

高等学校化学学报 ›› 2017, Vol. 38 ›› Issue (10): 1864.doi: 10.7503/cjcu20160907
收稿日期:2016-12-16
出版日期:2017-10-10
发布日期:2017-09-22
作者简介:联系人简介: 晋晓勇, 男, 博士, 副教授, 主要从事生化分析及生物传感器研究. E-mail:基金资助:
XU Kaige, ZHANG Di, LEI Jie, PENG Yage, PENG Juan, JIN Xiaoyong*( )
)
Received:2016-12-16
Online:2017-10-10
Published:2017-09-22
Contact:
JIN Xiaoyong
E-mail:jinxy588@163.com
Supported by:摘要:
通过油胺(Oleylamine)还原法制备了金纳米线(Au nanowires), 将其与酸化处理的多壁碳纳米管(MWCNTs)通过层层组装制备了Au nanowires-MWCNTs 复合结构修饰的玻碳电极(Au nanowires-MWCNTs/GCE). 电化学研究结果表明, 与单纯Au nanowires或MWCNTs修饰电极相比, Au nanowires-MWCNTs/GCE对葡萄糖表现出更优良的电催化性能. 以Au nanowires-MWCNTs/GCE为阳极, 电沉积Pt膜电极(Pt/GCE)为阴极, 构建了葡萄糖/O2燃料电池. 测试结果表明, 构建的燃料电池的开路电位(OCP)为0.57 V, 在0.44 V下最大功率密度(Pmax)为0.28 mW/cm2.
中图分类号:
TrendMD:
许凯歌, 张笛, 雷杰, 彭亚鸽, 彭娟, 晋晓勇. Au Nanowires-MWCNTs修饰电极对葡萄糖的催化氧化. 高等学校化学学报, 2017, 38(10): 1864.
XU Kaige, ZHANG Di, LEI Jie, PENG Yage, PENG Juan, JIN Xiaoyong. Au Nanowires-MWCNTs Modified Electrode for Catalyzing the Oxidization of Glucose†. Chem. J. Chinese Universities, 2017, 38(10): 1864.
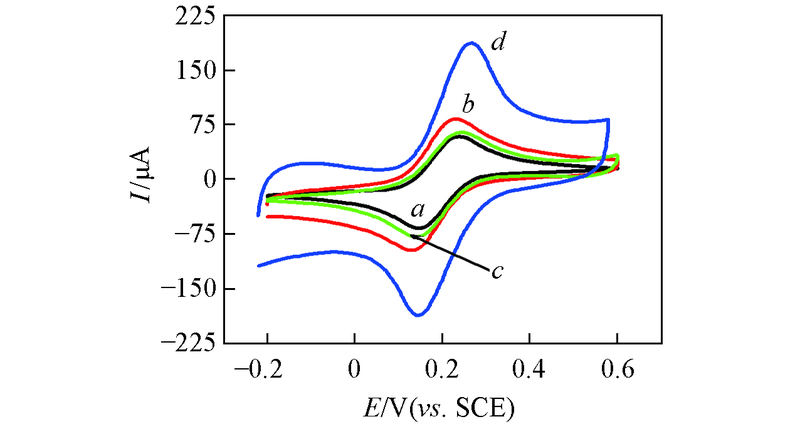
