

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (12): 2415.doi: 10.7503/cjcu20150508
李秀云1, 万川1, 杜士杰1, 李红1, 袁会珠2, 蒋家珍1, 肖玉梅1, 覃兆海1( )
)
收稿日期:2015-06-30
出版日期:2015-12-10
发布日期:2015-11-19
作者简介:联系人简介: 覃兆海, 男, 博士, 教授, 博士生导师, 主要从事新农药创制研究. E-mail:基金资助:
LI Xiuyun1, WAN Chuan1, DU Shijie1, LI Hong1, YUAN Huizhu2, JIANG Jiazhen1, XIAO Yumei1, QIN Zhaohai1,*( )
)
Received:2015-06-30
Online:2015-12-10
Published:2015-11-19
Contact:
QIN Zhaohai
E-mail:qinzhaohai@263.net
Supported by:摘要:
以吡啶芳酮为原料, 合成了11个含二芳基肟醚结构单元的甲氧丙烯酸酯类化合物, 通过氢核磁共振谱(1H NMR)、 碳核磁共振谱(13C NMR)和高分辨质谱(HRMS)等方法对其结构进行了确证. 离体杀菌活性测定结果表明, 该类化合物对供试的7种植物病原菌普遍表现出较高的抑制活性, 其中化合物5d和5g的活性与对照药剂嘧菌酯和肟菌酯相当. 进一步活体盆栽验证实验结果表明, 在400 mg/L浓度下, 部分化合物表现出很好的防治效果, 如化合物5h对小麦白粉病和黄瓜炭疽病的防治效果达100%. 另外, 该类化合物对稻梨孢的孢子萌发具有强烈的抑制作用, 在25 mg/L浓度下, 绝大多数化合物的抑制率达到100%, 但对灰葡萄孢的孢子萌发普遍无效. 研究结果显示该类化合物具有进一步开发的价值.
中图分类号:
TrendMD:
李秀云, 万川, 杜士杰, 李红, 袁会珠, 蒋家珍, 肖玉梅, 覃兆海. 二芳基肟苄醚类化合物的合成及杀菌活性. 高等学校化学学报, 2015, 36(12): 2415.
LI Xiuyun, WAN Chuan, DU Shijie, LI Hong, YUAN Huizhu, JIANG Jiazhen, XIAO Yumei, QIN Zhaohai. Synthesis and Fungicidal Activities of Biaryl Methanone O-Benzyl Oximes†. Chem. J. Chinese Universities, 2015, 36(12): 2415.
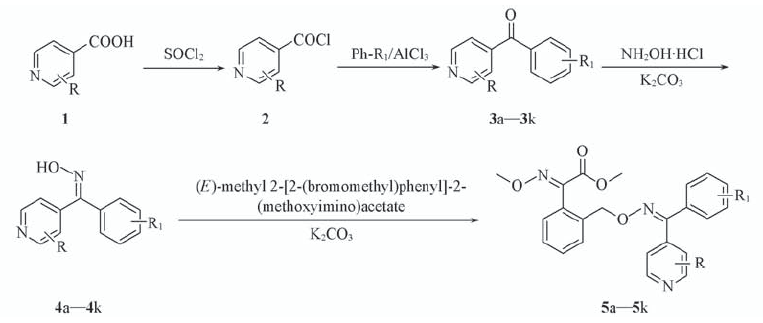
Scheme 1 Synthetic routes of compound 5 2a: R=H; 2b: R=Cl; 3a, 4a, 5a: R=H, R1=H; 3b, 4b, 5b: R=H, R1=4-Me; 3c, 4c, 5c: R=H, R1=4-OMe; 3d, 4d, 5d: R=H, R1=3-Cl-4-Me; 3e, 4e, 5e: R=H, R1=i-Pr; 3f, 4f, 5f: R=H, R1=4-Br; 3g, 4g, 5g: R=H, R1=2,3-2Cl; 3h, 4h, 5h: R=H, R1=4-t-Bu; 3i, 4i, 5i: R=2-Cl, R1=4-Me; 3j, 4j, 5j: R=2-Cl, R1=4-t-Bu; 3k, 4k, 5k: R=2-Cl, R1=4-Ph.
