

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (2): 20220464.doi: 10.7503/cjcu20220464
符芳媚1, 徐梦如1, 梁梓珊1, 黄斯锐1, 李晖1, 张浩然1,2, 李唯1,2, 郑明涛1,2, 雷炳富1,2( )
)
收稿日期:2022-07-08
出版日期:2023-02-10
发布日期:2022-09-15
通讯作者:
雷炳富
E-mail:tleibf@scau.edu.cn
基金资助:
FU Fangmei1, XU Mengru1, LIANG Zishan1, HUANG Sirui1, LI Hui1, ZHANG Haoran1,2, LI Wei1,2, ZHENG Mingtao1,2, LEI Bingfu1,2( )
)
Received:2022-07-08
Online:2023-02-10
Published:2022-09-15
Contact:
LEI Bingfu
E-mail:tleibf@scau.edu.cn
Supported by:摘要:
以邻苯二胺和盐酸多巴胺为前体, 利用磷酸调节反应体系的pH值(pH=7, 3, 1), 制备了荧光逐渐红移的碳点(CDs): CDs-7(绿光)、 CDs-3(橙光)和CDs-1(红光). 通过透射电子显微镜(TEM)、 拉曼光谱(Raman)、 傅里叶变换红外光谱(FTIR)、 X射线电子能谱(XPS)、 紫外-可见吸收光谱和荧光衰减曲线分析表明, 反应体系pH值的减小促进了前体碳化交联, 导致sp2共轭域尺寸和石墨化程度增加, 从而使CDs的荧光红移. 另外, 酸性环境有利于使CDs表面氧化形成羧基, 促进CDs荧光红移的同时改善了量子效率(QY). 利用CDs-1量子产率高(14.8%)和发光的溶剂依赖特性, 将其作为荧光探针分别检测了乙醇(EtOH)、 N,N-二甲基甲酰胺(DMF)和1,4-二氧六环(DIO)中的痕量水, 检出限分别为0.86%, 0.123%和0.023%, 表明CDs-1在痕量水的检测方面具有应用潜力.
中图分类号:
TrendMD:
符芳媚, 徐梦如, 梁梓珊, 黄斯锐, 李晖, 张浩然, 李唯, 郑明涛, 雷炳富. 共轭尺寸和表面氧化协同触发的红色荧光碳点用于有机溶剂中痕量水的检测. 高等学校化学学报, 2023, 44(2): 20220464.
FU Fangmei, XU Mengru, LIANG Zishan, HUANG Sirui, LI Hui, ZHANG Haoran, LI Wei, ZHENG Mingtao, LEI Bingfu. Conjugate Size and Surface Oxidation Synergistically Trigger Red Fluorescence in Carbon Dots for Detecting Trace Water in Organic Solvents. Chem. J. Chinese Universities, 2023, 44(2): 20220464.
| No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | ||
|---|---|---|---|---|---|---|---|---|---|
| DMF/EtOH | H2O | DMF/EtOH | H2O | ||||||
| 1 | 200 | 1800 | 0 | 0 | 10 | 200 | 1400 | 400 | 20 |
| 2 | 200 | 1794 | 6 | 0.3 | 11 | 200 | 1300 | 500 | 25 |
| 3 | 200 | 1788 | 12 | 0.6 | 12 | 200 | 1200 | 600 | 30 |
| 4 | 200 | 1780 | 20 | 1 | 13 | 200 | 1100 | 700 | 35 |
| 5 | 200 | 1740 | 60 | 3 | 14 | 200 | 1000 | 800 | 40 |
| 6 | 200 | 1700 | 100 | 5 | 15 | 200 | 900 | 900 | 45 |
| 7 | 200 | 1660 | 140 | 7 | 16 | 200 | 800 | 1000 | 50 |
| 8 | 200 | 1600 | 200 | 10 | 17 | 200 | 700 | 1100 | 55 |
| 9 | 200 | 1500 | 300 | 15 | 18 | 200 | 600 | 1200 | 60 |
Table 1 Preparation of DMF/EtOH detection solutions with different water contents ranging from 0—60%