Fig.3 CV curves of bare GCE(a), MWCNTs/GCE(b), Au nanowires/GCE(c), Au nanowires-MWCNTs/GCE(d) in 10.0 mmol/L K3[Fe(CN)6] solution containing 0.1 mol/L KCl at the scan rate of 100 mV/s
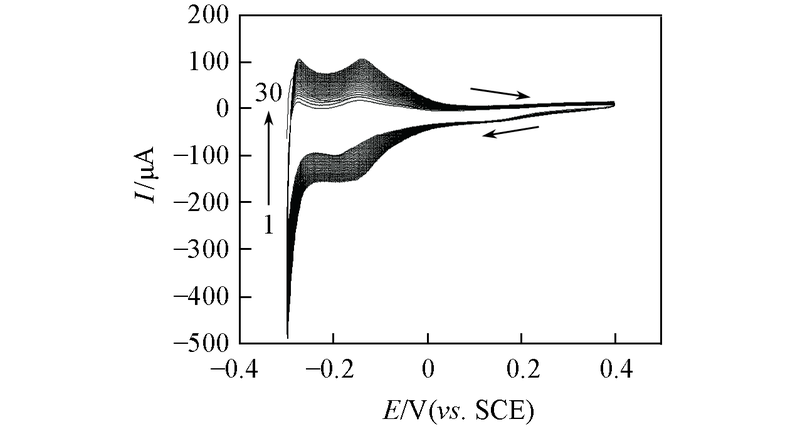
Fig.4 CV curves for electrodeposition process of the Pt film obtained from a solution mixture containing 1.8 × 10-3 mol/L H2PtCl6 and 0.5 mol/L H2SO4 between 0.4 and -0.3 V(vs. SCE) at 100 mV/s
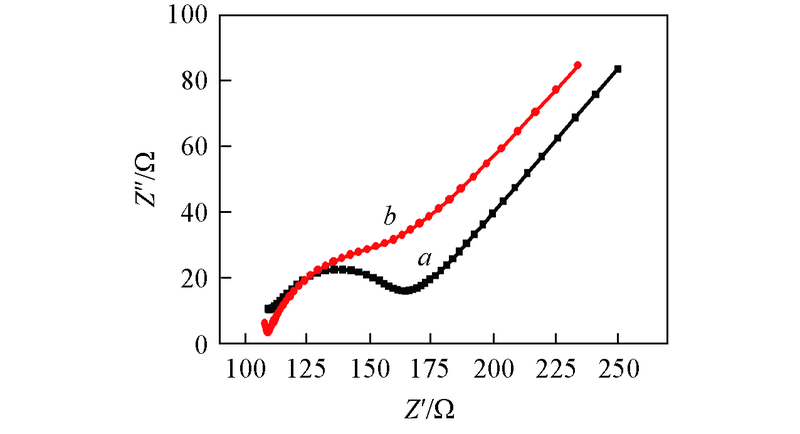
Fig.5 Electrochemical impedance spectra(EIS) in a mixture containing 5.0 mmol/L [Fe(CN)6]3-/4- and 0.1 mol/L KCl at bare GCE(a) and Pt/GCE(b), respectively
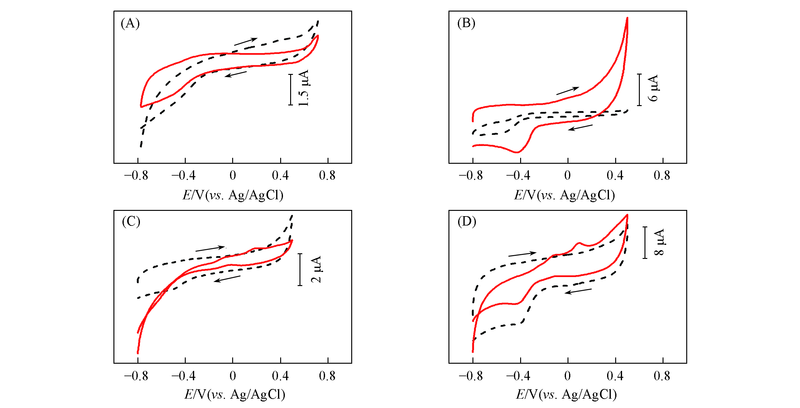
Fig.6 CV curves of bare GCE(A), MWCNTs/GCE(B), Au nanowires/GCE(C), Au nanowires-MWCNTs/GCE(LBL2)(D) in 0.5 mol/L NaOH in the absence(dash line) and presence(solid line) of 5 mmol/L glucose at the scan rate of 60 mV/s
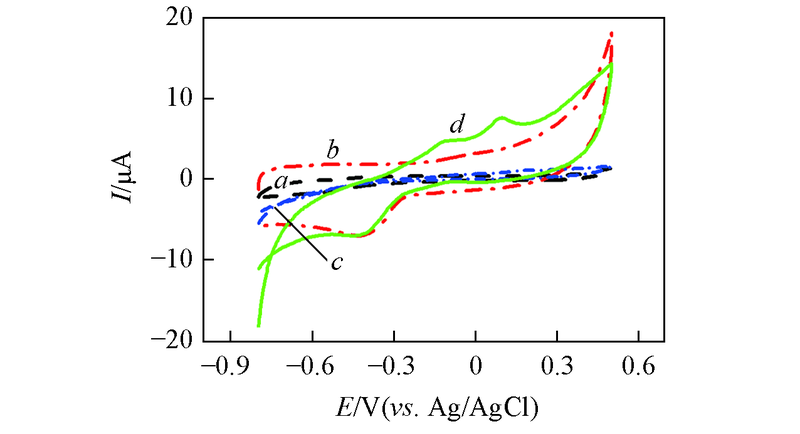
Fig.7 CV curves of bare GCE(a), MWCNTs/GCE(b), Au nanowires/GCE(c) and Au nanowires-MWCNTs/GCE(LBL2)(d) in 0.5 mol/L NaOH in the presence 5 mmol/L glucose at the scan rate of 60 mV/s
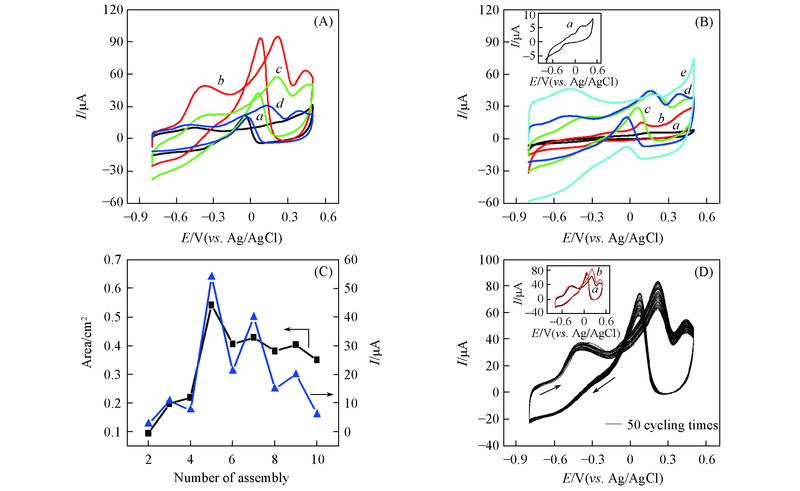