| Compd. | Appearance | Yield(%) | m.p./℃ | 1H NMR(300 MHz, CDCl3, δ) |
|---|---|---|---|---|
| 4c | White solid | 89 | 165—166 | 3.79, 3.81(m, 3H), 6.85(dd, J=8.9, 5.9 Hz, 2H), 7.15(d, J=8.9 Hz, 1H), |
| 7.35(dd, J=13.5, 7.5 Hz, 4H), 8.72(d, J=6.0 Hz, 2H), 10.04(s, 1H) | ||||
| 4d | White solid | 87 | 191—193 | 2.34—2.44(m, 3H), 7.14—7.49(m, 5H), 8.60(d, J=3.0 Hz, 1H), 8.76 |
| (d, J=6.0 Hz, 1H), 10.62(s, 1H) | ||||
| 4e | White solid | 89 | 178—179 | 1.23(s, 3H), 1.26(s, 3H), 2.86—3.00(m, 1H), 7.21(d, J=8.5 Hz, 2H), |
| 7.29—7.49(m, 5H), 8.75(d, J=5.8 Hz, 2H) | ||||
| 4i | White solid | 84 | 179—180 | 2.40(s, 3H), 7.11—7.46(m, 6H), 8.36(d, J=5.1 Hz, 1H), 8.52(d, J= |
| 3.0 Hz, 1H), 9.42(s, 1H) | ||||
| 4j | White solid | 79 | 187—188 | 1.34(s, 9H), 7.30—7.41(m, 4H), 7.51(d, J=8.5 Hz, 1H), 8.37(d, J= |
| 5.1 Hz, 1H), 8.52(d, J=6.0 Hz, 1H), 8.73(s, 1H) |
Table 1 Appearance, yields, melting points and 1H NMR data of compounds 4c—4e, 4i and 4j
| Compd. | Appearance | Yield(%) | m.p./℃ | 1H NMR(300 MHz, CDCl3, δ) |
|---|---|---|---|---|
| 4c | White solid | 89 | 165—166 | 3.79, 3.81(m, 3H), 6.85(dd, J=8.9, 5.9 Hz, 2H), 7.15(d, J=8.9 Hz, 1H), |
| 7.35(dd, J=13.5, 7.5 Hz, 4H), 8.72(d, J=6.0 Hz, 2H), 10.04(s, 1H) | ||||
| 4d | White solid | 87 | 191—193 | 2.34—2.44(m, 3H), 7.14—7.49(m, 5H), 8.60(d, J=3.0 Hz, 1H), 8.76 |
| (d, J=6.0 Hz, 1H), 10.62(s, 1H) | ||||
| 4e | White solid | 89 | 178—179 | 1.23(s, 3H), 1.26(s, 3H), 2.86—3.00(m, 1H), 7.21(d, J=8.5 Hz, 2H), |
| 7.29—7.49(m, 5H), 8.75(d, J=5.8 Hz, 2H) | ||||
| 4i | White solid | 84 | 179—180 | 2.40(s, 3H), 7.11—7.46(m, 6H), 8.36(d, J=5.1 Hz, 1H), 8.52(d, J= |
| 3.0 Hz, 1H), 9.42(s, 1H) | ||||
| 4j | White solid | 79 | 187—188 | 1.34(s, 9H), 7.30—7.41(m, 4H), 7.51(d, J=8.5 Hz, 1H), 8.37(d, J= |
| 5.1 Hz, 1H), 8.52(d, J=6.0 Hz, 1H), 8.73(s, 1H) |
| Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ | Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ |
|---|---|---|---|---|---|---|---|
| 5a | 85—87 | 78 | 404.1605(404.1605) | 5g | 103—104 | 70 | 472.0907(472.0825) |
| 5b | —* | 75 | 418.1710(418.1761) | 5h | —* | 75 | 460.2232(460.2231) |
| 5c | —* | 65 | 434.1710(434.1710) | 5i | 84—85 | 71 | 452.1374(452.1372) |
| 5d | 88—90 | 72 | 452.1374(452.1372) | 5j | —* | 74 | 494.1844(494.1841) |
| 5e | —* | 80 | 446.2078(446.2074) | 5k | —* | 67 | 514.1528(514.1528) |
| 5f | 104—106 | 68 | 482.0710(482.0710) |
Table 2 Melting points, yields and HRMS data of compounds 5a—5k
| Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ | Compd. | m.p./℃ | Yield(%) | HRMS(calcd.), m/z(M+H)+ |
