Qiuju2( ), ZHANG Zhiguo1(
), ZHANG Zhiguo1( )
)
Received:2023-03-10
Online:2023-07-10
Published:2023-04-18
Contact:
ZHOU Qiuju, ZHANG Zhiguo
E-mail:zhouqiuju@xynu.edu.cn;zgzhang@mail.buct.edu.cn
Supported by:摘要:
二维核磁技术(2D NMR)在复杂有机化合物结构的解析上起到重要作用. 通过分析2D NMR谱图关联信号可以得到分子内和分子间的相互作用信息. 有机光伏器件活性层材料之间相互作用的研究非常重要. 这种相互作用不仅可以用来分析活性层材料的聚集行为, 也可以用来分析活性层在加入第三组分后形貌及稳定性的变化, 为器件性能的研究提供强有力的工具. 本文对二维核磁技术进行了相关介绍; 总结了有机光伏领域中使用2D NMR研究分子间相互作用的相关工作; 最后, 展望了该技术的未来发展.
中图分类号:
TrendMD:
陈虹汝, 白阳, 周秋菊, 张志国. 二维核磁在有机光伏中的应用. 高等学校化学学报, 2023, 44(7): 20230104.
CHEN Hongru, BAI Yang, ZHOU Qiuju, ZHANG Zhiguo. Application of 2D NMR in Organic Photovoltaics. Chem. J. Chinese Universities, 2023, 44(7): 20230104.