Fig.8 CV curves of network film electrodes with odd numbers of layers(A) and even numbers of layers(B) in 0.5 mol/L NaOH+20.0 mmol/L glucose at the scan rate of 60 mV/s, the line graphs of the response current(▲) and effective electrode area(■) to the number of assembly layers(C) and the cycling CV curves of Au nanowires-MWCNTs/GCE(LBL5) for 50 cycling times in 0.5 mol/L NaOH+20.0 mmol/L glucose at the scan rate of 60 mV/s(D)(A) a. LBL3; b. LBL5; c. LBL7; d. LBL9. (B) a. LBL2; b. LBL4; c. LBL6; d. LBL8; e. LBL10. Inset of (D) shows the CV curves of the first cycling times(a) and the last cycling times(b).
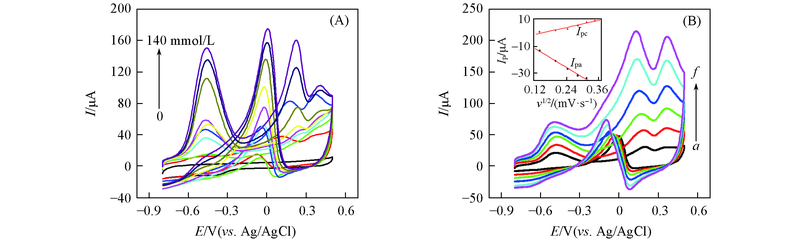
Fig.9 CV curves of Au nanowires-MWCNTs/GCE(LBL5) in 0.5 mol/L NaOH containing 0, 5.0, 10.0, 20.0, 40.0, 60.0, 80.0, 100.0, 120.0, 140.0 mmol/L glucose at the scan rate of 60 mV/s(A) and CV curves of Au nanowires-MWCNTs/GCE(LBL5) in 0.5 mol/L NaOH solution containing 20.0 mmol/L glucose at scan rates of 20(a), 40(b), 60(c), 80(d), 100(e), 120 mV/s(f)(B), respectivelyInset of (B) shows the plots of anodic and cathodic peak currents vs. v1/2.