|---|---|---|---|---|---|---|---|
| 5a | 85—87 | 78 | 404.1605(404.1605) | 5g | 103—104 | 70 | 472.0907(472.0825) |
| 5b | —* | 75 | 418.1710(418.1761) | 5h | —* | 75 | 460.2232(460.2231) |
| 5c | —* | 65 | 434.1710(434.1710) | 5i | 84—85 | 71 | 452.1374(452.1372) |
| 5d | 88—90 | 72 | 452.1374(452.1372) | 5j | —* | 74 | 494.1844(494.1841) |
| 5e | —* | 80 | 446.2078(446.2074) | 5k | —* | 67 | 514.1528(514.1528) |
| 5f | 104—106 | 68 | 482.0710(482.0710) |
| Compd. | 1H NMR(300 MHz, CDCl3, δ) | 13C NMR(75 MHz, CDCl3, δ) |
|---|---|---|
| 5a | 3.70(d, J=2.1 Hz, 3H), 3.98(d, J=0.9 Hz, 3H), 5.11(s, 2H), 7.20—7.13(m, 1H), 7.23(dd, J=4.4, 1.6 Hz, 1H), 7.44—7.30(m, 9H), 8.56(dd, J=4.4, 1.7 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 52.8, 63.2, 75.1, 122.1, 123.8, 127.6, 128.5, 129.3, 129.8, 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.7 |
| 5b | 2.37(d, J=15.0 Hz, 3H), 3.71(d, J=0.5 Hz, 3H), 3.98(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.13—7.22(m, 5H), 7.26(s, 1H), 7.28—7.42(m, 4H), 8.51(d, J=6.2 Hz, 1H), 8.67(d, J=5.1 Hz, 1H) | 21.4, 52.8(s), 63.7(s), 75.0, 127.2—128.1(m), 128.1— 130.0(m), 132.0, 135.9, 139.4, 140.0, 141.3, 149.3, 149.7, 154.9, 163.2 |
| 5c | 3.69(s, 3H), 3.83(s, 3H), 3.99(s, 3H), 5.12(s, 2H), 6.88—6.97(m, 2H), 7.12—7.20(m, 1H), 7.28—7.48(m, 7H), 8.55(d, J =5.6 Hz, 2H) | 52.7, 55.2, 63.7, 75.1, 113.7, 113.8, 122.4, 123.7, 123.8, 127.7, 128.4, 128.8, 129.0, 129.4, 129.7, 131.2, 135.9, 141.4, 144.4, 149.3, 149.8, 154.5, 160.2, 160.9, 163.1 |
| 5d | 2.32—2.41(m, 3H), 3.70(s, 3H), 3.97(d, J=2.1 Hz, 3H), 5.11(s, 2H), 7.12—7.18(m, 2H), 7.21(dd, J=4.4, 1.6 Hz, 1H), 7.26— 7.47(m, 6H), 8.56(dd, J=4.7, 1.4 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 20.1, 52.8, 63.7, 75.2, 122.0, 123.8, 126.3, 128.0, 128.1, 128.5, 128.8, 129.1, 129.4, 129.9, 131.5, 133.3, 135.6, 136.3, 140.9, 143.7, 149.2, 149.8, 154.0, 163.1 |
| 5e | 1.25(d, J=6.9 Hz, 6H), 2.77—3.05(m, 1H), 3.68(d, J=4.7 Hz, 3H), 3.97(s, 3H), 5.11(s, 2H), 7.10—7.52(m, 11H), 8.54(d, J=5.5 Hz, 1H), 8.67(d, J=5.5 Hz, 1H) | 23.8, 33.9, 52.7, 63.7, 75.0, 122.2, 123.8, 126.4, 127.7, 128.6, 129.4, 132.4, 135.9, 141.2, 144.2, 149.3, 149.8, 150.1, 150.8, 154.8, 163.1 |
| 5f | 3.71(d, J=1.7 Hz, 3H), 3.98(d, J=2.7 Hz, 3H), 5.10(s, 2H), 7.22—7.13(m, 3H), 7.31—7.23(m, 3H), 7.59—7.35(m, 5H), 8.57(dd, J=4.5, 1.6 Hz, 1H), 8.68(dd, J=3.0, 1.6 Hz, 1H) | 52.8, 63.7, 75.1, 122.1, 123.9, 127.7, 128.1—130.0(m), 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.2 |