| No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | ||
|---|---|---|---|---|---|---|---|---|---|
| DMF/EtOH | H2O | DMF/EtOH | H2O | ||||||
| 1 | 200 | 1800 | 0 | 0 | 10 | 200 | 1400 | 400 | 20 |
| 2 | 200 | 1794 | 6 | 0.3 | 11 | 200 | 1300 | 500 | 25 |
| 3 | 200 | 1788 | 12 | 0.6 | 12 | 200 | 1200 | 600 | 30 |
| 4 | 200 | 1780 | 20 | 1 | 13 | 200 | 1100 | 700 | 35 |
| 5 | 200 | 1740 | 60 | 3 | 14 | 200 | 1000 | 800 | 40 |
| 6 | 200 | 1700 | 100 | 5 | 15 | 200 | 900 | 900 | 45 |
| 7 | 200 | 1660 | 140 | 7 | 16 | 200 | 800 | 1000 | 50 |
| 8 | 200 | 1600 | 200 | 10 | 17 | 200 | 700 | 1100 | 55 |
| 9 | 200 | 1500 | 300 | 15 | 18 | 200 | 600 | 1200 | 60 |
| No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | ||
|---|---|---|---|---|---|---|---|---|---|
| DIO | H2O | DIO | H2O | ||||||
| 1 | 200 | 1800 | 0 | 0 | 12 | 200 | 1660 | 140 | 7 |
| 2 | 200 | 1798 | 2 | 0.1 | 13 | 200 | 1640 | 160 | 8 |
| 3 | 200 | 1794 | 6 | 0.3 | 14 | 200 | 1620 | 180 | 9 |
| 4 | 200 | 1790 | 10 | 0.5 | 15 | 200 | 1600 | 200 | 10 |
| 5 | 200 | 1786 | 14 | 0.7 | 16 | 200 | 1500 | 300 | 15 |
| 6 | 200 | 1780 | 20 | 1 | 17 | 200 | 1400 | 400 | 20 |
| 7 | 200 | 1760 | 40 | 2 | 18 | 200 | 1300 | 500 | 25 |
| 8 | 200 | 1740 | 60 | 3 | 19 | 200 | 1200 | 600 | 30 |
| 9 | 200 | 1720 | 80 | 4 | 20 | 200 | 1000 | 800 | 40 |
| 10 | 200 | 1700 | 100 | 5 | 21 | 200 | 800 | 1000 | 50 |
| 11 | 200 | 1680 | 120 | 6 | 22 | 200 | 600 | 1200 | 60 |
Table 2 Preparation of DIO detection solutions with different water contents ranging from 0—60%
| No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | No. | m(CDs⁃1)/μg | Volume/μL | Water content(%) | ||
|---|---|---|---|---|---|---|---|---|---|
| DIO | H2O | DIO | H2O | ||||||
| 1 | 200 | 1800 | 0 | 0 | 12 | 200 | 1660 | 140 | 7 |
| 2 | 200 | 1798 | 2 | 0.1 | 13 | 200 | 1640 | 160 | 8 |
| 3 | 200 | 1794 | 6 | 0.3 | 14 | 200 | 1620 | 180 | 9 |
| 4 | 200 | 1790 | 10 | 0.5 | 15 | 200 | 1600 | 200 | 10 |
| 5 | 200 | 1786 | 14 | 0.7 | 16 | 200 | 1500 | 300 | 15 |