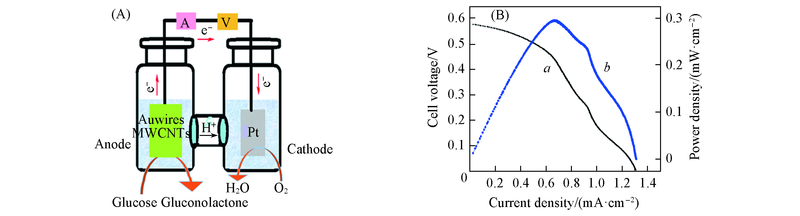
Fig.11 Schematic illustration for the principle of glucose/O2 fuel cell(A) and polarization curve(a) and power density curve(b) at room temperature for a glucose/O2 fuel cell(B)Anode: LBL5 in 0.5 mol/L NaOH cotaining 100.0 mmol/L glucose. Cathode: Pt/GCE in 0.1 mol/L PBS filled with O2.
| Mimic enzyme | Open circuit-potential/V | Maximum power density/(μW·cm-2) | Ref. |
|---|---|---|---|
| Co/NG | 0.79 | 150 | [ |
| Auglu/GCE | 0.916 | 307 | [ |
| 3D porous Pd | 0.650±0.005 | 5.7±0.4 | [ |
| Au nanowires | 0.425 | 126 | [ |
| MWCNT/(DPDE)Ⅲ | 0.64 | 182 | [ |
| Au nanowires-MWCNTs | 0.57 | 280 | This work |
Table 1 Performance comparison for glucose fuel cell among nanoparticles film electrodes
| Mimic enzyme | Open circuit-potential/V | Maximum power density/(μW·cm-2) | Ref. |
|---|---|---|---|
| Co/NG | 0.79 | 150 | [ |
| Auglu/GCE | 0.916 | 307 | [ |
| 3D porous Pd | 0.650±0.005 | 5.7±0.4 | [ |
| Au nanowires | 0.425 | 126 | [ |
| MWCNT/(DPDE)Ⅲ | 0.64 | 182 | [ |
| Au nanowires-MWCNTs | 0.57 | 280 | This work |
| [1] | Zebda A., Cosnier S., Alcaraz J. P., Holzinger M., Le G. A., Gondran C., Boucher F., Giroud F., Gorgy K., Lamraoui H., Cinquin P., Sci. Rep., 2013, 3, 1516—1520 |
| [2] | Tayhas R. P. G., Trends Biotechnol., 2004, 22(3), 99—100 |
| [3] | Zou Q., Liu J., Zhu G. B., Zhang X. H., Chen J. H., Acta Chimica Sinica, 2013, 71, 1154—1160 |
| (邹琼, 刘娟, 朱刚兵, 张小华, 陈金华. 化学学报, 2013, 71, 1154—1160) | |
| [4] | Devaraj M., Deivasigamani R. K., Jeyadevan S., Colloids Surf. B, 2013, 102, 554—561 |
| [5] | Lee Y. J., Park J. Y., Sensors, IEEE, 2010, 1875—1878 |
| [6] | Fujiwara N., Yamazaki S., Siroma Z., Ioroi T., Senoh H., Yasuda K., Electrochem. Commun., 2009, 11(2), 390—392 |
| [7] | Zhang W., Wang Z., Shen Y., Xi M., Chu X., Xi C., Chem. Res. Chinese Universities,2015, 31(6), 1007—1011 |
| [8] | Xiong T., Lin J. Y., Shang Z. J., Zhang X. T., Lin X., Tian W., Zhong Q. L., Ren B., Chem. J. Chinese Universities,2014, 35(11), 2460—2465 |
| (熊婷, 林剑云, 商中瑾, 张贤土, 林旋, 田伟, 钟起玲, 任斌. 高等学校化学学报, 2014, 35(11), 2460—2465) | |
| [9] | Ci S. Q., Wen Z. H., Mao S., Hou Y., Cui S. M., He Z., Chen J. H., Chem. Commun., 2015, 51(45), 9354—9357 |