| 5g | 3.71(d, J=2.6 Hz, 3H), 3.97(d, J=1.6 Hz, 3H), 5.11(s, 2H), 7.11—7.20(m, 3H), 7.25—7.30(m, 1H), 7.38(m, 4H), 7.50(dd, J= 5.2, 2.9 Hz, 1H), 8.57(dd, J=4.5, 1.5 Hz, 1H), 8.70(dd, J=3.0, 1.5 Hz, 1H) | 52.8, 63.7, 75.5, 121.7, 123.5, 126.8, 127.9, 128.6, 129.1, 129.4, 130.4, 131.1, 132.8, 133.6, 133.9, 134.8, 135.4, 139.9, 142.8, 149.2, 150.0, 152.7, 163.1 |
| 5h | 1.29(s, 9H), 3.62(d, J=5.5 Hz, 3H), 3.91(s, 3H), 5.09(s, 2H), 7.09—7.25(m, 3H), 7.26—7.45(m, 7H), 8.57(m, 2H) | 31.2, 34.7, 52.7, 63.6, 75.1, 77.0, 122.2, 123.7, 125.2, 127.5, 128.6, 129.3, 129.6, 132.0, 135.9, 141.1, 144.1, 149.3, 149.8, 152.3, 153.0, 154.7, 163.1 |
| 5i | 2.38(d, J=15.5 Hz, 3H), 3.73(d, J=4.6 Hz, 3H), 3.99(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.10—7.30(m, 7H), 7.33—7.49(m, 4H), 8.31(d, J=5.2 Hz, 1H), 8.45(d, J=5.1 Hz, 1H) | 21.4, 52.8, 63.8, 75.2, 120.9, 122.6, 124.2, 127.4, 127.8, 128.8, 129.2, 129.4, 131.4, 135.7, 139.7, 140.3, 144.3, 147.2, 149.2, 149.6, 151.7, 153.6, 154.0, 163.2 |
| 5j | 1.31(s, 9H), 3.72(s, 3H), 3.99(s, 3H), 5.09(s, 2H), 7.16(s, 2H), 7.24(dd, J=1.2, 0.7 Hz, 1H), 7.28—7.47(m, 7H), 8.45(dd, J=5.0, 0.6 Hz, 1H) | 31.2, 31.2, 31.3, 34.8, 52.8, 63.7, 75.0, 122.2, 123.9, 125.2, 127.3, 127.7, 128.4, 128.7, 128.9, 129.2, 129.3, 129.4, 131.9 , 135.9, 141.5, 149.6, 152.4, 153.2, 154.7, 163.2 |
| 5k | 3.71(s, 3H), 3.97(s, 3H), 5.19(s, 2H), 7.19(t, J=7.0 Hz, 2H), 7.27—7.52(m, 10H), 7.57(dd, J=7.8, 3.5 Hz, 4H), 8.38(m, 1H) | 52.8, 63.7, 75.4, 122.7, 124.3, 127.0, 128.0, 129.0, 129.5, 129.8, 133.1, 135.7, 140.0, 142.7, 144.1, 149.3, 149.9, 151.7, 153.4, 163.1 |
Table 3 NMR data of compounds 5a—5k
| Compd. | 1H NMR(300 MHz, CDCl3, δ) | 13C NMR(75 MHz, CDCl3, δ) |
|---|---|---|
| 5a | 3.70(d, J=2.1 Hz, 3H), 3.98(d, J=0.9 Hz, 3H), 5.11(s, 2H), 7.20—7.13(m, 1H), 7.23(dd, J=4.4, 1.6 Hz, 1H), 7.44—7.30(m, 9H), 8.56(dd, J=4.4, 1.7 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 52.8, 63.2, 75.1, 122.1, 123.8, 127.6, 128.5, 129.3, 129.8, 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.7 |
| 5b | 2.37(d, J=15.0 Hz, 3H), 3.71(d, J=0.5 Hz, 3H), 3.98(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.13—7.22(m, 5H), 7.26(s, 1H), 7.28—7.42(m, 4H), 8.51(d, J=6.2 Hz, 1H), 8.67(d, J=5.1 Hz, 1H) | 21.4, 52.8(s), 63.7(s), 75.0, 127.2—128.1(m), 128.1— 130.0(m), 132.0, 135.9, 139.4, 140.0, 141.3, 149.3, 149.7, 154.9, 163.2 |
| 5c | 3.69(s, 3H), 3.83(s, 3H), 3.99(s, 3H), 5.12(s, 2H), 6.88—6.97(m, 2H), 7.12—7.20(m, 1H), 7.28—7.48(m, 7H), 8.55(d, J =5.6 Hz, 2H) | 52.7, 55.2, 63.7, 75.1, 113.7, 113.8, 122.4, 123.7, 123.8, 127.7, 128.4, 128.8, 129.0, 129.4, 129.7, 131.2, 135.9, 141.4, 144.4, 149.3, 149.8, 154.5, 160.2, 160.9, 163.1 |