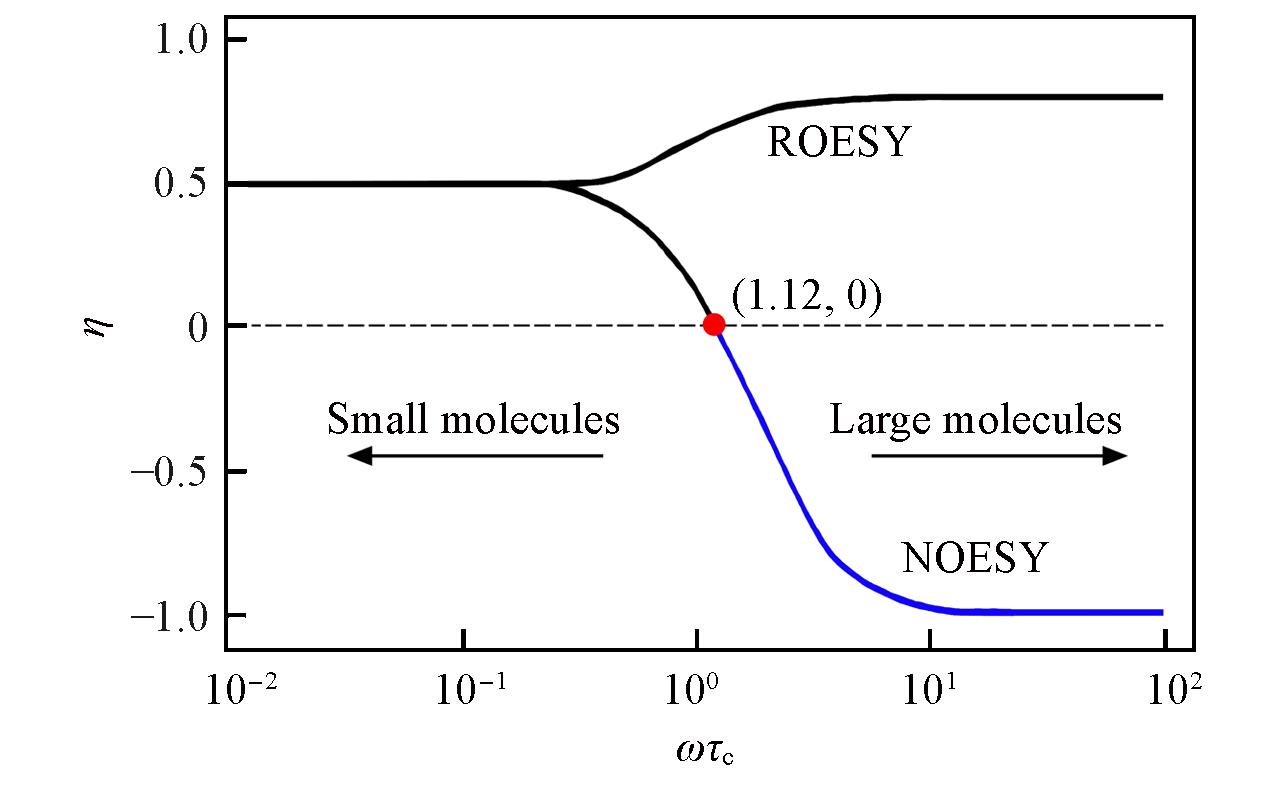
Fig.1 Enhancement factor η as a function of the effective correlation time ωτc for the standard nuclear Overhauser experiment(NOE) and for the spin⁃lock, or rotating⁃coordinate, variant(ROE)
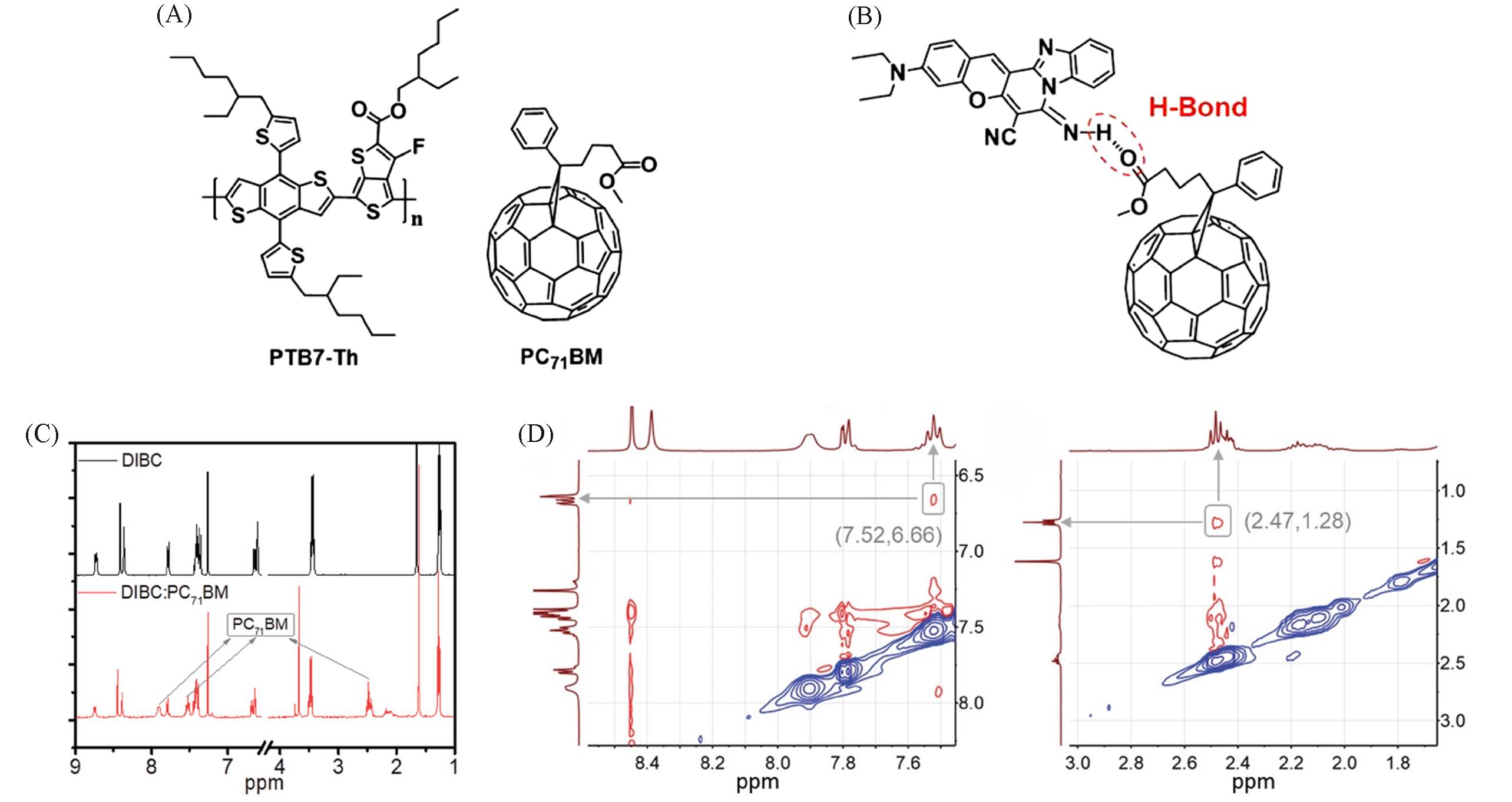
Fig.2 Molecule structures of PTB7⁃h and PC71BM(A), hydrogen⁃bond⁃linked DIBC and PC71BM(B), 1H NMR spectra of DIBC and DIBC∶PC71BM(C) and 2D 1HNMR spectra of DIBC∶PC71BM blend(D)[11]Copyright 2019, Wiley-VCH Verlag.
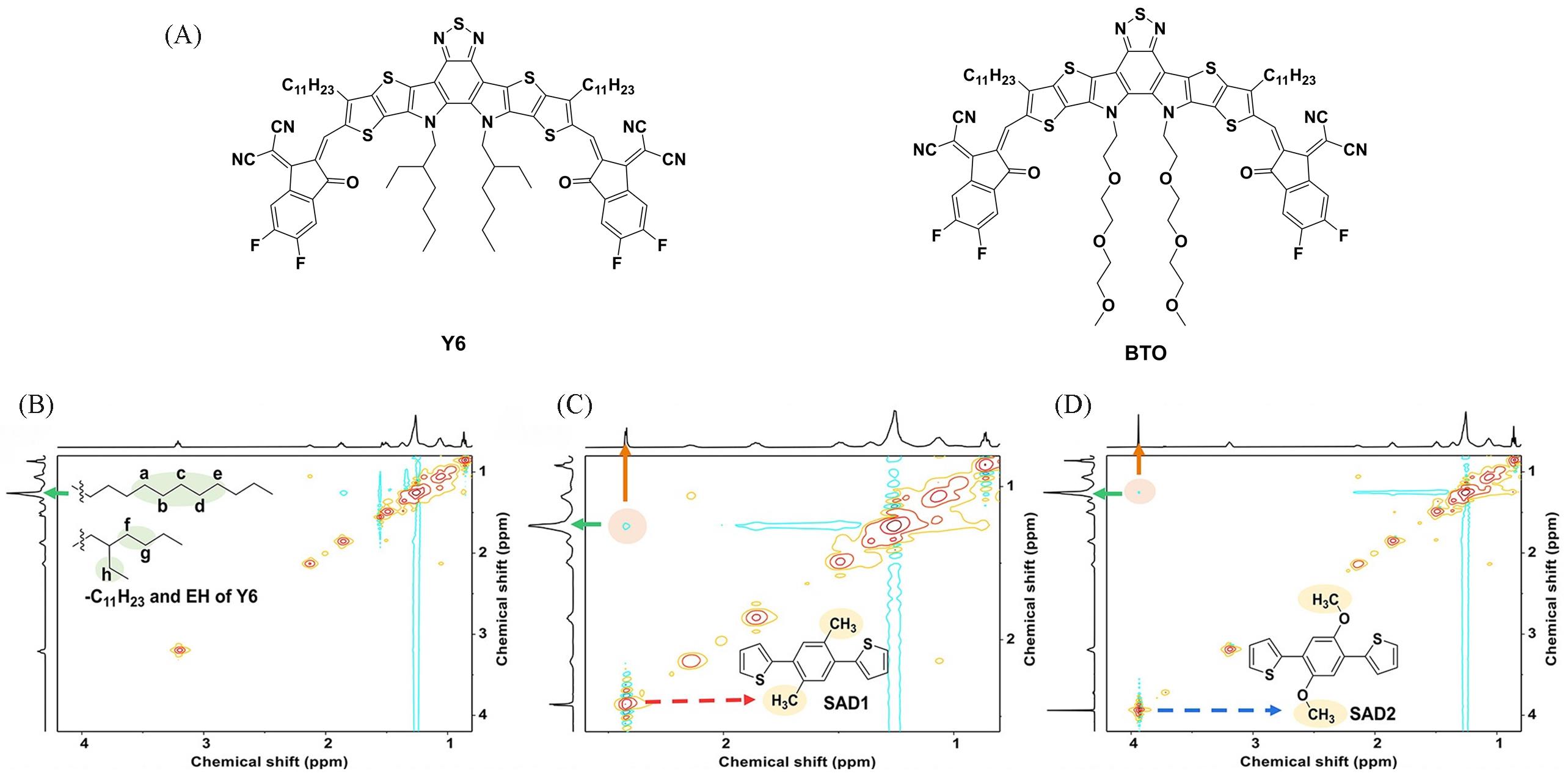
Fig.3 Chemical structures of BTO and Y6(A), 2D 1H⁃1H NMR spectra of Y6⁃0(B), Y6⁃1(C) and Y6⁃2(D) solutions[13]Copyright 2022, American Chemical Society.