| [10] | You X. X., Fan J. C., Huang W., Zhou Y. Z., Wei J. Y., Chem. J. Chinese Universities,2016, 37(9), 1694—1700 |
| (游向轩, 樊金串, 黄伟, 周由之, 魏珺谊. 高等学校化学学报, 2016, 37(9), 1694—1700) | |
| [11] | Navaeea A., Narimania M., Korania A., Ahmadib R., Salimia A., Soltanianc S., Electrochim. Acta, 2016, 208, 325—333 |
| [12] | An L., Zhao T. S., Shen S. Y., Wu Q. X., Chen R., J. Power Sources,2011, 196(1), 186—190 |
| [13] | Xu K., Li Y., Zhao N., Du W. X., Zeng W. W., Gao S., Cheng X. N., Yang J., Chem. J. Chinese Universities,2016, 37(8), 1476—1484 |
| (徐凯, 李毅, 赵南, 杜文修, 曾炜炜, 高帅, 程晓农, 杨娟. 高等学校化学学报, 2016, 37(8), 1476—1484) | |
| [14] | Kaura B., Srivastava R., Satpatib B., ACS Catalysis, 2016, 6(4), 2654—2663 |
| [15] | Hu J., Shao D., Chen C., Sheng G., Ren X., Wang X., J. Hazard. Mater., 2011, 185(1), 463—471 |
| [16] | Liu S. M., Zheng Y. D., Li W., Sun Y., Yue L. N., Zhao Z. J., Chem. J. Chinese Universities,2016, 37(2), 290—296 |
| (刘树敏, 郑裕东, 李伟, 孙乙, 岳丽娜, 赵振江. 高等学校化学学报, 2016, 37(2), 290—296) | |
| [17] | Elouarzaki K., Le Goff. A., Holzinger M., Thery J., Cosnier S., J. Am. Ceram. Soc., 2012, 134(34), 14078—14085 |
| [18] | Zhu N. N., Lin Y. Q., Yu P., Su L., Mao L. Q., Anal. Chim. Acta,2009, 650(1), 44—48 |
| [19] | Yang L., Zhang Y., Chu M., Deng W., Tan Y., Ma M., Su X., Xie Q., Yao S., Biosens. Bioelectron., 2014, 52, 105—110 |
| [20] | Wang P., Li F., Huang X., Li Y., Wang L., Electrochem. Commun., 2008, 10(2), 195—199 |
| [21] | Qin Q. X., Liu S. L., Plating and Finishing,2008, 30(7), 29—34 |
| (覃奇贤, 刘淑兰. 电镀与精饰, 2008, 30(7), 29—34) | |
| [22] | Huo Z. Y., Tsung C., Huang W. Y., Zhang X. F., Yang P. D., Nano Lett., 2008, 8(7), 2041—2044 |
| [23] | Yu A. M., Liang Z. J., Cho J. H., Caruso F., Nano Lett., 2003, 3(9), 1203—1207 |
| [24] | Kruusenberg I., Alexeyeva N., Tammeveski K., Carbon,2009, 47(3), 651—658 |
| [25] | Deo R. P., Wang J., Electrochem. Commun., 2004, 6(3), 284—287 |
| [26] | YeJ. S., Wen Y., Zhang W. D., Gan L. M., Xu G. Q., Sheu F. S., Electrochem. Commun., 2004, 6(1), 66—70 |
| [27] | Chen W., Chen S., Angew. Chem. Int. Ed., 2009, 48(24), 4386—4389 |
| [28] | Cherevko S., Chung C., Sens. Actuators B, 2009, 142(1), 216—223 |