| 5d | 2.32—2.41(m, 3H), 3.70(s, 3H), 3.97(d, J=2.1 Hz, 3H), 5.11(s, 2H), 7.12—7.18(m, 2H), 7.21(dd, J=4.4, 1.6 Hz, 1H), 7.26— 7.47(m, 6H), 8.56(dd, J=4.7, 1.4 Hz, 1H), 8.69(dd, J=4.5, 1.5 Hz, 1H) | 20.1, 52.8, 63.7, 75.2, 122.0, 123.8, 126.3, 128.0, 128.1, 128.5, 128.8, 129.1, 129.4, 129.9, 131.5, 133.3, 135.6, 136.3, 140.9, 143.7, 149.2, 149.8, 154.0, 163.1 |
| 5e | 1.25(d, J=6.9 Hz, 6H), 2.77—3.05(m, 1H), 3.68(d, J=4.7 Hz, 3H), 3.97(s, 3H), 5.11(s, 2H), 7.10—7.52(m, 11H), 8.54(d, J=5.5 Hz, 1H), 8.67(d, J=5.5 Hz, 1H) | 23.8, 33.9, 52.7, 63.7, 75.0, 122.2, 123.8, 126.4, 127.7, 128.6, 129.4, 132.4, 135.9, 141.2, 144.2, 149.3, 149.8, 150.1, 150.8, 154.8, 163.1 |
| 5f | 3.71(d, J=1.7 Hz, 3H), 3.98(d, J=2.7 Hz, 3H), 5.10(s, 2H), 7.22—7.13(m, 3H), 7.31—7.23(m, 3H), 7.59—7.35(m, 5H), 8.57(dd, J=4.5, 1.6 Hz, 1H), 8.68(dd, J=3.0, 1.6 Hz, 1H) | 52.8, 63.7, 75.1, 122.1, 123.9, 127.7, 128.1—130.0(m), 131.6, 134.8, 135.8, 141.2, 144.0, 149.3, 149.7, 154.8, 163.2 |
| 5g | 3.71(d, J=2.6 Hz, 3H), 3.97(d, J=1.6 Hz, 3H), 5.11(s, 2H), 7.11—7.20(m, 3H), 7.25—7.30(m, 1H), 7.38(m, 4H), 7.50(dd, J= 5.2, 2.9 Hz, 1H), 8.57(dd, J=4.5, 1.5 Hz, 1H), 8.70(dd, J=3.0, 1.5 Hz, 1H) | 52.8, 63.7, 75.5, 121.7, 123.5, 126.8, 127.9, 128.6, 129.1, 129.4, 130.4, 131.1, 132.8, 133.6, 133.9, 134.8, 135.4, 139.9, 142.8, 149.2, 150.0, 152.7, 163.1 |
| 5h | 1.29(s, 9H), 3.62(d, J=5.5 Hz, 3H), 3.91(s, 3H), 5.09(s, 2H), 7.09—7.25(m, 3H), 7.26—7.45(m, 7H), 8.57(m, 2H) | 31.2, 34.7, 52.7, 63.6, 75.1, 77.0, 122.2, 123.7, 125.2, 127.5, 128.6, 129.3, 129.6, 132.0, 135.9, 141.1, 144.1, 149.3, 149.8, 152.3, 153.0, 154.7, 163.1 |
| 5i | 2.38(d, J=15.5 Hz, 3H), 3.73(d, J=4.6 Hz, 3H), 3.99(d, J=3.1 Hz, 3H), 5.10(s, 2H), 7.10—7.30(m, 7H), 7.33—7.49(m, 4H), 8.31(d, J=5.2 Hz, 1H), 8.45(d, J=5.1 Hz, 1H) | 21.4, 52.8, 63.8, 75.2, 120.9, 122.6, 124.2, 127.4, 127.8, 128.8, 129.2, 129.4, 131.4, 135.7, 139.7, 140.3, 144.3, 147.2, 149.2, 149.6, 151.7, 153.6, 154.0, 163.2 |
| 5j | 1.31(s, 9H), 3.72(s, 3H), 3.99(s, 3H), 5.09(s, 2H), 7.16(s, 2H), 7.24(dd, J=1.2, 0.7 Hz, 1H), 7.28—7.47(m, 7H), 8.45(dd, J=5.0, 0.6 Hz, 1H) | 31.2, 31.2, 31.3, 34.8, 52.8, 63.7, 75.0, 122.2, 123.9, 125.2, 127.3, 127.7, 128.4, 128.7, 128.9, 129.2, 129.3, 129.4, 131.9 , 135.9, 141.5, 149.6, 152.4, 153.2, 154.7, 163.2 |
| 5k | 3.71(s, 3H), 3.97(s, 3H), 5.19(s, 2H), 7.19(t, J=7.0 Hz, 2H), 7.27—7.52(m, 10H), 7.57(dd, J=7.8, 3.5 Hz, 4H), 8.38(m, 1H) | 52.8, 63.7, 75.4, 122.7, 124.3, 127.0, 128.0, 129.0, 129.5, 129.8, 133.1, 135.7, 140.0, 142.7, 144.1, 149.3, 149.9, 151.7, 153.4, 163.1 |
