

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (12): 2749.doi: 10.7503/cjcu20200563
收稿日期:2020-08-14
出版日期:2020-12-10
发布日期:2020-12-09
通讯作者:
黄超
E-mail:huangchao@ynu.edu.cn
基金资助:
LIN Junjie, WANG Shuang, LI Weiqiang, CUI Xin, HUANG Chao( )
)
Received:2020-08-14
Online:2020-12-10
Published:2020-12-09
Contact:
HUANG Chao
E-mail:huangchao@ynu.edu.cn
Supported by:摘要:
建立了一种吡啶[2,3-d]嘧啶类化合物的高效合成方法. 以α,β?不饱和酮类化合物和1,3-二甲基-6-氨基脲嘧啶为原料, 在微波辅助无催化剂条件下, 于5~15 min内高效串联环化合成了28个具有分子多样性的吡啶[2,3-d]嘧啶类化合物, 其中21个化合物未见文献报道. 该方法具有原料简单易得、 高效绿色、 成键效率高及后处理简单等优点.
中图分类号:
TrendMD:
林俊洁, 王爽, 李伟强, 崔鑫, 黄超. 微波辅助无催化剂高效串联环化合成吡啶[2,3⁃d]嘧啶衍生物. 高等学校化学学报, 2020, 41(12): 2749.
LIN Junjie, WANG Shuang, LI Weiqiang, CUI Xin, HUANG Chao. Efficient Synthesis of Pyridine [2,3-d]pyrimidine Derivatives by Catalyst-free Tandem Cyclization Under Microwave Irradiation. Chem. J. Chinese Universities, 2020, 41(12): 2749.
| Compd. | Appearance | Yield*(%) | m. p./℃(ref.) | HRMS, m/z[M+H]+ | IR(KBr), |
|---|---|---|---|---|---|
| 3a | White solid | 88 | 245—246(262—264)[ | 344.1398 | 3067, 2906, 1705, 1663, 1555, 1420, 1363, 1004, 771, 701 |
| 3b | White solid | 85 | 220—223 | 358.1557 | 3327, 2988, 1658, 1550, 1364, 1092, 821, 777, 698 |
| 3c | White solid | 83 | 220—222 | 358.1555 | 3335, 2978, 1701, 1658, 1553, 1364, 1121, 747, 609 |
| 3d | Yellow solid | 85 | 260—262 | 371.1711 | 3376, 2977, 1702, 1655, 1553, 1417, 1095, 747, 610 |
| 3e | White solid | 79 | 231—233 | 358.1555 | 3284, 2980, 1704, 1657, 1364, 1095, 699, 608 |
| 3f | White solid | 78 | 232—234 | 358.1557 | 3384, 2987, 1605, 1690, 1655, 1369, 1193, 812, 748 |
| 3g | White solid | 81 | 214—216(217—219)[ | 372.1713 | 3379, 2988, 1705, 1658, 1655, 1363, 1093, 802, 749 |
| 3h | White solid | 78 | 253—254 | 374.1504 | 3356, 2976, 1655, 1551, 1416, 1367, 1243, 1178, 608, 569 |
| 3i | White solid | 82 | 201—203 | 388.1661 | 3357, 2989, 1706, 1678, 1261, 1083, 800, 608 |
| 3j | White solid | 81 | 228—231 | 404.1609 | 3354, 2968, 1704, 1654, 1600, 1089, 999, 787, 609 |
| 3k | White solid | 80 | 241—243(259—260)[ | 388.1661 | 3368, 2879, 1706, 1674, 1603, 1089, 998, 786, 605 |
| 3l | White solid | 88 | 241—243 | 362.1306 | 3389, 2989, 1708, 1661, 1550, 1361, 1159, 1001, 608, 562 |
| 3m | White solid | 85 | 251—253 | 378.1008 | 3354, 2968, 1655, 1567, 1365, 1094, 752, 609 |
| 3n | White solid | 85 | 231—233 | 422.0505 | 3375, 2968, 1655, 1549, 1360, 1112, 1001, 751, 609 |
| 3o | White solid | 84 | 235—237 | 376.1463 | 3389, 2988, 1666, 1567, 1546, 1153, 1096, 840, 799, 607 |
| 3p | White solid | 84 | 232—233(238—239)[ | 392.1167 | 3387, 2999, 1701, 1667, 1590, 1545, 1366, 1091, 799, 610 |
| 3q | White solid | 82 | 230—232 | 436.0662 | 3375, 2976, 1705, 1592, 1543, 1361, 1086, 1003, 823, 750, 609 |
| 3r | Yellow solid | 79 | 240—242 | 389.1252 | 3369, 2968, 1706, 1670, 1550, 1342, 1109, 521, 694, 609 |
| 3s | White solid | 77 | 214—215 | 369.1351 | 3378, 2899, 2025, 1704, 1360, 1120, 608, 536 |
| 3t | White solid | 78 | 215—217 | 360.1349 | 3397, 3066, 2948, 1710, 1670, 1551, 1367, 1224, 770, 746 |
| 3u | White solid | 84 | 220—222 | 383.1509 | 3389, 2967, 1633, 1556, 1367, 1128, 999, 671 |
| 3v | Yellow solid | 80 | 201—202 | 394.1555 | 3398, 2977, 1705, 1663, 1552, 1348, 1090, 767, 609 |
| 3w | Purple solid | 81 | 238—240 | 350.0964 | 3386, 2988, 1709, 1656, 1415, 1097, 730, 607 |
| 3x | Yellow solid | 81 | 217—218(213—215)[ | 350.0965 | 3389, 2977, 2025, 1656, 1424, 1094, 800, 608 |
| 5a | White solid | 70 | 165—166(171—178)[ | 268.1083 | 3367, 2980, 1705, 1655, 1122, 608 |
| 5b | White solid | 73 | 184—186 | 282.1235 | 3358, 2966, 1706, 1657, 1551, 1416, 1375, 754, 697, 550 |