| [29] | Rong L., Yang C., Qian Q., Xia X., Talanta,2007, 72(2), 819—824 |
| [30] | Song Y., Zhang D., Gao W., Xia X., Chem. Eur. J., 2005, 11(7), 2177—2182 |
| [31] | Dong S. J.,Chemistry Online, 1981, (12), 713—721 |
| (董绍俊. 化学通报, 1981, (12), 713—721) | |
| [32] | Xie F. Y., Huang Z., Chen C., Xie Q. J., Huang Y., Qin C., Liu Y., Su Z. H., Yao S. Z., Electrochem. Commun., 2012, 18, 108—111 |
| [33] | Zhao Y., Fan L. Z., Hong B., Zhang Y., Zhang M. S., Que Q. M., Ji J. Y., Energy Technology, 2016, 4(2), 249—255 |
| [1] | 仇心声, 吴芹, 史大昕, 张耀远, 陈康成, 黎汉生. 离子型交联磺化聚酰亚胺质子交换膜的制备及高温燃料电池性能[J]. 高等学校化学学报, 2022, 43(8): 20220140. |
| [2] | 李志光, 齐国栋, 徐君, 邓风. Sn-Al-β分子筛酸性在葡萄糖转化反应中作用的固体NMR研究[J]. 高等学校化学学报, 2022, 43(6): 20220138. |
| [3] | 陈长利, 米万良, 李煜璟. 单原子催化材料在电化学氢循环应用中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220065. |
| [4] | 罗昪, 周芬, 潘牧. 层级多孔碳载铂催化剂的制备及可达性[J]. 高等学校化学学报, 2022, 43(4): 20210853. |
| [5] | 曾晛阳, 赵熹, 黄旭日. 细胞松弛素B对葡萄糖/质子共转运蛋白GlcPSe的抑制机理[J]. 高等学校化学学报, 2022, 43(4): 20210822. |
| [6] | 夏大成, 周瑞, 涂博, 蔡志伟, 高难, 姬晓旭, 常钢, 任小明, 何云斌. 银/金纳米线阵列表面增强拉曼基底的制备及对孔雀石绿的高灵敏度检测[J]. 高等学校化学学报, 2022, 43(3): 20210731. |
| [7] | 刘杰, 李金晟, 柏景森, 金钊, 葛君杰, 刘长鹏, 邢巍. 降低直接甲醇燃料电池浓差极化的含磺化碳管阻水夹层的构建[J]. 高等学校化学学报, 2022, 43(11): 20220420. |
| [8] | 袁春玲, 姚晓条, 徐远金, 覃秀, 石睿, 成诗琦, 王益林. 双功能碳点用于葡萄糖的比色/比率荧光测定[J]. 高等学校化学学报, 2021, 42(8): 2428. |
| [9] | 蒲阳阳, 宁聪, 陆瑶, 刘莉莉, 李娜, 胡朝霞, 陈守文. 新型共混交联磺化聚醚醚酮/部分氟化磺化聚芳醚砜质子交换膜的制备与表征[J]. 高等学校化学学报, 2021, 42(6): 2002. |
| [10] | 曹凯悦, 彭金武, 李宏斌, 石埕荧, 王鹏, 刘佰军. 基于聚苯并咪唑/超支化聚合物的交联共混体系的高温质子交换膜[J]. 高等学校化学学报, 2021, 42(6): 2049. |
| [11] | 李柳, 孙仕勇, 吕瑞, GOLUBEVYevgeny Aleksandrovich, 王可, 董发勤, 段涛, KOTOVAOlga Borisovna, KOTOVAElena Leonidovna. 铁氨基黏土-葡萄糖氧化酶纳米复合催化剂的构筑及多酶级联反应研究[J]. 高等学校化学学报, 2021, 42(3): 803. |
| [12] | 王柏纯, 袁雨欣, 闫迎华, 丁传凡, 唐科奇. 葡萄糖-6-磷酸功能化亲水磁探针:有效分离富集糖肽/磷酸肽的双用途亲和材料[J]. 高等学校化学学报, 2021, 42(10): 3062. |
| [13] | 王欢, 所金泉, 王春艳, 王润伟. 氨基化树枝状介孔二氧化硅固定葡萄糖氧化酶用于检测葡萄糖[J]. 高等学校化学学报, 2020, 41(8): 1731. |
| [14] | 王跃民, 孟庆磊, 王显, 葛君杰, 刘长鹏, 邢巍. 铜,硫掺杂对铁氮碳氧还原催化剂性能的提升作用[J]. 高等学校化学学报, 2020, 41(8): 1843. |
| [15] | 叶晓栋, 齐国栋, 徐君, 邓风. Au负载SBA-15分子筛上葡萄糖氧化反应[J]. 高等学校化学学报, 2020, 41(5): 960. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||