| 6 | 200 | 1780 | 20 | 1 | 17 | 200 | 1400 | 400 | 20 |
| 7 | 200 | 1760 | 40 | 2 | 18 | 200 | 1300 | 500 | 25 |
| 8 | 200 | 1740 | 60 | 3 | 19 | 200 | 1200 | 600 | 30 |
| 9 | 200 | 1720 | 80 | 4 | 20 | 200 | 1000 | 800 | 40 |
| 10 | 200 | 1700 | 100 | 5 | 21 | 200 | 800 | 1000 | 50 |
| 11 | 200 | 1680 | 120 | 6 | 22 | 200 | 600 | 1200 | 60 |
| Element | Function group | Functional group proportion(%) | |||
|---|---|---|---|---|---|
| CDs⁃7 | CDs⁃3 | CDs⁃1 | |||
| C1s | C=C | 41.35 | 44.85 | 49.84 | |
| C—N | 25.97 | 45.42 | 8.18 | ||
| C—O | 29.19 | 6.30 | 37.13 | ||
| COOH | 3.49 | 3.44 | 4.84 | ||
| N1s | Pyridine N | 0 | 4.23 | 32.16 | |
| Pyrrolic N | 62.19 | 69.53 | 49.84 | ||
| Graphitic N | 27.40 | 9.29 | 13.62 | ||
| Amino N | 10.41 | 16.94 | 4.39 | ||
| O1s | C=O | 20.32 | 28.68 | 54.89 | |
| O—H | 79.48 | 71.32 | 45.11 | ||
Table 3 Data analysis of XPS C1s, N1s and O1s spectra of CDs-7, CDs-3 and CDs-1
| Element | Function group | Functional group proportion(%) | |||
|---|---|---|---|---|---|
| CDs⁃7 | CDs⁃3 | CDs⁃1 | |||
| C1s | C=C | 41.35 | 44.85 | 49.84 | |
| C—N | 25.97 | 45.42 | 8.18 | ||
| C—O | 29.19 | 6.30 | 37.13 | ||
| COOH | 3.49 | 3.44 | 4.84 | ||
| N1s | Pyridine N | 0 | 4.23 | 32.16 | |
| Pyrrolic N | 62.19 | 69.53 | 49.84 | ||
| Graphitic N | 27.40 | 9.29 | 13.62 | ||
| Amino N | 10.41 | 16.94 | 4.39 | ||
| O1s | C=O | 20.32 | 28.68 | 54.89 | |
| O—H | 79.48 | 71.32 | 45.11 | ||
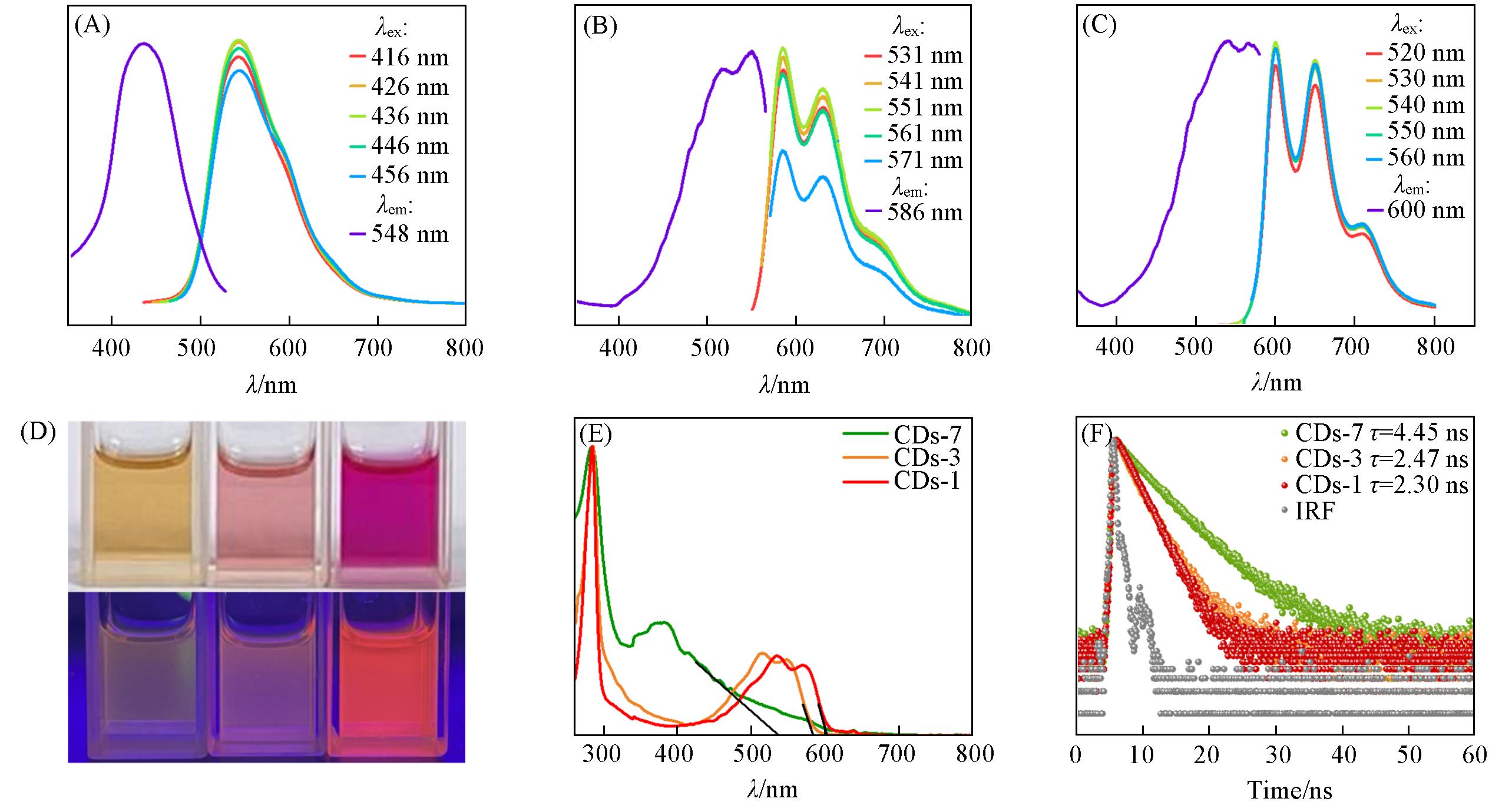
Fig.4 PL excitation and emission spectra under different excitation of CDs⁃7(A), CDs⁃3(B) and CDs⁃1(C) in ethanol, photographs of three types of CD ethanol solutions taken under daylight and 365 nm UV lamp irradiation(D), UV⁃Vis absorption spectra(E) and PL decay curves(F) of CDs⁃7, CDs⁃3 and CDs⁃1
| Sample | τ1/ns | B1 | τ2/ns | B2 | τ3/ns | B3 | τ(avg)/ns | X2 |
|---|---|---|---|---|---|---|---|---|
| CDs⁃7 | 0.5027 | 0.0484 | 3.647 | 0.0542 | 6.268 | 0.0223 | 4.50 | 1.0950 |
| CDs⁃3 | 0.6430 | 0.0370 | 2.417 | 0.0633 | 7.957 | 0.0012 | 2.47 | 1.1244 |
| CDs⁃1 | 2.1980 | 0.0956 | 9.565 | 0.0003 | — | — | 2.30 | 1.1970 |