| 5c | White solid | 71 | 186—188 | 296.1941 | 3378, 2980, 2025, 1702, 1667, 2593, 1419, 753, 608 |
| 5d | White solid | 78 | 185—186(180—182)[ | 298.1193 | 3301, 3129, 2987, 1626, 1504, 1155, 1063, 754, 539 |
Table 1 Appearance, yields, melting points, HRMS and IR data for compounds 3a—3x and 5a—5d
| Compd. | Appearance | Yield*(%) | m. p./℃(ref.) | HRMS, m/z[M+H]+ | IR(KBr), |
|---|---|---|---|---|---|
| 3a | White solid | 88 | 245—246(262—264)[ | 344.1398 | 3067, 2906, 1705, 1663, 1555, 1420, 1363, 1004, 771, 701 |
| 3b | White solid | 85 | 220—223 | 358.1557 | 3327, 2988, 1658, 1550, 1364, 1092, 821, 777, 698 |
| 3c | White solid | 83 | 220—222 | 358.1555 | 3335, 2978, 1701, 1658, 1553, 1364, 1121, 747, 609 |
| 3d | Yellow solid | 85 | 260—262 | 371.1711 | 3376, 2977, 1702, 1655, 1553, 1417, 1095, 747, 610 |
| 3e | White solid | 79 | 231—233 | 358.1555 | 3284, 2980, 1704, 1657, 1364, 1095, 699, 608 |
| 3f | White solid | 78 | 232—234 | 358.1557 | 3384, 2987, 1605, 1690, 1655, 1369, 1193, 812, 748 |
| 3g | White solid | 81 | 214—216(217—219)[ | 372.1713 | 3379, 2988, 1705, 1658, 1655, 1363, 1093, 802, 749 |
| 3h | White solid | 78 | 253—254 | 374.1504 | 3356, 2976, 1655, 1551, 1416, 1367, 1243, 1178, 608, 569 |
| 3i | White solid | 82 | 201—203 | 388.1661 | 3357, 2989, 1706, 1678, 1261, 1083, 800, 608 |
| 3j | White solid | 81 | 228—231 | 404.1609 | 3354, 2968, 1704, 1654, 1600, 1089, 999, 787, 609 |
| 3k | White solid | 80 | 241—243(259—260)[ | 388.1661 | 3368, 2879, 1706, 1674, 1603, 1089, 998, 786, 605 |
| 3l | White solid | 88 | 241—243 | 362.1306 | 3389, 2989, 1708, 1661, 1550, 1361, 1159, 1001, 608, 562 |
| 3m | White solid | 85 | 251—253 | 378.1008 | 3354, 2968, 1655, 1567, 1365, 1094, 752, 609 |
| 3n | White solid | 85 | 231—233 | 422.0505 | 3375, 2968, 1655, 1549, 1360, 1112, 1001, 751, 609 |
| 3o | White solid | 84 | 235—237 | 376.1463 | 3389, 2988, 1666, 1567, 1546, 1153, 1096, 840, 799, 607 |
| 3p | White solid | 84 | 232—233(238—239)[ | 392.1167 | 3387, 2999, 1701, 1667, 1590, 1545, 1366, 1091, 799, 610 |
| 3q | White solid | 82 | 230—232 | 436.0662 | 3375, 2976, 1705, 1592, 1543, 1361, 1086, 1003, 823, 750, 609 |
| 3r | Yellow solid | 79 | 240—242 | 389.1252 | 3369, 2968, 1706, 1670, 1550, 1342, 1109, 521, 694, 609 |
| 3s | White solid | 77 | 214—215 | 369.1351 | 3378, 2899, 2025, 1704, 1360, 1120, 608, 536 |
| 3t | White solid | 78 | 215—217 | 360.1349 | 3397, 3066, 2948, 1710, 1670, 1551, 1367, 1224, 770, 746 |
| 3u | White solid | 84 | 220—222 | 383.1509 | 3389, 2967, 1633, 1556, 1367, 1128, 999, 671 |
| 3v | Yellow solid | 80 | 201—202 | 394.1555 | 3398, 2977, 1705, 1663, 1552, 1348, 1090, 767, 609 |
| 3w | Purple solid | 81 | 238—240 | 350.0964 | 3386, 2988, 1709, 1656, 1415, 1097, 730, 607 |
| 3x | Yellow solid | 81 | 217—218(213—215)[ | 350.0965 | 3389, 2977, 2025, 1656, 1424, 1094, 800, 608 |
| 5a | White solid | 70 | 165—166(171—178)[ | 268.1083 | 3367, 2980, 1705, 1655, 1122, 608 |
| 5b | White solid | 73 | 184—186 | 282.1235 | 3358, 2966, 1706, 1657, 1551, 1416, 1375, 754, 697, 550 |
| 5c | White solid | 71 | 186—188 | 296.1941 | 3378, 2980, 2025, 1702, 1667, 2593, 1419, 753, 608 |
| 5d | White solid | 78 | 185—186(180—182)[ | 298.1193 | 3301, 3129, 2987, 1626, 1504, 1155, 1063, 754, 539 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(100 MHz), δ |
|---|---|---|
| 3a | 8.13—8.15(m, 2H), 7.52—7.50(m, 3H), 7.48—7.46(m, 4H), 7.37—7.33(m, 2H), 3.89(s, 3H), 3.39(s, 3H) | 159.5, 158.2, 154.2, 150.8, 150.6, 138.6, 136.2, 129.7, 127.9, 127.2, 126.8, 126.5, 117.4, 105.5, 29.1, 27.4 |