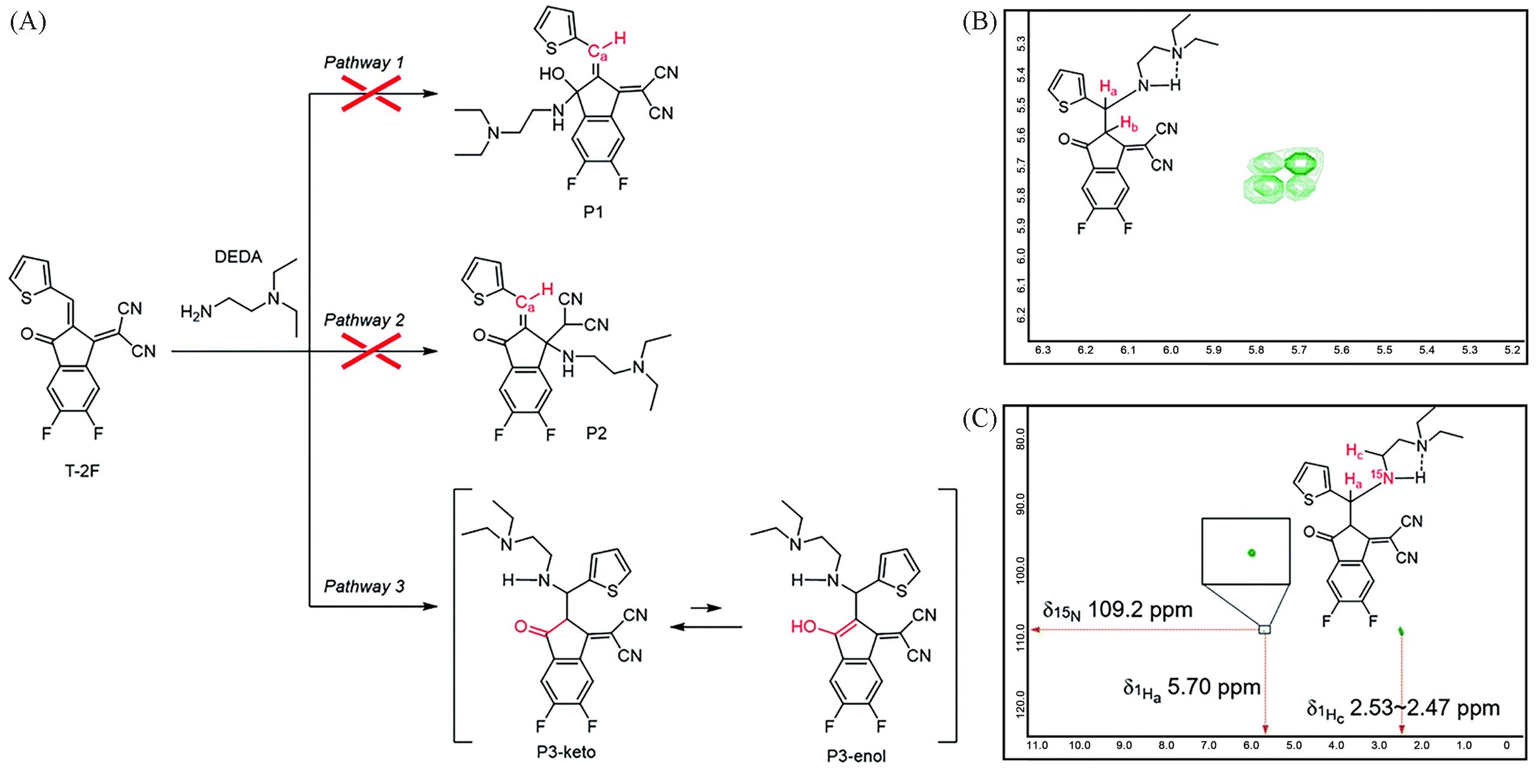
Fig.4 Plausible reaction pathways for the reaction between T⁃2F and DEDA(A), the 1H⁃1H COSY NMR spectrum of the product at -50 ℃(the image shows the magnified region between 5.97—5.52)(B) and 1H⁃15N HMBC NMR spectrum of the product at room temperature(C)[14]Copyright 2021, the Royal Society of Chemistry.