Table 4 PL lifetimes of CDs-7, CDs-3 and CDs-1*
| Sample | τ1/ns | B1 | τ2/ns | B2 | τ3/ns | B3 | τ(avg)/ns | X2 |
|---|---|---|---|---|---|---|---|---|
| CDs⁃7 | 0.5027 | 0.0484 | 3.647 | 0.0542 | 6.268 | 0.0223 | 4.50 | 1.0950 |
| CDs⁃3 | 0.6430 | 0.0370 | 2.417 | 0.0633 | 7.957 | 0.0012 | 2.47 | 1.1244 |
| CDs⁃1 | 2.1980 | 0.0956 | 9.565 | 0.0003 | — | — | 2.30 | 1.1970 |
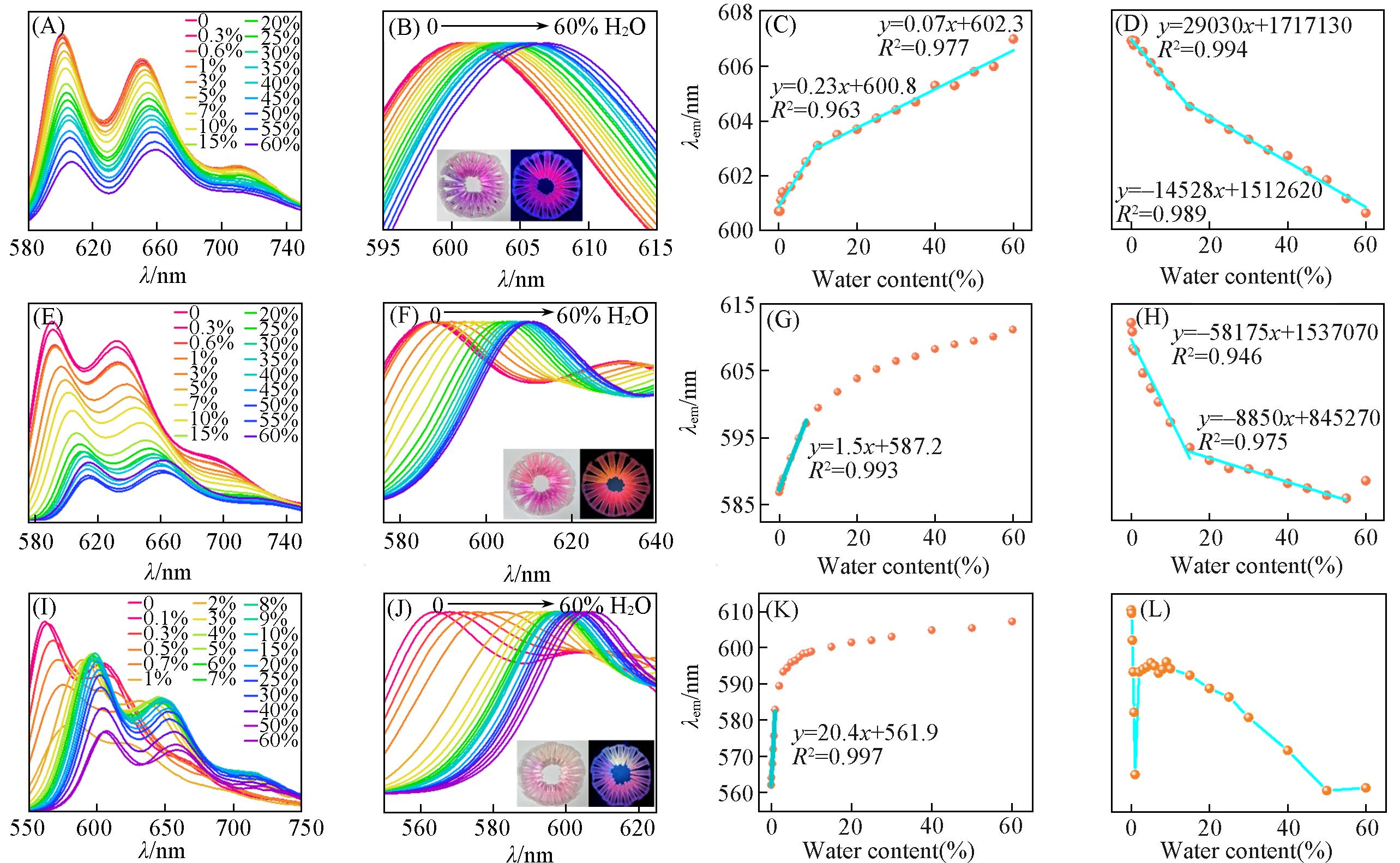
Fig.6 Emission spectra(A, E, I) and normalized emission spectra(B, F, J) of CDs⁃1 dispersed in EtOH(A, B), DMF(E, F) and DIO(I, J) with different water contents, linear relationship between emission peak and fluorescence intensity of CDs⁃1 dispersed in EtOH(C, D), DMF(G, H) and 1,4⁃dioxane (K, L) and water contentInsets of (B), (F) and (J): visual PL photos of CDs⁃1 dispersed in EtOH, DMF and DIO with various water contents excited at 365 nm.
| Material | Method | Response ranges and detection limit(%) | R2 | Ref. |
|---|---|---|---|---|
Ethylenediamine β⁃Resorcylic acid | Emission peak | 0—2, 0.37(Acetone) 0—2, 0.36(THF) | 0.970 0.973 | [ |
| o⁃Phenylenediamine | Emission peak | 0—3, 0.19(Acetone) 0—3, 0.13(THF) 0—3, 0.18(Acetonitrile) | 0.971 | [ |
| 2,5⁃Dihydroxyterephthalic acid | Ratiometric fluorescent | 0.2—6, 0.052(Ethanol) 0.5—8, 0.085(Acetonitrile) 0.8—8, 0.10(THF) 1—8, 0.18(1,4⁃Dioxane) | 0.996 0.992 0.997 0.992 | [ |
| 1,4⁃Phenylene diisocyanate | Intensity/Emission peak | 0—1.5, 0.006(DMF) 0—5, 0.004(Ethyl acetate) | 0.968 0.989 | [ |
| Ethylenediamine and Catechol | Intensity/Emission peak | 0.1—2.5, 0.07(Acetone) 2—20, 0.46(Methanol) | 0.995 0.993 | [ |
| o⁃Phenylenediamine and Dopamine HCl | Emission peak Intensity | 0—10, 4.11(EtOH) 0—7, 0.123(DMF) 0—1, 0.023(DIO) 0—15, 0.86(EtOH) 010, 0.33(DMF) | 0.963 0.993 0.997 0.994 0.946 | This work |
Table 5 Comparison of determination methods of trace water in organic solvents
| Material | Method | Response ranges and detection limit(%) | R2 | Ref. |
|---|---|---|---|---|
Ethylenediamine β⁃Resorcylic acid | Emission peak | 0—2, 0.37(Acetone) 0—2, 0.36(THF) | 0.970 0.973 | [ |