3b | 8.25—8.28(s, 1H), 7.57—7.50(m, 3H), 7.30—7.28(m, 2H), 7.24—7.22(m, 2H), 3.72(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.8, 157.7, 154.4, 151.5, 151.0, 137.2, 136.8, 136.4, 130.7, 129.0, 128.3, 128.0, 127.5, 117.8, 106.4, 99.5, 29.8, 28.0, 20.9 |
| 3c | 8.18—8.16(s, 1H), 7.57(s, 1H), 7.43—7.33(m, 7H), 3.72(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.5, 158.2, 154.1, 150.8, 150.6, 140.2, 138.7, 133.5, 128.7, 127.1, 126.8, 126.5, 117.0, 105.6, 29.1, 27.4, 20.4 |
3d | 7.90—7,87 (m, 2H), 7.48—7.44(m, 4H), 7.36—7.34(m, 2H), 7.27(s, 1H), 3.89(s, 3H), 3.39(s, 3H), 2.37—2.34(m, 6H) | 159.6, 158.4, 154.0, 150.7, 150.6, 138.9, 138.7, 136.2, 133.9, 129.3, 127.6, 127.1, 126.8, 126.8, 124.1, 117.1, 105.2, 29.1, 27.4, 19.0, 18.8 |
3e | 8.23—8.26(m, 3H), 7.58(d,J=8.0 Hz, 1H), 7.52—7.53(m, 3H), 7.29—7.27(m, 2H), 3.71(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.5, 158.1, 154.4, 150.8, 150.6, 137.1, 136.3, 135.6, 129.7, 127.9, 127.6, 126.8, 126.5, 117.5, 105.6, 29.1, 27.4, 20.0 |
3f | 8.15—8.13(m, 2H), 7.52—7.50(m, 3H), 7.47(s, 1H), 7.30—7.24(m, 3H), 7.24(s, 1H), 3.89(s, 3H), 3.40(s, 3H), 2.44(s, 3H) | 159.5, 158.1, 154.4, 150.8, 150.6, 137.1, 136.3, 135.6, 129.7, 127.9, 127.6, 126.8, 126.5, 117.5, 105.6, 29.1, 27.4, 20.4 |
3g | 8.02—8.04(m, 2H), 7.44(s, 1H), 7.32—7.27(m, 5H), 7.25(d, J=6.1 Hz, 1H), 3.88(s, 3H), 3.39(s, 3H), 2.44(d, J=3.8 Hz, 6H) | 159.6, 158.1, 154.2, 150.8, 150.6, 140.1, 137.0, 135.7, 133.5, 128.7, 127.8, 126.8, 126.4, 117.1, 105.3, 29.1, 27.4, 20.4, 20.4 |
3h | 8.26(d, J=9.0 Hz, 2H), 7.56(s, 1H), 7.43—7.41(m, 5H), 7.08(d, J=9.0 Hz, 2H), 3.84(s, 3H), 3.73(s, 3H), 3.19(s, 3H) | 160.9, 159.5, 157.8, 154.0 150.8, 150.6, 138.7, 128.7, 128.1, 127.1, 126.8, 126.8, 116.5, 113.3, 104.8, 54.4, 29.1, 27.4 |
3i | 8.04(d, J=8.2 Hz, 2H), 7.43(s, 1H), 7.31(d, J=8.7 Hz, 4H), 7.00(d, J=8.8 Hz, 2H), 3.88(d, J=0.5 Hz, 6H), 3.40(s, 3H), 2.43(s, 3H) | 159.7, 158.7, 158.1, 153.9, 150.9, 150.6, 140.1, 133.6, 130.7, 128.8, 128.4, 126.4, 117.3, 112.3, 105.2, 54.3, 29.1, 27.4, 20.4 |
3j | 8.10(d, J=8.9 Hz, 2H), 8.10(d, J=8.9 Hz, 2H), 7.38(s, 1H), 7.30(d, J=8.7 Hz, 2H), 7.00(m, 4H), 3.88—3.86(m, 8H), 3.39(s, 3H) | 160.9, 159.6, 158.7, 157.6, 153.8, 150.9, 150.6, 130.8, 128.8, 128.4, 128.0, 116.8, 113.3, 112.3, 104.8, 54.4, 54.3, 29.1, 27.4 |
3k | 8.10(d, J=8.9 Hz, 2H), 7.39(s, 1H), 7.28(d, J=8.1 Hz, 2H), 7.25(m, 2H), 7.01(d, J=8.9 Hz, 2H), 3.88(d, J=7.4 Hz, 6H), 3.39(s, 3H), 2.44(s, 3H) | 160.9, 159.6, 157.7, 154.1, 150.8, 150.6, 137.0, 135.8, 128.8, 128.0, 127.6, 126.8, 116.6, 113.3, 104.9, 54.4, 29.1, 27.4, 20.4 |
3l | 8.17—8.13(m, 2H), 7.49—7.48(m, 3H), 7.42(s, 1H), 7.34(m, 2H), 7.19(t, J=8.7 Hz, 2H), 3.88(s, 3H), 3.39(s, 3H) | 164.8, 162.3, 159.4, 157.1, 154.4, 150.8, 150.5, 138.5, 132.4, 132.4, 128.6, 128.5, 127.2, 126.9, 126.7, 117.0, 115.1, 114.9, 105.5, 29.1, 27.4 |
| 3m | 8.09—8.07(m, 2H), 7.48—7.47(m, 5H), 7.46—7.44(s, 1H), 7.35—7.33(m, 2H), 3.87(s, 3H), 3.39(s, 3H) | 159.4 156.9, 154.5, 150.8, 150.5, 138.4, 136.0, 134.7, 128.2, 127.8, 127.3, 126.9, 126.7, 117.1, 105.7, 29.2, 27.5 |
3n | 8.02—8.00(m, 2H), 7.65—7.63(m, 2H), 7.48—7.47(m, 3H), 7.44(s, 1H), 7.35—7.33(m, 2H), 3.87(s, 3H), 3.39(s, 3H) | 159.4, 157.0, 154.5, 150.8, 150.5, 138.4, 135.1, 131.2, 128.0, 127.3, 126.7, 126.7, 124.5, 117.1, 105.8, 29.2, 27.5 |
3o | 8.16—8.12(m, 2H), 7.41(s, 1H), 7.28(d, J=8.1 Hz, 2H), 7.23—7.17(m, 4H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 164.7, 162.2, 159.5, 157.0, 154.6, 150.8, 150.5, 137.2, 135.5, 132.5, 132.5, 128.6, 128.5, 127.6, 126.8, 117.1, 115.1, 114.9, 105.5, 29.1, 27.4, 20.4 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(100 MHz), δ |