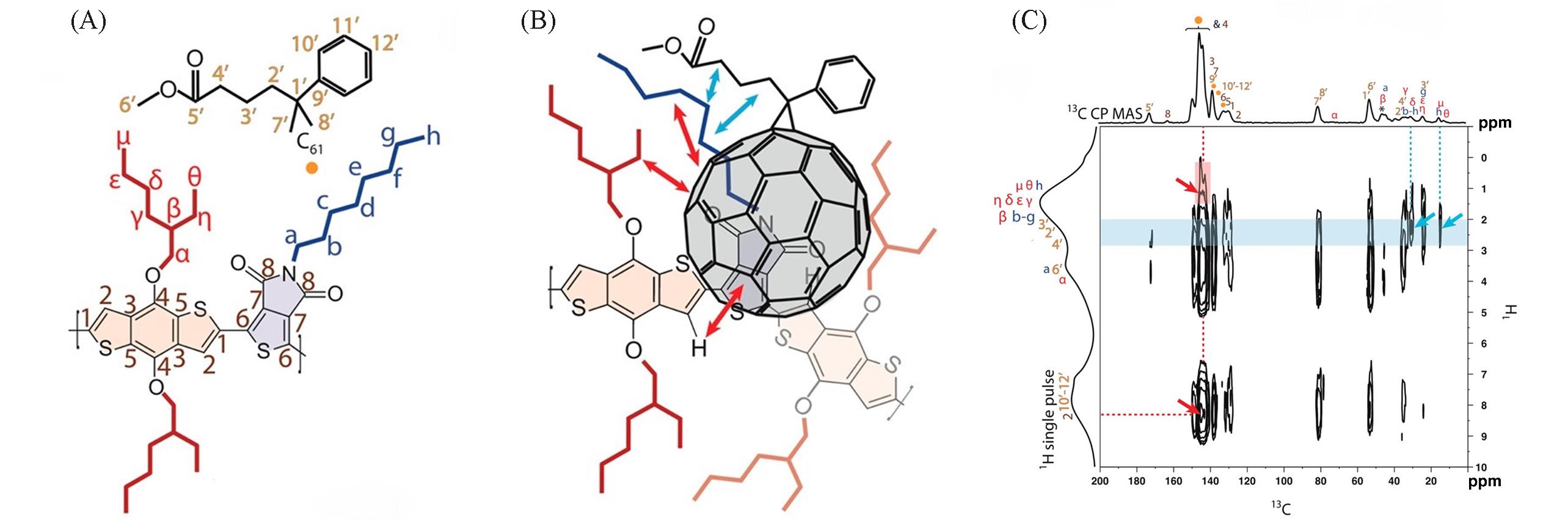
Fig.5 Molecular structures of EH/C8 and PC61BM(A), schematic of intermolecular interactions between the conjugated polymer and PC61BM(B), solid⁃state 2D 13C{1H}dipolar⁃mediated heteronuclear correlation(HETCOR) NMR spectrum acquired at room temperature for an 8%(mass fraction) EH/C8 in PC61BM blend under MAS conditions of 12.5 kHz with an 8 ms CP contact time(C)[16](C) One dimensional 13C{1H} CPMAS and single-pulse 1H MAS spectra are shown along the top horizontal axis and the left vertical axis, respectively.Copyright 2014, American Chemical Society.
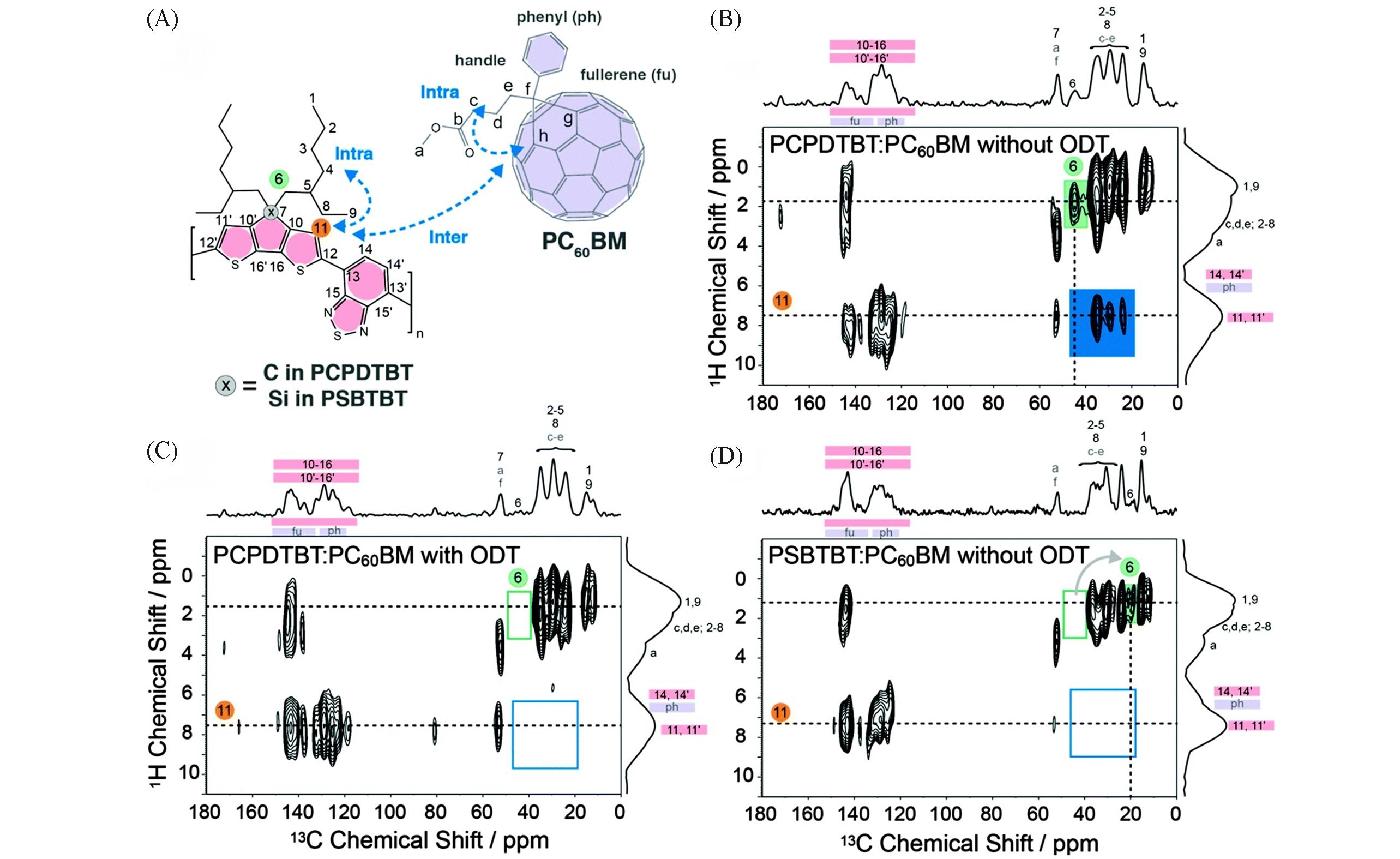