| o⁃Phenylenediamine | Emission peak | 0—3, 0.19(Acetone) 0—3, 0.13(THF) 0—3, 0.18(Acetonitrile) | 0.971 | [ |
| 2,5⁃Dihydroxyterephthalic acid | Ratiometric fluorescent | 0.2—6, 0.052(Ethanol) 0.5—8, 0.085(Acetonitrile) 0.8—8, 0.10(THF) 1—8, 0.18(1,4⁃Dioxane) | 0.996 0.992 0.997 0.992 | [ |
| 1,4⁃Phenylene diisocyanate | Intensity/Emission peak | 0—1.5, 0.006(DMF) 0—5, 0.004(Ethyl acetate) | 0.968 0.989 | [ |
| Ethylenediamine and Catechol | Intensity/Emission peak | 0.1—2.5, 0.07(Acetone) 2—20, 0.46(Methanol) | 0.995 0.993 | [ |
| o⁃Phenylenediamine and Dopamine HCl | Emission peak Intensity | 0—10, 4.11(EtOH) 0—7, 0.123(DMF) 0—1, 0.023(DIO) 0—15, 0.86(EtOH) 010, 0.33(DMF) | 0.963 0.993 0.997 0.994 0.946 | This work |
| 1 | Jia H. R., Wang Z. B., Yuan T., Yuan F. L., Li X. H., Li Y. C., Tan Z. A., Fan L. Z., Yang S. H., Adv. Sci., 2019, 6(13), 1900397 |
| 2 | Liu J. J., Geng Y. J., Li D. W., Yao H., Huo Z. P., Li Y. F., Zhang K., Zhu S. J., Wei H. T., Xu W. Q., Jiang J. L., Yang B., Adv. Mater., 2020, 33(38), 2007162 |
| 3 | Zhang M. L., Ma Y. R., Wang H. B., Wang B., Zhou Y. J., Liu Y., Shao M. W, Huang H., Lu F., Kang Z. H., ACS Appl. Mater. Interfaces, 2021, 13(4), 5877—5886 |
| 4 | Chen X. K., Zhang X. D., Xia L. Y., Wang H. Y., Chen Z., Wu, F. G., Nano Lett., 2018, 18(2), 1159—1167 |
| 5 | Sun X. C., Lei Y., Trac⁃Trends Anal. Chem., 2017, 89, 163—180 |
| 6 | Ding H., Zhou X. X., Zhang Z. H., Xia K. L., Zhao Y. P., J. Chinese Universities, 2021, 42(6), 2080—2084 |
| 丁辉, 周旋旋, 张子慧, 夏坤林, 赵云鹏. 高等学校化学学报, 2021, 42(6), 2080—2084 | |
| 7 | Li D. N., He H. X., Zhang H. R., Yang X., Kang Y. Y., Dong R. Y., Li W., Pan X. Q., Kou E. F., Liu Y. L., Lei B. F., Chin. J. Lumin., 2021, 42(5), 635—641 |
| 李冬娜, 何红想, 张浩然, 杨暹, 康云艳, 董日月, 李唯, 潘晓琴, 寇尔丰, 刘应亮, 雷炳富. 发光学报, 2021, 42(5), 635—641 | |
| 8 | Jiao Y. Y., Huang Q. Z., Wang J. S., He Z. H., Li Z. J., Appl. Catal. B: Environ., 2019, 247, 124—132 |
| 9 | Li Y. D., Xu X. K., Li W., Hu C. F., Zhuang J. L., Zhang X. J., Lei B. F., Liu Y. L., Chin. J. Lumin., 2021, 42(8), 1172—1181 |