3p | 8.07(d, J=8.7 Hz, 2H), 7.47(d, J=8.7 Hz, 2H), 7.43(s, 1H), 7.28(d, J=8.0 Hz, 2H), 7.25—7.25(m, 2H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 159.4, 156.8, 154.6, 150.9, 150.5, 137.2, 136.0, 135.4, 134.7, 128.2, 127.7, 127.6, 126.8, 117.2, 105.8, 29.2, 27.5, 20.4 |
3q | 8.00(d, J=8.7 Hz, 2H), 7.63(d, J=8.7 Hz, 2H), 7.43(s, 1H), 7.28(d, J=8.0 Hz, 2H), 7.25—7.23(m, 2H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 159.4, 156.9, 154.7, 150.9, 150.5, 137.2, 135.4, 135.2, 131.1, 128.0, 127.6, 126.8, 124.4, 117.2, 105.8, 29.2, 27.5, 20.4 |
| 3r | 8.55(d, J=9.0 Hz, 2H), 8.34(d, J=9.0 Hz, 2H), 7.79(s, 1H), 7.45—7.41(m, 5H), 3.74(s, 3H), 3.20(s, 3H) | 159.6, 155.3, 154.8, 151.6, 150.9, 148.5, 142.6, 139.1, 128.8, 128.3, 128.0, 127.5, 124.0, 119.0, 107.5, 29.9, 28.1 |
3s | 8.26—8.24(m, 2H), 7.81—7.79(m, 2H), 7.50—7.46(m, 4H), 7.35—7.35(m, 2H), 3.88(s, 3H), 3.39(s, 3H) | 159.2, 155.8, 154.9, 150.9, 150.3, 140.3, 138.1, 131.7, 127.5, 127.0, 127.0, 126.7, 117.8, 117.4, 113.0, 106.5, 29.2, 27.5 |
3t | 13.20(s, 1H), 7.85—7.82(m, 1H), 7.57(s, 1H), 7.52—7.47(m, 1H), 7.44—7.38(m, 1H), 7.35(s, 1H), 7.07—7.05(m, 1H), 6.97—6.91(m, 1H), 3.82(s, 3H), 3.39(s, 3H) | 159.2, 158.7, 154.8, 140.0, 139.4, 138.0, 132.6, 127.5, 127.0, 126.6, 118.6 117.8, 116.8, 116.4, 104.9, 29.5, 27.6 |
3u | 8.25(d, J=8.4 Hz, 2H), 7.80(d, J=8.4 Hz, 2H), 7.50(s, 1H), 7.30—7.26(m, 2H), 7.24(s, 1H), 3.88(s, 3H), 3.40(s, 3H), 2.45(s, 3H) | 159.2, 155.7, 155.1, 151.0, 150.4, 140.3, 137.5, 135.1, 131.7, 127.7, 127.0, 126.7, 118.0, 117.4, 113.0, 106.6, 29.2, 27.5, 20.4 |
3v | 8.59(s, 1H), 8.27—8.25(m, 1H), 7.96—7.88(m, 3H), 7.62(s, 1H), 7.57—7.54(m, 2H), 7.51—7.49(m, 3H), 7.40—7.38(m, 2H), 3.93(s, 3H), 3.40(s, 3H) | 159.5, 158.0, 154.2, 150.8, 150.5, 138.6, 133.5, 133.4, 132.2, 128.0, 127.7, 126.8, 126.7,126.7, 126.5, 125.6, 123.3, 117.5 105.5, 29.2, 27.4 |
3w | 8.14—8.12(m, 2H), 7.60(s, 1H), 7.60—7.50(m, 4H), 7.26—7.24(m, 1H), 7.16—7.14(m, 1H), 3.87(s, 3H), 3.43(s, 3H) | 159.3, 158.2, 151.0, 150.4, 146.71, 138.3, 136.0, 129.8, 127.9, 127.4, 126.5, 126.2, 125.9, 118.3, 105.7, 29.2, 27.5 |
3x | 7.73—7.72(m, 1H), 7.56—7.54(m, 1H), 7.48—7.46(m, 3H), 7.35—7.33(m, 3H), 7.16—7.14(m, 1H), 3.83(s, 3H), 3.37(s, 3H) | 159.3, 154.1, 153.3, 150.8, 150.5, 142.3, 138.4, 129.5, 127.5, 127.2, 126.8, 126.7, 126.4, 115.7, 105.1, 29.1, 27.4 |
| 5a | 8.60(d, J=4.9 Hz, 1H), 7.46—7.44(m, 3H), 7.30—7.26(m, 2H), 7.01(d, J=4.9 Hz, 1H), 3.78(s, 3H), 3.37(s, 3H) | 159.5, 153.5, 151.0, 150.3, 138.1, 127.2, 126.8, 121.0, 107.1, 29.2, 27.5 |
| 5b | 7.44—7.42(m, 3H), 7.29—7.26(m, 2H), 6.87(s, 1H), 3.77(s, 3H), 3.36(s, 3H), 2.61(s, 3H) | 161.6, 159.6, 153.4, 150.5, 138.4, 127.0, 126.7, 120.7, 104.5, 29.0, 27.3, 23.9 |
| 5c | 7.25—7.23(m, 4H), 6.85(s, 1H), 3.76(s, 3H), 3.36(s, 3H), 2.60(s, 3H), 2.41(s, 3H) | 161.5, 159.7, 153.6, 150.6, 150.6, 136.9, 135.4, 127.5, 126.7, 120.8, 104.6, 29.0, 27.4, 23.9, 20.3 |
5d | 13.23(s, 1H), 7.52(s, 1H), 7.39—7.37(m, 1H), 7.04—7.02(m, 1H), 6.98—6.94(m, 1H), 3.74(s, 3H), 3.47(s, 3H), 2.91(s, 3H) | 160.1, 159.1, 158.5, 153.7, 145.0, 149.3, 132.3, 126.3, 118.5, 117.7, 116.7, 116.2, 106.2, 29.4, 27.5, 22.1 |
Table 2 1H NMR and 13C NMR data for compounds 3a—3x and 5a—5d*
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(100 MHz), δ |
|---|---|---|
| 3a | 8.13—8.15(m, 2H), 7.52—7.50(m, 3H), 7.48—7.46(m, 4H), 7.37—7.33(m, 2H), 3.89(s, 3H), 3.39(s, 3H) | 159.5, 158.2, 154.2, 150.8, 150.6, 138.6, 136.2, 129.7, 127.9, 127.2, 126.8, 126.5, 117.4, 105.5, 29.1, 27.4 |