Fig.6 Chemical structures for PCPDTBT, PSBTBT and PC60BM(A), 2D 13C{1H} FSLG⁃HETCOR NMR spectra for PCPDTBT∶PC60BM(1∶2)(B), PCPDTBT∶PC60BM(1∶2) with 2.44% ODT(C) and PSBTBT∶PC60BM(1∶1.5)(D)[17]
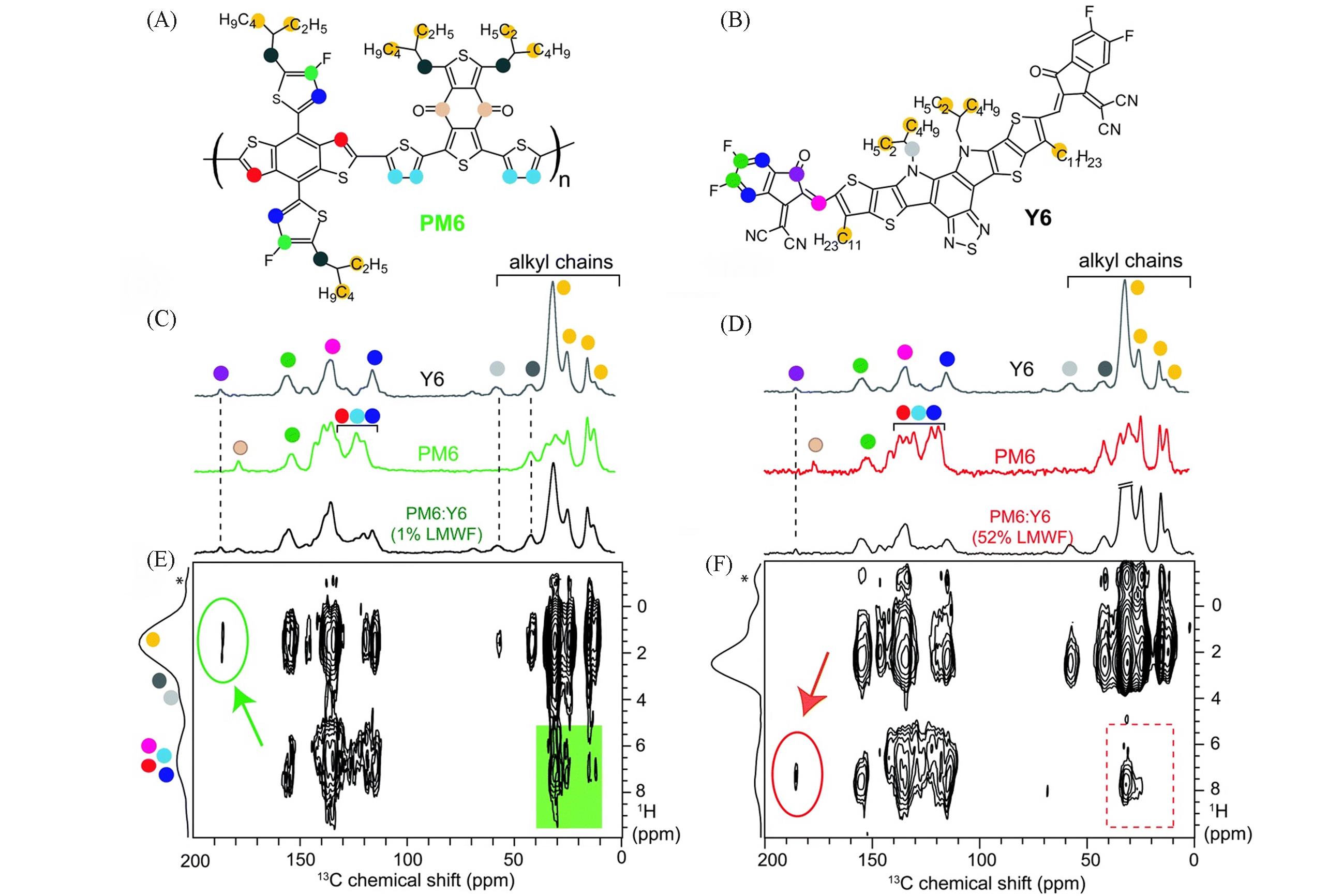
Fig.7 Structures of PM6(A) and Y6(B), comparison of 1D 13C{1H} CP⁃MAS spectra of neat Y6, PM6(1% LMWF) and PM6∶Y6 blends(C), comparison of 1D 13C{1H} CP⁃MAS spectra of neat Y6, PM6(52% LMWF), and PM6∶Y6 blends(D), 2D 13C⁃1H heteronuclear correlation NMR spectrum of 1% LMWF PM6∶Y6 blend(E), 2D 13C⁃1H heteronuclear correlation NMR spectrum of 52% LMWF PM6∶Y6 blend(F)[4]Spectra were acquired at 9.4 T(12.5 kHz MAS) with 4 ms of CP contact time. * Denotes the carrier frequency of 1H homonuclear decoupling. Copyright 2020, the Royal Society of Chemistry.