| 李亚东, 许晓凯, 李唯, 胡超凡, 庄健乐, 张学杰, 雷炳富, 刘应亮. 发光学报, 2021, 42(8), 1172—1181 | |
| 10 | Zhang Y. Q., Liu X. Y., Fan Y., Guo X. Y., Zhou L., Lv Y., Lin J., Nanoscale, 2016, 8(33), 15281—15287 |
| 11 | Li D., Jing P. T., Sun L. H., An Y., Shan X. Y., Lu X. H., Zhou D., Han D., Shen D. Z., Zhai Y. C., Qu S. N., Zbořil R., Rogach A. L., Adv. Mater., 2018, 30(13), 1705913 |
| 12 | Li D. N., Li, W., Zhang H. R., Zhang X. J., Zhuang J. L., Liu Y. L., Hu C. F., Lei B. F., ACS Appl. Mater. Interfaces, 2020, 12(18), 21009—21019 |
| 13 | Miao S. H., Liang K., Zhu J. J., Yang B., Zhao D. Y., Kong B., Nano Today, 2020, 33, 100879 |
| 14 | Gao D., Liu A. M., Zhang Y. S., Zhu Y. D., Wei D., Sun J., Luo H. R., Fan H. S., Chem. Eng. J., 2021, 415, 128984 |
| 15 | Dai R. Y., Chen X. P., Oyang N., Hu Y. P., Chem. Eng. J., 2021, 431, 134172 |
| 16 | Jiang K., Sun S., Zhang L., Lu Y., Wu A. G., Cai C. Z., Lin H. W., Angew. Chem. Int. Ed., 2015, 54(18), 5360—5363 |
| 17 | Gao D., Zhang Y. S., Liu A. M., Zhu Y. D., Chen S. P., Wei D., Sun. J., Guo Z. Z., Fan H. S., Chem. Eng. J., 2020, 388, 12499 |
| 18 | Zhu Z. J., Zhai Y. L., Li Z. H., Zhu P. Y., Mao S., Zhu C. Z., Du D., Belfiore L. A., Tang J. G., Lin Y. H., Mater. Today, 2019, 30, 52—79 |
| 19 | Wang L., Li W. T., Yin L. Q., Liu Y. J., Guo H. Z., Lai J. W., Han Y., Li G., Li M., Zhang J. H., Vajtai R., Ajayan P. M., Wu M. H., Sci. Adv., 2020, 6(40), eabb6772 |
| 20 | Chao D. Y., Lyu W., Liu Y. B., Zhou L., Zhang Q. R., Deng R. P., Zhang H. J., J. Mater. Chem. C, 2018, 6(28), 7527—7532 |
| 21 | Wang D. J., Zhao H. W., Li H. J., Sun S. G., Xu Y. Q., J. Mater. Chem. C, 2016, 4(47), 11050—11054 |
| 22 | Li Y., Fan K. C., Ban H. T., Yang M. J., Sens. Actuator B: Chem., 2015, 222, 151—158 |
| 23 | Jung H. S., Verwilst P., Kim W. Y., Kim J. S., Chem. Soc. Rev., 2016, 45(5), 1242—1256 |
| 24 | Tang S. Y., Chen D., Yang Y. S., Wang C. X., Li X. M., Wang Y. R., Gu C. J., Cao Z., J. Colloid Interface Sci., 2022, 617, 182—192 |
| 25 | Li D.N., Kou E. F., Li W., Zhang H. R., Zhang X. J., Zhuang J. L., Liu Y. L., Hu C. F., Zheng Y. L., Yang Q. C., Lei B. F., Chem. Eng. J., 2021, 425, 131653 |