3b | 8.25—8.28(s, 1H), 7.57—7.50(m, 3H), 7.30—7.28(m, 2H), 7.24—7.22(m, 2H), 3.72(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.8, 157.7, 154.4, 151.5, 151.0, 137.2, 136.8, 136.4, 130.7, 129.0, 128.3, 128.0, 127.5, 117.8, 106.4, 99.5, 29.8, 28.0, 20.9 |
| 3c | 8.18—8.16(s, 1H), 7.57(s, 1H), 7.43—7.33(m, 7H), 3.72(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.5, 158.2, 154.1, 150.8, 150.6, 140.2, 138.7, 133.5, 128.7, 127.1, 126.8, 126.5, 117.0, 105.6, 29.1, 27.4, 20.4 |
3d | 7.90—7,87 (m, 2H), 7.48—7.44(m, 4H), 7.36—7.34(m, 2H), 7.27(s, 1H), 3.89(s, 3H), 3.39(s, 3H), 2.37—2.34(m, 6H) | 159.6, 158.4, 154.0, 150.7, 150.6, 138.9, 138.7, 136.2, 133.9, 129.3, 127.6, 127.1, 126.8, 126.8, 124.1, 117.1, 105.2, 29.1, 27.4, 19.0, 18.8 |
3e | 8.23—8.26(m, 3H), 7.58(d,J=8.0 Hz, 1H), 7.52—7.53(m, 3H), 7.29—7.27(m, 2H), 3.71(s, 3H), 3.19(s, 3H), 2.38(s, 3H) | 159.5, 158.1, 154.4, 150.8, 150.6, 137.1, 136.3, 135.6, 129.7, 127.9, 127.6, 126.8, 126.5, 117.5, 105.6, 29.1, 27.4, 20.0 |
3f | 8.15—8.13(m, 2H), 7.52—7.50(m, 3H), 7.47(s, 1H), 7.30—7.24(m, 3H), 7.24(s, 1H), 3.89(s, 3H), 3.40(s, 3H), 2.44(s, 3H) | 159.5, 158.1, 154.4, 150.8, 150.6, 137.1, 136.3, 135.6, 129.7, 127.9, 127.6, 126.8, 126.5, 117.5, 105.6, 29.1, 27.4, 20.4 |
3g | 8.02—8.04(m, 2H), 7.44(s, 1H), 7.32—7.27(m, 5H), 7.25(d, J=6.1 Hz, 1H), 3.88(s, 3H), 3.39(s, 3H), 2.44(d, J=3.8 Hz, 6H) | 159.6, 158.1, 154.2, 150.8, 150.6, 140.1, 137.0, 135.7, 133.5, 128.7, 127.8, 126.8, 126.4, 117.1, 105.3, 29.1, 27.4, 20.4, 20.4 |
3h | 8.26(d, J=9.0 Hz, 2H), 7.56(s, 1H), 7.43—7.41(m, 5H), 7.08(d, J=9.0 Hz, 2H), 3.84(s, 3H), 3.73(s, 3H), 3.19(s, 3H) | 160.9, 159.5, 157.8, 154.0 150.8, 150.6, 138.7, 128.7, 128.1, 127.1, 126.8, 126.8, 116.5, 113.3, 104.8, 54.4, 29.1, 27.4 |
3i | 8.04(d, J=8.2 Hz, 2H), 7.43(s, 1H), 7.31(d, J=8.7 Hz, 4H), 7.00(d, J=8.8 Hz, 2H), 3.88(d, J=0.5 Hz, 6H), 3.40(s, 3H), 2.43(s, 3H) | 159.7, 158.7, 158.1, 153.9, 150.9, 150.6, 140.1, 133.6, 130.7, 128.8, 128.4, 126.4, 117.3, 112.3, 105.2, 54.3, 29.1, 27.4, 20.4 |
3j | 8.10(d, J=8.9 Hz, 2H), 8.10(d, J=8.9 Hz, 2H), 7.38(s, 1H), 7.30(d, J=8.7 Hz, 2H), 7.00(m, 4H), 3.88—3.86(m, 8H), 3.39(s, 3H) | 160.9, 159.6, 158.7, 157.6, 153.8, 150.9, 150.6, 130.8, 128.8, 128.4, 128.0, 116.8, 113.3, 112.3, 104.8, 54.4, 54.3, 29.1, 27.4 |
3k | 8.10(d, J=8.9 Hz, 2H), 7.39(s, 1H), 7.28(d, J=8.1 Hz, 2H), 7.25(m, 2H), 7.01(d, J=8.9 Hz, 2H), 3.88(d, J=7.4 Hz, 6H), 3.39(s, 3H), 2.44(s, 3H) | 160.9, 159.6, 157.7, 154.1, 150.8, 150.6, 137.0, 135.8, 128.8, 128.0, 127.6, 126.8, 116.6, 113.3, 104.9, 54.4, 29.1, 27.4, 20.4 |
3l | 8.17—8.13(m, 2H), 7.49—7.48(m, 3H), 7.42(s, 1H), 7.34(m, 2H), 7.19(t, J=8.7 Hz, 2H), 3.88(s, 3H), 3.39(s, 3H) | 164.8, 162.3, 159.4, 157.1, 154.4, 150.8, 150.5, 138.5, 132.4, 132.4, 128.6, 128.5, 127.2, 126.9, 126.7, 117.0, 115.1, 114.9, 105.5, 29.1, 27.4 |
| 3m | 8.09—8.07(m, 2H), 7.48—7.47(m, 5H), 7.46—7.44(s, 1H), 7.35—7.33(m, 2H), 3.87(s, 3H), 3.39(s, 3H) | 159.4 156.9, 154.5, 150.8, 150.5, 138.4, 136.0, 134.7, 128.2, 127.8, 127.3, 126.9, 126.7, 117.1, 105.7, 29.2, 27.5 |
3n | 8.02—8.00(m, 2H), 7.65—7.63(m, 2H), 7.48—7.47(m, 3H), 7.44(s, 1H), 7.35—7.33(m, 2H), 3.87(s, 3H), 3.39(s, 3H) | 159.4, 157.0, 154.5, 150.8, 150.5, 138.4, 135.1, 131.2, 128.0, 127.3, 126.7, 126.7, 124.5, 117.1, 105.8, 29.2, 27.5 |
3o | 8.16—8.12(m, 2H), 7.41(s, 1H), 7.28(d, J=8.1 Hz, 2H), 7.23—7.17(m, 4H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 164.7, 162.2, 159.5, 157.0, 154.6, 150.8, 150.5, 137.2, 135.5, 132.5, 132.5, 128.6, 128.5, 127.6, 126.8, 117.1, 115.1, 114.9, 105.5, 29.1, 27.4, 20.4 |
| Compd. | 1H NMR(400 MHz), δ | 13C NMR(100 MHz), δ |