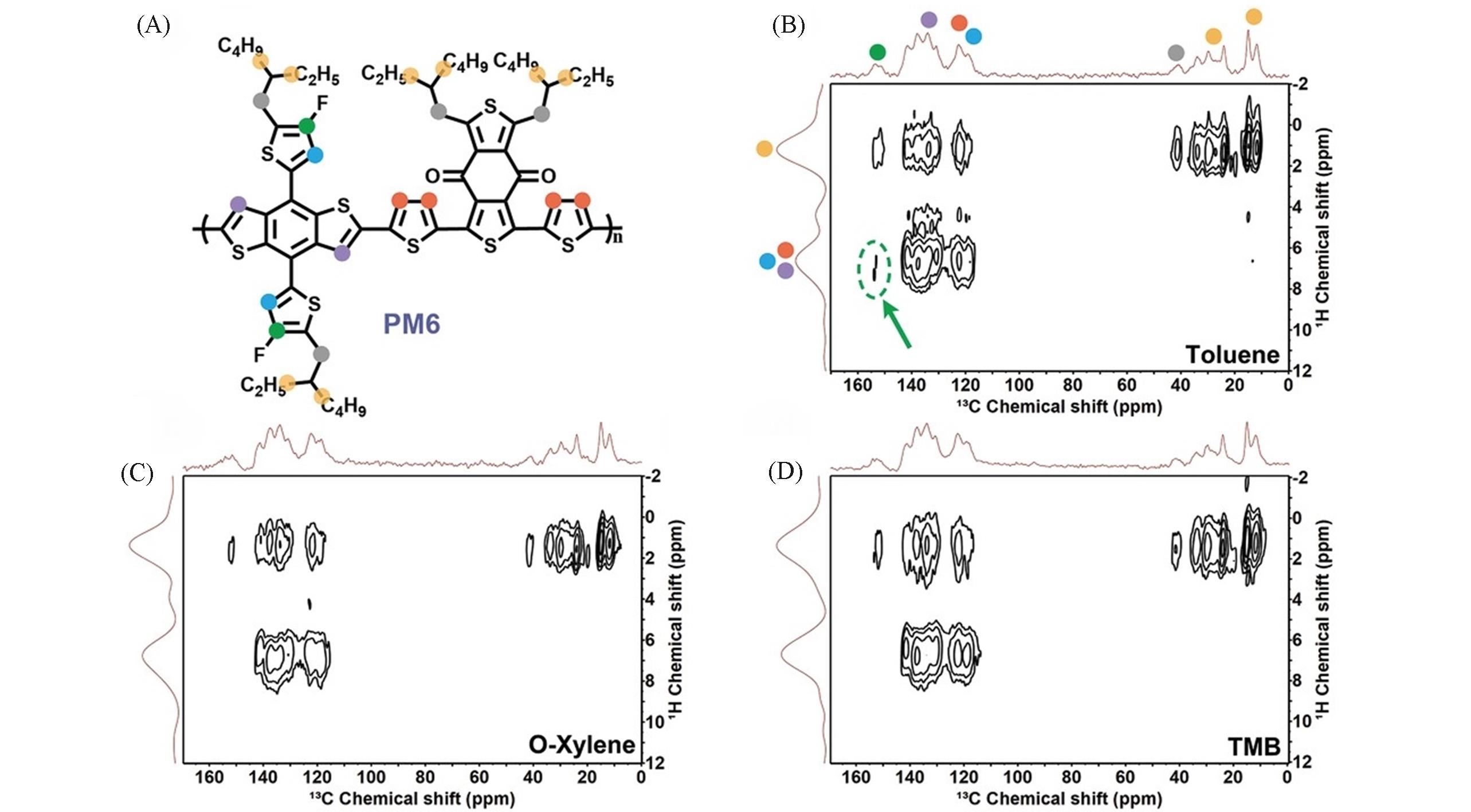
Fig.8 Chemical structure of PM6(A), comparison of solid⁃state 2D 13C⁃1H heteronuclear correlation NMR spectra of PM6 polymer processed from toluene(B), o⁃xylene(C) and TMB(D) acquired at 9.4 T(12.5 kHz MAS) using 4 ms of CP contact time[22]Copyright 2022, Wiley-VCH Verlag.
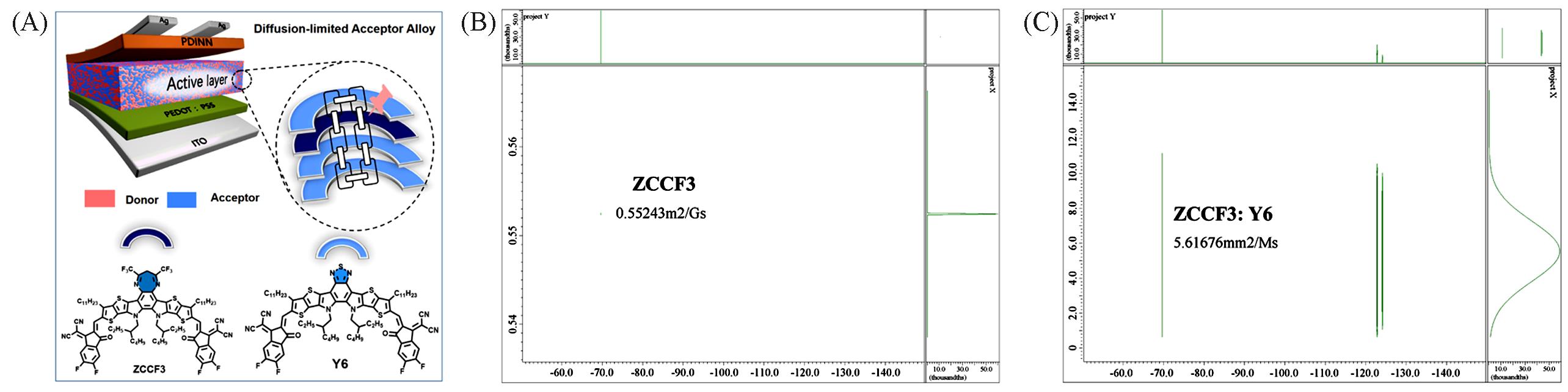
Fig.9 Device structure and the intermolecular interaction between ZCCF3 and Y6(A), 19F DOSY spectra of ZCCF3(B) and ZCCF3∶Y6 in CDCl3(C)[34]Copyright 2023, Wiley-VCH Verlag.