| 26 | Cao Y. Q., Chen L. G., Liu D. Q., Wang B. W., Sci. China: Mater., 2018, 62(6), 823—830 |
| 27 | Chen Z. E., Qi Q. L., Zhang H., Spectroc. Acta Pt. A: Molec. Biomolec. Spectr., 2020, 238, 118384 |
| 28 | Zhou S. H., Li Q., Zhang T., Pang D. W., Tang H. W., Chem. J. Chinese Universities, 2019, 40(8), 1593—1599 |
| 周思慧, 李琼, 张婷, 庞代文, 唐宏武. 高等学校化学学报, 2019, 40(8), 1593—1599 | |
| 29 | Miao X., Qu D., Yang D. X., Nie B., Zhao Y. K., Fan H. Y., Sun Z. C, Adv. Mater., 2017, 30(1), 1704740 |
| 30 | Guo J. Z., Lu Y. S., Xie A. Q., Li G., Liang Z. B., Wang C. F., Yang X. F., Chen S., Adv. Funct. Mater., 2022, 32(20), 2110393 |
| 31 | Zhang Q., Wang R. Y., Feng B. W., Zhong X. X., Ostrikov K., Nat. Commun., 2021, 12(1), 6856 |
| 32 | Ji C. Y., Han Q. R., Zhou Y. Q., Wu J. J., Shi W. Q., Gao L. P., Leblanc R. M., Peng Z. L., Carbon, 2022, 192, 198—208 |
| 33 | Tetsuka H., Nagoya A., Fukusumi T., Matsui T., Adv. Mater., 2016, 28(23), 4632—4638 |
| 34 | Samanta S., Roy P., Kar P., Macromol Res., 2016, 24(4), 342—349 |
| 35 | Liu Y. H., Zhu C., Gao Y., Yang L., Xu J. Y., Zhang X. T., Lu C., Wang Y. F., Zhu Y. Z., Appl. Surf. Sci., 2020, 510, 145437 |
| 36 | Gao D., Zhang Y. S., Wu K., Min H. Y., Wei D., Sun J., Yang H. Q., Fan H. S., Biosens. Bioelectron., 2021, 200, 113928 |
| 37 | Huang H., Li C. G., Shi Z., Feng S. H., Chem. J. Chinese Universities, 2019, 40(8), 1579—1585 |
| 黄贺, 李春光, 施展, 冯守华. 高等学校化学学报, 2019, 40(8), 1579—1585 | |
| 38 | Su W., Guo R. H., Yuan F. L., Li Y. C., Li X. H., Zhang Y., Zhou S. X., Fan L. Z., J. Phys. Chem. Lett., 2020, 11(11), 4345 |
| 39 | Hu Y. P., Gao Z. J., J. Hazard. Mater., 2020, 288, 122073 |
| 40 | Yuan F. L., Yuan T., Sui L. Z., Wang Z. B., Xi Z. F., Li Y. C., Li X. H., Fan L. Z., Tan Z. A., Chen A. M., Jin M. X., Yang S. H., Nat. Commun., 2018, 9, 2249 |
| 41 | Wang B. Y., Yu J. K., Sui L. Z., Zhu S. J., Tang Z. Y., Yang B., Lu S. Y., Adv. Sci., 2020, 8(1), 2001453 |
| 42 | Sk M. A., Ananthanarayanan A., Huang L., Lim K. H., Chen P., J. Mater. Chem. C, 2014, 2(34), 6954—6960 |