3p | 8.07(d, J=8.7 Hz, 2H), 7.47(d, J=8.7 Hz, 2H), 7.43(s, 1H), 7.28(d, J=8.0 Hz, 2H), 7.25—7.25(m, 2H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 159.4, 156.8, 154.6, 150.9, 150.5, 137.2, 136.0, 135.4, 134.7, 128.2, 127.7, 127.6, 126.8, 117.2, 105.8, 29.2, 27.5, 20.4 |
3q | 8.00(d, J=8.7 Hz, 2H), 7.63(d, J=8.7 Hz, 2H), 7.43(s, 1H), 7.28(d, J=8.0 Hz, 2H), 7.25—7.23(m, 2H), 3.87(s, 3H), 3.39(s, 3H), 2.44(s, 3H) | 159.4, 156.9, 154.7, 150.9, 150.5, 137.2, 135.4, 135.2, 131.1, 128.0, 127.6, 126.8, 124.4, 117.2, 105.8, 29.2, 27.5, 20.4 |
| 3r | 8.55(d, J=9.0 Hz, 2H), 8.34(d, J=9.0 Hz, 2H), 7.79(s, 1H), 7.45—7.41(m, 5H), 3.74(s, 3H), 3.20(s, 3H) | 159.6, 155.3, 154.8, 151.6, 150.9, 148.5, 142.6, 139.1, 128.8, 128.3, 128.0, 127.5, 124.0, 119.0, 107.5, 29.9, 28.1 |
3s | 8.26—8.24(m, 2H), 7.81—7.79(m, 2H), 7.50—7.46(m, 4H), 7.35—7.35(m, 2H), 3.88(s, 3H), 3.39(s, 3H) | 159.2, 155.8, 154.9, 150.9, 150.3, 140.3, 138.1, 131.7, 127.5, 127.0, 127.0, 126.7, 117.8, 117.4, 113.0, 106.5, 29.2, 27.5 |
3t | 13.20(s, 1H), 7.85—7.82(m, 1H), 7.57(s, 1H), 7.52—7.47(m, 1H), 7.44—7.38(m, 1H), 7.35(s, 1H), 7.07—7.05(m, 1H), 6.97—6.91(m, 1H), 3.82(s, 3H), 3.39(s, 3H) | 159.2, 158.7, 154.8, 140.0, 139.4, 138.0, 132.6, 127.5, 127.0, 126.6, 118.6 117.8, 116.8, 116.4, 104.9, 29.5, 27.6 |
3u | 8.25(d, J=8.4 Hz, 2H), 7.80(d, J=8.4 Hz, 2H), 7.50(s, 1H), 7.30—7.26(m, 2H), 7.24(s, 1H), 3.88(s, 3H), 3.40(s, 3H), 2.45(s, 3H) | 159.2, 155.7, 155.1, 151.0, 150.4, 140.3, 137.5, 135.1, 131.7, 127.7, 127.0, 126.7, 118.0, 117.4, 113.0, 106.6, 29.2, 27.5, 20.4 |
3v | 8.59(s, 1H), 8.27—8.25(m, 1H), 7.96—7.88(m, 3H), 7.62(s, 1H), 7.57—7.54(m, 2H), 7.51—7.49(m, 3H), 7.40—7.38(m, 2H), 3.93(s, 3H), 3.40(s, 3H) | 159.5, 158.0, 154.2, 150.8, 150.5, 138.6, 133.5, 133.4, 132.2, 128.0, 127.7, 126.8, 126.7,126.7, 126.5, 125.6, 123.3, 117.5 105.5, 29.2, 27.4 |
3w | 8.14—8.12(m, 2H), 7.60(s, 1H), 7.60—7.50(m, 4H), 7.26—7.24(m, 1H), 7.16—7.14(m, 1H), 3.87(s, 3H), 3.43(s, 3H) | 159.3, 158.2, 151.0, 150.4, 146.71, 138.3, 136.0, 129.8, 127.9, 127.4, 126.5, 126.2, 125.9, 118.3, 105.7, 29.2, 27.5 |
3x | 7.73—7.72(m, 1H), 7.56—7.54(m, 1H), 7.48—7.46(m, 3H), 7.35—7.33(m, 3H), 7.16—7.14(m, 1H), 3.83(s, 3H), 3.37(s, 3H) | 159.3, 154.1, 153.3, 150.8, 150.5, 142.3, 138.4, 129.5, 127.5, 127.2, 126.8, 126.7, 126.4, 115.7, 105.1, 29.1, 27.4 |
| 5a | 8.60(d, J=4.9 Hz, 1H), 7.46—7.44(m, 3H), 7.30—7.26(m, 2H), 7.01(d, J=4.9 Hz, 1H), 3.78(s, 3H), 3.37(s, 3H) | 159.5, 153.5, 151.0, 150.3, 138.1, 127.2, 126.8, 121.0, 107.1, 29.2, 27.5 |
| 5b | 7.44—7.42(m, 3H), 7.29—7.26(m, 2H), 6.87(s, 1H), 3.77(s, 3H), 3.36(s, 3H), 2.61(s, 3H) | 161.6, 159.6, 153.4, 150.5, 138.4, 127.0, 126.7, 120.7, 104.5, 29.0, 27.3, 23.9 |
| 5c | 7.25—7.23(m, 4H), 6.85(s, 1H), 3.76(s, 3H), 3.36(s, 3H), 2.60(s, 3H), 2.41(s, 3H) | 161.5, 159.7, 153.6, 150.6, 150.6, 136.9, 135.4, 127.5, 126.7, 120.8, 104.6, 29.0, 27.4, 23.9, 20.3 |
5d | 13.23(s, 1H), 7.52(s, 1H), 7.39—7.37(m, 1H), 7.04—7.02(m, 1H), 6.98—6.94(m, 1H), 3.74(s, 3H), 3.47(s, 3H), 2.91(s, 3H) | 160.1, 159.1, 158.5, 153.7, 145.0, 149.3, 132.3, 126.3, 118.5, 117.7, 116.7, 116.2, 106.2, 29.4, 27.5, 22.1 |
| Compd. | Product | Yieldb(%) | Time/min | Compd. | Product | Yieldb(%) | Time/min |
|---|---|---|---|---|---|---|---|
| 3a | 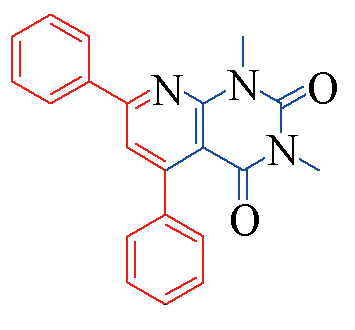 | 88 | 10 | 3h | 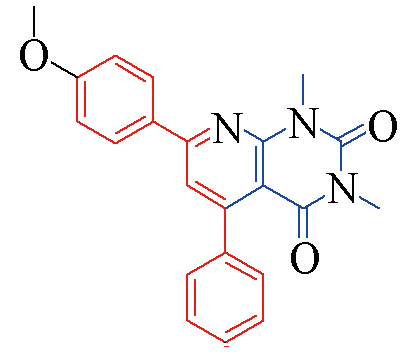 | 78 | 13 |
| 3b | 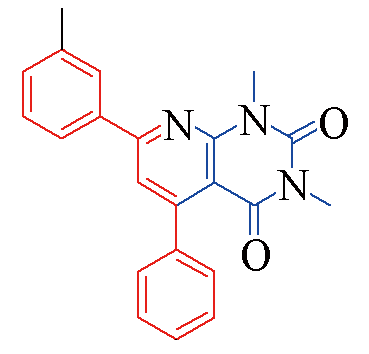 | 85 | 11 | 3i | 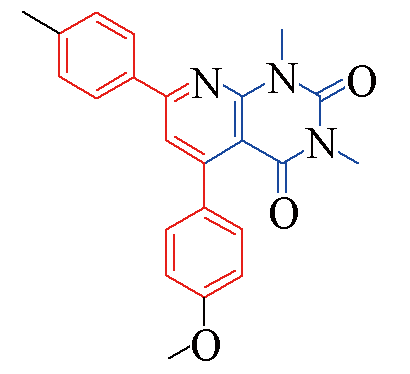 | 82 | 15 |
| 3c | 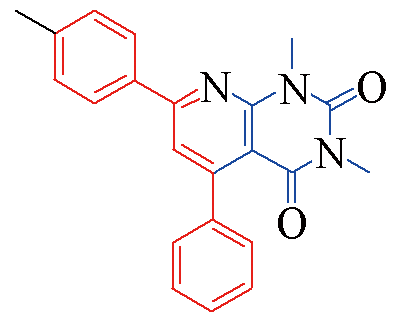 | 83 | 11 | 3j | 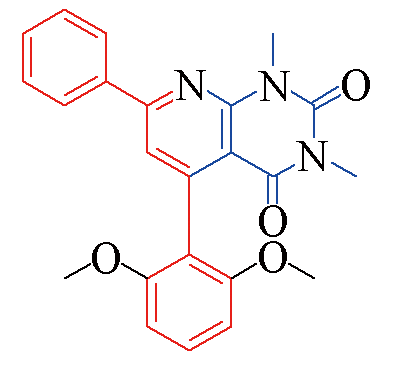 | 81 | 15 |
| 3d | 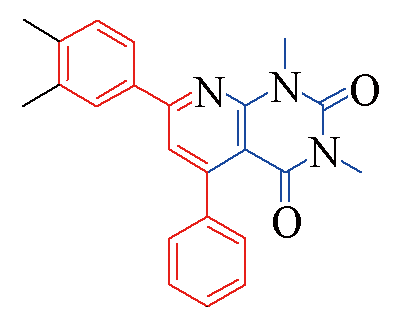 | 85 | 12 | 3k | 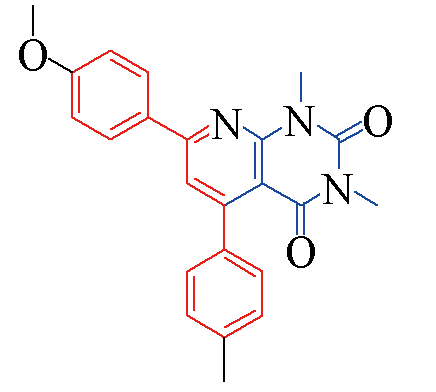 | 80 | 13 |