| 1 | Jeener J., Meier B. H., Bachmann P., Ernst R. R., J. Chem. Phys., 1979, 71(11), 4546—4553 |
| 2 | Zhao T., Qin H., Zhang H., Qu L., Anal. Chem., 2018, 46(3), 461 |
| 3 | Fiaux J., Bertelsen E. B., Horwich A. L., Wüthrich K., Nature, 2002, 418(6894), 207—211 |
| 4 | Karki A., Vollbrecht J., Gillett A. J., Xiao S. S., Yang Y., Peng Z., Schopp N., Dixon A. L., Yoon S., Schrock M., Ade H., Reddy G. N. M., Friend R. H., Nguyen T. Q., Energy Environ. Sci., 2020, 13(10), 3679—3692 |
| 5 | Brown S. P., Spiess H. W., Chem. Rev., 2001, 101(12), 4125—4156 |
| 6 | Andrew E. R., Bradbury A., Eades R. G., Nature, 1958, 182(4650), 1659—1659 |
| 7 | Cadars S., Lesage A., Pickard C. J., Sautet P., Emsley L., J. Phys. Chem. A, 2009, 113(5), 902—911 |
| 8 | Nieuwendaal R. C., Ro H. W., Germack D. S., Kline R. J., Toney M. F., Chan C. K., Agrawal A., Gundlach D., VanderHart D. L., Delongchamp D. M., Adv. Funct. Mater., 2012, 22(6), 1255—1266 |
| 9 | Lamber J. B., Mazzola E. P., Ridge C. D., Xiang J., Zhou Q., Nuclear Magnetic Resonance Spectroscopy, Chemical Industry Press, Beijing, 2021, 250—254 |
| 10 | Liu Y., Wang F., Food and Drugs, 2007, 9(8), 39—42 |
| 11 | Du X., Lu X., Zhao J., Zhang Y., Li X., Lin H., Zheng C., Tao S., Adv. Funct. Mater., 2019, 29(30), 1902078 |
| 12 | Chen H., Zhang R., Chen X., Zeng G., Kobera L., Abbrent S., Zhang B., Chen W., Xu G., Oh J., Kang S. H., Chen S., Yang C., Brus J., Hou J., Gao F., Li Y., Li Y., Nat. Energy, 2021, 6(11), 1045—1053 |
| 13 | Li C., Gu X., Chen Z., Han X., Yu N., Wei Y., Gao J., Chen H., Zhang M., Wang A., Zhang J., Wei Z., Peng Q., Tang Z., Hao X., Zhang X., Huang H., J. Am. Chem. Soc., 2022, 144(32), 14731—14739 |
| 14 | Kyeong M., Lee J., Daboczi M., Stewart K., Yao H., Cha H., Luke J., Lee K., Durrant J. R., Kim J. S., Hong S., J. Mater. Chem. A, 2021, 9(23), 13506—13514 |
| 15 | Jia X., Wolak J., Wang X., White J. L., Macromolecules, 2003, 36(3), 712—718 |
| 16 | Graham K. R., Cabanetos C., Jahnke J. P., Idso M. N., Labban A. E., Ndjawa G. N., Heumueller T., Vandewal K., Salleo A., Chmelka B. F., Amassian A., Beaujuge P. M., McGehee M. D., J. Am. Chem. Soc., 2014, 136(27), 9608 |
| 17 | Etzold F., Howard I. A., Forler N., Melnyk A., Andrienko D., Hansen M. R., Laquai F., Energy Environ. Sci., 2015, 8(5), 1511—1522 |
| 18 | Shu J., Dudenko D., Esmaeili M., Park J. H., Puniredd S. R., Chang J. Y., Breiby D. W., Pisula W., Hansen M. R., J. Am. Chem. Soc., 2013, 135(30), 11075 |
| 19 | Gitsas A., Floudas G., Mondeshki M., Lieberwirth I., Spiess H. W., Iatrou H., Hadjichristidis N., Hirao A., Macromolecules, 2010, 43(4), 1874—1881 |
| 20 | Jasinska⁃Walc L., Villani M., Dudenko D., van Asselen O., Klop E., Rastogi S., Hansen M. R., Koning C. E., Macromolecules, 2012, 45(6), 2796—2808 |
| 21 | Rothwell W. P., Waugh J. S., J. Chem. Phys., 1981, 74(5), 2721—2732 |
| 22 | Liu Y., Zhou K., Zhou X., Xue W., Bi Z., Wu H., Ma Z., Ma W., Macromol. Rapid Commun., 2022, 43(22), 2100871 |
| 23 | Luginbuhl B. R., Raval P., Pawlak T., Du Z., Wang T., Kupgan G., Schopp N., Chae S., Yoon S., Yi A., Kim H. J., Veaceslav C., Brédas J. L., Nguyen T. Q., Reddy G. N. M., Adv. Mater., 2022, 34(6), 2105943 |