| Compd. | CL | BB | RS | PC | PA | PI | BC |
|---|---|---|---|---|---|---|---|
| 5a | 51.6 | 31.5 | 50.3 | 42.7 | 32.2 | 62.3 | 9.6 |
| 5b | 46.2 | 32.6 | 48.3 | -5.7 | 33.6 | 68.6 | 27.4 |
| 5c | 43.0 | 23.2 | 45.1 | 50.0 | 30.6 | 59.1 | 26.0 |
| 5d | 66.2 | 54.5 | 61.3 | 59.0 | 49.9 | 76.6 | 32.9 |
| 5e | 48.1 | 13.9 | 45.1 | 47.2 | 21.2 | 66.2 | 9.6 |
| 5f | 52.2 | 34.7 | 44.0 | -6.2 | 33.3 | 68.1 | 37.0 |
| 5g | 50.8 | 31.5 | 46.0 | 36.5 | 43.3 | 71.3 | 27.4 |
| 5h | 49.8 | 26.7 | 33.3 | 44.4 | 36.6 | 41.0 | 4.1 |
| 5i | 43.3 | 5.3 | 26.1 | 11.7 | 34.4 | 39.9 | 5.5 |
| 5j | 33.3 | 1.6 | 9.9 | 17.9 | 26.4 | 25.6 | - |
| 5k | 6.8 | -25.7 | -7.2 | 6.1 | -8.3 | 8.0 | 2.7 |
| Azoxystrobin | 54.3 | 33.7 | 52.6 | 29.2 | 35.0 | 81.9 | 37.0 |
| Trifloxystrobin | 60.8 | 11.7 | 40.5 | 24.7 | 42.1 | 70.5 | 60.3 |
Table 4 In vitro fungicidal activity(inhibition ratio, %) of compounds 5a—5k(50 mg/L)*
| Compd. | CL | BB | RS | PC | PA | PI | BC |
|---|---|---|---|---|---|---|---|
| 5a | 51.6 | 31.5 | 50.3 | 42.7 | 32.2 | 62.3 | 9.6 |
| 5b | 46.2 | 32.6 | 48.3 | -5.7 | 33.6 | 68.6 | 27.4 |
| 5c | 43.0 | 23.2 | 45.1 | 50.0 | 30.6 | 59.1 | 26.0 |
| 5d | 66.2 | 54.5 | 61.3 | 59.0 | 49.9 | 76.6 | 32.9 |
| 5e | 48.1 | 13.9 | 45.1 | 47.2 | 21.2 | 66.2 | 9.6 |
| 5f | 52.2 | 34.7 | 44.0 | -6.2 | 33.3 | 68.1 | 37.0 |
| 5g | 50.8 | 31.5 | 46.0 | 36.5 | 43.3 | 71.3 | 27.4 |
| 5h | 49.8 | 26.7 | 33.3 | 44.4 | 36.6 | 41.0 | 4.1 |
| 5i | 43.3 | 5.3 | 26.1 | 11.7 | 34.4 | 39.9 | 5.5 |
| 5j | 33.3 | 1.6 | 9.9 | 17.9 | 26.4 | 25.6 | - |
| 5k | 6.8 | -25.7 | -7.2 | 6.1 | -8.3 | 8.0 | 2.7 |
| Azoxystrobin | 54.3 | 33.7 | 52.6 | 29.2 | 35.0 | 81.9 | 37.0 |
| Trifloxystrobin | 60.8 | 11.7 | 40.5 | 24.7 | 42.1 | 70.5 | 60.3 |
| Compd. | 400 mg/L(Spraying assay) | 25 mg/L(Spores germ. assay) | ||||
|---|---|---|---|---|---|---|
| CDM | WPM | CSR | CA | RB | CGM | |
| 5a | 0 | 0 | 0 | 0 | 100 | 0 |
| 5b | 0 | 0 | 85 | 95 | 100 | 0 |
| 5c | 0 | 0 | 70 | 60 | 80 | 0 |
| 5d | 70 | 0 | 0 | 0 | 100 | 0 |
| 5e | 0 | 95 | 50 | 0 | 100 | 0 |
| 5f | 0 | 0 | 80 | 0 | 100 | 0 |
| 5g | 35 | 0 | 0 | 0 | 100 | 0 |
| 5h | 0 | 100 | 0 | 100 | 80 | 0 |
| 5i | 0 | 0 | 80 | 0 | 80 | 0 |
| 5j | 0 | 0 | 0 | 0 | 100 | 0 |
| 5k | 0 | 50 | 0 | 0 | 0 | 0 |
| Azoxystrobin | 0 | 100 | 100 | 80 | 80 | 0 |
| Trifloxystrobin | 50 | 100 | 90b | 100 | 100 | 0 |
Table 5 In vivo fungicidal activity(inhibition ratio, %) of compounds 5a—5k*