| 3e | 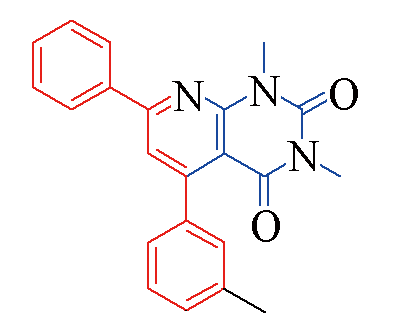 | 79 | 10 | 3l | 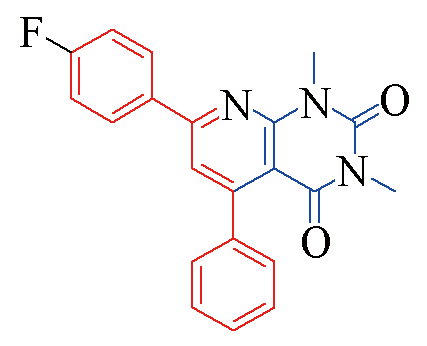 | 88 | 10 |
| 3f | 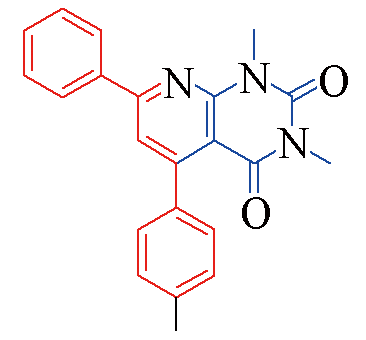 | 78 | 11 | 3m | 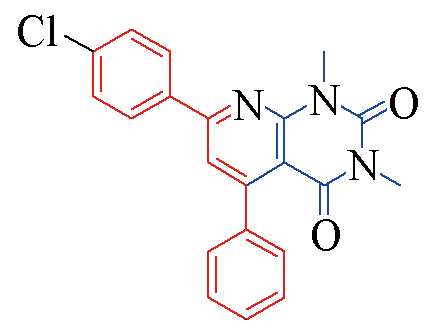 | 85 | 10 |
| 3g | 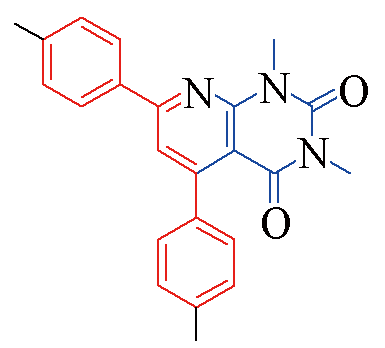 | 81 | 14 | 3n | 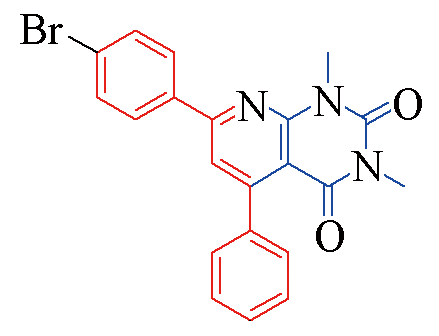 | 85 | 11 |
| Compd. | Product | Yieldb(%) | Time/min | Compd. | Product | Yieldb(%) | Time/min |
| 3o | 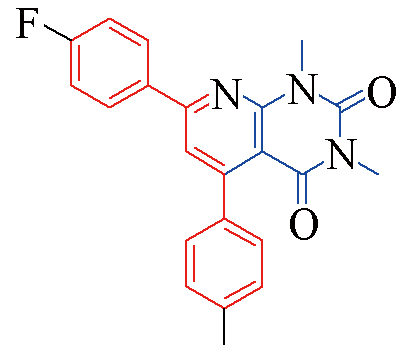 | 84 | 12 | 3t | 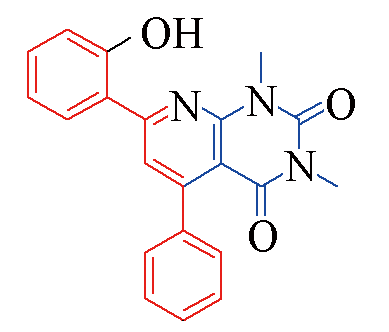 | 78 | 15 |
| 3p | 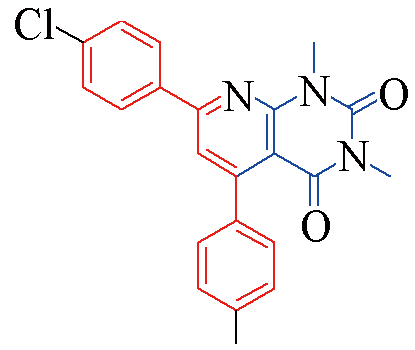 | 81 | 13 | 3u | 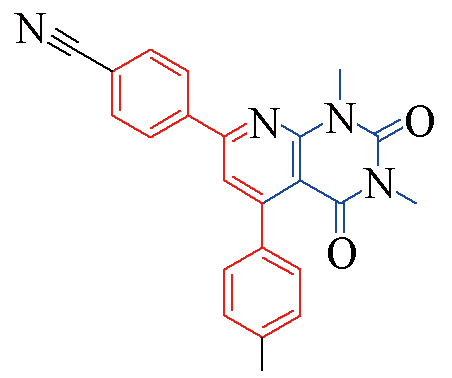 | 84 | 13 |
| 3q | 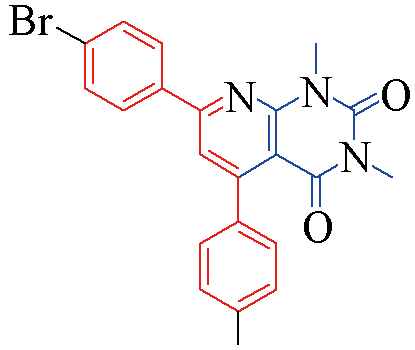 | 82 | 12 | 3v | 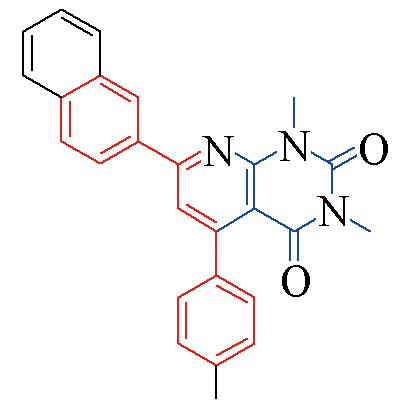 | 80 | 13 |
| 3r | 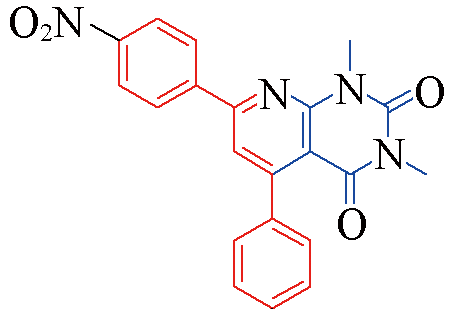 | 79 | 12 | 3w | 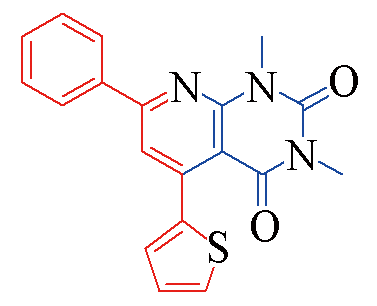 | 81 | 11 |
| 3s | 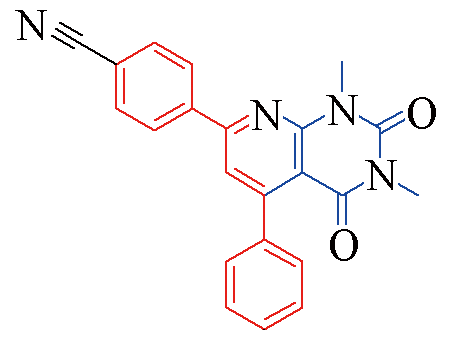 | 77 | 11 | 3x | 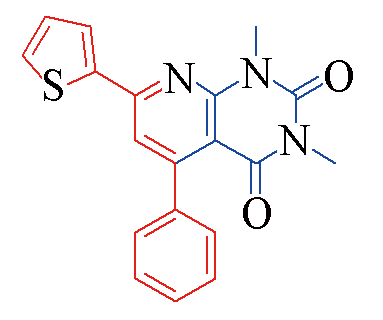 | 83 | 10 |
Table 4 Synthesis of pyridine [2,3-d]pyrimidinone derivatives from chalconesa
| Compd. | Product | Yieldb(%) | Time/min | Compd. | Product | Yieldb(%) | Time/min |
|---|---|---|---|---|---|---|---|
| 3a |  | 88 | 10 | 3h |  | 78 | 13 |
| 3b |  | 85 | 11 | 3i |  | 82 | 15 |
| 3c |  | 83 | 11 | 3j |  | 81 | 15 |
| 3d |  | 85 | 12 | 3k |  | 80 | 13 |
| 3e |  | 79 | 10 | 3l |  | 88 | 10 |
| 3f |  | 78 | 11 | 3m |  | 85 | 10 |
| 3g |  | 81 | 14 | 3n |  | 85 | 11 |
| Compd. | Product | Yieldb(%) | Time/min | Compd. | Product | Yieldb(%) | Time/min |
| 3o |  | 84 | 12 | 3t |  | 78 | 15 |
| 3p |  | 81 | 13 | 3u |  | 84 | 13 |
| 3q |  | 82 | 12 | 3v |  | 80 | 13 |
| 3r |  | 79 | 12 | 3w |  | 81 | 11 |
| 3s |  | 77 | 11 | 3x |  | 83 | 10 |
| 1 | Zhang H. J., Wang S. B., Wen X., Li J. Z., Quan Z. S., Med. Chem. Res., 2016, 25(7), 1287—1298 |
| 2 | Dongre R. S., Meshram J. S., Selokar R. S., Almalki F. A., Ben H. T., New J. Chem., 2018, 42(19), 15610—15617 |
| 3 | Gao X. G., Cen L. Q., Li F. Y., Wen R., Yan H. R., Yao H., Zhu S. L., Biochem. Biophys. Res. Commun., 2018, 505(3), 761—767 |
| 4 | Buron F., Merour J. Y., Akssira M., Guillaumet G., Routier S., Eur. J. Med. Chem., 2015, 95, 76—95 |
| 5 | Shimizu M., Takase Y., Nakamura S., Katae H., Minami A., Antimicrob.Agents Chemother., 1975, 8(2), 132—138 |
| 6 | Acosta P., Insuasty B., Ortiz A., Abonia R., Sortino M., Zacchino S. A., Quiroga J., Arabian J. Chem., 2016, 9(3), 481—492 |
| 7 | Veeraswamy B., Madhu D., Dev G. J., Poornachandra Y., Kumar G. S., Kumar C. G., Narsaiah B., Bioorg. Med. Chem. Lett., 2018, 28(9), 1670—1675 |
| 8 | Fang F., Xue L. M., Cong J., Tian C., Wang X. W., Liu J., Zhang Z. L., Chem. J. Chinese Universities, 2019, 40(10), 2111—2120 (方芳, 薛良敏, 丛 婧, 田 超, 王孝伟, 刘俊义, 张志丽. 高等学校化学学报, 2019, 40(10), 2111—2120) |
| 9 | Abbas S. E. S., George R. F., Samir E. M., Aref M. M. A., Abdel⁃Aziz H. A., Future Med. Chem., 2019, 11(18), 2395—2414 |
| 10 | Cheung A. W. H., BanneB R., Bose J., Kim K., Li S., Marcopulos N., Orzechowski L., Sergi J. A., Thakkar K. C., Wang B. B., Yun W., Zwingelstein C., Berthel S., Olivier A. R., Bioorg. Med. Chem. Lett., 2012, 22(24), 7518—7522 |
| 11 | Lacbay C. M., Mancuso J., Lin Y. S., Bennett N., G€otte M., Tsantrizos Y. S., J. Med. Chem., 2014, 57(17), 7435—7449 |
| 12 | Wawzonek S., J. Org. Chem., 1976, 41(19), 3149—3151 |
| 13 | Zhang F. R., Li C. M., Liang X. Z., Green Chem., 2018, 20(9), 2057—2063 |
| 14 | Shi D. Q., Zhou Y., Liu H., J. Heterocycl. Chem., 2010, 47, 131—135 |
| 15 | Wang Z. S., Wang M. Y., Xu S. S., Gu M. M., Meng Z. Y., Li C., Cai P. J., Rong L. C., Synth. Commun., 2016, 46(23), 1887—1892 |