| 24 | Forgo P., Vincze I., Kover K. E., Steroids, 2003, 68(4), 321 |
| 25 | Adhikari S., Daftardar S., Fratev F., Rivera M., Sirimulla S., Alexander K., Boddu S. H. S., Int. J. Pharm., 2018, 545(1/2), 357—365 |
| 26 | Kitagishi H., Saito M., Mao Q., Kiriyama A., Negi S., Kano K., Chem. Asian J., 2019, 14(19), 3320—3328 |
| 27 | Mazzini S., Musco G., Ragg E., Penco S., Magn. Reson. Chem., 1994, 32(3), 139—150 |
| 28 | Ujihara T., Hayashi N., J. Nat. Prod., 2016, 79(1), 66—73 |
| 29 | Pletneva E. V., Laederach A. T., Fulton D. B., Kostić N. M., J. Am. Chem. Soc., 2001, 123(26), 6232—6245 |
| 30 | Li S., Zhang R., Zhang M., Yao J., Peng Z., Chen Q., Zhang C., Chang B., Bai Y., Fu H., Ouyang Y., Zhang C., Steele J. A., Alshahrani T., Roeffaers M. B. J., Solano E., Meng L., Gao F., Li Y., Zhang Z. G., Adv. Mater., 2023, 35(2), 2206563 |
| 31 | Ghasemi M., Balar N., Peng Z., Hu H., Qin Y., Kim T., Rech J. J., Bidwell M., Mask W., McCulloch I., You W., Amassian A., Risko C., O'Connor B. T., Ade H., Nat. Mater., 2021, 20(4), 525—532 |
| 32 | Liang Y., Zhang D., Wu Z., Jia T., Lüer L., Tang H., Hong L., Zhang J., Zhang K., Brabec C. J., Li N., Huang F., Nat. Energy, 2022, 7(12), 1180—1190 |
| 33 | Ghasemi M., Hu H., Peng Z., Rech J. J., Angunawela I., Carpenter J. H., Stuard S. J., Wadsworth A., McCulloch I., You W., Ade H., Joule, 2019, 3(5), 1328—1348 |
| 34 | Zhang C., Zhang M., Zhou Q., Chen S., Kim S., Yao J., Zhang Z., Bai Y., Chen Q., Chang B., Fu H., Xue L., Wang H., Yang C., Zhang Z. G., Adv. Funct. Mater., 2023, doi: 10.1002/adfm.202214392 |
| [1] | 王英杰, 潘泽晖, 陶正煜, 王妍, 童碧海, 冯敏强, 宋明星. 分子间相互作用对苯甲酮类聚集诱导延迟荧光材料性能的影响[J]. 高等学校化学学报, 2023, 44(4): 20220562. |
| [2] | 马卓远, 汪大洋. 分子自组装单层膜的表面浸润性研究现状和展望[J]. 高等学校化学学报, 2021, 42(4): 1031. |
| [3] | 于泳博, 刘翠, 宫利东. 从头算和ABEEMσπ/MM对(CH3OH)n(n=3~12)和[Na(CH3OH)n]+(n=3~6)体系的研究[J]. 高等学校化学学报, 2016, 37(8): 1468. |
| [4] | 司朋飞, 罗发亮, 海梅. 聚L-乳酸/4,4'-二羟基二苯硫醚共混物的分子间相互作用及结晶和熔融行为[J]. 高等学校化学学报, 2015, 36(1): 188. |
| [5] | 袁伟, 任清江, 孙恒达, 李慧, 程延祥, 马东阁. 外围取代基团对卟啉铂(Ⅱ)配合物发光性能的影响[J]. 高等学校化学学报, 2014, 35(6): 1229. |
| [6] | 侯海云, 黄银蓉, 白博峰, 杨靖. 293.15 K时咪唑醋酸盐-乙醇二元体系的体积性质及分子间相互作用[J]. 高等学校化学学报, 2014, 35(1): 121. |
| [7] | 孟素慈, 殷秀莲, 马晶, 谢吉民. 有机π共轭配体溶剂化效应与分子间相互作用的理论研究[J]. 高等学校化学学报, 2012, 33(11): 2492. |
| [8] | 梁雪, 孙涛, 王一波. 苯-卤素(X2, X=F, Cl, Br, I)相互作用本质的对称性匹配微扰理论研究[J]. 高等学校化学学报, 2012, 33(03): 541. |
| [9] | 李荣强, 徐承凤, 韦鲁滨, 王金本. 含Gemini表面活性剂结构单元的新型两亲聚电解质的合成及聚集行为[J]. 高等学校化学学报, 2010, 31(10): 2024. |
| [10] | 赵建新,乔义涛,冯菁,罗昭峰,袁直 . 共聚物-锌与多肽的相互作用[J]. 高等学校化学学报, 2008, 29(3): 658. |
| [11] | 凌笑梅,刘一,赖先银,张媛,刘晓明,屠鹏飞,赵玉英,,崔景荣 . 采用毛细管电泳方法以凝血酶为靶筛选天然药物提取化合物[J]. 高等学校化学学报, 2007, 28(2): 234. |
| [12] | 汪朝旭, 张敬畅, 曹维良 . HCN(HNC)与NH3, H2O和HF分子间相互作用的理论研究[J]. 高等学校化学学报, 2007, 28(2): 320. |
| [13] | 林宪杰,徐为人,王建武,刘成卜 . 对甲氧基苯甲醛肟晶体结构、 红外光谱及分子间相互作用的实验与理论研究[J]. 高等学校化学学报, 2006, 27(5): 897. |
| [14] | 李宝宗, 国永敏, 庞文琴. AlPO4-5分子筛模板剂的量子化学计算[J]. 高等学校化学学报, 2002, 23(4): 636. |
| [15] | 居学海, 肖鹤鸣, 贡雪东. N-甲硝胺二聚体分子间相互作用的理论研究[J]. 高等学校化学学报, 2002, 23(3): 440. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||