| Compd. | 400 mg/L(Spraying assay) | 25 mg/L(Spores germ. assay) | ||||
|---|---|---|---|---|---|---|
| CDM | WPM | CSR | CA | RB | CGM | |
| 5a | 0 | 0 | 0 | 0 | 100 | 0 |
| 5b | 0 | 0 | 85 | 95 | 100 | 0 |
| 5c | 0 | 0 | 70 | 60 | 80 | 0 |
| 5d | 70 | 0 | 0 | 0 | 100 | 0 |
| 5e | 0 | 95 | 50 | 0 | 100 | 0 |
| 5f | 0 | 0 | 80 | 0 | 100 | 0 |
| 5g | 35 | 0 | 0 | 0 | 100 | 0 |
| 5h | 0 | 100 | 0 | 100 | 80 | 0 |
| 5i | 0 | 0 | 80 | 0 | 80 | 0 |
| 5j | 0 | 0 | 0 | 0 | 100 | 0 |
| 5k | 0 | 50 | 0 | 0 | 0 | 0 |
| Azoxystrobin | 0 | 100 | 100 | 80 | 80 | 0 |
| Trifloxystrobin | 50 | 100 | 90b | 100 | 100 | 0 |
| [1] | Yang G.L.,Chem. Ind., 2013, 31(12), 45—47 |
| (杨光亮. 化学工业, 2013, 31(12), 45—47) | |
| [2] | Zhang G. S., Pesticides Science and Administration, 2003, 24(12), 30—34 |
| (张国生. 农药科学与管理, 2003, 24(12), 30—34) | |
| [3] | Wang L., Shi Y. X., Li B. J., Liu C. L., Xiang W. S., Pesticides Science and Administration, 2008, 29(9), 24—27 |
| (王丽, 石延霞, 李宝聚, 刘长令, 向文胜. 农药科学与管理, 2008, 29(9), 24—27) | |
| [4] | Guan A. Y., Li H. C., Zhang J. B., Sun X. F., Wang L. Z., Fine and Specialty, 2012, 20(4), 24—28 |
| (关爱莹, 李慧超, 张金波, 孙旭峰, 王立增. 精细与专用化学品, 2012, 20(4), 24—28) | |
| [5] | Zhao P., China Agrochemicals, 2012, 1, 24—30 |
| (赵平. 中国农药. 2012, 1, 24—30) | |
| [6] | Masaji O., Chikako O., Takafumi T., Agricultural Microbicides Containing Methoxyiminoacetate Derivatives, JP 05255012A, 1993-10-05 |
| [7] | Nakafawa Y., Mitani A., Sano H., Hamamura H., Ando T., Sugiura T., Ito S., Preparation of 2-Pyridinecarboxaldehyde Oxime Heterocyclylmethyl Ether Compounds and Agricultural or Horticultural Fungicides, EP 20020700611, 2002-08-29 |
| [8] | Yuan L. P., Chen L., Sheng Z., Zhang Y. B., Huang J. M., Wang M., Qin X. S., Cao J., Li X. L., Preparation of Substituted Propiophenone Oxime Derivatives as Agricultural Bactericides, CN 1640868A, 2005-07-20 |
| (袁莉萍, 陈亮, 沈宙, 张一宾, 黄建明, 王敏, 秦旭升, 曹谨, 栗秀丽. 取代苯丙酮肟衍生物及其制备方法和应用, CN 1640868A, 2005-07-20) | |
| [9] | Dai H., Liu J. B., Miao W. K., Wu S. S., Qin X., Zhang X., Wang T. T., Fang J. X., Chin. J. Org. Chem., 2011, 31(10), 1662—1667 |
| (戴红, 刘建兵, 苗文科, 吴珊珊, 秦雪, 张欣, 王婷婷, 方建新. 有机化学, 2011, 31(10), 1662—1667) | |
| [10] | Mu C. W., Qin Z. H., Mod. Pestic., 2003, 2(2), 1—6 |
| (慕长炜, 覃兆海. 现代农药, 2003, 2(2), 1—6) | |
| [11] | Zhao W. G., Wang J. G., Yuan D. K., Luo T. J., Li Z. M., Pestic., 2002, 41(7), 8—11 |
| (赵卫光, 王建国, 袁德凯, 罗铁军, 李正名. 农药, 2002, 41(7), 8—11) | |
| [12] | Xu J. Y., Dong W. L., Xiong L. X., Li Z. M., Chem. J. Chinese Universities,2012, 33(2), 298—302 |