| 16 | Farahmand T., Hashemian S., Sheibani A., J. Mol. Struct., 2020, 1206, 1—7 |
| 17 | Upadhyay A., Sharma L. K., Singh V. K., Singh R. K. P., Tetrahedron Lett., 2016, 57(50), 5599—5604 |
| 18 | Chate A. V., Kulkarni A. S., Jadhav C. K., Nipte A. S., Bondle G. M., J. Heterocycl. Chem., 2020, 57(5), 2184—2193 |
| 19 | Mamaghani M., Sheykhan M., Sadeghpour M., Tavakoli F., Monatsh Chem., 2018, 149(8), 1437—1446 |
| 20 | Jiang L., Ye W., Su W., Yu C., Chem. Res. Chinese Universities, 2019, 35(1), 21—25 |
| 21 | Kappe C. O., van der Eycken E., Chem. Soc. Rev., 2010, 39(4), 1280—1290 |
| 22 | Polshettiwar V., Varma R. S., Chem. Soc. Rev., 2008, 37(8) , 1546—1557 |
| 23 | Appukkuttan P., Mehta V. P., van der Eycken E., Chem. Soc. Rev., 2010, 39(5), 1467—1477 |
| 24 | Sharma A., Appukkuttan P., van der Eycken E., Chem. Commun., 2012, 48(11), 1623—1637 |
| 25 | Jiang Z., Liu J., Zhou F., Zhang J., Liu Z., Zhang C., Chen H., Chem. Res. Chinese Universities, 2018, 34(6), 918—922 |
| 26 | Sun H. S., Wang J. Q., Guo C., Shen L. J., Chin. J. Org. Chem., 2013, 33(10), 2220—2225(孙宏顺, 王建强, 郭成, 沈临江. 有机化学, 2013, 33(10), 2220—2225) |
| 27 | Polshettiwar V., Varma R. S., Acc.Chem. Res., 2008, 41(5), 629—639 |
| 28 | Kokel A., Schafer C., Torok B., Green Chem., 2017, 19(16), 3729—3751 |
| 29 | Bai H. R., Sun R. R., Chen X. B., Yang L. J., Huang C., ChemistrySelect, 2018, 3(17), 4635—4638 |
| 30 | Bai H., Sun R., Liu S., Yang L., Chen X., Huang C., J. Org. Chem., 2018, 83(20), 12535—12548 |
| 31 | Li J., Duan W., Pan X., Ye Y., Huang C., ChemistrySelect, 2019, 4(12), 3281—3285 |
| 32 | Li J. S., Li X. H., Duan W. W., Huang C., Jouranl of Yunnan Minzu Universtiy(Natural Sciences Edition), 2019, 117(05), 444—451 (李济森, 李新汉, 段文文, 黄超. 云南民族大学学报(自然科学版), 2019, 117(05), 444—451) |
| 33 | Majumdar K., Ponra S., Ghosh D., Synthesis, 2011, 2011(7), 1132—1136 |
| 34 | Abdelrazek F. M., Gomha S. M., Abdel⁃aziz H. M., Farghaly M. S., Metz P., Abdel⁃Shafy A., J. Heterocycl. Chem., 2020, 57(4), 1759—1769 |
| [1] | 闭格宁, 肖小华, 李攻科. 微波辅助萃取多物理场耦合模型的构建及验证[J]. 高等学校化学学报, 2022, 43(3): 20210739. |
| [2] | 马东玮, 田润赛, 刘振江, 冯源源, 丁泓宇, 冯季军. Na掺杂Li2-xNaxMnSiO4/C正极材料的微波辅助合成与电化学性能[J]. 高等学校化学学报, 2019, 40(6): 1280. |
| [3] | 代佳男, 杨子祺, 魏忠林, 曹军刚, 梁大鹏, 段海峰, 林英杰. TBAC/TBHP体系下6-氨基甲酰基取代的啡啶类化合物的合成[J]. 高等学校化学学报, 2019, 40(4): 712. |
| [4] | 甘思平, 李国华, 翟佳欣, 张雪明, 朱萌萌, 胡恩言, 张晓蕊, 张静茹. 氮化硼纳米片负载Pd(OAc)2催化剂的制备及催化微波辅助Heck反应[J]. 高等学校化学学报, 2019, 40(11): 2314. |
| [5] | 苏蕊, 汪亿晗, 陈长宝, 孙秀丽, 刘淑莹, 杨洪梅. 微波辅助离子液体分散液-液微萃取DART-Orbitrap质谱法测定环境水中的溴代阻燃剂[J]. 高等学校化学学报, 2018, 39(9): 1934. |
| [6] | 徐源, 陈艳华, 丁兰. 微波辅助法一步合成荧光碳点用于水中三价铁离子的检测[J]. 高等学校化学学报, 2018, 39(7): 1420. |
| [7] | 王林香. Na+, Li+, Bi3+掺杂CaWO4:Eu3+荧光粉的制备及发光特性[J]. 高等学校化学学报, 2018, 39(1): 25. |
| [8] | 胡耀娟, 黄梦丹, 陈昌云, 张长丽. 微波辅助法制备氢氧化镍-石墨烯纳米复合结构及在葡萄糖检测中的应用[J]. 高等学校化学学报, 2016, 37(3): 468. |
| [9] | 佟拉嘎, 任广生, 党晓峰, 林世静, 荣华. 微/纳米ZnO粉体在Glu-BF4离子液体水溶液中的诱导生长[J]. 高等学校化学学报, 2016, 37(11): 1939. |
| [10] | 陈海燕, 丁兰, 刘密兰. 微波辅助合成分子印迹聚合物用于萃取蜂蜜中的氯霉素[J]. 高等学校化学学报, 2015, 36(1): 67. |
| [11] | 吴琼, 梅文杰, 吴韦黎, 陈燕华, 曾玲莉, 郑文杰. SiC管中微波辅助芳烃钌(Ⅱ)化合物[(η6-C6H6)Ru(H2iiP)Cl]Cl的合成[J]. 高等学校化学学报, 2013, 34(8): 1863. |
| [12] | 李莉, 李恩帅, 高宇, 赵月红, 禚娜, 路露. 三维球状ZnO纳米簇的微波辅助合成与微波增强光催化降解染料[J]. 高等学校化学学报, 2012, 33(07): 1528. |
| [13] | 张朝晖, 张明磊, 徐添珍, 罗丽娟, 杨潇, 姚守拙. 微波辅助核-壳型Cr(Ⅲ)离子印迹聚合物的制备及在尿液中的分离应用[J]. 高等学校化学学报, 2010, 31(9): 1734. |
| [14] | 朱晓楠, 魏士刚, 王浩南, 孙颖, 姜春竹, 王健, 王璐, 王丽英, 宋大千. 微波辅助提取-气相色谱质谱联用测定肉桂中的挥发油[J]. 高等学校化学学报, 2009, 30(7): 1300. |
| [15] | 郇延富,张志权,费强, 冯国栋,牟颖,. PPESO3监测微波辅助酶解蛋白的进程[J]. 高等学校化学学报, 2009, 30(1): 54. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||