| 43 | Moniruzzaman M., Kim J., Sens. Actuator B: Chem., 2019, 295, 12—21 |
| 44 | Chao D. Y., Lyu W., Liu Y. B., Zhou L., Zhang Q. R., Deng R. P., Zhang H. J., J. Mater. Chem. C, 2018, 6, 7527—7532 |
| 45 | Qin Y. J., Bai Y. J., Huang P. C., Wu, F. Y., ACS Appl. Nano Mater., 2021, 4, 10674—10681 |
| 46 | Bai J. J., Cui J. J., Ma Y. D., Zhao W. H., Wang Y. L., Li Z. Z., Opt. Mater., 2022, 123, 111927 |
| 47 | Wang X. Y., Teng X. M., Sun X. B., Pan, W., Wang, J. P., Spectroc. Acta Pt. A: Molec. Biomolec. Spectr., 2021, 267, 120547 |
| [1] | 赵永梅, 穆叶舒, 洪琛, 罗稳, 田智勇. 双萘酰亚胺衍生物用于检测水溶液中的苦味酸[J]. 高等学校化学学报, 2022, 43(3): 20210765. |
| [2] | 唐倩, 但飞君, 郭涛, 兰海闯. 喹啉酮-香豆素类Hg2+比色荧光探针的合成及应用[J]. 高等学校化学学报, 2022, 43(2): 20210660. |
| [3] | 李成, 周森森, 蒋锡群. 乏氧光学影像探针的设计与应用[J]. 高等学校化学学报, 2022, 43(12): 20220558. |
| [4] | 杨燕玲, 叶德举. 碳酸酐酶靶向探针的研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220557. |
| [5] | 姚善昆, 丁伟忠, 吴延平, 陈韵聪, 郭子建. 硫代部花菁类染料生物成像及诊疗的研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220568. |
| [6] | 徐心昱, 张乐天, 曹晖, 马原, 刘柳卉, 宋国胜, 张晓兵. 脂质响应型探针用于动脉粥样硬化成像及治疗的研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220383. |
| [7] | 王迪, 钟克利, 汤立军, 侯淑华, 吕春欣. 席夫碱共价有机框架的合成及对I ‒ 的识别[J]. 高等学校化学学报, 2022, 43(10): 20220115. |
| [8] | 袁春玲, 姚晓条, 徐远金, 覃秀, 石睿, 成诗琦, 王益林. 双功能碳点用于葡萄糖的比色/比率荧光测定[J]. 高等学校化学学报, 2021, 42(8): 2428. |
| [9] | 李安然, 赵冰, 阚伟, 宋天舒, 孔祥东, 卜凡强, 孙立, 殷广明, 王丽艳. 基于菲并咪唑的ON⁃OFF⁃ON双比色荧光探针及细胞成像[J]. 高等学校化学学报, 2021, 42(8): 2403. |
| [10] | 黄珊, 姚建东, 宁淦, 肖琦, 刘义. 石墨烯量子点荧光探针对碱性磷酸酶活性的高效检测[J]. 高等学校化学学报, 2021, 42(8): 2412. |
| [11] | 丁辉, 周旋旋, 张子慧, 夏坤林, 赵云鹏. 无溶剂法大量制备高效红光碳点及其在白光器件中的应用[J]. 高等学校化学学报, 2021, 42(6): 2080. |
| [12] | 孙海珠, 杨国夺, 杨柏. 碳点的设计合成、 结构调控及应用[J]. 高等学校化学学报, 2021, 42(2): 349. |
| [13] | 杨新杰, 赖艳琼, 李秋旸, 张艳丽, 王红斌, 庞鹏飞, 杨文荣. 基于环状DNA-银纳米簇荧光探针对微囊藻毒素-LR的传感检测[J]. 高等学校化学学报, 2021, 42(12): 3600. |
| [14] | 谌委菊, 陈诗雅, 薛曹叶, 刘波, 郑晶. 缺氧响应荧光探针的成像及治疗应用[J]. 高等学校化学学报, 2021, 42(11): 3433. |
| [15] | 颜范勇, 孙中慧, 庞纪平, 江英霞, 陈圆. 苯并噻嗪衍生物功能化的碳点用于检测银杏叶茶中的槲皮素[J]. 高等学校化学学报, 2020, 41(8): 1768. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||