| (徐俊英, 董卫莉, 熊丽霞, 李正名. 高等学校化学学报,2012, 33(2), 298—302) | |
| [13] | Pei J. J., Ouyang G. P., Zou L. B., Fine Chemical Intermediates,2014, 44(1), 1—9 |
| (裴娟娟, 欧阳贵平, 邹骆波. 精细化工中间体,2014, 44(1), 1—9) | |
| [14] | Massolini G., Kitsos M., Gandini C., Caccialanza G., Pestic. Sci., 1989, 26(2), 209—214 |
| [15] | Shuman R. F., Amstutz E. D., Recueil des Travaux Chimiques des Pays-Bas, 1965, 84(4), 441—459 |
| [16] | Massolini G., Kitsos M., Gandini C., Caccialanza G., Pestic. Sci., 1989, 26(2), 209—214 |
| [17] | Otsuka K., Ishiyama N., Watanabe M., Seri K., Sanai K., Muraoka K., Pyridylketoxime Ethers as Calmodulin Antagonists and Their Preparation EP 366006A1,1990-05-02 |
| [18] | Xu J. G., Zhao X. M., Han X. W., Du Y. G., Pestic. Biochem. Physiol., 2007, 87(3), 220—228 |
| [19] | Li M., Zhang J. B., Yang J. C., Li Z. N., Liu C. L., Li Z. M., Chem. J. Chinese Universities,2009, 30(7), 1348—1352 |
| (李淼, 张金波, 杨吉春, 李志念, 刘长令, 李正名. 高等学校化学学报,2009, 30(7), 1348—1352) |
| [1] | 李康明, 李延赛, 易阳杰, 徐雷涛, 叶姣, 欧晓明, 李建明, 胡艾希. 5-吡唑甲酰胺类衍生物的设计、 合成与生物活性[J]. 高等学校化学学报, 2020, 41(4): 716. |
| [2] | 董心睿, 夏喆, 王桢学, 边强, 李华斌. 含1,2,4,5-四取代苯基的吡唑-4-甲酰胺类化合物的设计、 合成及生物活性[J]. 高等学校化学学报, 2020, 41(12): 2759. |
| [3] | 谢瑞龙, 宋越, 杨新玲, 汪梅子, 凌云. 新型瑞香酮类似物的合成及杀菌活性[J]. 高等学校化学学报, 2014, 35(7): 1451. |
| [4] | 杨海葵, 许万福, 段安娜, 游文玮, 赵培亮. ,4-三唑环的亚胺及酰亚胺类化合物的合成及生物活性[J]. 高等学校化学学报, 2014, 35(3): 555. |
| [5] | 于岚岚, 毛烨炫, 白希希, 冉瑜, 李爱荣, 朱艳艳, 于斐, 屈凌波. 具有双活性序列的新型抗菌肽的设计及性质[J]. 高等学校化学学报, 2013, 34(5): 1166. |
| [6] | 于岚岚, 冉瑜, 白希希, 李爱荣, 朱艳艳, 覃韵, 屈凌波. 新型抗菌肽的设计、活性研究及与磷脂相互作用的计算模拟[J]. 高等学校化学学报, 2012, 33(12): 2681. |
| [7] | 姚红伟, 崔灿, 李永强, 王力钟, 李正名, 赵卫光. N-[(2-烷氧基)-取代苯乙基]-苯乙酰胺类化合物的合成及生物活性[J]. 高等学校化学学报, 2012, 33(07): 1481. |
| [8] | 冯启 刘智力 王明忠 熊丽霞 于淑晶 李正名. 含七氟异丙基的氯虫酰胺类似物的设计合成及生物活性[J]. 高等学校化学学报, 2011, 32(1): 74. |
| [9] | 李欢欢 王振军 王力钟 李正名 赵卫光. α-取代苯乙酰胺类化合物的合成及生物活性[J]. 高等学校化学学报, 2011, 32(1): 79. |
| [10] | 李洋, 张志国, 迟会伟, 罗艳梅, 刘长令, 李正名. 2-酰基-氰基乙酸衍生物的合成及生物活性[J]. 高等学校化学学报, 2010, 31(9): 1798. |
| [11] | 李淼, 张金波, 杨吉春, 李志念, 刘长令, 李正名. 新型含吡唑环的N-甲氧基氨基甲酸甲酯类化合物的设计、合成及生物活性[J]. 高等学校化学学报, 2009, 30(7): 1348. |
| [12] | 王艳华, 杜锡光, 吴晓燕, 朱超光, 杜大峰, 朱东升. 新型双大环二正丁基锡羧酸酯的合成、晶体结构及生物活性[J]. 高等学校化学学报, 2008, 29(9): 1781. |
| [13] | 邵玲, 张青, 周欣, 金钟, 刘建兵, 方建新 . 新型含噻唑和三唑环的亚胺类化合物的合成及生物活性研究[J]. 高等学校化学学报, 2007, 28(2): 270. |
| [14] | 慕长炜,袁会珠,李楠,傅滨,肖玉梅,马永强,齐淑华,覃兆海 . 4-[3-(吡啶-4-基)-3-取代苯基丙烯酰]吗啉类化合物的合成及杀菌活性[J]. 高等学校化学学报, 2007, 28(10): 1902. |
| [15] | 金钟, 刘伟, 霍爱红, 刘建兵, 方建新. 3-芳基-1-二茂铁基-2-(1H-1,2,4-三唑-1-基)-2-丙烯酮类衍生物的选择性还原、产物结构表征及其生物活性研究[J]. 高等学校化学学报, 2005, 26(9): 1637. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||